Abstract
Background:
Colorectal cancers (CRC) with deficient mismatch repair system and/or microsatellite instability-high (dMMR/MSI-H) phenotype represent about 12% of CRC. dMMR/MSI-H CRC is due to a germline mutation (Lynch syndrome, LS) or an age-related epigenetic mechanism, mostly by hypermethylation of MLH1 promoter (sporadic cases). It is well recognized that patients with LS have a high lifetime risk of various dMMR/MSI-H cancers, but there are no data concerning the risk of a second cancer in sporadic dMMR/MSI-H CRC.
Objectives:
The main objective of this study was to determine the risk of having another primary cancer (APC) in patients with dMMR/MSI-H CRC. We also collected these tumors to determine their MMR phenotype.
Design:
We used a prospective cohort of 484 well-characterized patients with dMMR/MSI-H CRC to describe their risk of having APC.
Methods:
We evaluated the occurrence of APC (before or after the occurrence of the dMMR/MSI-H CRC) according to LS versus sporadic status, whatever the stage or the tumor site. The characteristics of the two groups, with and without APC, and LS versus sporadic status were compared.
Results:
Among the 484 patients with dMMR/MSI-H CRC, we identified 116 patients with a previous or a second primary tumor (24.0%), with an average of 1.4 tumors per patient in addition to the dMMR/MSI-H CRC. The most frequent tumor sites were skin (24.7%) and breast (18.5%). Regarding the occurrence of APC, we found no difference between patients with LS-related dMMR/MSI-H CRC group (25.0%) and those with sporadic dMMR/MSI-H CRC (24.8%). No risk factor was associated with the occurrence of APC in our cohort, in the LS or sporadic cases subgroup. In the sporadic group, 3.8% of APC had a dMMR/MSI-H phenotype as compared to 50.0% in the LS group.
Conclusion:
It seems important to follow patients with a history of dMMR/MSI-H CRC due to the high risk of second tumors, even in sporadic cases. Second cancers in patients with sporadic dMMR/MSI-H CRC are rarely associated with a dMMR/MSI-H phenotype.
Plain language summary
Why was the study done? Colorectal cancers (CRC) with deficient mismatch repair system and/or microsatellite instability-high (dMMR/MSI-H) phenotype represent about 12% of CRC. dMMR/MSI-H CRC is due to a germline mutation (Lynch syndrome, LS) or an age-related epigenetic mechanism (sporadic cases). It is well recognized that patients with LS have a high lifetime risk of various cancers but there are no data concerning the risk of a second cancer in sporadic dMMR/MSI-H CRC. What did the researchers do? In a prospective cohort of 484 well-characterized patients with a dMMR/MSI-H CRC, we described their risk of having another primary cancer (APC) according to the LS versus sporadic status. What did the researchers find? Among the 484 patients with dMMR/MSI-H CRC, we identified 24% of patients with a previous or a second primary tumor. Regarding the occurrence of APC, we found no difference between patients with LS-related dMMR/MSI-H CRC group and those with sporadic dMMR/MSI-H CRC. What do the findings mean? It seems important to follow patients with a history of dMMR/MSI-H CRC due to the high risk of second tumors, even in sporadic cases.
Keywords
Introduction
Colorectal cancers (CRC) represent the third most frequent cancers worldwide, with nearly 2 million new cases each year. 1 Approximately 12% of CRC have a microsatellite instability-high (MSI-H) and/or deficient mismatch repair system (dMMR) status. 2 Nowadays, it is recommended to determine MMR and/or MSI status for all CRC cases to screen for Lynch syndrome (LS) and establish therapeutic management. 3 The dMMR/MSI-H phenotype is associated with germline mutation of an MMR gene (LS) or a somatic inactivation of one MMR gene, mostly MLH1, by hypermethylation of its promoter (sporadic cases). MSI-H or microsatellite stable (MSS) status is determined by evaluating MSI using molecular tests on tumor DNA (polymerase chain reaction or next-generation sequencing (NGS)), and MMR status using immunohistochemistry (IHC) of the four MMR proteins (MLH1, PMS2, MSH2, and MSH6). Indeed, dMMR/MSI-H status is associated with a good prognosis in non-metastatic CRC but also chemoresistance to adjuvant 5-fluorouracil in stage II colon cancer.4 –6 In metastatic settings, dMMR/MSI-H mCRC have high sensitivity to immune checkpoint inhibitors (ICI), and pembrolizumab is the standard of care in first-line setting. 7
With improvements in cancer management, the survival of patients with dMMR/MSI-H CRC is increasing, leading to a higher risk of them developing a second primary cancer (SPC, cancer after diagnosis of the first primary cancer). The incidence of another primary cancer (APC, cancer before or after the diagnosis of a first primary cancer) has been analyzed in several studies, and most concluded that patients with a history of cancer have a higher risk of developing APC, ranging from 14% to 36% higher than that in the general population.8 –10 The risk of SPC differs according to the site of the first cancer. Concerning colon cancer, the standard incidence ratio of SPC ranges from 1.07 to 1.20, and the most common tumors are colorectal, endometrial, and small bowel cancers. 10 Nevertheless, in these studies, hereditary diseases that predispose patients to cancer, like LS or familial adenomatous polyposis, were not considered, which induces a major bias in the estimation of SPC risk.
In a cohort of 1273 patients with an LS-related primary cancer with a proven MMR mutation, 318 (25.7%) had an SPC, and CRC was the most frequent subsequent malignancy (43.0%). However, the MMR/MSI status of the SPC was not determined, leading to doubts about the involvement of LS. 11 The cumulative risk of any metachronous cancer after CRC in LS populations varies from 20% to 90%, depending on the duration of follow-up and the period of the study, with the oldest studies reporting the highest risk.12,13 Only one unpublished series compared the risk of a second CRC after a first localized sporadic dMMR CRC with that in a group with proficient MMR (pMMR) CRC. A second CRC developed in 1.91% of the sporadic dMMR CRC group versus 0.83% of the pMMR CRC group, and the median time to the second CRC was shorter in the sporadic dMMR CRC group (1.13 vs 2.38 years). 14 These data suggest an increased risk of SPC among patients with a first dMMR/MSI-H CRC, whatever the germline or sporadic origin, but the MMR status of these tumors remains to be determined. We can hypothesize that sporadic dMMR/MSI-H CRC patients could develop a SPC due to MLH1 promoter hypermethylation.
The aim of this study was to determine, in a well-characterized large cohort of dMMR/MSI-H CRC patients, the frequency and characteristics of other tumors occurring in these patients, including the MMR and MSI status of the other tumors.
Patients and methods
Population
From 2003 to 2020, all consecutive patients with a CRC classified as MSI-H by molecular testing (Pentaplex panel) by the Cancer Biology Department of Poitiers University Hospital were included in this multicenter prospective cohort.15 –18
As previously described, tumor DNA was extracted in routine clinical practice from formalin-fixed paraffin-embedded tumor tissue using the DNeasy Blood and Tissue DNA isolation kit (Qiagen, Hilden, Germany). 15 MSI status was assessed by analyzing microsatellite loci comprising five mononucleotide markers BAT-25, BAT-26, NR21, NR24, and NR27 (Promega, ref MD1641, Madison, WI, USA), as previously described (Pentaplex panel).15,16
MMR status was determined for all tumors using MMR IHC of the four MMR proteins from formalin-fixed paraffin-embedded tumor tissue using antibodies directed against MLH1, MSH2, MSH6, and PMS2 proteins (Ventana Medical Systems, Tucson, AZ, USA), as previously described.16,18 A tumor was considered dMMR if there was a loss of nuclear staining of at least one of the four MMR proteins.
All dMMR/MSI-H CRC cases were included in the study, whatever the tumor stage (stage 0–IV), but discordant cases between MSI and MMR status were excluded.
The study was approved by the ethics committee of Poitiers University Hospital (Comité de Protection des Personnes Ouest III, number DC-2008-565). This study was conducted in accordance with current French law and with the ethical principles of the Helsinki Declaration of 1975 and its subsequent revisions. All living patients provided oral non-opposition to the use of their data. All patient details were de-identified.
Patients’ and CRC characteristics
The main characteristics of patients include sex and age at CRC diagnosis. The main characteristics of the first dMMR/MSI-H CRC include tumor site, TNM stage at diagnosis, and tumor grade as well as treatments (surgery, chemotherapy, and/or ICI).
LS versus sporadic dMMR/MSI-H CRC cases were determined as previously published. 4 Briefly, proven LS was defined as the presence of an MMR gene germline mutation. Suspected LS was defined as (1) loss of MSH6 and/or MSH2 protein expression, (2) loss of PMS2 protein expression with no loss of MLH1 protein expression, or (3) loss of MLH1 protein expression with no BRAF mutation and no MLH1 promoter hypermethylation. Cancers were considered sporadic dMMR/MSI-H CRC if there was a loss of MLH1 protein expression with a BRAF mutation or MLH1 promoter hypermethylation. All other tumors were considered unclassified regarding the molecular mechanism underlying the MMR deficiency.
We determined the number of patients with a history of cancer, meaning with a diagnosis of a tumor before the dMMR/MSI-H CRC diagnosis, and the number of patients with a new diagnosis of cancer, meaning the diagnosis of a subsequent tumor after the dMMR/MSI-H CRC diagnosis. Previous or SPCs are described as “APC.” APCs were identified through computerized medical records and the regional cancer multidisciplinary team meetings. In France, it is mandatory to register all new cancer cases at a multidisciplinary team meeting, which is available to all physicians.
Determination of mismatch repair deficiency and MSI of other primary cancers
The MMR status of APC was determined using the MMR IHC of the four MMR proteins, as previously described.16,18 MSI status was determined using NGS, as recently published. 19 Briefly, a panel of eight microsatellite loci was analyzed to determine the level of MSI using a test termed CaBio-MSID. A tumor was considered MSI-H if the CaBio-MSID score was equal to or greater than 0.50 (at least four unstable markers).
Statistical analysis
All data were updated in December 2023. Median follow-up was calculated by reverse Kaplan-Meier estimation, and patients lost to follow-up were censored at the date of the last news.
Continuous variables were described with medians and ranges, and qualitative variables with frequencies and percentages. Missing data were not taken into account for the calculations of medians and percentages. The characteristics of the two groups, with and without APC, were compared with the Mann-Whitney test for continuous variables and the Chi-square test or Fisher’s exact test for qualitative variables. Given that more than 400 patients were included, the statistical results and comparisons between patients with and without APC can be considered reliable.
A p-value of less than 0.05 was considered statistically significant. All analyses were performed using Statview© software (SAS Institute, Cary, NC, USA). STROBE guidelines were used to prepare the manuscript.
Results
Patient and tumor characteristics
This cohort included 484 patients, 273 females (56.4%) and 211 males (43.6%), with a dMMR/MSI-H CRC (Figure 1 and Table 1). The mean age at dMMR/MSI-H CRC diagnosis was 68.1 ± 14.2 years. Most patients had moderately differentiated tumors (46.7%) and ascending colon adenocarcinoma (80.0%). Most tumors were stage II or III dMMR/MSI-H (42.9% and 29.0%, respectively) CRC. RAS and BRAF mutations were observed in 15.5% and 52.2%, respectively.

Flow diagram.
Characteristics of patients with and without APC.

APC, another primary cancer; BRAF, V-Raf Murine Sarcoma Viral Oncogene Homolog B; CRC, colorectal cancer; LS, Lynch syndrome; MLH, MutL Homolog; MSH, MutS Homolog; PMS2, postmeiotic segregation increased 2; RAS, rat sarcoma virus.
Most tumors had a loss of MLH1 and PMS2 proteins (82.8%), mostly due to hypermethylation of the MLH1 gene promoter (84.8%). Among the 484 patients, 29.5% had proven or suspected LS (Table 1).
Surgical resection of the primary dMMR/MSI-H CRC was performed in 93.6% of the cases (n = 453). All but one patient with stage 0–III tumors had primary tumor resection (one patient with a locally advanced unresectable T4 tumor did not undergo surgery). Among patients with stage II dMMR/MSI-H CRC, 21.6% had adjuvant chemotherapy compared with 77.1% for stage III.
Analysis of other primary cancers
The median age at which patients were diagnosed with dMMR/MSI-H CRC was 70.3 years, and the median follow-up period after diagnosis was 38.6 months. We identified 116 patients (24.0%) with at least one APC. Overall, 162 APCs were observed with an average of 1.4 cancers per patient. Among these patients, 76.7% had one APC, 16.4% had two APCs, 4.3% had three APCs, and 2.6% had four or more APCs. In most patients, the APC was diagnosed before the diagnosis of the dMMR/MSI-H CRC (59.8%) versus 33.3% after the dMMR/MSI-H CRC diagnosis and 6.9% both before and after the dMMR/MSI-H CRC diagnosis. The proportion of patients with APC and LS-related CRC was similar to that of patients with APC and sporadic CRC (25.0% vs 24.8%).
The characteristics of the patients and primary dMMR/MSI-H CRC in cases with APC were not different from those without APC (Table 1). Among patients with APC, we compared patients with dMMR/MSI-H CRC related to LS (proven or suspected LS) versus patients with sporadic dMMR/MSI-H CRC (Supplemental Table 1). In the sporadic group, patients were older and more likely to have ascending colon cancer and BRAF-mutated dMMR/MSI-H CRC. Half of the APCs were found after the diagnosis of CRC in the LS group (50.0%), compared with only 26.0% in the sporadic group (p = 0.06). Comparing patients with and without APC in the dMMR/MSI-H sporadic group, the BRAF mutation was significantly less frequent in patients with APC than in patients without APC (66.6% vs 81.1%, p = 0.01; Table 2). By contrast, there was no statistical difference among LS-related dMMR/MSI-H CRC patients with and without APC (Supplemental Table 2).
Characteristics of patients with and without APC in the sporadic group.

APC, another primary cancer; BRAF, V-Raf Murine Sarcoma Viral Oncogene Homolog B; CRC, colorectal cancer; IHC, immunochemistry; LS, Lynch syndrome; MLH, MutL homolog; MSH, MutS homolog; PMS2, postmeiotic segregation increased 2; RAS, rat sarcoma virus.
The number of second cancers was higher in the LS group than in the sporadic group (1.64 vs 1.32; Table 3). Sites of APC were globally similar in both groups. The most frequent APCs in the whole cohort were breast (18.5%) and skin cancers (24.7%). We found more breast cancers (22.3% vs 11.1%) and prostate cancers (11.6% vs 0%) in the sporadic group than in the LS group. By contrast, endometrial (5.5% vs 2.9%), urothelial (7.4% vs 2.9%), rectal (7.4% vs 1.9%), and skin (31.5% vs 22.3%) tumors were more frequent in LS. In addition, there were two sebaceous adenomas and one sebaceous carcinoma in the LS group versus none in the sporadic group.
Location of second primary tumors in each dMMR/MSI-H CRC population.

Five patients had an undetermined status for LS versus sporadic dMMR/MSI-H CRC.
APC, another primary cancer; CRC, colorectal cancer; dMMR, deficient mismatch repair; ENT, ears–nose–throat; HCC, hepatocellular carcinoma; LS, Lynch syndrome; MSI-H, microsatellite instability-high; MSH, MutS homolog.
Finally, we looked at the MMR and MSI status of the available APC. The APC was predominantly pMMR (78.3%) and MSS (80.3%; Table 4). Half of the APC among patients with Lynch-related dMMR/MSI-H CRC were dMMR/MSI-H. By contrast, in the sporadic dMMR/MSI-H CRC group, only two APCs were identified as dMMR/MSI-H in two different patients. For the first patient, it was a well-differentiated stage IIIc endometrioid adenocarcinoma with a papillary contingent. For the second patient, it was a well-differentiated stage III rectal adenocarcinoma. In these two tumors, there was loss of MLH1 and PMS2, and MLH1 gene promoter hypermethylation.
MMR IHC and MSI status of APC.
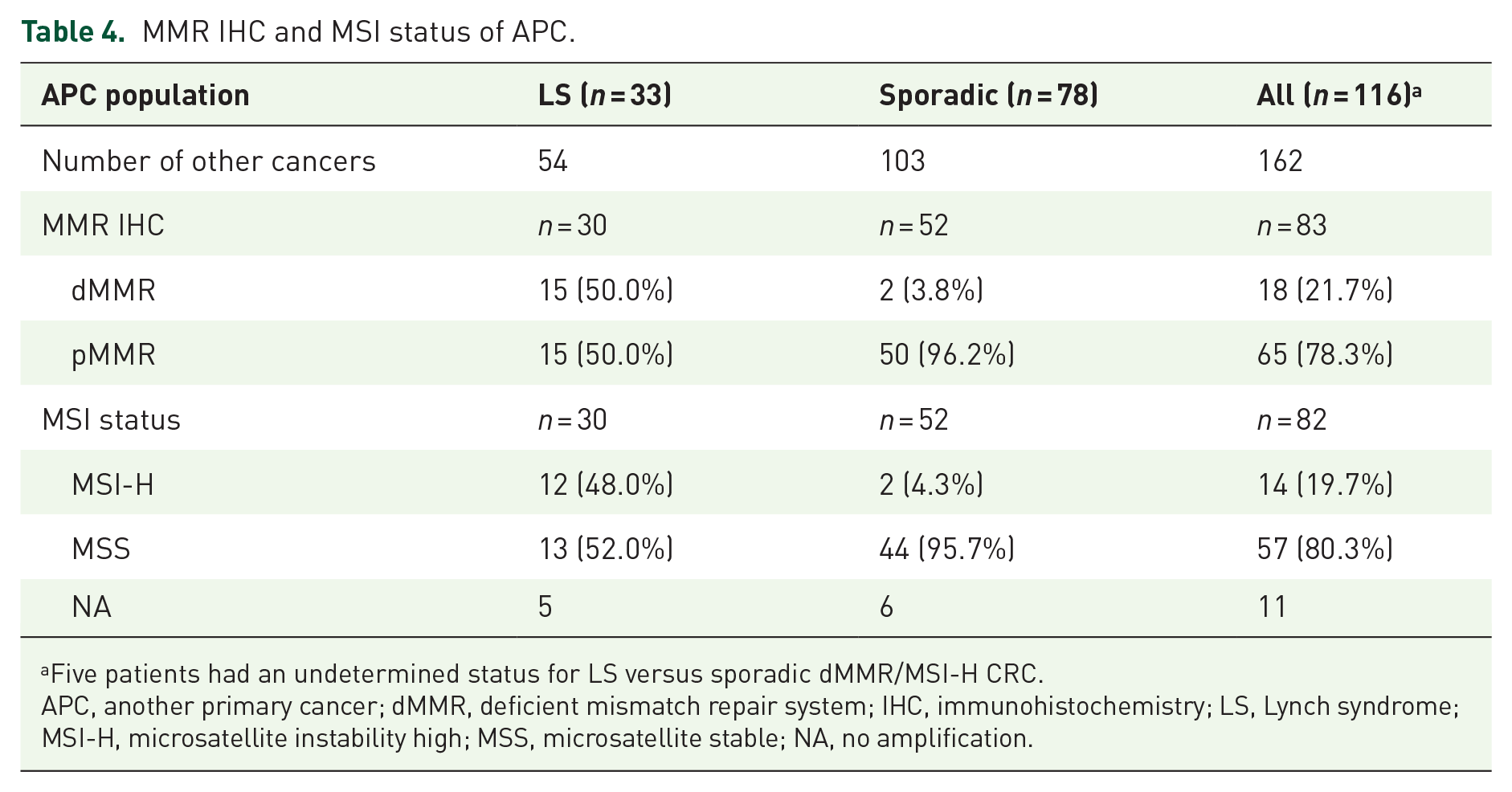
Five patients had an undetermined status for LS versus sporadic dMMR/MSI-H CRC.
APC, another primary cancer; dMMR, deficient mismatch repair system; IHC, immunohistochemistry; LS, Lynch syndrome; MSI-H, microsatellite instability high; MSS, microsatellite stable; NA, no amplification.
Discussion
Our study analyzed the risk of another cancer occurring in patients with dMMR/MSI-H CRC, including both sporadic and LS cases. We identified a high risk of having another cancer of about 25% in both LS patients and sporadic cases. Surprisingly, the percentage of patients with APC was no higher in LS than in sporadic cases, but LS patients had a higher number of APC per patient. In addition, we found no strong predictive factors for APC in the sporadic or LS group when we compared patients with and without APC. Finally, only half of the APCs were dMMR/MSI-H in the LS group, and there were very few in the sporadic group.
The characteristics of our series of dMMR/MSI-H CRC are in accordance with the characteristics of other dMMR/MSI-H CRC series that included both LS and sporadic cases. The mean age of patients (68.1 years) is lower than the mean age at CRC diagnosis in France (72 years) due to the high proportion of LS in dMMR/MSI-H tumors. 20 The ratio of patients with LS to those with sporadic cancer, respectively, 29.5% versus 70.5%, is comparable to data in the literature. 2 By contrast, our study found 52.2% of tumors with a BRAF mutation compared with around 30%–40% of BRAF-mutated dMMR/MSI-H CRC tumors reported in the literature, probably due to selection bias in these series with an underrepresentation of sporadic dMMR/MSI-H CRC. 21
Our study found 24.0% of dMMR/MSI-H CRC patients with APC, surprisingly with no difference between LS and sporadic cases (25.0% vs 24.8%). Even though there is a well-known significant excess risk of cancer in patients with LS, improvements in cancer prevention in patients with LS (colonoscopy with chromoendoscopy and prophylactic hysterectomy with bilateral salpingo-oophorectomy) have probably decreased this risk of LS-related cancer. Nevertheless, we found a higher average number of APCs per patient in LS than in sporadic cases (1.64 vs 1.32). Only one unpublished series analyzed the risk of a second CRC after a first localized CRC, including 714 patients with a dMMR CRC (17.4%), of which 575 (14.6%) were deemed sporadic dMMR CRC. 14 A second primary CRC was identified in 1.91% of the sporadic dMMR CRC group versus 0.83% of the pMMR CRC group, with a significant difference (hazard ratio (HR) = 2.57, 95% confidence interval (CI) 1.28–5.17, p = 0.008). The median time to the appearance of the second CRC was shorter in the sporadic dMMR CRC group, 1.13 versus 2.38 years. This unpublished series had some limitations: only MMR status was determined, no MSI testing was done, and we have no data on the MMR or MSI status of the second primary CRC. In addition, there were no data concerning tumors other than CRC. To our knowledge, there are no other data concerning APC in dMMR/MSI-H CRC patients, especially in sporadic cases, nor are there data comparing LS versus sporadic groups or evaluating all tumor sites.
When we analyzed the sequence of APC, we noticed that in the sporadic group, two-thirds (66.8%) of the APC were diagnosed before the dMMR/MSI-H CRC, while the distribution was more homogeneous in the LS group (43.3% before and 50.0% after the dMMR/MSI-H CRC). This is quite understandable since patients in the sporadic group were older (73.3 vs 58.5 years), therefore, carrying a higher risk of having a previous cancer. This is also consistent with the finding that prostate and breast cancers were more frequent in the sporadic group than in the LS group.
The distribution of tumor sites between the sporadic and LS groups was expected. The most frequent tumor sites were the most common sites in the general population, such as skin (24.7%) and breast (18.5%) tumors. LS-related cancers were more frequent in the LS group than in the sporadic group: gastric cancers (1.8% vs 0), urothelial cancers (7.4% vs 2.9%), endometrial cancers (5.5% vs 2.9%), and CRCs (16.7% vs 9.7%). Skin tumors were more frequent in LS (31.5% vs 22.3%), and sebaceous tumors (n = 3) were only observed in the LS group, revealing a rare syndrome with digestive and cutaneous dMMR/MSI-H tumors, Muir-Torre syndrome. 22 Nevertheless, most skin tumors observed in LS were squamous and basal cell carcinoma, as was the case in sporadic CRC.
We found that 24.8% of patients with sporadic dMMR/MSI-H CRC had at least one other primary malignancy. This risk is higher than that in CRC overall, for which the mean risk of having APC was 7.0%, according to the American Cancer Registry. 9 Indeed, we can suppose that sporadic dMMR/MSI-H CRC patients carry a higher risk of APC with dMMR/MSI-H status due to MLH1 promoter hypermethylation. Nevertheless, this cannot be the case since only two APCs were identified as dMMR/MSI-H (3.8%). Indeed, we have no clear explanation as to why sporadic dMMR/MSI-H CRC patients carry a high risk of developing another tumor. In addition to the fact that sporadic dMMR/MSI-H CRC occurs in the elderly, perhaps there are other contributing cancer risk factors such as being overweight, a sedentary lifestyle, sun damage, smoking, or alcohol consumption. Our study raises the question of cancer screening programs for patients with sporadic dMMR/MSI-H CRC, given the high incidence of secondary tumors in this population. Of course, CRC patients considered cured must have not only endoscopy follow-up but also breast cancer screening, as recommended. Regarding the risk of other cancers, our results need to be confirmed by large prospective studies before routine clinical practices can be changed.
We found 25.0% of APC in LS-related dMMR/MSI-H CRC, which is in accordance with the previous series. Møller et al. 11 analyzed the cumulative incidence of SPC in LS patients after a first diagnosis of cancer. The average number of SPC per patient was 1.4, and CRC was the most frequent location of subsequent malignancies in this study (43.0%). In a Finnish LS population, the cumulative risk of any metachronous cancer was 90% after treatment of the first CRC, but the series is old (1995) and probably overrated the risk of metachronous tumors. 12 Indeed, a more recent cohort study evaluated the risk of extracolonic SPC after a first LS-related CRC and showed a 10-year-cumulative risk of 1.9% for kidney tumor, 1.6% for bladder cancer, 0.66% for stomach cancer, 0.92% for small intestine cancer, and for men 2.74% for prostate cancer and for women 12.12% and 1.94% of endometrium and breast cancer, respectively. 13 In our study, it is worth noting that in the LS group, only half of the APCs were dMMR/MSI-H, suggesting that cancer screening in LS decreases the risk of LS-related cancer. However, these patients could have had “standard” cancers, that is to say, cancers observed in the general population, such as breast and skin tumors, which accounted for 11.1% and 31.5% of tumors observed in our series, respectively. As expected, APC with dMMR/MSI-H status belonged to the LS spectrum (gastric cancers, urothelial cancers, endometrial cancers, and CRCs). Finally, our results will have a limited impact on current clinical practices in patients with LS since follow-up strategies are already well defined by international guidelines. Nevertheless, it is worth noting that published data on the MMR status of secondary cancer after a first LS-related CRC are limited, and our study highlighted that half of these cancers are not dMMR/MSI-H and thus not related to LS.
By contrast, in the sporadic dMMR/MSI-H CRC group, only two APC had dMMR/MSI-H status (3.8%) with hypermethylation of the MLH1 promoter, suggesting in rare settings that some patients could have had two dMMR/MSI-H tumors due to MLH1 promoter hypermethylation. Our hypothesis that patients with sporadic dMMR/MSI-H CRC could “frequently” have dMMR/MSI-H SPC due to MLH1 promoter hypermethylation is not confirmed by our study.
As compared to LS patients, patients with a sporadic dMMR/MSI-H CRC with APC were older, more frequently had ascending colon cancer, and more frequently had BRAF-mutated tumors, which was expected. There was no statistical difference between LS-related dMMR/MSI-H CRC patients with APC and those without APC. By contrast, in the sporadic dMMR/MSI-H group, the BRAF mutation was significantly less frequent in patients with APC than in patients without APC. We have no clear explanation as to why, but this difference may be purely coincidental.
The main limitation of this study is the retrospective evaluation of APC, which could have led us to miss some APC. Nevertheless, since it is mandatory to discuss all cancer cases in France during multidisciplinary team meetings, the number of missed APCs is probably very low. Another limitation is a single regional population, which may not be representative of broader patient demographics. We can highlight many strengths of our study, including a well-characterized prospective cohort, the large number of patients, and its originality, in that it is the first study to evaluate the MMR/MSI-H status of APC. To our knowledge, no study has evaluated the proportion of dMMR/MSI-H versus pMMR/MSS second cancers in LS or non-LS patients with a first dMMR/MSI-H CRC. Our study raises the question of cancer screening programs for patients with sporadic dMMR/MSI-H CRC, given the high incidence of other tumors in this population. Nevertheless, before changing routine clinical practices, our results need to be confirmed by large prospective studies.
Conclusion
In this cohort, patients with dMMR/MSI-H CRC had a high rate of another primary tumor, those with LS as well as those with sporadic dMMR/MSI-H CRC. Only half of the other primary tumors in patients with LS-related dMMR/MSI-H CRC were associated with a dMMR/MSI-H phenotype (LS-related cancers) and were very rare in other primary tumors associated with sporadic dMMR/MSI-H CRC. This study raises the question of cancer screening in patients with a history of sporadic dMMR/MSI-H CRC.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251347375 – Supplemental material for Second malignancies in patients with deficient mismatch repair system/microsatellite instability-high colorectal cancer
Supplemental material, sj-docx-1-tag-10.1177_17562848251347375 for Second malignancies in patients with deficient mismatch repair system/microsatellite instability-high colorectal cancer by Paul Franques, Pierre Moreau, Camille Evrard, Arnaud Chong-Si-Tsaon, Éric Frouin, Marie-Luce Auriault, Valérie Moulin, Audelaure Junca, Julie Godet, Lucie Lebeau, Roxane Aguillon, Gaëlle Tachon, Lucie Karayan-Tapon and David Tougeron in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
