Abstract
Gastroesophageal reflux disease (GERD) remains a challenging condition, even in the third millennium. For much of the past century, Schwartz’s dictum—“No acid, no ulcer”—has shaped our approach to acid-related diseases, making acid suppression the cornerstone of therapy. Proton pump inhibitors (PPIs) are widely regarded as the standard treatment for GERD. However, they provide only symptomatic relief and do not address the underlying disease. Moreover, nearly 50% of patients experience limited or no response to PPIs in clinical practice. Recent advances in understanding GERD’s pathophysiology, particularly the role of impaired mucosal integrity, have led to innovative therapeutic strategies. Among these, medical devices designed to prevent reflux or coat the esophageal mucosa and form a stable protective barrier represent a significant breakthrough. Esophageal mucosal protection is emerging as a promising approach, especially for patients who do not respond adequately to PPIs. While mucosal-protective agents such as sucralfate and irsogladine have long been available, their formulations have not been well-suited for esophageal protection. The rapid transit time of liquids through the esophagus (typically just a few seconds, even in a supine position) limits the duration of contact between active ingredients and the mucosa. However, hyaluronic acid and chondroitin sulfate-based medical devices have revolutionized the field by enabling active ingredients to adhere to the esophageal lining, ensuring prolonged contact and enhanced protection. Further advancements have led to the development of three distinct formulations (Esoxx™ One, Esoxx Defence, and Esoxx Protection), incorporating additional components, that is, Poloxamer 407, aluminum hydroxide, or natural remedies such as Aloe vera and honey. Each of these formulations offers unique physicochemical properties tailored to address both typical and atypical GERD symptoms. By leveraging the novel therapeutic approach of mucosal protection, these innovations aim to improve treatment outcomes and enhance patients’ overall quality of life.
Plain language summary
Gastro-esophageal reflux disease (GERD), a condition where stomach acid or food flows back into the esophagus, is highly prevalent in the Western world and strongly impairs quality of life. It can cause symptoms like heartburn (a burning feeling in your chest), regurgitation (feeling like food is coming back up), and sometimes even coughing or a sore throat. GERD is still a tough condition to manage, even today. For many years, doctors have focused on reducing stomach acid to treat GERD, with proton pump inhibitors (PPIs) being the standard treatment. However, PPIs only help with the symptoms and don’t tackle the root cause of the disease. Additionally, about half of patients don’t experience enough relief from PPIs. New research into the causes of GERD has led to the development of new treatments, especially those designed to protect the lining of the esophagus. These treatments aim to prevent acid from damaging the esophagus or to create a protective barrier around it. Recently introduced medical devices containing hyaluronic acid (HA) and chondroitin sulfate (CS) that stick to the esophagus offer good protection of the esophageal mucosa, ultimately aiming to provide better results and a better quality of life for patients.
Keywords
Introduction
Gastroesophageal reflux disease (GERD) is a common and chronic condition, affecting 10%–30% of the Western population. 1 This condition is characterized by the reflux of gastric contents into the esophagus, leading to symptoms and/or mucosal damage, as well as potential complications.2–4 The pathogenesis of GERD is multifactorial, involving an interplay of anatomical, physiological, and lifestyle factors.2,4 Recent studies have emphasized two key contributors in the onset of GERD symptoms: impairment of esophageal epithelial barrier and neuro-immune modulation, which may lead to increased reflux hypersensitivity or hyperalgesia.5–7
Given the multifactorial nature of GERD pathogenesis, management strategies must be similarly multifaceted.8–10 Current pharmacologic approaches to address this clinically challenging condition are limited. Reflux inhibitors represent a promise unfulfilled, effective prokinetics are lacking and antidepressants, despite being effective in selected patients, give rise to adverse events in up to 32% of patients.11–14 Antisecretory drugs (H2-receptor antagonists, H2RAs, and proton pump inhibitors (PPIs) remain therefore the mainstay of medical treatment for GERD. They act indirectly by reducing the amount and concentration of gastric secretion available for reflux, thus lessening the aggressive power of the refluxed material.15,16 PPIs also reduce the size of the acid pocket and increase the pH (from 1 to 4) of its content. 17 The clinical efficacy of these drugs has been clearly shown in many studies and the superiority of PPIs over H2RAs has been established beyond doubt. 18 The greater pharmacodynamic effect of PPIs depends on their ability to block the final step in the production of acid, regardless the secretory stimulus. Moreover, PPIs are relatively more effective during the daytime than the nighttime and this leads to a better control of post-prandial reflux events 18 and represent therefore the standard of care.18–20
While PPIs remain the cornerstone of GERD treatment,18–20 they come with limitations.8,21–25 The effectiveness of PPI therapy in patients with esophageal symptoms ranges from 40% to 80% but—compared to those with typical GERD manifestations—is less pronounced in subjects with extraesophageal symptoms.3,26 Studies have shown that 50% of patients with atypical GERD symptoms do not respond to 8–12 weeks of PPI therapy, and 15% show only a partial response.25,27,28 Consequently, there is growing interest in the development and utilization of novel treatments (be they drugs or medical devices) as add-on or alternative medications to traditional pharmacotherapy.8,29,30 In particular, medical devices offer the potential for topical therapy, reduced use of systemic therapies and, likely, improved patient outcomes.
Furthermore, mucosal lesions caused by GERD may be treated with PPI therapy in combination with other active substances or devices with the aim of enhancing their healing effect and bolstering esophageal mucosal defenses.31–33 These devices exert mucosal defensive action by creating a film over the esophageal mucosa and acting as a mechanical barrier against the noxious components of the refluxate.31–33 Each product, based on its specific formulation, may be tailored for patients with different clinical characteristics and natural history. 32 Some studies have indeed shown that adding alginate-containing formulations to PPI therapy increases the response rate in patients with nonerosive reflux disease (NERD) as well as those with laryngopharyngeal reflux (LPR, for review see Scarpignato et al. 30 ). Furthermore, mucosal protectants (such as antacids, alginates, and sucralfate) have been shown to be more effective than placebo in treating mild reflux symptoms and offer a viable alternative to acid suppression in this patient population. 34 A Class III medical device, Poliprotect™, has recently been introduced to the market. It consists of a polysaccharide fraction derived from Aloe vera, Malva sylvestris, and Althea officinalis, along with a flavonoid fraction from Glycyrrhiza glabra and Matricaria recutita. This device, known for its long-lasting mucoadhesive properties, has been shown to reduce esophageal mucosal damage induced by an acid-pepsin-bile solution. 35 A recent randomized controlled trial evaluated the efficacy and safety of Poliprotect (administered five times a day for the first 2 weeks, followed by on-demand use) compared to omeprazole in alleviating heartburn and epigastric pain or burning. The study found that Poliprotect was non-inferior to standard-dose omeprazole in relieving symptoms of heartburn and epigastric burning in patients without erosive esophagitis (EE) or gastroduodenal lesions. 36 It is worth mentioning that the comparison was made with omeprazole 20 mg, a relatively low dose that exhibits considerable inter-individual variability. 37
An innovative development in the treatment of acid-related diseases, including GERD, is the introduction of H+,K+-ATPase inhibitors, known as potassium-competitive acid blockers (P-CABs). These drugs block the K+ exchange channel of the proton pump, leading to fast, competitive, and reversible inhibition of acid secretion. P-CABs provide a rapid and more significant increase in intra-gastric pH compared to delayed-release PPIs, while maintaining similar or even stronger antisecretory effects. 38 The duration of their action depends on the drug’s half-life. In the treatment of severe reflux esophagitis (C&D according to the Los Angeles classification), vonoprazan (the first P-CAB) has shown superiority over PPIs. However, the benefits of P-CABs for NERD and extra-esophageal manifestations of GERD have yet to be fully established. 39
Over the past decade, several investigations have highlighted the role of medical devices containing hyaluronic acid (HA) and chondroitin sulfate (CS), with or without an antacid component, in the treatment of GERD. In experiments, performed on a 3D reconstructed human esophageal epithelium, formulations combining HA and CS have demonstrated a barrier-protective effect, improving esophageal epithelial integrity thus preventing the transepithelial penetration of small, toxic, and acidic molecules from the lumen. 40 This topical protective effect arises from their ability to adhere to esophageal mucosa and form a long-lasting protective film over the epithelial layer. 40
The aim of this paper is to outline the complexities of GERD spectrum and provide a rationale for incorporating the recently developed medical devices into the therapeutic armamentarium. By recognizing the diversity and overlap of GERD subtypes, and evaluating the efficacy and tolerability profile of these devices, clinicians can better tailor treatment strategies to address the specific needs of each patient, ultimately enhancing the quality of life for individuals living with this chronic and challenging condition.
Epidemiology of GERD
The prevalence of GERD has progressively increased over the past few decades. However, the occurrence of gastroesophageal reflux symptoms varies significantly across countries, even when similar definitions are applied. According to the latest review, 41 GERD prevalence estimates range from 18.1% to 27.8% in North America, 8.8% to 25.9% in Europe, 2.5% to 7.8% in East Asia, 8.7% to 33.1% in the Middle East, 11.6% in Australia, and 23.0% in South America. When considering only studies that used a weekly frequency of heartburn or regurgitation to define GERD, a recent meta-analysis reported a pooled prevalence of 13.3%. 1 GERD prevalence was notably higher in individuals aged 50 or older, smokers, NSAID users, and those who were obese, although these associations were relatively modest.
A prospective European study, the ProGERD study, found that 32.8% of patients with heartburn also experienced extra-esophageal symptoms. The prevalence was higher among those with erosive reflux disease (34.9%) compared to those with NERD (30.5%). 42 The most common GERD-associated disorders included chest pain (14.5%), chronic cough (13%), laryngeal disorders (10.4%), and asthma (4.8%). 42 However, this study only considered patients with concomitant heartburn, leaving out those who experienced only extra-esophageal symptoms. As a result, determining the prevalence of extra-esophageal symptoms in GERD patients without typical symptoms is more difficult, as diagnosing these cases is challenging. It is likely that the true prevalence of extra-digestive GERD is higher than previously estimated. For example, the incidence of LPR in published studies ranges from 5% to 30% (for review see Stabenau and Johnston 43 ). However, due to the absence of a gold standard for diagnosing LPR, accurately assessing its prevalence and incidence remains problematic. In particular, some symptoms like chronic cough and wheezing have a significantly lower likelihood of direct reflux etiology compared with typical symptoms. 44 The complex pathophysiology of LPR and atypical GERD, which extends beyond gastroesophageal reflux to include autonomic nerve dysfunction and neural hypersensitivity, further complicates the issue. 45
The clinical spectrum of GERD
GERD is a heterogeneous condition with a diverse and wide spectrum of symptoms and reflux profiles. 4 The frequency of clinical manifestations can vary from occasional episodes to daily symptoms. 46 GERD presents with a broad spectrum of symptoms that are typically classified into two categories: typical symptoms (such as heartburn, regurgitation, and non-cardiac chest pain) and atypical or extra-esophageal symptoms (Figure 1). 46 Extra-esophageal GERD encompasses a variety of conditions. 26 Pulmonary manifestations include asthma, bronchitis, microaspiration, and pulmonary fibrosis. 47 Ear, nose, and throat (ENT) symptoms comprise hoarseness, chronic cough, laryngitis. GERD may contribute to extraesophageal syndromes through either direct or indirect mechanisms.48–50 Specifically, damage may result from the direct exposure of gastric acid to the mucosa of the tracheobronchial tree, laryngopharynx (including the vocal cords), middle ear, and nasal sinuses, or from the macro- and micro-aspiration of refluxed gastroduodenal contents. 48

Clinical presentation of gastroesophageal reflux disease: typical and atypical symptoms.
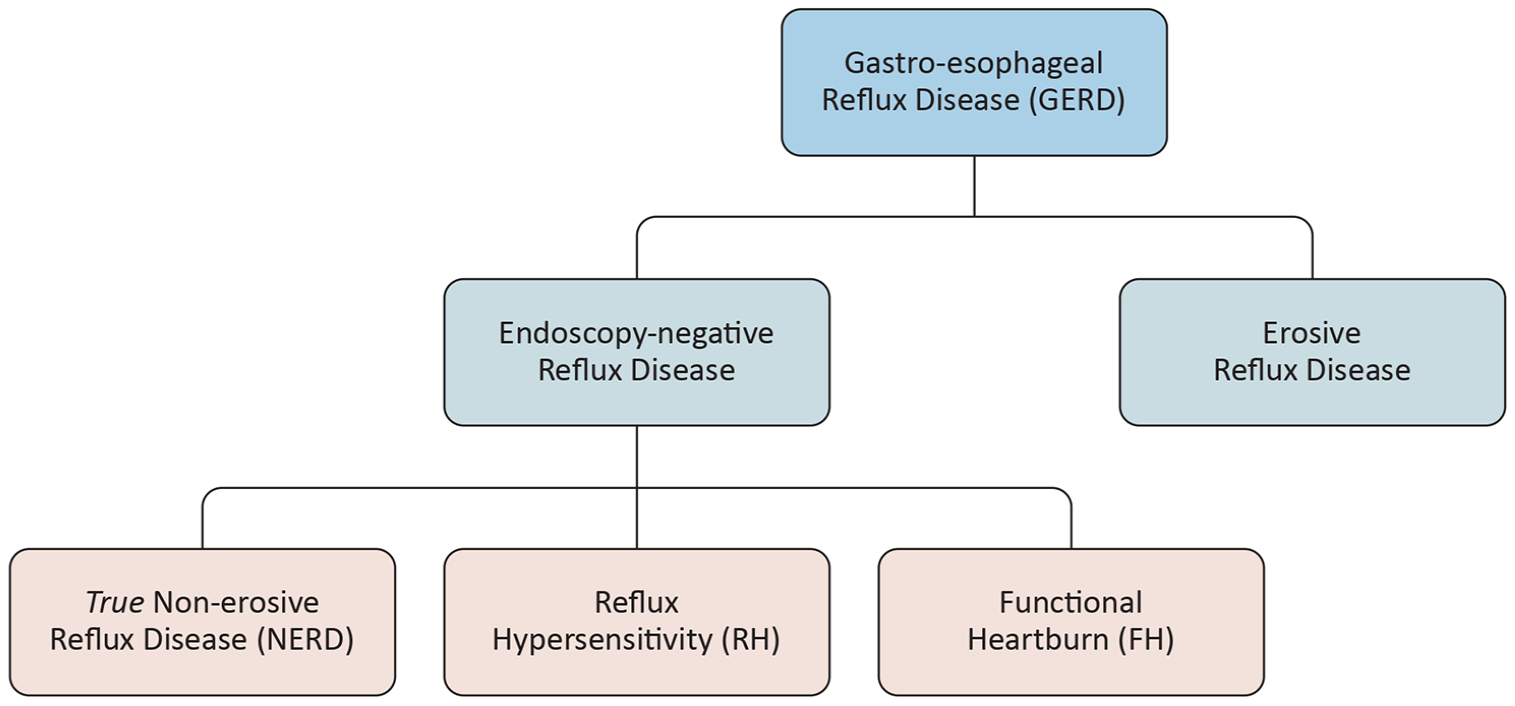
NERD is the more common phenotype of GERD, accounting for 70%–80% of cases. It includes patients who experience typical symptoms but show no macroscopic mucosal damage at endoscopy. 51 In contrast, 20%–30% of patients may show esophageal erosions during endoscopy or develop complications such as Barrett Esophagus or esophageal stenosis. 52
In clinical practice, GERD is empirically diagnosed based on typical esophageal symptoms and positive response to acid suppression.53,54 However, some GERD patients may present with atypical symptoms, which can be extraesophageal or mixed in nature. 55 When refluxate reaches the proximal esophagus or even the laryngopharynx, it can lead to a condition known as LPR syndrome.45,56 The larynx is significantly sensitive to acid and pepsin than the esophagus, with even a few reflux episodes being sufficient to cause substantial inflammation and damage to the epithelial lining. 57 Unlike the esophageal mucosa, this damage is often irreversible. 58 An Italian study found that both patients with erosive and non-erosive reflux disease experience at least one extra-esophageal symptom, particularly laryngeal symptoms, with similar prevalence rates (72% vs 79%, respectively). 59
A comprehensive diagnosis of GERD typically involves a combination of clinical evaluation, response to acid suppression therapy, endoscopy, and functional tests such as pH-impedance monitoring.44,54,60,61 When the evidence for GERD remains inconclusive, high-resolution manometry and advanced impedance metrics can provide clinicians with a more accurate diagnosis.62–66 This approach is particularly useful for patients with persistent symptoms (both typical and atypical), who show no evidence of esophageal mucosal damage, especially when a trial of acid suppression has been ineffective.25,67,68 Patients with endoscopy-negative reflux disease represent indeed a heterogeneous population, including those with true NERD, reflux hypersensitivity (RH), or functional heartburn (FH). 51 Only patients in whom acid is the primary trigger for symptom can benefit from acid suppression therapy. 54
This diagnostic approach uncovers a broad phenotypic spectrum of GERD, ranging from endoscopy-negative reflux disease to EE (Figure 2).4,69,70 Esophagitis refers to inflammation of the distal esophageal mucosa, which, if left untreated, can progress to erosions or ulcers. Esophageal erosions are the most common consequence of esophageal injury. 71
Phenotypic varieties of gastroesophageal reflux disease. In clinical practice, overlap between subgroups is possible.
The use of pH-impedance monitoring has revealed a high prevalence of esophageal functional disorders in patients with endoscopy-negative reflux disease.4,51 In addition to true NERD (presence of symptoms and abnormal pH-impedance recording), RH is characterized by esophageal symptoms in the absence of clear structural, inflammatory, or functional abnormalities. Although the underlying mechanisms for esophageal hypersensitivity remain unclear, RH patients typically experience more frequently weakly acidic reflux events, heightened sensitivity compared to those with FH, and a greater incidence of proximal reflux, factors that may contribute to their symptoms.72,73 Notably, functional disorders such as functional dyspepsia and irritable bowel syndrome are more frequently associated with FH than with other phenotypes of non-erosive reflux disease.74,75
Patient journey in GERD
The “patient’s journey” refers to the full experience a patient undergoes, starting from the recognition of symptoms or a health issue, through diagnosis, treatment, and ongoing management, and extending to follow-up care and long-term recovery or adaptation. It encompasses all interactions with healthcare providers, medical systems, and services, addressing both the physical and emotional aspects of the healthcare process. 76 This concept is often used to analyze and improve the overall patient experience, outcomes, and healthcare delivery. By understanding how patients navigate the healthcare system, the goal is to optimize their experiences, enhancing both health outcomes and patient satisfaction.77,78
GERD is a complex condition with a wide range of clinical manifestations and varying levels of severity. Treating GERD requires a personalized approach, tailored to the individual patient’s symptoms, disease severity, and response to therapy.28,79,80 Serious complications, including EE, Barrett’s esophagus, and esophageal cancer, underscore the importance of effective management. 52
General practitioners, as frontline healthcare providers, play a crucial role in managing GERD. 10 However, for more complex cases, the expertise of gastroenterologists and other specialists is essential. Collaborative care across multiple medical disciplines is necessary to address the diverse and multifaceted nature of the condition. 81 In clinical practice, gastroenterologists typically assess symptoms and medical history and may perform an upper gastrointestinal (GI) endoscopy when indicated to confirm a GERD diagnosis. 82 In patients with macroscopically normal esophageal mucosa, additional functional tests are needed to differentiate between true NERD, RH, or FH. Notably, pH-impedance monitoring can quantify both acid and non-acid reflux, while also correlating symptoms with reflux events.44,65,83,84 Measuring baseline impedance is particularly informative, as it provides insight into mucosal integrity, enabling a comprehensive evaluation of the patient's condition.84–86 Esophageal mucosal impedance can also be measured during endoscopy using specially designed catheters that pass through the operative channel.87,88 However, this technique is typically available only at referral centers.
Patients presenting with chronic cough, laryngitis, and upper airway symptoms suggestive of GERD are often referred to an ENT specialist. After a thorough evaluation, the specialist may suspect extraesophageal GERD and recommend further diagnostic testing, which may include a consultation with a gastroenterologist.26,56,66,89 In this context, the ENT specialist plays a key role in assessing and managing the impact of GERD on both the upper respiratory and digestive systems, providing valuable insights that allow a more comprehensive and coordinated treatment approach for GERD.
In conclusion, categorizing GERD patients into distinct phenotypic groups can help personalize treatment strategies, leading to more effective management. However, GERD therapy remains challenging, as acid suppression (even with co-medications) is not always sufficient. A significant proportion of GERD patients (20%–40%)25,82,90 continue to experience symptoms despite medical treatment. In such cases, alternative therapies or mucosal protectants, either alone or in combination with acid suppressants, may achieve symptom relief and promote mucosal healing.32,78
HA and CS-based medical devices
The recent advances in understanding the pathophysiology of GERD, particularly the role of impaired mucosal integrity, have driven the development of novel medical devices designed to prevent reflux or coat the esophageal mucosa and form a stable protective film.73,84,91–95 These devices can be broadly classified into two main categories 8 :
• Raft-forming, alginate-based preparations, which create a mechanical barrier against reflux and primarily act in the stomach.
• Mucosal protectants, such as HA and CS-based formulations, which primarily exert their effects in the esophagus.
Both types of medical devices are generally well tolerated, with mucosal protectants typically being more palatable. However, a key concern with these devices is the bioavailability of the active ingredients (namely HA and CS) at the level of the esophageal mucosa. The short transit time of liquids through the esophagus (typically less than 16 s, even in a supine position) 96 limits the duration of contact between the active ingredients and the mucosa.
To address this challenge, viscous liquid formulations that adhere to and coat the mucosal surface are used. These formulations not only help limit the contact of refluxed acid and pepsin with the epithelial cells 97 but also serve as a vehicle to deliver drugs locally within the esophagus. 98 In response to these considerations, bioadhesive formulations have been developed to prolong their contact with the mucosal lining.
Esoxx™ One
A Class III medical device, Esoxx™ One (Alfasigma, Bologna, Italy), was specifically designed and developed.99,100 It is composed of a 1:2.5 ratio mixture of low molecular weight (80–100 kDa) HA and low molecular weight CS (10–20 kDa), dispersed in a bioadhesive carrier (Poloxamer 407). This combination forms a macromolecular complex that coats the esophageal mucosa, acting as a mechanical barrier to protect against the harmful components of refluxate.
Esoxx One is composed of two well-recognized physiological substances. One of them, HA, is a biologically active compound widely present in the body, playing a crucial role in cellular regulation through its interaction with specific receptors. 101 As a versatile glycosaminoglycan, it is a fundamental component of most extracellular matrices and is involved in essential physiological functions such as tissue regeneration, wound healing, morphogenesis, and structural organization of the matrix. 102 The biological significance of HA largely stems from its hydrophilic and hydrodynamic characteristics, which enable it to retain water and provide structural support. Hydrogels, which consist of cross-linked hydrophilic polymers, utilize these properties to act as scaffolds for tissue repair and regeneration, gradually breaking down through enzymatic degradation once healing is complete. 101 Moreover, the molecular weight of HA determines its effect on angiogenesis. The low molecular weight form promotes the development of new blood vessels and triggers a signaling cascade that drives endothelial cell growth and migration. Conversely, the native high molecular weight version inhibits angiogenesis, preventing blood vessel formation. 101 Topical applications of HA have been widely used to manage recurrent aphthous ulcers in the oral mucosa103,104 providing rapid symptom relief. This therapeutic effect is likely enhanced by its dose-dependent anti-inflammatory properties, which further contribute to its effectiveness. 105
CS is a naturally occurring glycosaminoglycan found within the extracellular matrix that surrounds cells. It is particularly abundant in cartilage, skin, blood vessels, ligaments, and tendons, where it plays a crucial role as a key component of proteoglycans. 106 Research indicates that CS contributes to various biological processes, including inflammation regulation, cell proliferation, differentiation, migration, tissue development, organ formation, wound healing, and even responses to infections. 107 These functions stem from its ability to interact with a diverse range of molecules, such as matrix components, growth factors, protease inhibitors, cytokines, chemokines, and adhesion molecules, through both specific and non-specific saccharide domains along its chains. 107 Additionally, this compound possesses immune-modulatory, 108 anti-inflammatory,107,108 and antioxidant 109 properties. Beyond these broad interactions, CS can also bind selectively to bioactive molecules like pepsin. Studies have demonstrated that it can reduce peptic activity in both in vitro 110 and in vivo111,112 settings. Historically, it has even been explored as a potential treatment for peptic ulcers. 113
Poloxamer 407, composed of ethylene oxide and propylene oxide blocks, is a hydrophilic, non-ionic surfactant known for its thermo-reversible properties, making it highly valuable in drug formulation. At room temperature, it remains in a fluid state, facilitating easy administration, while at body temperature, it transitions into a gel-like form, enabling sustained release of pharmaceutical compounds. 114 Formulations incorporating Poloxamer 407 have been shown to improve the solubility of drugs with low water solubility and provide extended-release profiles in various pharmaceutical applications. 115 Additionally, its adhesive properties enhance the retention time of active agents within the gastrointestinal tract. Studies using an optical fiber spectrofluorimetric method in mice have demonstrated strong adhesion to the esophagus, allowing for effective drug diffusion into the mucosal lining. 114
In accordance with European Council Directive 93/42/EEC, 116 the National Health Institute in Rome has designated this bioadhesive formulation as a class III medical device, intended for human use in the treatment or relief of diseases. Generally, medical devices in this category function through physical mechanisms, such as mechanical action, serving as a protective barrier, or providing structural support or replacement for organs and bodily functions.
This medical device was studied in in vitro and ex vivo models to evaluate its filming and barrier protective activity.
The mucoadhesive properties of Esoxx One were assessed for its ability to adhere to a partially purified mucin layer type II from pig stomach. The formulation remained attached to a 12% (w/w) mucin layer, even when the support was rotated 90° or when the film was rinsed with water to simulate washout during swallowing. 117
The rheological behavior was evaluated using the amplitude sweep technique. The flow curves showed that Esoxx One exhibited a Newtonian-like behavior, with viscosity remaining unaffected by mechanical stress. 117 This characteristic suggests that the product could stay attached to the esophageal surface during ingestion, despite the mechanical stresses that typically reduce the viscosity of liquid formulations. In the rheological comparison, the formulation demonstrated the ability to interact with mucin, as evidenced by a rheological synergy. Specifically, the viscosity of the formulation/mucin mixture was higher than the sum of the viscosities of the individual components (formulation and pure mucin). This synergy points to a strong interaction between the liquid formulation and mucin, implying bioadhesive behavior with mucin, the primary component of the esophageal lining. 117
The film-forming and barrier effects of this medical device were studied using a 3D reconstructed human esophageal epithelium model. This model is developed after 5 days of air-lifted culture of the K510 human epithelial cell line (derived from squamous cell carcinoma) in a chemically defined medium, and it fully replicates the morphology of the esophageal epithelium. 118 The film-forming capacity was evaluated by tracking the kinetics of transepithelial caffeine passage, while the barrier function was assessed by measuring transepithelial electrical resistance (TEER) and lucifer yellow (LY) permeability as markers of paracellular passage.40,117,119,120 Compared to placebo (an identical formulation without the functional ingredients, i.e., HA and CS), caffeine passage after 15 min was significantly reduced, with this difference persisting at both 1 and 2 h. TEER remained unchanged after application of Esoxx One, indicating preserved barrier integrity, while LY permeability was reduced by both the formulation and placebo, suggesting a decrease in intercellular gaps or a strengthening of cell–cell junctions. 120
The mucosal protective effects of Esoxx One were further evaluated by measuring the expression of the tight junction protein, claudin-4, and the back diffusion of H+ ions. Exposure of tissue to 0.1 M HCl or simulated gastric fluid (SGF) led to a significant reduction in claudin-4 expression (by 90% and 50%, respectively). However, pre-treatment with Esoxx One completely prevented this decline in expression. As a result, the back diffusion of H+ ions following HCl or SGF application was significantly reduced by the mucoadhesive formulation, as indicated by pH measurements taken from both the apical and basolateral surfaces. These findings suggest that the reduction in H+ ion back diffusion is due to the preservation of mucosal integrity, as reflected by the maintenance of tight junction protein expression, rather than a direct neutralizing effect of Esoxx One. Notably, the acid-neutralizing capacity (ANC) of the formulation was minimal, measuring only 3.73 ± 0.18 mEq. 120
An ex vivo experimental study using a swine model demonstrated that perfusing the esophageal lumen with Esoxx One prevents the increase in mucosal permeability and tissue damage induced by acid and/or pepsin. 121 Building on these findings, two double-blind, placebo-controlled studies showed that short-term treatment with Esoxx One provides significant and rapid symptom relief in patients with GERD.122,123
In the first study, 52% of patients in the Esoxx One group achieved complete symptom relief, compared to only 10% in the placebo group (p < 0.01). Furthermore, the time to complete symptom relief was significantly shorter in the Esoxx One group (38 min) compared to the placebo group (65 min). 122 In the subsequent randomized, double-blind, placebo-controlled, two-way crossover study, 20 patients with endoscopy-negative reflux disease, who had symptoms unresponsive to antisecretory treatment were given one spoon of Esoxx One four times daily (away from meals, with a double dose before bedtime). After just 2 weeks of treatment, there was a marked and statistically significant reduction in the total Symptom Severity Index score, as well as in the individual scores for heartburn and regurgitation. 123 The substantial reduction in regurgitation episodes is particularly relevant, given that regurgitation tends to be less responsive to acid suppression than heartburn in GERD patients. 124 This suggests that persistent regurgitation may be a contributing factor to incomplete treatment response.
Several underlying mechanisms contribute to the failure of PPI treatment. They include patient-related (e.g., lack of compliance), physician-related (e.g., misdiagnosis), and drug-related (e.g., short duration of action) mechanisms.125,126 Currently, the most effective approach to deal with PPI-refractory reflux disease is making a precise diagnosis, by adding a functional evaluation (e.g., high-resolution manometry and pH-impedance recording) to upper GI endoscopy. Including biopsy (and subsequent histological examination) of the “macroscopically-normal” mucosa during endoscopic examination can help to identify microscopic mucosal damage.127,128 Microscopic esophagitis has proven to be a reliable diagnostic marker, distinguishing FH from GERD, and could help guide more appropriate treatment. 91 However, these methods, while effective, are time-consuming and costly, making them impractical for routine clinical practice.
An alternative, simpler approach could involve combination therapy, where drugs with different mechanisms of action are added to PPIs. Until very recently, only alginate-containing formulations129,130—given as add-on medications—proved to be capable of improving symptom control in endoscopy-negative patients. For those with motility-related symptoms, such as nausea, postprandial fullness, and early satiety, a trial with prokinetics may be warranted. 131 A recent meta-analysis confirmed that combining prokinetics with PPIs is more effective than PPIs alone in managing GERD. 132 However, safety issues and the dichotomy between symptoms and delayed gastric emptying in the prokinetic effect (where drugs can improve symptoms without accelerating emptying rate or vice-versa or affect neither) 133 make this treatment option challenging and unpredictable. In contrast, adding mucosal protectants, which are both effective and safe, to PPIs could offer a promising alternative.
To test this hypothesis, a double-blind, placebo-controlled trial evaluated the efficacy and safety of Esoxx One in combination with acid suppression, compared to acid suppression alone, in patients with endoscopy-negative reflux disease. 134 This design was chosen to reflect real-world clinical practice outside referral centers, where advanced diagnostic tools are not typically available. The results of this study demonstrated that adding Esoxx One (administered 1 h after each meal and at bedtime for 14 consecutive days) to acid suppression led to significantly higher symptom relief in GERD patients compared to acid suppression alone. Both primary and secondary endpoints were achieved in a larger proportion of patients. Additionally, the combination therapy was well-tolerated, with the total number of adverse events comparable to that in the placebo group. 134 The synergistic effect of Esoxx One with PPIs observed in this study suggests that adding mucosal protection to acid suppression may benefit a broader group of NERD patients, providing both symptom relief and improved health-related quality of life, and potentially reducing the incidence of treatment failures. While PPIs are effective in providing symptom relief over time in both erosive and non-erosive disease, as shown in studies comparing PPIs with P-CABs135,136 this combination therapy may achieve in 2 weeks the same symptom relief that PPIs typically provide in 4 weeks. For patients seeking quicker symptom relief, this time-dependent therapeutic advantage could be particularly valuable.
Following a case report suggesting that adding Esoxx One to intensive acid suppression accelerates the healing of severe esophagitis, 137 a randomized clinical trial was conducted to evaluate the effects of combining mucosal protection with PPIs in patients with C and D esophagitis. 31 After 4 weeks, 100% of patients in the combined treatment group showed a downgrading of mucosal lesions, compared to 80% in the PPI-only group (p < 0.01). Additionally, the proportion of patients experiencing symptom relief, including heartburn, retrosternal pain, odynophagia, and dysphagia, was significantly higher in those receiving both mucosal protection and acid suppression. 31 Notably, in this study, the standard PPI dose was used (while a double dose is often prescribed for severe esophagitis), and the treatment duration was only 4 weeks, rather than the typical 8–12 weeks.138,139
During endoscopy, biopsy samples were collected and analyzed by immunohistochemistry to quantify the expression of Ki67 (a marker of cell proliferation), as well as claudin-1 and claudin-4. As expected, Ki67 expression was elevated, and tight junction proteins were reduced in the erosive mucosa of the esophagus. After therapy, Ki67 expression decreased, while levels of claudin-1 and claudin-4 increased. Notably, in tissues from patients treated with Esoxx One in combination with PPIs, these changes were significantly more pronounced, indicating a more complete restoration of mucosal integrity. 31
A meta-analysis of three studies involving 181 patients with erosive GERD, published in Russian, assessed the efficacy of combination therapy. All studies followed a uniform design, with primary endpoints including complete epithelialization of esophageal erosions and full resolution of heartburn, measured 28 days after starting therapy. The results showed that combination therapy with Esoxx One (10 mL, four times daily) and pantoprazole (40 mg daily) was significantly more effective than monotherapy for healing of esophageal erosions at 28 days (Relative Risk (RR): 1.267, 95% CI: 1.082–1.483, p = 0.003). However, there was no significant difference between the groups in terms of complete resolution of heartburn on day 28 (RR: 1.638, 95% CI: 0.660–4.067, p = 0.287). 140
In addition to its effectiveness in reflux esophagitis, Esoxx One was also found to be efficacious in radiation-induced esophagitis, a common complication and dose-limiting factor in oncologic treatments. In an open-label study, 41 patients undergoing radio- or radio-chemotherapy for lung, gastric, or esophageal cancer received the medical device either as a standalone treatment or in combination with supportive therapy (antacids, antisecretory compounds, anti-inflammatory drugs, opioids). In the combination group, Esoxx One was administered either concurrently with supportive drugs or as an adjuvant treatment following inadequate response to the supportive therapy. Symptom relief was substantial in nearly all patients, allowing 95% of them to successfully complete their oncologic treatment. 141
In addition to typical GERD symptoms, extra-esophageal manifestations can also benefit from esophageal mucosal protection. In a Polish open-label study, 51 patients with LPR symptoms, such as throat clearing, hoarseness, and cough after eating or while lying down, and laryngoscopic signs, including redness, vocal fold edema, and posterior commissure hypertrophy, were evaluated. 142 The Reflux Symptom Index (RSI) and Reflux Finding Score (RFS) were assessed before and after 2 weeks of treatment with Esoxx One, in combination with PPIs. While patients were not completely symptom-free, a significant reduction in symptoms was observed across the entire patient population. After treatment, the RFS fell below the diagnostic threshold for LPR. Compared to baseline, 98% of patients showed substantial improvement in laryngeal lesions. 142 While these promising results are encouraging, they should be confirmed in larger, well-designed clinical trials.
Although originally developed for esophageal mucosal protection, coating of the gastric mucosa is inevitable following esophageal emptying, making its efficacy for gastric complaints unsurprising. In a retrospective, double-blind, randomized placebo-controlled study, the effects of Esoxx One were evaluated in patients with endoscopy-confirmed gastritis. 143 The study assessed symptoms (upper abdominal pain/discomfort, measured by the visual analog scale (VAS)) and mucosal lesions (blood oozing, hyperemia, and edema) before and after therapy. Compared to placebo, the treatment group showed a significant reduction in VAS pain after 5 weeks of therapy (p < 0.001). Additionally, 68% of patients exhibited endoscopic healing of mucosal lesions, while 24% showed moderate improvement. The improvement in mucosal lesions was consistent with the reduction in dyspeptic symptoms. 143 While these findings suggest potential for expanding the clinical use of Esoxx One beyond esophageal conditions, caution is warranted in interpreting the results, as the study lacked appropriate histological characterization of gastritis and testing for Helicobacter pylori infection.
Esoxx™ Defence
The success of mucosal protection combined with acid suppression has led to the development of a modified medical device, Esoxx Defence, which incorporates a buffering agent. Among the available antacids, aluminum hydroxide is one of the most effective, rapidly neutralizing gastric acid (ANC: 29 mEq/15 mL) and increasing intra-gastric pH. 144 The ratio between the active ingredients of the medical device is 1:20:40 for CS, HA, and aluminum hydroxide, respectively. This antacid also exhibits pH-dependent binding and inactivation of pepsin, as well as bile-binding capacity comparable to that of colestyramine.144,145 These pharmacological properties counterbalance two key aggressive factors of the refluxate (i.e., gastric acid and pepsin), which can damage not only the esophageal mucosa but also the upper airways.57,146,147 Furthermore, aluminum hydroxide exerts site- and cyto-protective effects through the synthesis and release of endogenous prostaglandins. 148 When administered to GERD patients, antacids reduce esophageal acid exposure and are effective in providing symptom relief for both occasional heartburn and short-term treatment.145,149 Due to its poor absorption, aluminum hydroxide is more suitable for long-term treatment compared to PPIs. While PPIs are very effective, they are not without adverse effects, some of which are plausible and predictable while others are rare and idiosyncratic.19,150,151 Although much of the evidence linking PPI use to serious long-term conditions is weak, with very low odd ratios,89,152 the potential risks associated with this drug class cannot be ignored. In contrast, aluminum hydroxide, being short-acting and poorly absorbed, avoids the hypergastrinemia 153 and disruption of gut microbiota154,155 commonly seen with PPIs.
To leverage the physiological esophageal clearing mechanism of salivary secretion, 156 a melt-in-mouth tablet formulation was chosen. This type of tablet differs from other fast-dissolving forms, such as orodispersible tablets. While orodispersible tablets disintegrate into smaller particles upon contact with saliva, melt-in-mouth tablets dissolve or melt more smoothly into a liquid form.
This type of formulation offers several advantages157,158:
•
•
•
•
The potential of Esoxx Defence to restore impaired mucosal integrity in GERD was evaluated in 32 patients with EE greater than Los Angeles grade B. 159 Baseline mucosal impedance, a marker of mucosal integrity, was measured at the distal (3 cm), mid (9 cm), and proximal (15 cm) esophagus before and after 2 weeks of therapy with this medical device. Patients were randomized in a 1:1 ratio to receive either Esoxx Defence (six tablets per day, taken after each meal, mid-morning, mid-afternoon, and at bedtime) or an oral antacid or alginate (as needed) for 14 ± 2 days. Secondary outcomes included symptom relief, palatability, safety, and tolerability. While baseline impedance was similar between groups, after treatment significant improvements in distal, mid, and proximal esophageal impedance were observed only in the Esoxx Defence group. The medical device demonstrated favorable results in terms of symptom relief, palatability, safety, and tolerability. 159
In the same cohort of patients from the study, the authors conducted a thorough symptom evaluation using the Reflux Disease Questionnaire (RDQ) and assessed health-related quality of life (HRQL) through the GERD-HQRL questionnaire. Initially, mean RDQ and GERD-HQRL values were similar between the groups. However, after treatment, a significant reduction in scores was observed only in the patients treated with Esoxx Defence. 160 These findings suggest that 2 weeks of therapy with the medical device can effectively improve both GERD-related symptoms and quality of life in patients with severe esophagitis.
Alginate-containing formulations are among the most widely used over-the-counter treatments for GERD, typically providing quick and effective symptom relief.145,149 However, this is not always the case. To evaluate the effectiveness of Esoxx Defence in GERD patients who have a poor response to alginates, an open-label study was conducted with 40 patients, 22 of whom were on stable PPI therapy. 161 The primary endpoint was the reduction in weekly heartburn episodes, while secondary endpoints included GERD-related symptoms, patient satisfaction, and safety. The GERD Impact Scale (GIS) questionnaire was administered at baseline and again after 7 and 14 days of treatment. Results showed a significant (p < 0.0001) and progressive decrease in both the number of days with heartburn episodes and the GIS score compared to baseline, with improvements observed during both the first and second weeks of treatment. The reductions in heartburn episodes and GIS scores were similar in patients both on and off PPI therapy. The treatment was safe, well-tolerated, and received high patient satisfaction, with 46.2% rating it as “very good” and 43.6% as “good.” 161 These results suggest that Esoxx Defence can overcome the refractoriness to other GERD medications, often observed in clinical practice.
Extra-esophageal manifestations of GERD pose significant diagnostic and therapeutic challenges, often proving refractory to PPIs, even when combined with additional medications. A randomized trial was conducted to assess the effectiveness of Esoxx Defence in alleviating atypical GERD symptoms, such as hoarseness, cough, throat clearing, sore throat, voice changes, globus sensation, and postnasal drip. 162 Patients in the trial were given Esoxx Defence in addition to omeprazole (40 mg daily), or omeprazole alone for 6 weeks. After this period, responders to the combination therapy were randomized to either continue Esoxx Defence alone or receive no further medication. Results showed a progressive decrease in the RSI for both groups, with patients receiving the combination treatment showing a greater reduction (7.9 ± 7.0) compared to those on omeprazole alone (12.3 ± 8.9). The difference however did not reach statistical significance. Nevertheless, a statistically significant improvement was observed for certain individual RSI items in the Esoxx Defence plus omeprazole group. For patients who responded to combination therapy, the RSI score further decreased under continued treatment, while patients without any therapy did not experience any improvement. 162 These findings suggest that combining Esoxx Defence with acid suppression may offer a rational approach to managing extra-esophageal GERD symptoms. Despite the promising results, the study’s main limitation lies in its patient selection process, which was based solely on symptoms rather than objective measures of reflux and extra-esophageal manifestations. This, combined with the potential for a type II error (due to lack of statistical significance), underscores the need for a larger, more rigorous clinical trial to confirm these preliminary findings.
Burning mouth syndrome (BMS) is a condition characterized by a chronic, often unexplained sensation of burning or discomfort in the mouth, without visible oral abnormalities. The sensation can affect the tongue, lips, gums, or roof of the mouth. It’s considered a complex disorder with multiple possible etiologies, and it can significantly impact the quality of life. 163 Treatment of BMS is challenging due to its complex and often multifactorial nature, the primary goal being symptom relief. 164 BMS and GERD are distinct conditions, but they can be interconnected in certain cases. While there is no definitive causal link between the two, both research and clinical experience suggest that extra-esophageal manifestations of GERD, particularly LPR, may contribute to or exacerbate BMS symptoms.165,166 When stomach acid reaches the mouth, it can irritate the oral mucosa, leading to discomfort, a burning sensation, or dryness. This acid exposure may also affect the tongue and palate, causing a burning feeling, especially in individuals who are already susceptible to BMS. In some instances, the acidic exposure may disrupt the protective mechanisms of the oral tissues, increasing their sensitivity.165,166
In a study of 81 patients with BMS, 76 (93.8%) reported experiencing multiple pharyngeal reflux events during hypopharyngeal-esophageal pH-impedance monitoring. Thirty-two of these patients had both LPR and GERD. 165 In another large study involving 500 patients with established GERD, 11.2% identified BMS as their primary symptom, a prevalence comparable to that of chronic cough and pharyngitis. 167 Additionally, BMS was present in 124 patients with both typical and atypical GERD symptoms. Of these, 82 patients were treated for 3 months with Esoxx Defence (a tablet taken three times daily, after breakfast, after lunch, and at bedtime). Among them, 31.7% reported slight improvement, while 28% experienced almost complete remission of oral symptoms. 167 While these results may not appear particularly striking, they are still noteworthy, especially considering the difficulty in treating BMS, which is often refractory to most therapies.
Esoxx™ Protection
The clinical evidence presented above clearly shows that HS and CS-based formulations (with or without antacids) are highly effective in treating both erosive and non-erosive reflux disease. However, while Esoxx One and Esoxx Defence provide clinically significant symptom relief, they are not fully effective in addressing the extra-esophageal manifestations of GERD, including LPR and BMS.
The esophagus harbors a diverse microbial community, with Gram-positive bacteria (mainly Streptococcus) being more common in healthy individuals, while Gram-negative bacteria are more prevalent in those with GERD or Barrett’s esophagus.22,168,169 Gram-negative bacteria release lipopolysaccharides, which can activate the Tool-like receptors 4 (TLR4) and nuclear factor kappa B (NF-κB) pathways, triggering an increase in pro-inflammatory cytokine production. 168 In patients with acute reflux esophagitis, inflammation of the esophagus is predominantly characterized by T-lymphocytes, with papillary and basal cell hyperplasia occurring before surface cell erosion. 170 These observations suggest that reflux-induced inflammation may be driven by cytokines rather than by direct acid-induced chemical injury. 171 Additionally, refluxed acid and bile help stabilize hypoxia-inducible factor-2α (a key transcription factor that plays a critical role in cellular and systemic responses to hypoxia) in esophageal epithelial cells. This stabilization enhances the production of pro-inflammatory cytokines, attracting T-lymphocytes and other immune cells that contribute to esophageal damage. 171 These observations point to a potential link between esophageal dysbiosis and inflammation.
Oxidative stress refers to the imbalance between reactive oxygen species (ROS) and the body’s ability to neutralize them with antioxidants. In both GERD and LPR, the oxidative stress plays a central role in mucosal tissue damage to mucosal tissues and symptom development. 172 Preclinical studies have shown that exposure to gastric refluxate induces ROS production as a response to chemical insults. These ROS, including superoxide anions, hydrogen peroxide, and hydroxyl radicals, can damage cellular components such as lipids, proteins, and DNA, leading to cell death, inflammation, and tissue remodeling. 172 Under normal conditions, the body’s antioxidant enzymes (such as superoxide dismutase (SOD), glutathione peroxidase, and catalase) neutralize ROS. 173 However, chronic reflux exposure can impair this defense system, resulting in sustained oxidative damage. In children with reflux disease, the severity of EE has been shown to correlate with the intensity of oxidative stress, with a linear relationship between tissue SOD levels and the De Meester score. 174 In patients with LPR, low blood levels of antioxidant enzymes and elevated oxidative stress have also been reported. 175 Additionally, reduced levels of nitric oxide metabolites in exhaled breath concentrate of children with LPR further suggest an increased oxidative stress in the airways. 176
Given these pathophysiological insights, targeting both inflammation and oxidative stress mechanisms in digestive and extra-digestive GERD could offer promising new strategies for managing this chronic and challenging condition. While, anti-inflammatory drugs, whether steroidal or non-steroidal, are highly effective, they are systemic medications (not permitted as ingredients of medical devices 116 ) and came with significant adverse events. 177 To address inflammation and oxidative stress more safely, a new medical device, namely, Esoxx Protection, has been developed specifically for extra-esophageal manifestations of reflux disease and airway protection. This formulation incorporates Aloe vera and honey to further enhance therapeutic outcomes.
Aloe vera, a succulent plant widely known for its therapeutic properties, is often used in traditional medicine for various ailments. Its pharmacological properties are primarily attributed to its bioactive compounds, which include polysaccharides, glycoproteins, anthraquinones, saponins, and enzymes. Aloe vera displays a wide range of pharmacological effects, 178 many of which could be relevant in the treatment of GERD:
• Anti-inflammatory effect, mainly due to the polysaccharide acemannan.
• Antioxidant activity, attributed to its content in vitamin C, vitamin E, and beta-carotene.
• Mucosal protective thanks to the mucilage content present in the gel and to aloin (an anthraquinone compound) and barbaloin, which enhance mucosal integrity. These anthraquinone glycosides also display a laxative action provided they are given (and absorbed) in sufficient amount to stimulate bowel motility and reduce colonic water absorption.
• Demulcent properties (i.e., it forms a protective film) that may help soothe and protect the irritated lining of the esophagus.
• Wound-healing properties, due to stimulation of growth factor and collagen, which also might contribute to healing of esophageal damage.
• Antimicrobial (antibacterial, antifungal, and antiviral) activity.
• An acid buffering capacity has been also reported.
In a pilot, controlled trial, 79 patients with GERD symptoms referred for upper GI endoscopy were randomized to receive one of three treatments: Aloe vera syrup (standardized to 5.0 mg of polysaccharide per mL) at a dose of 10 mL per day, omeprazole (20 mg daily), or ranitidine (150 mg twice daily) for 4 weeks. The frequency of heartburn, regurgitation, belching, dysphagia, nausea, vomiting, and flatulence was assessed at 2 and 4 weeks. Aloe vera significantly reduced all symptoms except vomiting, with efficacy comparable to that of omeprazole and ranitidine. 179
Honey has been used for medicinal purposes for centuries, particularly in the treatment of gastrointestinal conditions. 180 It contains a range of beneficial compounds, including small amounts of proteins, enzymes, amino acids, minerals, trace elements, vitamins, aroma compounds, and polyphenols. 181 Current research suggests that darker honeys (such as buckwheat, heather, and honeydew) and multifloral varieties tend to have more pronounced therapeutic properties than lighter or unifloral honeys. The pharmacological properties of honey, including anti-inflammatory, antioxidant, antimicrobial, and soothing effects, make it an effective remedy for conditions like GERD and LPR. 182
Honey reduces intracellular ROS generation and helps restore intracellular glutathione levels. 183 It may also reduce inflammation by inhibiting the production of nitric oxide and prostaglandin E2. 184 Honey contributes to the management of GERD symptoms by coating the esophagus and stomach lining, thereby preventing the upward flow of food and gastric juice. Furthermore, honey may stimulate tissue regeneration in the lower esophageal sphincter, helping to reduce the likelihood of acid reflux. 185 Honey can be used alongside conventional therapies in treating reflux esophagitis. 186
Similar to Aloe vera, honey acts as a natural demulcent, forming a protective coating over mucous membranes. When consumed, honey coats the lining of the throat and esophagus, providing a soothing layer that helps alleviate the burning and irritation commonly associated with acid reflux. The thick, viscous texture of honey not only reduces discomfort in the throat but also helps alleviate irritation in the larynx and vocal cords, which is often seen in individuals with LPR. This protective action can also help calm a persistent cough and reduce hoarseness. 187 Additionally, honey is a natural humectant, meaning it attracts and retains moisture. For individuals with LPR, dry throat and irritated mucous membranes are common symptoms of reflux. Honey’s ability to hydrate the throat can offer relief from dryness and promote healing. 187
A recent, randomized, placebo-controlled trial investigated the effects of Manuka honey, which originates from New Zealand or Australia, in patients with GERD. Manuka honey is known for its high antioxidant capacity, attributed to its significant content of polyphenols, particularly flavonoids, phenolic acids, 179 and methylglyoxal, a compound found in trace amount in other honeys. 180 The study included 35 GERD patients, all of whom had heartburn or dyspeptic symptoms and were referred from General Practitioners. Participants were given Manuka honey (5 g three times daily) or placebo in addition to PPIs for a duration of 4 weeks. After 2 weeks of treatment, 86.7% of patients in the Manuka honey group experienced symptom improvement, compared to only 26.7% in the placebo group. At the 4-week, symptom improvement was seen in 100% of the Manuka group versus 40% in the placebo group. Additionally, 73.3% of those in the Manuka group showed a downgrading of mucosal lesions on endoscopy, while only 33.3% of placebo-treated patients showed similar improvements. 181 It is important to note that, due to the variability in composition among different types of honey, the results of this study may not be directly applicable to other honey varieties. Nonetheless, the findings are promising and highlight the potential of honey, either on its own or in combination with other active ingredients, to address some unmet needs in GERD treatment.
The film-forming effect of Esoxx Protection was evaluated and compared to Esoxx One using a 3D reconstructed human esophageal epithelium model, as described above. This was done by measuring the kinetics of transepithelial caffeine passage. 119 Both devices showed a significant reduction in caffeine permeation at 1 and 2 h, compared to the negative control. After 1 h, Esoxx Protection demonstrated a significantly lower caffeine permeation rate (0.19% caffeine/min) than Esoxx One (0.30% caffeine/min, p < 0.0001). By 2 h, however, the permeation rates between the two devices were comparable. 119 The LY flux data at 15 min supported the caffeine permeation results. While Esoxx Protection (11.0%, p < 0.05) significantly reduced LY flux compared to the untreated control (15.8%), Esoxx One (14.9%, NS) did not show a significant reduction. 119 These findings suggest that Esoxx Protection reduces intercellular gaps or strengthens cell–cell junctions.
Demonstrating the potential synergy of combining Aloe vera and honey with HA and CS is challenging due to the unique properties of these two natural remedies. However, a clinical study in patients with LPR is currently underway to explore the therapeutic advantages of this combination (Esoxx Protection).
Discussion
GERD continues to be a challenging and difficult-to-treat condition even in the third millennium. Current pharmacologic treatments offer limited solutions. Reflux inhibitors, while promising, remain largely ineffective, and effective prokinetics are still lacking. Although antidepressants may benefit select patients, they are associated with adverse events in up to 32% of cases.11–14 As a consequence, antisecretory drugs (H2RAs and PPIs) remain the cornerstone of medical management for GERD. These drugs work indirectly by reducing the volume and concentration of gastric secretions available for reflux, thereby diminishing the harmful effects of the refluxed material.15,16 The clinical efficacy of PPIs has been well-established in numerous studies, with their superiority over H2RAs firmly demonstrated. 18
As previously discussed, PPI-refractory reflux disease—both erosive and non-erosive—does exist.24,25,188–190 Current pharmacologic options for managing PPI-refractory GERD are also limited. These include switching to a PPI with less dependence on the CYP2C19 enzyme, adding an H2RA at night, using alginate-based formulations, or, as a last resort, trialing a gamma-aminobutyric acid agonist like baclofen. In cases where hypersensitivity or anxiety is suspected, neuromodulation with antidepressants may be beneficial for PPI non-responders.25,28,188 Although research does not strongly support the routine use of prokinetic therapies for GERD, their addition could be worthwhile in patients with concurrent motility-related symptoms or established gastroparesis. 131
Recent experimental and clinical studies have revealed that—in patients with GERD—esophageal mucosal integrity is impaired and that this feature represents a hallmark of the disease.191,192 This understanding of GERD pathophysiology has led to the development of a new therapeutic approach: esophageal mucosal protection. While drugs with mucosal protective activities, such as sucralfate and irsogladine (the latter not marketed in Europe) have long been available, the current formulations were not suitable for esophageal protection. 8 The HA and CS-based medical devices have been advanced in the field since they allow the active ingredients to adhere to the esophageal mucosa, effectively coating the esophageal lining and providing a sufficient contact time with the esophageal mucosa.
A thorough review of the literature shows that the efficacy of PPIs in treating extra-digestive GERD is less consistent compared to their effectiveness in patients with typical symptoms. 3 In extra-digestive GERD, the complexity of patient presentations is often matched by the challenge of diagnosing reflux as the underlying cause of their symptoms. Diagnostic tools such as upper GI endoscopy and pH-impedance monitoring are limited by poor sensitivity, while laryngoscopy suffers from poor specificity in identifying reflux in this patient population. 193 An empirical trial of PPIs may be an appropriate initial approach for both diagnosing and managing potential extra-digestive symptoms linked to reflux. However, symptom resolution often requires higher PPI doses and longer treatment durations than those typically used for conventional GERD. 194 It is important to note that while PPI therapy and twice-daily dosing for extra-digestive GERD are not officially approved indications, they are nonetheless recommended by both gastroenterology195,196 and other specialty guidelines.197–199
Several drugs, including prokinetics200–202 and N-acetylcysteine, 203 have been tested as add-on treatments, but their results have been inconsistent. In contrast, mucosal protectants appear to be a more promising option for improving treatment efficacy in these patients.
The Esoxx One and Esoxx Defence medical devices are especially suitable for patients unresponsive to PPI therapy or for those with extra-esophageal manifestations of reflux disease. In addition to being used in combination with PPIs, these devices can be used alone in mild cases of GERD or after discontinuing acid suppression with the hope to maintain remission and prevent recurrence. The Esoxx Protection, including Aloe vera and honey, appears more suitable for patients with oral and/or ENT manifestations of GERD, such as cough, throat clearing, hoarseness, and sore throat (Figure 3).

Medical devices with esophageal mucosal protective activity. Formulations and suggested therapeutic use in patients with gastroesophageal reflux disease.
Conclusion
A personalized treatment strategy tailored to each patient’s specific phenotype is essential for effective GERD management. While PPIs remain the cornerstone of therapy, combining acid suppression with mucosal protection—especially in PPI-resistant cases—emerges as a promising approach. The use of HA- and CS-based medical devices, each with its unique composition and therapeutic activities, holds the potential for more effective management of both typical GERD symptoms and extra-esophageal manifestations. This comprehensive approach could significantly improve treatment outcomes and enhance patients’ overall quality of life.
