Abstract
The vast majority of colorectal cancers (CRCs) are proficient mismatch repair (pMMR) and microsatellite stable, and their immune microenvironment appears as a “cold tumor,” which is not sensitive to single immunotherapy based on immune checkpoint inhibitors (ICIs). The utilization of ICIs in pMMR advanced CRC is still in the exploratory phase. Cutaneous metastasis from colorectal carcinoma is extremely rare, presenting with diverse clinical manifestations, and there is a lack of standard treatment options for such cases. Patients with skin metastasis from CRC usually progress rapidly and are associated with a dismal prognosis. Herein, we report the case of a 66-year-old woman with extensive cutaneous metastasis of pMMR advanced rectal carcinoma. The patient presented to the abdominal oncology clinic with a complaint of erythema on the right lower limb, perineum, and abdominal skin. The patient underwent radical surgery for rectal carcinoma 3 years before the presentation. The histologic examination revealed low-grade squamous cell subepithelial adenocarcinoma. The patient was treated with sintilizumab in combination with fruquintinib, which exhibited remarkable efficacy and improved the patient’s quality of life significantly. Previous cases of cutaneous metastasis of colorectal carcinoma were retrieved to characterize the clinicopathological features. For the rare subset of patients with skin metastasis from CRC, immunotherapy combined with anti-angiogenic targeted therapy may be considered.
Introduction
Colorectal cancer (CRC) is the most common malignant tumor of the digestive system in clinical practice. According to the World Health Organization, in 2022, CRC ranked third in incidence and second in mortality among all neoplasms globally. The incidence and mortality rates of CRC in China ranked second and fourth among all malignant tumors, respectively, accounting for approximately 10% of the annual number of diagnosed cancers and cancer-related deaths. 1 The most common metastatic sites are the lungs and the liver.2–4 Skin metastasis is extremely rare, with studies indicating an incidence rate of about 4%. 5 It usually appears in the late stages of the disease, progresses rapidly, and has a poor prognosis, with more than two-thirds of patients dying within the first 6 months after being diagnosed with skin metastasis. 6 Skin metastases can present in various forms and have no obvious specific symptoms in the early stage, making them easy to miss or misdiagnose. Currently, there is a lack of standard treatment protocols for skin metastases of CRC, which are mostly derived from case reports.
Herein, we present the clinical case of a 66-year-old female patient with proficient mismatch repair (pMMR) rectal cancer. She had metastases on the skin of her abdomen, perineum, lower limbs, and neck, diagnosed 3 years after completion of treatment of the underlying disease. Histological examination of the skin lesions revealed adenocarcinoma, and expression of the same immunohistochemical markers was also found in the primary tumor and the cutaneous metastases. After receiving immunotherapy and targeted therapy, the patient was in partial response (PR). This article reviews the diagnosis and treatment process of this case to provide a reference for clinical diagnosis and treatment, as the incidence of CRC with skin metastases is very low, and there is a lack of standard treatment programs. The reporting of this study conforms to the CARE statement (Supplemental Material).
Case report
A 66-year-old female patient presented with abdominal distension and underwent colonoscopy in July 2020, which revealed a large irregular mass located 15 cm from the anus. Histological examination suggested adenocarcinoma. Rectal cancer radical surgery was performed on August 4, 2020. The morphological pattern was described as adenocarcinoma G3, growing into the myenteron and invading the adjacent fat tissue, and cancer emboli in the vessels were noted (Figure 1(a) and (b)). Immunohistochemistry revealed expression of specific CRC markers: CK20 and SATB-2 (Figure 1(c) and (d)), and showed that mismatch repair (MMR) proteins were intact. Of the 14 adjacent lymph nodes examined, tumor elements were visible in 10 (including 1 mesenteric root lymph node and 9 mesenteric lymph nodes), and 2 additional cancer nodules were observed. The patient was diagnosed with rectal cancer with lymph node metastases (pT3N2bM0, stage IIIc) and underwent six cycles of adjuvant chemotherapy with capecitabine from August 2020 to December 2020.
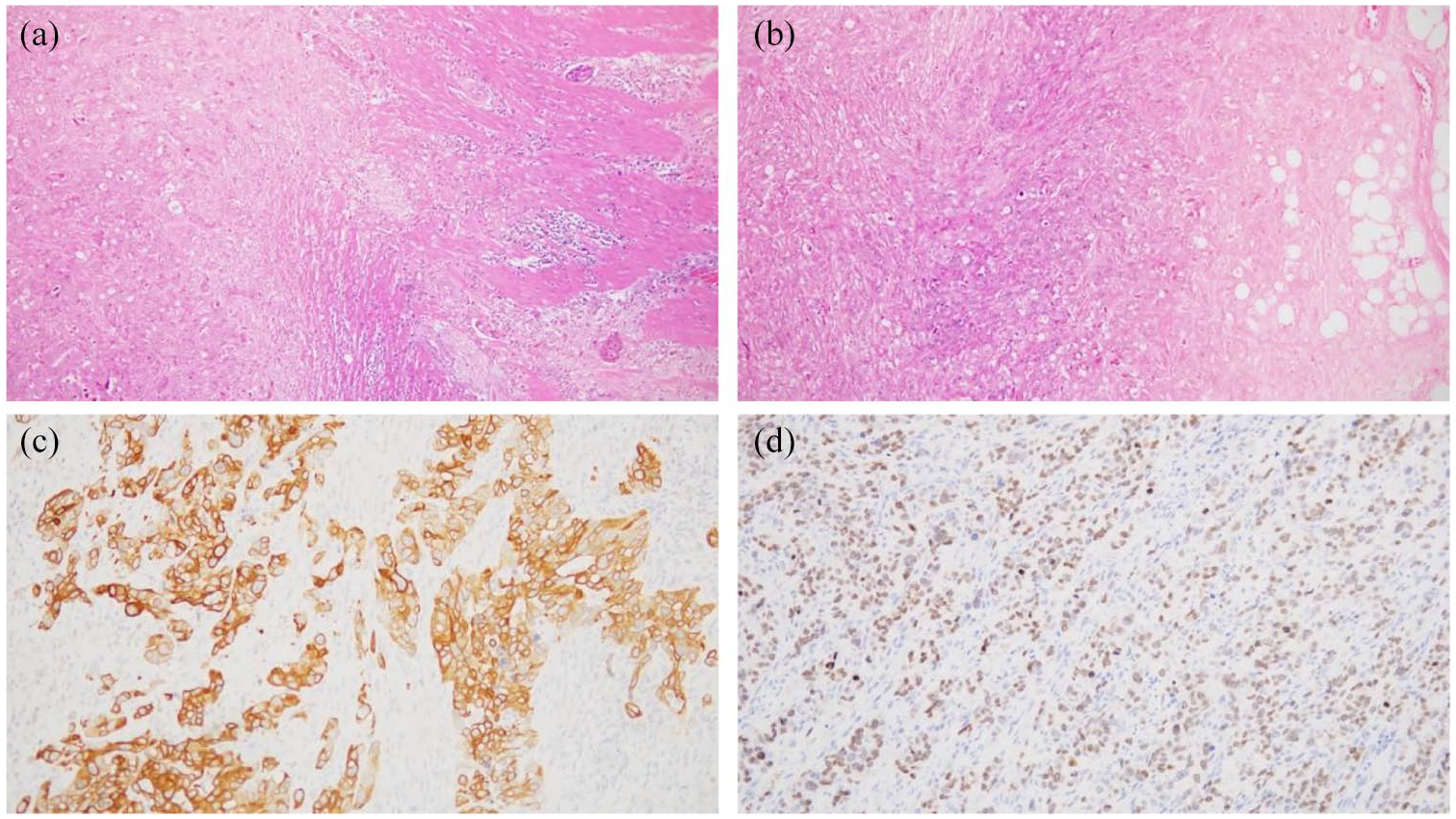
Histological (a, b) (magnification ×10) and Immunohistochemistry (IHC) examination (c, d) (magnification ×40) of the primary tumor. Proliferation of adenocarcinoma masses with predominance of low-grade structures is detected in the rectum wall. The tumor grows into the myenteron and invades the adjacent fat tissue (a, b). IHC study revealed the expression of CK20 (c) and SATB-2 (d).
In November 2021, the patient was found to have enlarged left cervical, right iliac perivascular, and retroperitoneal lymph nodes. A left cervical lymph node biopsy was performed. The cytology showed that heterogeneous cells were present in the left cervical lymph node, which was considered to be a metastatic adenocarcinoma (Figure 2). Given the disease progression (cT3N2bM1b, stage IVB), the patient continued to take capecitabine until April 2022. In April 2022, a computed tomography (CT) scan showed that the above lymph nodes were enlarged compared to the previous scan. To control disease progression, the patient received six cycles of chemotherapy (5-fluorouracil combined with irinotecan) alongside bevacizumab until July 2022. Following disease stabilization, she subsequently underwent seven cycles of bevacizumab plus capecitabine as maintenance therapy until November 2022.
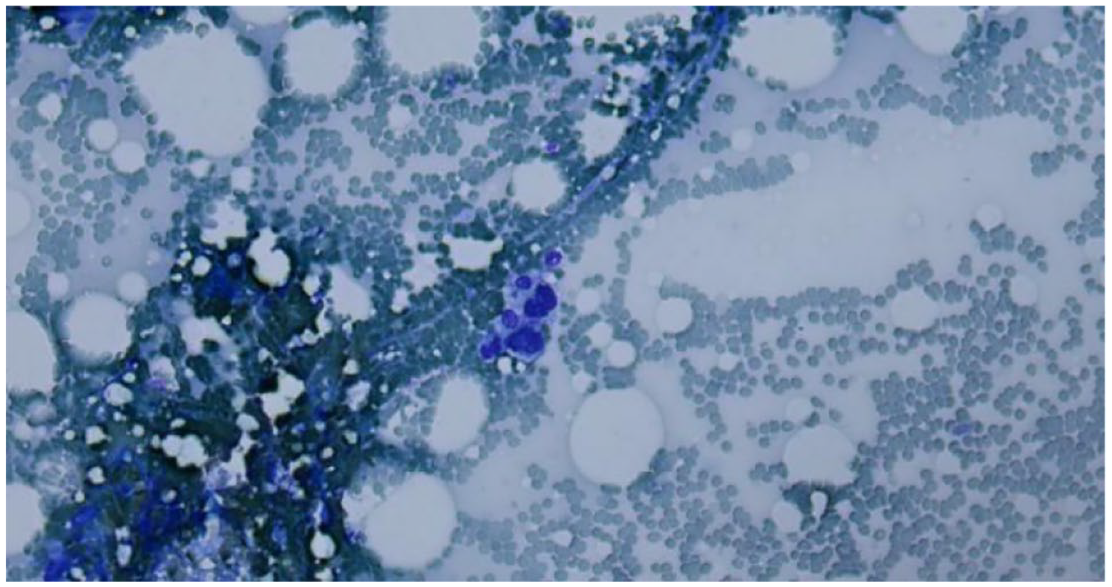
Cytology of the left cervical lymph node (magnification ×10). Heterogeneous cells are present, which are considered to be a metastatic adenocarcinoma.
In January 2023, the patient developed edema of the right lower limb, perineum, and abdomen, thought to be a drug-related side effect. No further treatment or investigations were carried out. The patient was kept under dynamic observation. Due to the apparent increase in tumor markers, six cycles of chemotherapy with 5-fluorouracil and irinotecan, combined with bevacizumab, were administered from February 2023 to May 2023. However, the edema of these regions significantly worsened from June 2023. The patient was referred to the vascular surgery department and underwent a lower limb peripheral lymphatics-small vein anastomosis in September 2023. However, there was still no significant improvement in the perineum and abdominal erythema. The skin lesions presented as non-sunken edema with a hard texture, erythema, and scattered subcutaneous hard nodules on palpation (Figure 3(a)). To clarify the nature of the skin lesions, a diagnostic biopsy was performed in the perineal groin region in October 2023. Histologic examination revealed low-grade squamous cell subepithelial adenocarcinoma (Figure 4(a)), and immunohistochemistry revealed the expression of a specific CRC marker: CK20 (Figure 4(b)). In combination with the patient’s history of rectal cancer and the results of immunohistochemistry, the cause of CRC was first considered. The KRAS gene exon 2 G13D mutation was found. No mutations in the BRAF and NRAS genes were found by standard genetic testing. In addition, a CT scan revealed metastases in the soft tissues of the left back of the neck (Figure 5(a)). Given the presence of multiple cutaneous metastatic lesions and extensive skin involvement in the abdomen, perineum, and right lower limb, palliative radiotherapy was deemed unfeasible. Consequently, the patient received one cycle of oxaliplatin (150 mg) combined with raltitrexed (3 mg) on October 28, 2023. After the chemotherapy, the patient developed a grade III gastrointestinal side effect, and the patient was not willing to continue with the chemotherapy. Meanwhile, the erythema in the perineum and right lower limb gradually increased. On November 17, 2023, the patient initiated fruquintinib-targeted therapy (5 mg orally once daily for 3 weeks followed by a 1-week treatment break), and sintilizumab (200 mg intravenously every 3 weeks) was added in December 2023. After one cycle of fruquintinib combined with sintilizumab, the color of the patient’s perineal and abdominal skin gradually lightened. After three cycles, the texture of the skin gradually softened, and the color further lightened. CT imaging showed a PR. The patient received a total of 14 cycles of sintilizumab combined with fruquintinib from December 11, 2023 to October 12, 2024. During this period, the patient’s skin on the abdominal and perineum regions had ulceration with itching (Figure 3(b)). The patient’s skin on the abdomen, perineum, and right lower limb improved significantly after treatment, with swelling subsiding, ulcer healing, and old scarring (Figure 3(c)-(f)). The metastatic subcutaneous mass on the back of the neck gradually shrank, and the tumor markers continued to decline and stabilized at low levels (Figure 6). The images showed that the right iliac perivascular lymph nodes and retroperitoneal lymph nodes remained steady, the metastases in the soft parts of the left back of the neck decreased further (Figure 5(b)–(f)), and the effectiveness of the treatment reached PR.

Rectal cancer metastases to the skin. (a) Image showing first signs of metastatic process on the right lower limb, perineum, and abdominal skin, presented as non-sunken edema with a hard texture, erythema, and scattered subcutaneous hard nodules on palpation (November 2023). (b) Image showing ulceration and itching of the skin metastatic lesions during treatment (December 2023). (c) Image showing the skin lesions improving gradually after treatment with sintilizumab combined with fruquintinib (January 3, 2024). (d) January 24, 2024. (e) February 2024. (f) November 2024.

Histological (a) and IHC (b) assays of cutaneous metastasis of rectal adenocarcinoma (magnification ×10). Histologic examination revealed low-grade squamous cell subepithelial adenocarcinoma (a). Our IHC study revealed the expression of CK20 (b).

Rectal cancer metastases to the left back of the neck. (a) CT scans showing first signs of metastatic process (October 25, 2023). (b) CT scans showing the skin lesion shrinking gradually (January 19, 2024). (c) April 9, 2024. (d) June 19, 2024. (e) August 28, 2024. (f) October 30, 2024.
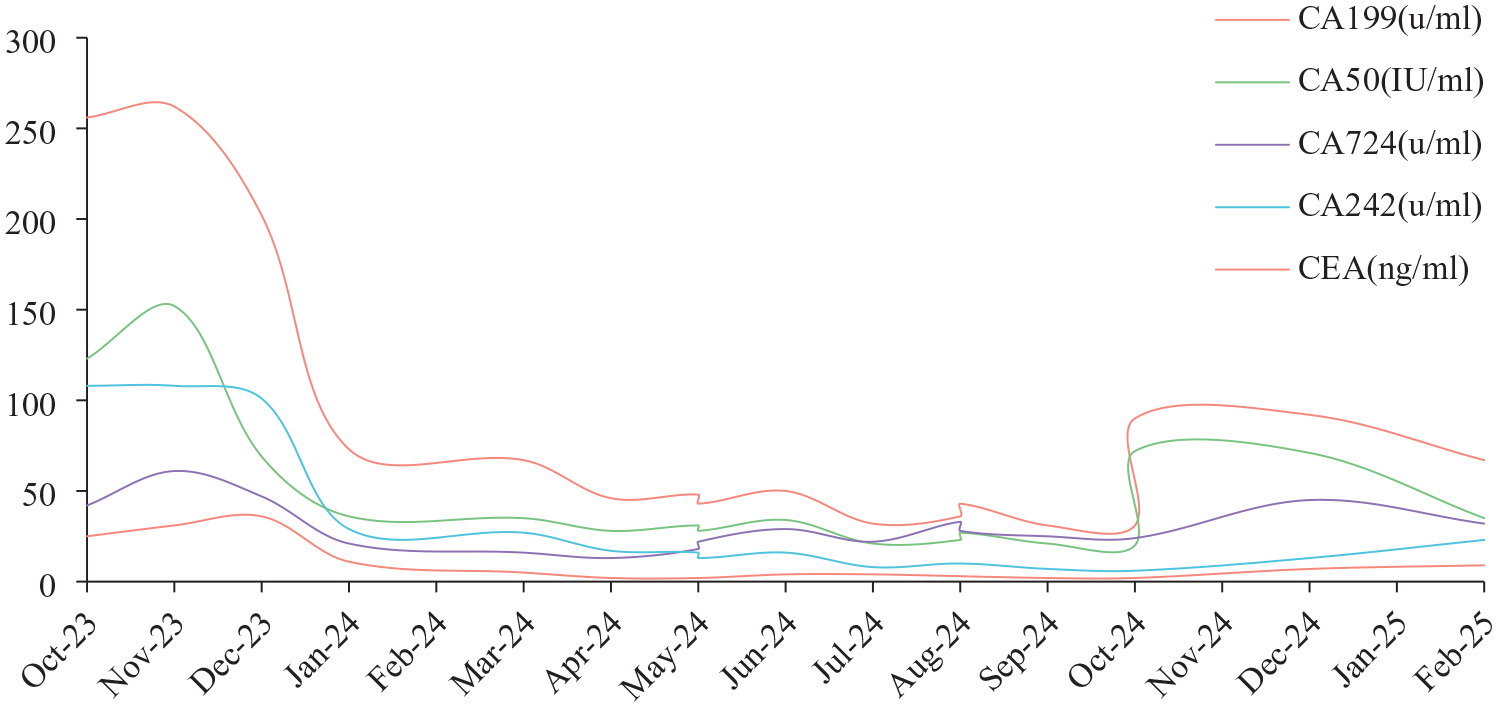
Changes in tumor markers between October 2023 and February 2025.
The patient was admitted to the hospital on October 29, 2024, with “acute abdominal pain with vomiting for 1 day,” and hematologic tests showed that the alanine aminotransferase was 770 U/L, the glutamine aminotransferase was 1173 U/L, the blood amylase was 647 U/L, and the urine amylase was 2995 U/L. At once, the CT scan showed a slightly filled pancreas with exudation from the peripancreatic and mesenteric fat spaces and thickening of the anterior fascia of the left kidney. The diagnosis was considered to be acute immune-related pancreatitis and acute immune-related hepatitis. Given the potentially severe and life-threatening nature of immune-related pancreatitis, along with the need for appropriate management of the adverse events, the patient discontinued fruquintinib and immunotherapy. After hormonal shock and symptomatic treatment, the patient’s symptoms improved, aminotransferases essentially normalized, amylases decreased significantly, and CT suggested inflammation resolved. Meanwhile, following the discontinuation of sintilizumab and fruquintinib, the patient’s tumor markers showed a gradual increase. Consequently, the patient resumed two cycles of fruquintinib-targeted therapy (5 mg orally once daily for 3 weeks followed by a 1-week treatment break) from December 2024 to January 2025. The tumor markers subsequently declined gradually. However, a follow-up CT scan on February 18, 2025, revealed a slight enlargement of the right iliac perivascular lymph nodes and bilateral axillary lymph nodes compared to previous imaging, while lesions in other regions remained stable. As a result, the patient received one cycle of reduced-dose sintilizumab (100 mg intravenously) combined with fruquintinib on February 20, 2025, without experiencing any treatment-related adverse events. As of March 15, 2025, the patient’s condition has remained stable. The results regarding the diagnosis and treatment of the patient are presented in Figure 7.

Timeline of the diagnosis and treatment of the patient.
Discussion
Typically, relatively few malignant tumors metastasize to the skin, and the most common malignant tumors that metastasize to the skin include lung cancer, breast cancer, CRC, and melanoma. 7 Studies have shown that skin involvement occurs in approximately 4% of patients with CRC. Skin metastases of CRC usually occur during the first 2 years of observation. 8 A study showed that about 57% of patients with skin metastasis of CRC had concurrent metastasis to other sites such as the adrenal glands, lungs, liver, brain, prostate, and lymph nodes, as well as bones and muscle tissues. 9 In this case, the patient’s skin metastasis occurred 38 months after the diagnosis of CRC, and lymph node metastases were present at the time of skin metastasis. The significantly longer time interval compared to the 2-year period mentioned in the aforementioned study may be attributed to the fact that the patient only had distant lymph node metastases prior to the development of cutaneous metastases, coupled with well-controlled disease progression. This suggests that delayed skin metastasis in CRC may correlate with limited metastatic burden and effective systemic control, highlighting the need for dynamic risk stratification beyond the conventional 2-year surveillance window.
The regional distribution of skin metastasis is related to the location of the primary tumor. Skin metastasis of CRC commonly occurs in the abdomen, perineum, and perianal skin, and can also occur in various locations of the skin of the whole body, such as the chest and back, scalp, face, and limbs.10–13 Cutaneous metastases can present in various forms, mostly as a single skin lesion, but rarely as multiple lesions. 9 These include persistent erythema, subcutaneous or intradermal nodules, papules, nonhealing ulcers, single or multiple cysts, and granulomatous or fibromatous lesions.14,15 Typically, there are rapidly growing, single or multiple, hard, flesh-colored, round, or oval nodules with limited scleroderma-like changes, which may be accompanied by pain, itching, and ulceration. In addition, it may also present with atypical manifestations, including hyperpigmentation or hypopigmentation, lymphedema, and significant pain. In this case, the cutaneous metastases occurred at general sites. 15 The patient’s skin metastasis initially presented as edema of the abdomen, perineum, and the right lower limb, gradually developing into erythema. The lesion presented a hard texture, and scattered subcutaneous hard nodules were palpable, accompanied by ulceration, itching, and erosion. Besides, a CT scan revealed a subcutaneous soft tissue mass on the left side of the neck. However, the initial clinical presentation was atypical, and there was no significant clinical improvement after local treatment of the symptoms. Later, it gradually progressed to exhibit more specific symptoms. The progression from nonspecific edema to ulcerative nodules underscores the diagnostic challenge of CRC skin metastasis, necessitating heightened suspicion for occult malignancy in patients with persistent cutaneous lesions unresponsive to local therapies.
There is limited research on the mechanism of skin metastasis of CRC. One study reported a case of skin metastasis of rectal cancer with a clinical presentation resembling the case discussed in this article. This case may have resulted in extensive skin metastasis through three major mechanisms simultaneously: lymphatic or hematogenous dissemination, direct tumor extension, and implantation of tumor cells during surgery. 10
The pathology of cutaneous metastases is essentially the same type of pathology as that of the primary. However, there may be differences in the mutational profiles of the primary tumor and metastasis to the skin. A case report of skin metastases from rectal cancer identified that somatic mutations in the APC, TP53, and PTPN11 genes were revealed in primary rectal adenocarcinoma, but another pathogenic TP53 mutation and a frameshift variant in the DYNC1I1 gene were found in cutaneous metastases. 12 The differences may be associated with both the heterogeneity of the malignant neoplasm and the evolution of the primary tumor on top of previous treatment. When a CRC skin metastasis is detected, it is recommended to carry out an immunohistochemical study, such as CK20, CDX2, and SATB2, 11 including detection of the mismatch repair deficiency (dMMR) and evaluation of HER2 status, as well as a molecular genetic study, and compare the results with the available data on the primary tumor. In this case, the morphological pattern of the primary lesion was described as low-grade adenocarcinoma. Immunohistochemistry revealed expression of CK20 and SATB-2, and showed that MMR proteins were intact. Meanwhile, histologic examination of the cutaneous metastatic lesions revealed low-grade squamous cell subepithelial adenocarcinoma with immunohistochemistry staining positive for CK20. The pathological features of the cutaneous metastases were consistent with those of the primary tumor. In addition, standard genetic testing of the cutaneous metastatic lesions identified the KRAS gene exon 2 G13D mutation, with no mutations detected in the BRAF and NRAS genes. Unfortunately, further genetic testing to analyze the molecular genetic characteristics of the patient and to compare the primary lesion with the cutaneous metastatic lesions could not be performed, as both the postoperative specimen of rectal cancer and the biopsy specimen of the cutaneous metastases are currently unavailable. Despite this limitation, the conserved CK20 expression across primary and metastatic sites implies clonal evolution rather than de novo transformation, aligning with prior reports on CRC metastatic fidelity. 12
Due to the low incidence, there is a lack of standard protocols for the treatment of metastatic skin cancer, mostly derived from case reports. Surgical excision of skin metastases from CRC is currently recommended when feasible, with the goal of reducing the tumor burden and improving quality of life. For unresectable lesions, palliative radiotherapy and systemic chemotherapy are possible options, and there is no consensus or guideline to make recommendations for relevant treatment options.15–17
pMMR or microsatellite stable (MSS) CRCs account for approximately 95% of metastatic colorectal cancers (mCRCs). 18 Immune checkpoint inhibitors (ICIs) are effective in the treatment of CRC patients with dMMR or microsatellite instability-high, but studies have shown that pMMR-MSS mCRC is largely ineffective in the treatment of single ICIs. 19 To enhance immunotherapy efficacy in patients with pMMR-MSS mCRC, numerous studies have initially explored a variety of immune combination therapy strategies, and there are worthwhile “bright spots” in the direction of advanced CRC posterior line therapy and perioperative period treatment of locally advanced CRC, among which multi-target tyrosine kinase inhibitors (TKIs), mainly vascular endothelial growth factor receptors (VEGFR), combined with programmed death-1 (PD-1) inhibitors, is one of the most clinically promising combination regimens at present.
The Japanese REGONIVO study was the first exploratory phase Ib study to report the use of nivolumab in combination with low-dose regorafenib in the treatment of refractory MSS colorectal and gastric cancers, with an Objective Response Rate (ORR) as high as 33.3%, a median progression-free survival (mPFS) of 7.9 months, and a 1-year OS rate of 68.0% in MSS mCRC. 20 Fruquintinib, an oral multi-kinase inhibitor, is a potent, highly selective small-molecule inhibitor of VEGFR1, VEGFR2, and VEGFR3. Based on the results of the FRESCO study and according to the guidelines of the Chinese Society of Clinical Oncology in 2019, fruquintinib has been recommended as a treatment in Chinese mCRC patients who experienced tumor progression following two or more prior chemotherapy regimens.21,22 In addition, three additional antibody therapeutics developed by Chinese companies (tislelizumab, sintilizumab, and camrelizumab) became available in China in January 2020, 23 all of which have been proven to be highly selective, fully humanized monoclonal antibodies that have potential in blocking the interaction between PD-1 and its ligands. 24 Based on the REGONIVO combination strategy, Gou et al. attempted a similar regimen using fruquintinib combined with several PD-1 inhibitors as a third-line therapy for mCRC patients with MSS, with an ORR of 11.1%, a DCR of 62.2%, a mPFS of 3.8 months, and a median OS of 14.9 months. In the sintilizumab plus fruquintinib group, the ORR was 7.4% and DCR was 62.9%. The PFS and OS were observed at 3.8 and 11.7 months. 25 Although this study did not replicate the high ORR of the Japanese REGONIVO study, the combination of sintilizumab and fruquintinib shows a numerical improvement in PFS and OS compared to previous randomized controlled trials for standard third-line monotherapies (e.g., regorafenib, fruquintinib, or TAS-102). Besides, Liu et al. conducted a retrospective analysis comparing fruquintinib-sintilizumab (FP) versus TAS-102-bevacizumab (TB) in refractory mCRC. The FP regimen demonstrated superior efficacy, with ORR 16.67% versus 8.82%, DCR 93.06% versus 73.53%, and significantly prolonged mOS (19.4 vs 11.6 months; p < 0.001). 26
In this case of rectal carcinoma with cutaneous and multiple lymph node metastases, the patient received later-line therapy with sintilizumab plus fruquintinib, a decision driven by the lack of standard regimens for skin metastases and prior evidence supporting immune-angiogenic synergy. Cost-effectiveness and accessibility of sintilizumab in resource-limited settings further justified this approach. The patient achieved a PR, and PFS has reached 15 months. The patient’s condition was stable even after discontinuing the fruquintinib and the immunotherapy for immune-related pancreatitis for 4 months. Notably, the 15-month PFS in our case exceeds these benchmarks, potentially attributable to sustained angiogenic inhibition by fruquintinib overcoming transient immune activation seen in prior trials. 25 The patient developed G3–4 immune-related pancreatitis and hepatitis after 14 cycles of PD-1 inhibitor therapy, and the patient’s symptoms improved significantly after timely treatment.
Skin metastases from CRC usually appear in advanced stages of the disease and are a marker of poor prognosis. It is reported that the average survival of patients after diagnosis of cutaneous metastasis from CRC is 18 months, ranging from approximately 1–34 months.7,27 A study retrieved and analyzed 50 patients with skin metastases from rectal cancer published between 1966 and 2023, and the median survival time from the discovery of skin metastases to the patient’s death was 8.5 months. 12 Our patient’s survival beyond 15 months (ongoing) not only validates the efficacy of this combinatorial approach but also redefines prognostic expectations for CRC skin metastasis, advocating its consideration in similar resource-constrained settings. Moreover, the patient harbors a KRAS G13D mutation, which has been associated with immune evasion through tumor microenvironment remodeling in prior studies. 28 Notably, the combination of sintilizumab and fruquintinib demonstrated a partial reversal of this resistance mechanism via synergistic inhibition of angiogenesis and activation of anti-tumor immunity. 29
Previous case reports used conventional treatment regimens, and this patient with skin metastases applied immunotherapy combined with small-molecule TKI, which resulted in a longer period of disease control. For pMMR advanced CRC, the site of metastasis may also be an important factor affecting the efficacy of immunotherapy. In the future, for patients with pMMR advanced CRC, we need to optimize and screen further the advantageous population that may benefit from immunotherapy.
Supplemental Material
sj-pdf-1-tag-10.1177_17562848251338673 – Supplemental material for Partial response to posterior line immunotherapy for more than 15 months in a pMMR patient with cutaneous metastasis of rectal carcinoma: a case report
Supplemental material, sj-pdf-1-tag-10.1177_17562848251338673 for Partial response to posterior line immunotherapy for more than 15 months in a pMMR patient with cutaneous metastasis of rectal carcinoma: a case report by Jing-Jing Li, Peng-Fei Xu and Yan-Li Nie in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
