Abstract
Background:
As no direct comparison is available between drugs to prevent endoscopic postoperative recurrence (POR) in Crohn’s disease (CD), hierarchizing these therapeutic options remains challenging.
Objectives:
We aimed to compare the effectiveness of treatments to prevent CD endoscopic POR.
Design:
Systematic review and network meta-analysis using a random-effects model.
Data sources and methods:
We include studies comparing treatments to prevent CD POR according to PRISMA guidelines. The primary endpoint was endoscopic POR (Rutgeerts score ⩾i2). Surface under the cumulative ranking (SUCRA) was used to hierarchize the treatments.
Results:
Twenty studies were included (2414 patients). Overall heterogeneity was moderate (τ = 0.34). Ustekinumab (odds ratio (OR) = 0.23 (0.07–0.70); OR = 0.29 (0.08–0.99)), vedolizumab (OR = 0.17 (0.05–0.59); OR = 0.22 (0.06–0.85)), infliximab (OR = 0.18 (0.36–0.88); OR = 0.23 (0.09–0.54)), and adalimumab (OR = 0.17 (0.07–0.42); OR = 0.22 (0.08–0.59)) were more effective to prevent endoscopic POR than placebo or 5-ASA, respectively, contrary to thiopurines (OR = 0.52 (0.22–1.24); OR = 0.66 (0.25–1.76)). Adalimumab (OR = 0.33 (0.15–0.74)) and infliximab (OR = 0.34 (0.13–0.87)) were more effective than thiopurines. While no difference was observed between the four biologics, adalimumab (SUCRA = 0.81), infliximab (SUCRA = 0.80), vedolizumab (SUCRA = 0.79), and ustekinumab (SUCRA = 0.72) had the highest likelihood of being the most effective drug, contrary to thiopurines (SUCRA = 0.41), 5-ASA (SUCRA = 0.24), or placebo (SUCRA = 0.16).
Conclusion:
This network meta-analysis confirms the efficacy of anti-TNF agents, vedolizumab, and ustekinumab in preventing endoscopic CD POR without any difference between them. When a prophylactic therapy is needed, biologics should be preferred to 5-ASA or thiopurines.
Trial registration:
PROSPERO registration number CRD42024555528.
Plain language summary
After surgery for Crohn’s disease (CD), many patients experience a return of inflammation in the gut, even if they have no symptoms at first. This is called endoscopic postoperative recurrence (POR), and it can lead to complications over time. To reduce this risk, doctors may prescribe medications right after surgery. However, it has been unclear which drugs work best, because few studies have directly compared them. In this study, we reviewed and analyzed the results of 20 studies including over 2,400 patients with CD who had undergone bowel surgery. We compared different medications used to prevent endoscopic POR. These included biologic drugs like infliximab, adalimumab, vedolizumab, and ustekinumab, as well as older treatments like thiopurines and 5-ASA (a type of anti-inflammatory drug). We found that the biological therapies were significantly more effective than placebo and 5-ASA at preventing inflammation from returning. Specifically, infliximab, adalimumab, vedolizumab, and ustekinumab all reduced the risk of endoscopic POR. Adalimumab and infliximab were also more effective than thiopurines. There was no clear difference in effectiveness between the four biologics. In contrast, thiopurines and 5-ASA were less effective and had results similar to placebo in many cases. In conclusion, if a patient with CD needs medication to prevent disease recurrence after surgery, biological therapies are likely to offer the most protective effect. Older drugs like 5-ASA and thiopurines may not be as effective in this situation.
Introduction
Crohn’s disease (CD) is a chronic inflammatory disorder that can cause bowel damage such as strictures or fistulas, and lead to altered patients’ quality of life and disability. 1 Although the widespread use of biological therapies during the past decades has contributed to reduce the need for surgery, bowel resection is still required in more than 25% of patients 10 years after the diagnosis. 2 Since surgery is not curative, postoperative recurrence (POR) remains a major concern in patients with CD. The landmark historical cohort from Rutgeerts et al. 3 described the natural history (i.e., without any prophylactic treatment) of endoscopic POR (reappearance of endoscopic lesions without any symptoms) and reported that 90% of patients experienced endoscopic POR within 5 years after surgery, with about half of them developing CD-related symptoms (i.e., clinical POR). Of note, the risk of endoscopic POR is estimated at 75% within the first year after surgery in patients who did not receive any preventive treatment, 4 which is highly predictive of symptomatic recurrence (clinical POR). 3 To mitigate this risk, several strategies have been studied and showed their benefits. The POCER trial demonstrated that a strategy based on systematic colonoscopy at 6 months after surgery, combined with therapeutic escalation in case of endoscopic POR, is associated with better outcomes. 5 Due to the high risk of recurrence, initiating prophylactic medication to prevent endoscopic POR is probably the best option for most patients.6,7 However, 10%–15% of CD patients in the historical cohort from Leuven did not experience any clinical recurrence within 5 years after surgery, despite not receiving any prophylactic medication. 3 To date, the stratification of patients based on their risk factors is still imperfect to distinguish high-risk patients who will benefit from a systematic prophylactic treatment from low-risk patients, as the performances of the identified risk factors remain disappointed. Nevertheless, when preventive treatment is decided, several options are available, ranging from conventional therapies (5-ASA,8–11 thiopurines 12 ) to biologics (anti-TNF agents,13–16 vedolizumab (REPREVIO), ustekinumab 17 ). Because only a few head-to-head comparisons of drug efficacy to prevent endoscopic POR are available, prioritizing these therapeutic options remains challenging.
We conducted a systematic literature review to identify studies comparing the efficacy of drugs in preventing endoscopic POR after ileo-cecal resection for CD, followed by a network meta-analysis, comparing the effectiveness of all available therapies.
Methods
Search strategy and selection criteria
These systematic reviews and network meta-analyses were performed according to the PRISMA guidelines 1 and were registered on the website of the International Prospective Register of Systematic Reviews (PROSPERO; registration number: CRD42024555528). Two authors (M.H. and A.B.) searched independently MEDLINE (via PubMed) and CENTRAL (Cochrane) for articles published up to July 7, 2025, with no additional time restriction. Previous meta-analyses were also screened to identify additional references that may have been missed by our systematic search. No language restrictions were applied. Disagreements were resolved through discussion between the authors until a consensus was reached. Both prospective and retrospective studies were eligible for inclusion in this network meta-analysis if led in patients over 18 years old, comparing at least two treatment arms for the prevention of POR of CD after ileo-colonic resection. An endoscopic evaluation of POR using Rutgeerts’ score had to be available within the first year following bowel continuity restoration. Data were collected in an electronic case report form to enable subsequent analysis. The quality of the selected studies was evaluated independently by two authors (M.H. and A.B.) using the Cochrane Risk of Bias tools (RoB2 for randomized trials and ROBINS-I for non-randomized studies).
Data collection
Study data were collected and managed using REDCap (Research Electronic Data Capture) tools (For more details, please see Supplemental Appendix). In each study, the following data were collected: number of patients, timing of treatment and endoscopy, Rutgeerts’ score, type of study, and whether patients were selected on their risk of recurrence. For the studies retrieved from scientific meetings and not fully published so far, data were extracted from the abstract or after a request to the authors.
Outcome
In accordance with international guidelines for postoperative CD, the primary endpoint was endoscopic POR defined as Rutgeerts’ score ⩾i2.18,19
Statistical analyses
We performed a network meta-analysis using the frequentist model, with the statistical package “netmeta” V.0.9–0 in R V.4.0.2, R core team, Vienna (Austria). We explored direct and indirect comparisons of efficacy between available drugs and placebo. Possible bias/inconsistency20,21 was evaluated by comparison of direct evidence with the indirect evidence. Heterogeneity among studies was evaluated by I2 tests, with values greater than 50% suggesting substantial heterogeneity as well as by τ² (global statistical heterogeneity across all comparisons). As previously described, we used estimates of τ² of approximately 0.04, 0.16, and 0.36 to consider the level of heterogeneity as low, moderate, and high. A random-effects model was used to give a more conservative estimate of the effect of each drug, allowing for heterogeneity between studies. A sensitivity analysis was led, including only randomized studies. Results are expressed in odds ratios with 95% confidence intervals. The relative ranking of drugs for the prevention of endoscopic recurrence was calculated with their surface under the cumulative ranking (SUCRA). The SUCRA represents the percentage of efficacy achieved by an agent compared with an imaginary agent that is always the best without uncertainty: a higher SUCRA means a higher ranking for efficacy outcomes. SUCRA rankings: SUCRA is helpful for hierarchy but should be interpreted with caution, because small differences may not reflect clinically meaningful superiority.
Role of the funding source
There was no funding source for this study.
Results
Bibliographical research and article selection
The search equations and strategies generate 9700 citations and are available in Supplemental Appendix 1. After removing duplicates (n = 1274), 8426 studies were screened for eligibility (Figure 1). Seven previous systematic reviews were also screened.2–8

Search strategy according to PRISMA guidelines.
A total of 20 studies (including 15 randomized controlled trials) were selected for this meta-analysis, encompassing a total of 41 arms (2414 patients). Among them, 32 were active arms representing 2109 patients, while 9 arms evaluated the efficacy of a placebo. The investigated treatments were conventional (5-ASA, antibiotics, thiopurines) or biological therapies (adalimumab, infliximab, ustekinumab, vedolizumab). Immunosuppressants and antibiotics (metronidazole) were administered in association with other drugs to, respectively, 347 (16.4%) and 172 (8.2%) patients. Overall, 555 (between 0% and 84% within the studies) patients had been exposed to at least one biological therapy prior to surgery, and 591 (8.0%–51.5% within the studies) had required at least one previous surgery. For 16 studies, the presence of risk factors was part of the inclusion or exclusion criteria. In most of the analyzed studies, prophylactic medications were started within 4 weeks after surgery, and endoscopic evaluation was performed at 6 months.
Evaluation of risk of bias and heterogeneity
Risk of bias and support for judgment are detailed in Figure 2 for randomized controlled trials and Figure 3 for observational studies. Overall heterogeneity was moderate, with a τ² = 0.34 (global statistical heterogeneity across all comparisons), I2 = 63% (35%; 79%).
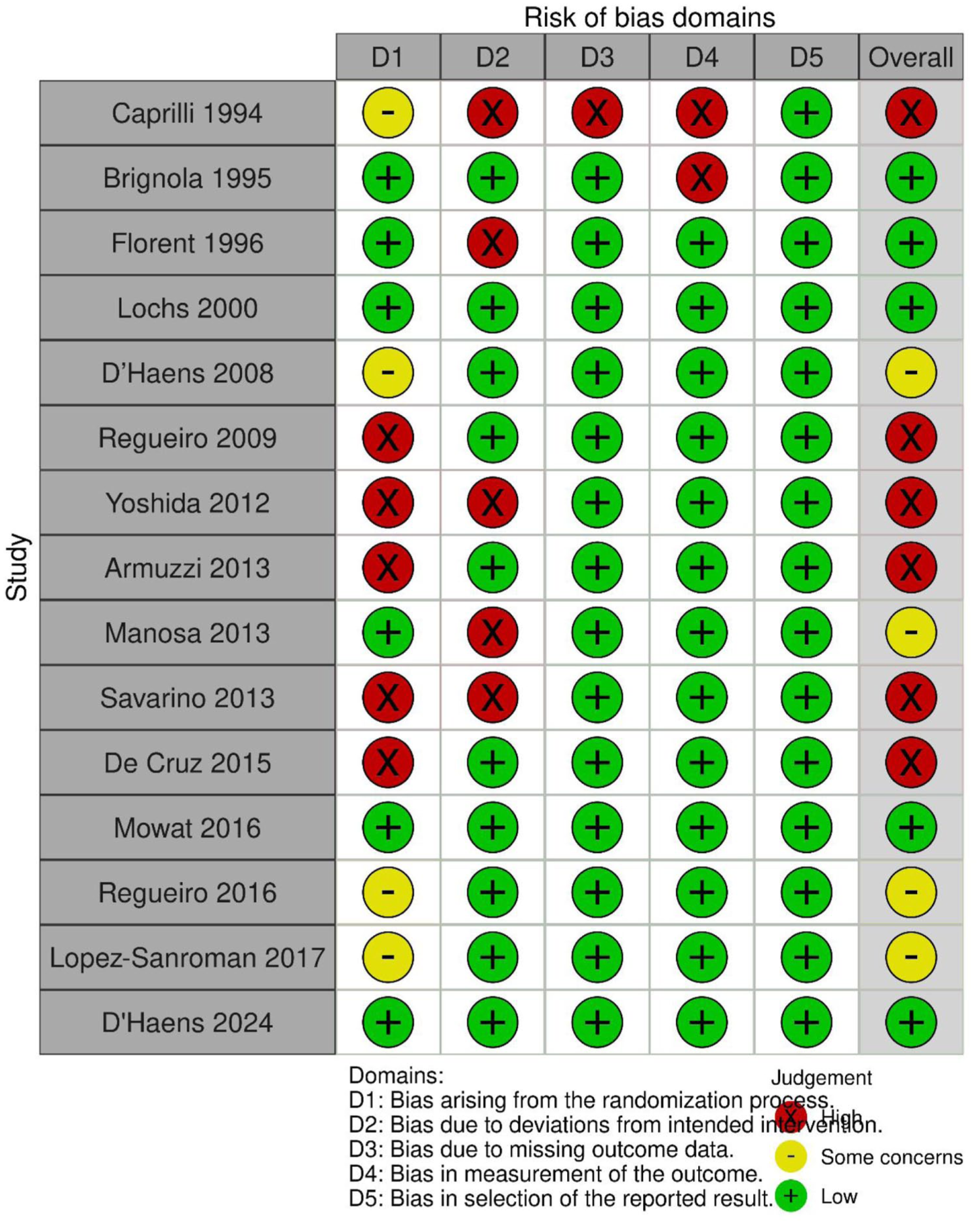
Risk of bias summary: authors’ judgments about each risk of bias item for each included randomized controlled trial.

Risk of bias summary: authors’ judgments about each risk of bias item for each included observational study. Allez et al. have an unknown risk due to insufficient information; this study was retrieved through scientific meetings’ abstracts.
Prevention of endoscopic POR
The network plot is displayed in Figure 4. Details regarding the studies included in the meta-analysis are available in Supplemental Tables S1 and S2. Ustekinumab (OR = 0.23 (0.07–0.70)), vedolizumab (OR = 0.17 (0.05–0.59)), infliximab (OR = (0.18 (0.36–0.88)), and adalimumab (OR = 0.17 (0.07–0.42)) were superior to placebo to prevent endoscopic POR. On the other hand, antibiotics alone (OR = 1.52 (0.27–8.57)), 5-ASA (OR = 0.79 (0.42–1.48)), and thiopurines (RR = 0.52 (0.22–1.24)) were not significantly superior to placebo (Table 1). Ustekinumab (OR = 0.29 (0.08–0.99)), vedolizumab (OR = 0.22 (0.06–0.85)), infliximab (OR = (0.23 (0.09–0.54)), and adalimumab (OR = 0.22 (0.08–0.59)) were more effective than 5-ASA. Adalimumab (OR = 0.33 (0.15–0.74)) and infliximab (OR = 0.34 (0.13–0.87)) showed higher efficacy than thiopurines. No difference was found between biological therapies included in this analysis (adalimumab, infliximab, ustekinumab, vedolizumab; Table 1). The medications were ranked from the most to the least effective as follows: adalimumab (SUCRA = 0.81), infliximab (SUCRA = 0.80), vedolizumab (SUCRA = 0.79), ustekinumab (SUCRA = 0.72), thiopurines (SUCRA = 0.41), 5-ASA (SUCRA = 0.24), placebo (SUCRA = 0.16), or antibiotics (SUCRA = 0.08; Figure 5). The sensitivity analysis conducted on randomized controlled trials led to the following SUCRA ranking: infliximab (SUCRA = 0.81), adalimumab (SUCRA = 0.80), vedolizumab (SUCRA = 0.76), thiopurines (SUCRA = 0.50), 5-ASA (SUCRA = 0.28), antibiotics (SUCRA = 0.19), and placebo (SUCRA = 0.16).
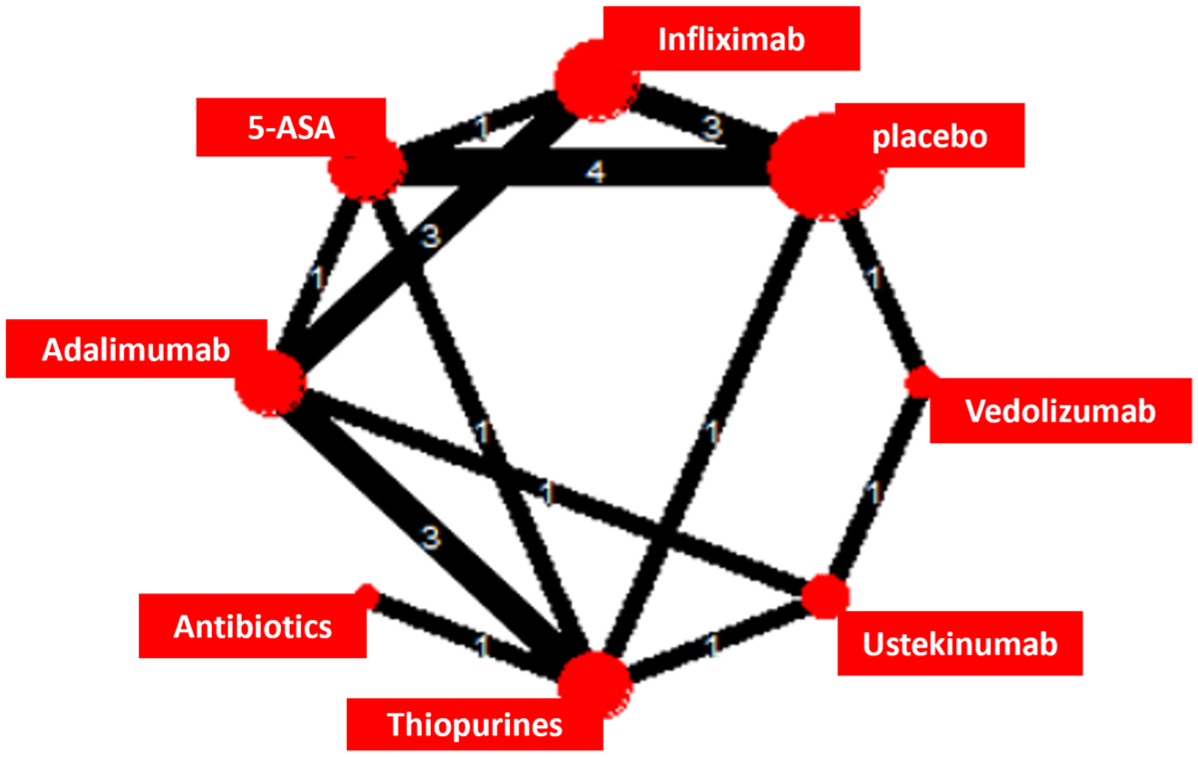
Network plot of the meta-analysis.
League table reporting the efficacy of medications for preventing endoscopic postoperative recurrence in Crohn’s disease.

(Upper part of the table reports direct comparison while lower part of the table is reporting indirect comparisons). Bold values with green highlights indicate statistically significant differences.

SUCRA ranking of the studied treatment.
Discussion
This network meta-analysis confirms the efficacy of anti-TNF agents, vedolizumab, and ustekinumab in preventing endoscopic POR in patients with CD, with no significant difference between these treatments. Our results suggest that biologics represent the most effective therapeutic option for preventing endoscopic POR when prophylactic therapy is indicated. Eventually, the use of 5-ASA and thiopurines should be limited in this setting owing to the availability of more effective alternatives.
Transitivity is a key assumption in network meta-analysis. In ours, thanks to a standardized postoperative management, most included studies were comparable regarding interventions (timing of treatment initiation), comparators (despite differences in study design between interventional and observational designs), and outcomes (consensual definition of endoscopic POR). However, the presence and number of potential risk factors for endoscopic POR varied across studies and must be considered when interpreting the results. In addition, as antibiotics were sometimes used as an add-on therapy, we adjusted our comparisons on this factor to limit its potential impact. Overall heterogeneity remained moderate with τ² = 0.34. Despite the limited number of direct comparisons, the consistency (agreement between direct and indirect comparisons) was acceptable. Nonetheless, only a few studies included in this network meta-analysis were at low risk of bias, warranting caution when interpreting the findings.
In this network meta-analysis, we confirmed the efficacy of anti-TNF agents to prevent endoscopic POR compared to placebo, 5-ASA, and thiopurines. This result was expected and aligns with a previous study showing strong evidence with anti-TNF therapy in this context.13–16,22,23 Notably, our meta-analysis is the first to include data on vedolizumab and ustekinumab. Inclusion of the REPREVIO randomized controlled trial allowed us to confirm the efficacy of vedolizumab. Interestingly, we also highlighted the efficacy of ustekinumab despite the absence of a dedicated randomized controlled trial. 17 We did not observe any signal suggesting the superiority of one biologic over another, which is consistent with results from a head-to-head randomized controlled trial 24 and other network meta-analysis 25 in bio-naïve patients with CD, where no clear therapeutic hierarchy has been established. At the time of data collection, no data were available on anti-IL23 agents such as risankizumab, even though this therapeutic class appears promising due to a favorable efficacy/safety profile and may emerge as an additional option in the postoperative setting. Furthermore, no JAK inhibitors have yet been evaluated as prophylactic treatment after surgery. Given their safety profiles and the availability of alternatives with similar efficacy, widespread use of JAK inhibitors as prophylactic treatment is not currently supported.
The question of optimal patient selection for early treatment to prevent endoscopic POR remains controversial and is beyond the scope of our analysis. However, when prophylactic treatment is deemed necessary, our data support the use of biological therapy, as we found no evidence of superiority of 5-ASA and thiopurines over placebo. While 5-ASA has shown some marginal efficacy in preventing POR, the effect size is small and of questionable clinical relevance. 26 Thiopurines have demonstrated some benefit, but the effect is modest (approximately a 15% absolute reduction in recurrence) 12 or limited to specific subpopulations. 27 As with the evolving management of luminal CD, the use of thiopurines as monotherapy should be discouraged in favor of more effective therapeutic alternative.
Several limitations have to be acknowledged and should be taken into account to avoid overinterpreting the results of our meta-analysis. These include heterogeneity in study quality, variable risk of bias, and differences in baseline risk of recurrence across studies. However, our work is the first network meta-analysis in the postoperative CD, including data on ustekinumab and vedolizumab efficacy, providing valuable guidance for clinical practice.
In conclusion, this network meta-analysis confirms the efficacy of anti-TNF agents but also vedolizumab and ustekinumab in preventing endoscopic CD POR, with no clear superiority among these options. When a prophylactic treatment is indicated, biological therapies are the best therapeutic option to prevent endoscopic POR in CD. Due to lower efficacy, the use of 5-ASA and thiopurines should be limited in this situation. Our findings highlight the need for further high-quality, head-to-head comparative trials (particularly including newer therapeutic classes) to improve the evidence base for treatment decisions and support clinicians in preventing POR in patients with CD.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251374240 – Supplemental material for Systematic review and network meta-analysis comparing the efficacy of conventional therapy and biologics to prevent endoscopic postoperative recurrence in patients with Crohn’s disease
Supplemental material, sj-docx-1-tag-10.1177_17562848251374240 for Systematic review and network meta-analysis comparing the efficacy of conventional therapy and biologics to prevent endoscopic postoperative recurrence in patients with Crohn’s disease by Marianne Hupé, Bruno Pereira and Anthony Buisson in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848251374240 – Supplemental material for Systematic review and network meta-analysis comparing the efficacy of conventional therapy and biologics to prevent endoscopic postoperative recurrence in patients with Crohn’s disease
Supplemental material, sj-docx-2-tag-10.1177_17562848251374240 for Systematic review and network meta-analysis comparing the efficacy of conventional therapy and biologics to prevent endoscopic postoperative recurrence in patients with Crohn’s disease by Marianne Hupé, Bruno Pereira and Anthony Buisson in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-3-tag-10.1177_17562848251374240 – Supplemental material for Systematic review and network meta-analysis comparing the efficacy of conventional therapy and biologics to prevent endoscopic postoperative recurrence in patients with Crohn’s disease
Supplemental material, sj-docx-3-tag-10.1177_17562848251374240 for Systematic review and network meta-analysis comparing the efficacy of conventional therapy and biologics to prevent endoscopic postoperative recurrence in patients with Crohn’s disease by Marianne Hupé, Bruno Pereira and Anthony Buisson in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

