Abstract
Background:
In patients with inflammatory bowel disease (IBD) who need intestinal resection, prior data suggest that earlier surgical intervention may be associated with improved outcomes. However, surgery is often deferred for additional trials of advanced therapies, which potentially shifts patients from a fit to a frail preoperative state.
Objectives:
This study aimed to evaluate clinical changes that occur in the year prior to intestinal resection in patients with IBD.
Design:
Retrospective cohort study.
Methods:
This was a multi-hospital retrospective study of patients ⩾18 years old who underwent initial IBD-related intestinal resection between January 1, 2018 and May 31, 2023. Clinical characteristics and radiographical skeletal muscle mass were compared using the Wilcoxon Signed-Rank test for continuous variables and McNemar’s test for categorical variables.
Results:
A total of 170 patients were included (120 with Crohn’s disease, 40 with ulcerative colitis, and 10 with indeterminate colitis), with a median disease duration of 7.4 years (interquartile range (IQR) 3.3–13.8). Median age at surgery was 32.6 years (IQR 25.9–44.9), and 51% were female. In the month prior to surgery as compared to the 6–12 months prior, individuals were more likely to have an IBD-related hospitalization (31% vs 5%, p < 0.01), malnutrition (30% vs 18%, p < 0.01), or an infection (74% vs 28%, p < 0.01). Though not statistically significant, there was an increase in the proportion of individuals who developed a venous thromboembolism in the month prior to surgery, and a decrease in skeletal muscle mass among those with two computerized tomography scans in the year prior to surgery (n = 6, 126 vs 111 cm2; p = 0.06).
Conclusion:
In the 6–12 months prior to an IBD-related intestinal resection, as compared to the month prior, individuals were less likely to be malnourished, have an infection, or need hospitalization for IBD. This suggests that minimizing delays to surgery may lead to improved outcomes.
Plain language summary
Keywords
Introduction
Inflammatory bowel disease (IBD), including Crohn’s disease (CD) and ulcerative colitis (UC), is a chronic immune-mediated inflammatory condition affecting roughly 3 million Americans. 1 It often requires lifelong therapy, significantly impacts quality of life, and contributes to high healthcare utilization. The goals of treatment are not only aimed at improving clinical symptoms and quality of life, but also at achieving endoscopic remission to minimize the long-term consequences of undertreated inflammation, including infection, malignancy, and the need for surgery.
Although there have been several recent advances in IBD management, including a growing number of available advanced therapies, approximately 10% of individuals with UC and 26% with CD will ultimately require surgical resection within 10 years of an initial diagnosis, with an even larger number needing surgery within their lifetime. 2 Although surgery is often considered when advanced therapies fail to control inflammation, recent data suggest improved outcomes with earlier surgical intervention. A recent study by Agrawal et al. 3 found that early ileocolonic resection, as opposed to continuing medical therapy, among individuals with ileal/ileocolonic CD was associated with a 39% reduction in the need for corticosteroids and a 51% reduction in the need for future CD-related surgery. Similar findings were also seen in a study by Bewtra et al. 4 in which patients with UC had improvement in 5-year all-cause mortality when undergoing elective colectomy as compared to continuing antitumor necrosis factor therapy. Of note, the greatest benefit to early surgical intervention in both studies was observed among the cohort of older adults with IBD.
Earlier surgery, compared to continuing medical therapy among those who are likely to require surgery, may be associated with improved clinical outcomes by limiting the time with ongoing inflammation. This may reduce the risk of malnutrition, preoperative infection, physical decline, and the need for emergency surgery. 5 The notion of earlier surgery challenges the current paradigm of care and has significant implications given the growing number of therapies available to treat IBD.
While prior studies have evaluated outcomes of early versus delayed surgery, there is limited research characterizing the changes that occur in the year prior to surgery. Thus, to further understand the potential ramifications of delays in surgery, we aimed to evaluate clinical characteristics, including changes in muscle mass that occur in the year prior to surgery. Furthermore, given the rising number of older adults with IBD, as well as the increased risk of postoperative adverse events in this population, we looked to examine changes in preoperative characteristics between younger and older adults, and to compare adults with CD and UC. 6
Materials and methods
Study design and population
We conducted a single-center, multi-hospital, retrospective study of all patients aged 18 years or older with IBD who underwent initial IBD-related surgery between January 1, 2018 and May 31, 2023. The study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental Table 1). 7 We analyzed patient characteristics in the 6–12 months prior to surgery, compared to the immediate preoperative period (1 month). Patients must have been seen in one of our IBD clinics during both time frames to be eligible for inclusion. In addition, patients were excluded if the resection indication was not related to ongoing inflammation (dysplasia, malignancy). Patients were identified consecutively through electronic health records based on procedural and diagnostic coding.
Baseline demographics
We collected baseline demographic data from the electronic medical record (EMR), including age at resection, sex, race, and ethnicity. IBD diagnosis was identified using International Classification of Diseases (ICD) codes for CD, UC, or indeterminate colitis (ICD-9 codes 555, 556, 558.9; ICD-10 codes K50, K51, K52.3), and was manually verified on chart review. Prior use of 5-aminosalicylic acid, biologics, or small-molecule inhibitors was derived from both medication history in the EMR as well as chart review to confirm accuracy. Intestinal resection was recorded as elective or emergent, as delineated in the preoperative note.
IBD characteristics
Disease severity was categorized as “mild,” “moderate,” or “severe” based upon the first endoscopic score within the year prior to surgery (mild: Mayo 1 or SES-CD 3–6, moderate: Mayo 2 or SES-CD 7–15, severe: Mayo 3 or SES-CD >16). 8 In the case where endoscopic data was not available, disease severity was based upon documentation of clinical severity in provider notes (written by IBD subspecialists).
Clinical characteristics
Clinical characteristics were obtained both in the 6–12 and 0–1 months prior to intestinal resection. Values were captured at the first timepoint within each of these two time windows. Continuous variables included body mass index (BMI, measured as kg/m2), hemoglobin (g/dL), creatinine (mg/dL), and C-reactive protein (CRP, mg/L). Albumin was measured as a categorical variable, separated into values <3, 3–3.5, or >3.5 g/dL. Systemic corticosteroid use was defined as a categorical variable including any usage of oral prednisone, hydrocortisone, methylprednisolone, and budesonide within the same time frame.
Outcomes of interest
Patients with diagnoses of malnutrition were identified using ICD codes for malnutrition, inadequate energy intake, unintentional weight loss, underweight, and cachexia (Supplemental Table 2), as have been previously used. 9 Presence of an infection and/or abscess was captured by the need for antibiotics, excluding topical medications and rifaximin. Presence of venous thromboembolism (VTE) was identified by ICD codes for pulmonary embolism or deep vein thrombosis (Supplemental Table 3), as has been previously validated. 10 Last, to assess the need for a nonelective hospitalization (excluding direct admissions and planned surgical procedures), we captured any nonelective IBD-related hospitalization 30 days before the first IBD clinic visit within the 6–12 month time frame, as well as during the 0–1 month time frame.
Muscle mass
Sarcopenia refers to a loss of muscle mass, strength, or function, and can occur due to ongoing inflammation in IBD. 11 Changes in muscle mass were captured using computerized tomography (CT) images among individuals with more than one CT scan in the year prior to surgery, with scans performed at least 30 days apart. Given the limited number of patients with two CT scans in the year prior to surgery in our main study population, we also performed a secondary analysis. This secondary analysis expanded the population to include all adult patients undergoing an IBD-related intestinal resection within our study period (did not have to be the first intestinal resection) who had two preoperative CT scans performed at least 30 days apart. Prior surgery needed to be at least 1 year prior to limit any residual muscle mass changes that can occur in the early postoperative period.
Image analysis was conducted by a radiology trainee and overseen by a board-certified abdominal radiologist with IBD subspecialty training. Abdominopelvic CT examinations were analyzed using the picture archiving communication system (Visage, San Diego, CA, USA) and the freely available software package FireVoxel (Build 435A; https://firevoxel.org). Muscle mass was calculated as skeletal muscle mass, using the cross-sectional area of skeletal muscle at the superior L3 vertebral body endplate, as has been previously validated.12–14 The muscles at the L3 level include the transversus abdominis, internal and external oblique, erector spinae, psoas, and quadratus lumborum muscles.12,13
Data analysis
Continuous data were reported as medians with interquartile ranges (IQR), while categorical variables were expressed as frequencies (%). Clinical characteristics at 6–12 and 0–1 months before resection were compared using the Wilcoxon Signed-Rank test for continuous variables and the McNemar’s or McNemar-Bowker’s test for categorical variables, with pairwise deletion used to handle the few variables with missing data. For our primary analysis, the Holm-Bonferroni method was used to account for multiple comparisons using a more stringent p-value. 15
Further stratified comparisons were performed for disease duration (<7 vs ⩾7 years, based on the median), age (<50 vs ⩾50 years), and IBD subtype (CD vs UC) using Wilcoxon-Rank Sum (for continuous variables) and Pearson’s Chi-squared or Fisher’s exact tests (for categorical variables). Age 50 was used as a cutoff given the bimodal peak incidence of IBD, as well as studies suggesting a benefit to earlier surgery among adults ⩾50 years.3,4,6 Differences in changes between subgroups were evaluated by comparing the magnitude of change using absolute differences and corresponding 95% confidence intervals using a two-proportion z-test.
Changes in muscle mass were also analyzed using the Wilcoxon Signed-Rank test, with a p-value <0.05 considered to be statistically significant. All statistical analyses were performed using Stata/BE 18.0 (StataCorp, College Station, TX, USA) and R 4.2.1 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Patient demographics
There were 595 patients identified who underwent IBD resection during our study interval, of whom 242 patients were seen in the IBD clinic between 6–12 months prior to resection and 0–1 month prior to resection (Figure 1). Of these, 170 patients met eligibility criteria for inclusion in the study, of which 51% were female (Table 1). The median age at time of surgery was 32.6 years (IQR 25.9–44.9), 35 patients (21%) were ⩾50 years of age, and 72% of patients identified as White.
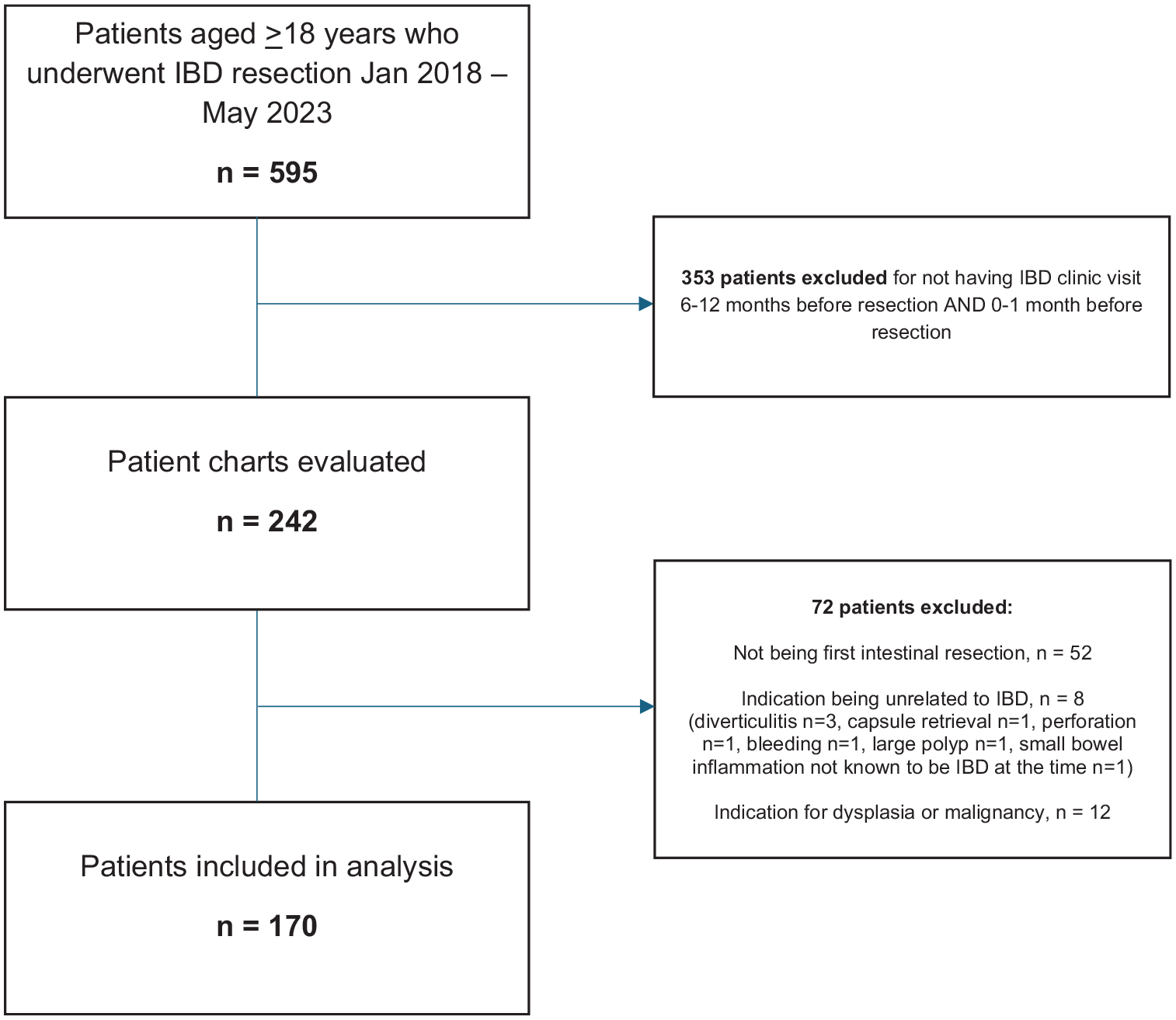
Flow diagram of participants.
Population baseline characteristics at time of surgery (N = 170).
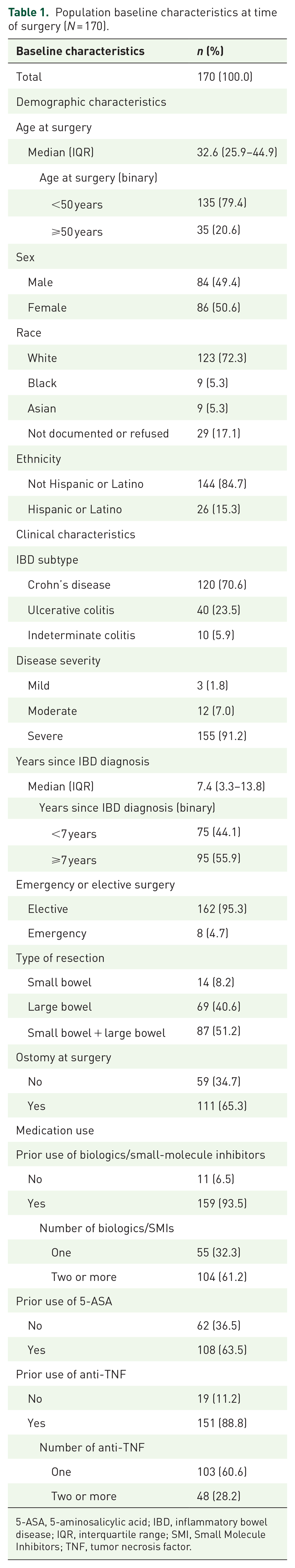
5-ASA, 5-aminosalicylic acid; IBD, inflammatory bowel disease; IQR, interquartile range; SMI, Small Molecule Inhibitors; TNF, tumor necrosis factor.
The majority of patients (n = 120, 71%) had CD, 40 (24%) had UC, and 10 patients (6%) had indeterminate colitis. There was a median of 7.4 years between the time of IBD diagnosis and surgical resection (IQR 3.3–13.8), and 155 (91%) of patients had severe disease. Most resections were considered elective (n = 162, 95%), while eight patients (5%) underwent emergent resections (six with CD, two with UC). Most patients (n = 159, 94%) had prior use of a biologic or small-molecule inhibitor before surgery, with 61% exposed to two or more (Table 1).
Differences between 0–1 versus 6–12 months prior to surgery
In the month prior to surgery, as compared to the 6–12 months prior to surgery, individuals had similar levels of creatinine and hemoglobin (Table 2). Further, although albumin only decreased slightly between the two time frames (0–1 month: 3.9 g/dL vs 6–12 months: 4.0 g/dL), the proportion of patients with an albumin <3.0 more than doubled (0–1 month: 2.9% vs 6–12 months: 1.2%). There was also a significant rise in CRP over time (0–1 month: 15.0 mg/L vs 6–12 months: 5.0 mg/L, p < 0.01).
Comparison of clinical characteristics 6–12 and 0–1 month prior to surgery among all patients (N = 170).

p-Values calculated by Wilcoxon Signed-Rank test for continuous variables and McNemar’s test or McNemar-Bowker’s tests for categorical variables. Bolded values indicate statistical significance (p < 0.05).
Missing values: BMI (25), hemoglobin (55), creatinine (62), albumin (97), CRP (115).
p-Value not significant after adjustment for multiple comparisons (Holm-Bonferroni method).
Need for IBD hospitalization was captured within 30 days of the first IBD clinic visit in the 6–12 month time frame, as well as during the 0–1 month time frame.
BMI, body mass index; CRP, C-reactive protein; IBD, inflammatory bowel disease; IQR, interquartile range; VTE, venous thromboembolism.
In additionAdditionally, there was nearly a twofold increase in the presence of malnutrition over time (0–1 month: 30% of individuals vs 6–12 months: 18% of individuals, p < 0.01), though BMI did not significantly change (0–1 month: 23.1 kg/m2 vs 6–12 months: 23.5 kg/m2, p = 0.19, Table 2). Furthermore, the proportion of patients requiring antibiotics for presumed infection more than doubled over the year prior to surgery, with 74% of individuals having an infection requiring antibiotics in the 0–1 month prior to surgery, as compared to 28% in the 6–12 months prior to surgery (p < 0.01). There was also a significant increase in the need for nonelective IBD-related hospitalization, with 31% of individuals hospitalized in the 0–1 month prior to surgery, as compared to 5% of individuals hospitalized within 30 days prior to first IBD clinic visit in the 6–12 months prior to surgery (p < 0.01). In addition, we observed a numerical increase in the rate of VTE over time, though this was not statistically significant (0–1 month: 3% of individuals vs 6–12 months: 1% of individuals, p = 0.38). Finally, there was a decrease in systemic steroid use over time (0–1 month: 39% of individuals vs 6–12 months: 54% of individuals, p < 0.01).
On subgroup analysis, we found that the proportion of individuals with malnutrition, infection requiring antibiotics, and IBD-related hospitalization increased from the 6–12 months prior to surgery to the 0–1 month preoperative period, independent of disease duration (Supplemental Table 4). Furthermore, the magnitude of change was similar across these subgroups (Supplemental Table 5).
Individuals ⩾50 versus <50 years old
On subgroup analysis, we compared 35 individuals aged ⩾50 years (median 59.5 years, IQR 56.5–67.3) to 135 individuals <50 years of age (median 29.8 years, IQR 25.0–35.7). We found similar baseline characteristics, including the percentage of female individuals, race/ethnicity, prior use of a biologic, and other IBD-related characteristics (Supplemental Table 6).
Between the month prior to surgery compared to 6–12 months prior to surgery, both age groups had a significant increase in the presence of infection requiring antibiotics (in ⩾50 years old, 0–1 month: 77% vs 6–12 months: 31%, p < 0.01; in <50 years old, 0–1 month: 73% vs 6–12 months: 27%, p < 0.01), and the need for IBD hospitalization (in ⩾50 years old, 0–1 month: 23% vs 6–12 months: 0%, p < 0.01; in <50 years old, 0–1 month: 33% vs 6–12 months: 7%, p < 0.01, Table 3). Rates of malnutrition also increased over time in adults ⩾50 years (0–1 month: 23% vs 6–12 months: 14%, p = 0.45) as well as in those <50 years (0–1 month: 32% vs 6–12 months: 19%, p < 0.01), though in the former group, this was not statistically significant. Similarly, systemic corticosteroid use decreased over time among older adults (0–1 month: 23% vs 6–12 months: 43%, p = 0.09), as well as among younger adults (0–1 month: 44% vs 6–12 months: 56%, p = 0.04). There were also increased rates of VTE in the immediate preoperative period for both the older population (0–1 month: 9% vs 6–12 months: 3%, p = 0.50) and the younger population (0–1 month: 2% vs 6–12 months: 1%, p = 1.00, Table 3). Furthermore, when comparing the magnitude of change across these clinical characteristics, no statistically significant differences were observed between age subgroups (Supplemental Table 5).
Comparison of clinical characteristics 6–12 and 0–1 month prior to surgery by older versus younger adults with IBD (N = 170).

p-Values calculated by Wilcoxon Signed-Rank test for continuous variables and McNemar’s test or McNemar-Bowker’s tests for categorical variables. Bolded values indicate statistical significance (p < 0.05).
Missing values for patients <50 years: BMI (18), hemoglobin (42), creatinine (48), albumin (73), CRP (87). Missing values for patients ⩾50 years: BMI (7), hemoglobin (13), creatinine (14), albumin (24), CRP (28).
Need for IBD hospitalization was captured within 30 days of the first IBD clinic visit in the 6–12 month time frame, as well as during the 0–1 month time frame.
BMI, body mass index; CRP, C-reactive protein; IBD, inflammatory bowel disease; IQR, interquartile range; VTE, venous thromboembolism.
CD versus UC
Similarly, when comparing the 120 patients with CD and 40 patients with UC, demographic data were similar, though patients with UC had a decreased median duration from time of diagnosis to surgery (CD: 8.6 years vs UC: 5.9 years, p = 0.04; Supplemental Table 7). When assessing the month prior to surgery as compared to the 6–12 months prior, both individuals with CD and UC had a significant rise in rates of infection (in CD, 0–1 month: 73% vs 6–12 months: 23%, p < 0.01; in UC, 0–1 month: 75% vs 6–12 months: 48%, p = 0.03), malnutrition (in CD, 0–1 month: 27% vs 6–12 months: 15%, p = 0.02; in UC, 0–1 month: 45% vs 6–12 months: 23%, p = 0.049), and the need for IBD hospitalization (in CD, 0–1 month: 23% vs 6–12 months: 5%, p < 0.01; in UC, 0–1 month: 58% vs 6–12 months: 5%, p < 0.01, Table 4). Finally, corticosteroid use decreased over time among patients with both CD (0–1 month: 33% vs 6–12 months: 43%, p = 0.07) and UC (0–1 month: 63% vs 6–12 months: 80%, p = 0.19). Notably, when comparing overall rates, individuals with CD were more likely to develop an infection requiring antibiotics in the year prior to surgery compared to those with UC (+23.3%, 95% CI: 6.9%–39.8%), whereas individuals with UC were more likely to be hospitalized in the year prior to surgery compared to those with CD (+34.2%, 95% CI: 17.2%–51.1%; Supplemental Table 5).
Comparison of clinical characteristics 6–12 and 0–1 month prior to surgery by IBD subtype (only UC and CD patients).

p-Values calculated by Wilcoxon Signed-Rank test for continuous variables and McNemar’s test or McNemar-Bowker’s tests for categorical variables. Bolded values indicate statistical significance (p < 0.05).
Missing values for CD patients: BMI (21), Hemoglobin (46), Creatinine (52), Albumin (76), CRP (90). Missing values for UC patients: BMI (3), Hemoglobin (7), Creatinine (7), Albumin (14), CRP (17).
Need for IBD hospitalization was captured within 30 days of the first IBD clinic visit in the 6–12 month time frame, as well as during the 0–1 month time frame.
BMI, body mass index; CRP, C-reactive protein; IBD, inflammatory bowel disease; IQR, interquartile range; VTE, venous thromboembolism.
Muscle mass
Overall, six patients (3.5%) in our study had two CT scans in the year prior to surgery that were at least 30 days apart. The median time interval between scans was 2.9 months (IQR 2.7–3.5), with muscle mass decreasing over this period (126.0 vs 111.1 cm2, p = 0.06). We observed similar results in our secondary analysis: of the 23 patients who had an intestinal resection (did not need to be the first intestinal resection) and 2 preoperative CT scans, muscle mass similarly declined (123.6 vs 117.8 cm2, p = 0.43) over a median time interval of 3.5 months (IQR 2.7–6.1).
Discussion
In this retrospective study of adults with IBD undergoing an initial intestinal resection, we found that individuals were almost twice as likely to develop malnutrition, more than twice as likely to have an abscess or infection requiring antibiotics, and more than six times as likely to have an unplanned hospitalization in the month prior to surgery as compared to 6–12 months prior. These results remained consistent when stratifying by disease duration, chronological age, and IBD subtype. Although limited by the number of events, more than twice as many individuals developed VTE in the month prior to surgery as compared to 6–12 months earlier, with muscle mass also declining over time in the preoperative state.
Despite the increasing number of medical therapies available to treat IBD, surgery remains common, particularly in cases of refractory disease. More recently, data also suggest that earlier surgery may improve clinical outcomes among individuals with IBD, including reduced corticosteroid use, decreased need for future surgeries, and even reduced mortality.3,4 However, due to the growing armamentarium of medical therapies, patient preferences, and barriers in access to care, earlier surgery may be deferred for continued trials of medications. This can contribute to the increased risk of adverse postoperative outcomes observed among individuals with IBD, as prolonged periods of ongoing inflammation can lead to the development of malnutrition, decline in functional status, and the need for emergency surgery. 5
Malnutrition may increase the risk of an adverse postoperative outcome in individuals with IBD, with studies showing a 22% increased risk of 30-day adverse events, as well as more than tripling the rate of postoperative infectious complications.5,16 To improve malnutrition in the preoperative state, some data have shown a possible benefit with preoperative enteral or parenteral nutrition. 17 Although the treatment of malnutrition is important to improve postoperative outcomes, a key focus should also be on prevention; ongoing inflammation itself can precipitate the development of malnutrition. This is driven by increased energy requirements due to the hypercatabolic inflammatory state, malabsorption from intestinal inflammation, and decreased food intake (including from potential diet restriction due to symptoms). 18 In our study, we see evidence of this, as 18% of individuals had a diagnosis of malnutrition in the 6–12 months prior to surgery, as compared to 30% in the month prior to surgery.
In addition, ongoing inflammation can lead to the development of infections and/or abscesses in the preoperative state. 16 This can occur through several mechanisms, including transmural intestinal inflammation leading to intraabdominal abscesses or fistulae, impairment of innate immune mechanisms (which can also be worsened by malnutrition), as well as the use of immunosuppressive agents, including corticosteroids. 19 Furthermore, the presence of a preoperative infection has been shown to increase the risk of postoperative complications by up to twofold in some studies.5,20 Thus, minimizing time with ongoing inflammation may have additional benefits, particularly among individuals who may eventually need surgery. In our study, we noted a significant increase in the rates of infection over time, with 74% of patients having an abscess or infection requiring antibiotics in the month prior to surgery, compared to only 28% in the 6–12 months prior to surgery. On subgroup analysis, findings were similar when stratified by median disease duration, chronological age, and IBD subtype. However, individuals with CD were significantly more likely than those with UC to develop an infection requiring antibiotics from the 6–12 months to the 0–1 month preoperative period.
Sarcopenia (which is the loss of muscle mass, strength, and function) can also occur as a result of ongoing inflammation in IBD. 11 It has been shown to increase the risk of postoperative outcomes among individuals with IBD, including higher rates of anastomotic leak, surgical site infection, rehospitalization, and need for total parenteral nutrition.14,21,22 Despite being limited by the number of individuals with two preoperative CT scans, we observed a decline in muscle mass of 14.9 cm2 over a median of 2.9 months in our study population, and by 5.8 cm2 over a median of 3.5 months in our expanded cohort. Although these findings were not statistically significant, our study was likely underpowered to detect small differences and may have been limited by the relatively short interval between CT scans. However, with the recent ability to assess muscle mass during intestinal ultrasound, future studies may have the ability to assess longitudinal changes in muscle mass over time. 23
By highlighting these clinical changes that occur in the preoperative period, our findings support data-driven approaches to shared decision-making and underscore the potential benefits of earlier surgical referral. Beyond timing alone, these changes also present an opportunity to implement targeted prehabilitation strategies. In IBD, muscle loss is driven by chronic inflammation, anabolic resistance, oxidative stress, and malnutrition, factors that may be mitigated through prehabilitation. 24 Though specific data is needed in the IBD patient population, randomized trials among individuals without IBD have shown that such programs have the potential to reduce postoperative morbidity, hospital stays, and improve quality of life.14,25–27
One strength of our study is the utilization of manual chart review, which allowed for the collection of more granular patient details and confirmation of accuracy. Data collection from multiple hospitals across the institution, including a large tertiary care center with specialized IBD care, also contributes to the accuracy of information collected. Another strength is our use of CT images to quantify muscle mass over time, which has not been previously evaluated. Further, although select laboratory values were missing, we had complete data on all clinically relevant factors included in our analysis, which further reinforces the robustness of our findings.
Despite these strengths, several limitations exist. While we included 170 individuals, there were only 35 adults ⩾50 years old, limiting the power of our subgroup analyses. Further, as this was not prospectively collected, we relied on clinical notes and diagnosis codes, rather than uniform assessments (e.g., malnutrition) among all individuals. Future analysis using validated assessments for malnutrition (such as GLIM or MUST) would ensure greater consistency across patients.28,29 Additionally, a high proportion of individuals were White, which limits the ability to discern racial disparities that can contribute to differences in surgical outcomes.30,31 Finally, although some of the factors assessed may have driven the ultimate decision for surgery, our study is the first to highlight the changes that occur in the preoperative state, which may contribute to an increased risk for adverse postoperative outcomes.
Conclusion
In sum, we found that rates of malnutrition and preoperative infection more than doubled in the year prior to initial surgical resection; this was consistent across IBD subtypes (UC and CD), age groups (<50 vs ⩾50 years), and disease duration (<7 vs ⩾7 years). Further, as these factors are associated with an increased risk of adverse postoperative outcomes, our study builds upon prior findings and is one of the first to detail the rising incidence of these important clinical factors in the year prior to surgery. Taken together, these results support earlier discussions among individuals who may eventually require surgery (e.g., those with severe or refractory disease), as this may reduce the risk of developing such conditions, thereby lowering the risk of an adverse postoperative outcome. While future prospective studies using validated assessments are needed to further characterize the preoperative changes that occur in the year prior to surgery, our findings (1) provide data to support shared decision-making, (2) highlight the potential benefit of earlier surgical referral, and (3) help lay the groundwork for identifying individuals with IBD who may benefit from targeted preoperative interventions (e.g., prehabilitation).
Supplemental Material
sj-docx-1-tag-10.1177_17562848251365036 – Supplemental material for Risk of malnutrition increases in the year prior to surgery among patients with inflammatory bowel disease
Supplemental material, sj-docx-1-tag-10.1177_17562848251365036 for Risk of malnutrition increases in the year prior to surgery among patients with inflammatory bowel disease by Vasantham Chaudhary, Frank R. Chung, Olivia Delau, Bari Dane, Irving Levine, Xucong Meng, Joshua Chodosh, Andre da Luz Moreira, Jessica N. Simon, Jordan E. Axelrad, Seymour Katz, John Dodson, Aasma Shaukat and Adam S. Faye in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
