Abstract
Background:
The Operative Link on Gastritis Assessment (OLGA) and Operative Link on Gastric Intestinal Metaplasia Assessment (OLGIM) are established classification systems used to evaluate atrophic gastritis and intestinal metaplasia, respectively.
Objectives:
We evaluated the association of OLGA and OLGIM scores and the risk of gastric cancer (GC) in only prospective cohort studies.
Design:
Systematic review and meta-analysis.
Data sources and methods:
We systematically searched four databases for prospective cohorts that evaluated the use of OLGA and OLGIM staging systems in predicting the risk of GC. We primarily compared OLGA/OLGIM III–IV versus OLGA/OLGIM 0–II categories and GC events. Pooled risk ratios (RR) and absolute risk differences with their 95% confidence intervals (CIs) were calculated.
Results:
Eight studies were included (n = 12,526). The mean age of the patients ranged from 48.2 to 64.9 years. OLGA III–IV and OLGIM III–IV were associated with the development of GC in comparison to their 0–II categories (RR 32.31, 95% CI 9.14–114.21 and RR 12.38, 95% CI 5.75–26.65, respectively). OLGA III–IV and OLGIM III–IV were associated with an increase in the absolute risk of GC of 4% and 5%, respectively. The risk remained significant if we only included countries with high incidence of GC, and was greater if we excluded one study that included mostly patients with autoimmune gastritis. OLGA II and OLGIM II were associated with higher risk of high-grade dysplasia (HGD) and GC in comparison with OLGA 0–I and OLGIM 0–I, respectively.
Conclusion:
Higher stages in OLGA and OLGIM systems are associated with a significantly increased risk of developing HGD and GC, validating these scoring systems for the assessment of GC risk and the design of endoscopic surveillance programs.
Trial PROSPERO registration:
CRD42024565771.
Introduction
Despite declining trends in incidence and mortality, gastric cancer (GC) remains one of the most common cancers worldwide.1,2 The prognosis of patients with GC mostly depends on the stage of diagnosis, with better survival at early stages. 3 However, in Western countries most of the cases are diagnosed in advanced stages, which leads to a poor prognosis.4,5
To improve the early detection of GC, strategies that include screening and endoscopic surveillance should be implemented, mainly in the highest-risk population. The Operative Link on Gastritis Assessment (OLGA) system and the Operative Link on Gastric Intestinal Metaplasia (OLGIM) system were developed for this purpose: to evaluate and stratify the severity and distribution of chronic atrophic gastritis (CAG) with or without intestinal metaplasia (IM), identifying patients at higher risk of GC.6,7
A previous systematic review and meta-analysis, that included mostly case-control studies, described that there is an association between the stages III/IV of the OLGA and OLGIM system with an increased risk of GC. 8 For this reason, the European Society for Gastrointestinal Endoscopy (ESGE) and the American Gastroenterological Association (AGA) recommended that these high-risk individuals should be surveilled with a high-quality endoscopy.9,10 However, newly published evidence suggests that not only patients with OLGA or OLGIM advanced stages have a higher risk of GC. Lee et al. 11 found that patients with OLGIM II also had an increased risk of developing dysplasia and CG, so these patients could also benefit from endoscopic surveillance.
In recent years, new prospective cohorts have been published in which the usefulness of the OLGA and OLGIM systems has been evaluated, making it necessary to update a systematic review and meta-analysis prioritizing the inclusion of only prospective cohort studies. Prospective cohort studies allow for more accurate and systematic follow-up, ensuring that the progression of gastric lesions is documented longitudinally, thus providing stronger evidence for the predictive validity of these staging systems and avoiding selection bias and retrospective misclassification. The aim of this review was to analyze data from prospective cohorts to quantify the association between advanced stages of OLGA and OLGIM scores with the risk of developing GC. Furthermore, we look for clarifying the risk of OLGA and OLGIM stage II in the development of GC.
Materials and methods
This systematic review was reported following the 2020 PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) statement 12 and was registered on the PROSPERO database (CRD42024565771).
Search strategy
We searched the following four electronic databases: PubMed, Embase, Web of Science, and Scopus; from inception to January 21, 2025. We used the following keywords: “Stomach Neoplasms,” “operative link on gastritis assessment,” and “operative link on gastritis intestinal metaplasia assessment.” The search strategies are available in the Supplemental Material. There were no restrictions on language or publication date. After the electronic searches were complete, manual searching was performed. Titles that seemed relevant were identified by hand. In addition, citation searching was performed using the citation search function in Google Scholar. 13
Eligibility criteria
We included prospective cohorts that enrolled adult patients (⩾18 years) without previous GC undergoing a baseline endoscopy with stomach mapping biopsies who were categorized according to the OLGA and OLGIM systems and then followed by endoscopy. Studies were excluded if they were abstracts, case-control, or cross-sectional designs; included patients with previous GC; followed only high-risk patients of developing GC; and were similar to others or duplicated.
We also excluded studies that recruited only patients with restricted atrophic gastritis or autoimmune gastritis (AIG). OLGA and OLGIM systems were developed for assessing GC risk in patients with CAG, in which both the antrum and the corpus mucosa are involved. 14 In contrast, in AIG, the antrum mucosa is spared 15 ; therefore, OLGA and OLGIM systems would not allow us to accurately assess the true risk of cancer development in these patients.
Study selection
Articles were downloaded from electronic search to EndNote X8 software. After removing duplicate records, selected studies were uploaded to Rayyan QCRI (https://rayyan.qcri.org/). Two authors screened the studies according to the inclusion and exclusion criteria (H.B.-G. and D.C.-H.). Disagreements were resolved by a third researcher (A.V.H.).
Outcomes
The primary outcome was GC at any stage. Secondary outcome was high-grade dysplasia (HGD). Also, we explored the association of OLGA/OLGIM II versus 0–I stages with the development of the composite secondary outcome of HGD and GC.
Data extraction
Two researchers (H.B.-G. and D.C.-H.) extracted data independently on a previously designed Microsoft Excel® spreadsheet. The following data were extracted: author name, year and country of publication, sample size, history of first-degree familiarity of GC, Helicobacter pylori (Hp) infection, smoking history, presence of AIG, OLGA/OLGIM stage, and outcomes.
Risk of bias assessment
To evaluate risk of bias on cohort studies we employed the Newcastle-Ottawa Scale (NOS) tool. 16 Studies were divided in three categories: low risk of bias (8–9 points), moderate risk of bias (5–7 points), and high risk of bias (0–4 points).
Statistical analysis
All meta-analyses were conducted using inverse variance random-effects models. The Paule Mandel method was used to calculate the between-study variance tau.2,17 To account for studies where zero events occurred in one or both exposure groups, we applied the treatment-arm continuity correction method, in which a small constant (0.5) was added to all cells of the contingency table. This approach prevents computational issues when calculating relative risks and maintains the stability of effect estimates while minimizing bias. 18 All outcomes were pooled using risk ratio (RR) with their 95% confidence intervals (CIs). Heterogeneity was assessed by using the Chi-square test (threshold p < 0.10) and I2 statistic. The amount of statistical heterogeneity was defined as low if I2 < 30%, moderate if I2 is 30%–60%, and high if I2 > 60%. Sensitivity analyses were performed excluding the cohorts that included mostly patients with AIG, including only cohorts from a high incidence of GC, and computing absolute risk differences (RD) instead of RR. We performed all meta-analyses using the meta package from R 3.6.3 (http://www.r-project.org/). A two-tailed p < 0.05 was considered statistically significant.
Results
Study selection
A total of 919 records were retrieved from electronic searches. After the removal of 422 duplicates, 497 studies underwent title/abstract and full-text screening. After the screening of studies by title/abstract, 477 articles were excluded. After the full-text assessment of 20 articles, 13 articles were excluded. Two records were also retrieved from manual searching. One record was excluded after full-text inspection because there was no outcome measure of interest. Finally, we included eight studies for analysis, including seven from electronic searches11,19–24 and one from a manual search 25 (Figure 1).
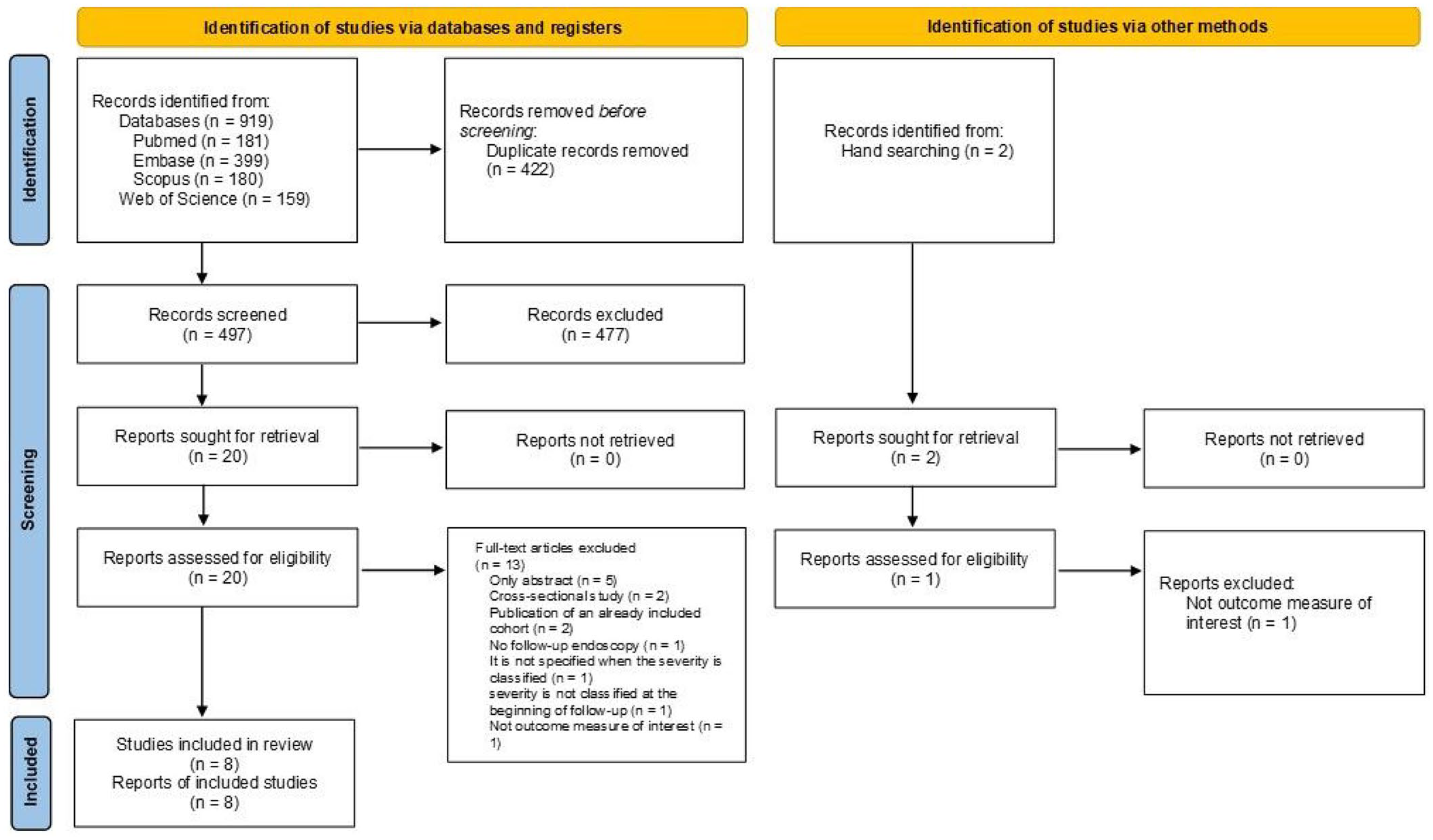
Flow diagram of study selection.
Study characteristics
The main characteristics of the eight studies are summarized in Table 1. A total of 12,526 patients were included (sample sizes ranged from 79 to 7436). The mean age of the patients ranged from 48.2 to 64.9 years, and 46% were male. Seven studies were written in English11,19–23,25 and one in Chinese. 24 Four studies reported information about GC in relatives,11,22–24 five studies reported the status of Hp infection,11,19,21–23 and three studies reported a history of tobacco use.11,22,23 In two cohorts, patients with AIG were included along with patients with CAG22,23; representing 1.6% of the total number of patients (206 patients). The follow-up time ranged from 3 to 10 years. The majority of GC cases were diagnosed in stages I and II.
Main characteristics of the included cohort studies.

Baseline infection by Hp.
Two cases of GC were diffuse adenocarcinoma.
Previous infection of Hp.
Two cases detected at baseline.
One case detected at baseline and one case detected within 12 months from the index endoscopy.
One case of diffuse adenocarcinoma in a patient without intestinal metaplasia.
Active or former smoker.
GC, gastric cancer; HGD, high-grade dysplasia; Hp, Helicobacter pylori; LGD, low-grade dysplasia; NA, not available; OLGA, Operative Link on Gastritis Assessment; OLGIM, Operative Link on Gastric Intestinal Metaplasia Assessment.
Risk of bias assessment
According to the NOS assessment, all studies had a moderate risk of bias due to the lack of adjustment for confounders (Table S1). None of the studies was scored as high-risk of bias.
OLGA and OLGIM score system and the risk of GC
Five studies evaluated the risk of GC using the OLGA staging system (8978 patients).19,22–25 During follow-up, 30 GC were diagnosed (incidence of 0.3%) with a mean follow-up of 4.4 years. OLGA III–IV stages were associated with the development of GC contrasted with OLGA 0–II categories (RR 32.31, 95% CI 9.14–114.21), with low heterogeneity (I2 = 23%; Figure 2(a)). When we compared the pooled risk of GC between OLGA II versus 0–I, the effect was not significantly different (RR 3.21, 95% CI 0.47–21.75; Figure 2(b)).

OLGA stages and risk of HGD and GC. (a) Forest plot for studies assessing the association of OLGA stages with the risk of GC. (b) Forest plot for studies assessing the association of OLGA II versus 0–I stages with the risk of GC. (c) Forest plot for studies assessing the association of OLGA stages with the risk of HGD and GC. (d) Forest plot for studies assessing the association of OLGA II versus 0–I stages with the risk HGD and GC.
Six studies evaluated the OLGIM staging system for predicting GC (4393 patients).11,20–23,25 During follow-up, 34 GC were diagnosed (incidence of 0.8%) with a mean follow-up of 4.0 years. OLGIM III–IV stages were associated with the development of GC in comparison to their 0–II categories (RR 12.38, 95% CI 5.75–26.65), with low heterogeneity (I2 = 0%; Figure 3(a)). OLGIM II was associated with an increased risk of GC contrasted with OLGIM 0–I (RR 9.99, 95% CI 3.39–29.42; Figure 3(b)).

OLGIM stages and risk of GC. (a) Forest plot for studies assessing the association of OLGIM stages with the risk of GC. (b) Forest plot for studies assessing the association of OLGIM II versus 0–I stages with the risk of GC. (c) Forest plot for studies assessing the association of OLGIM stages with the risk of HGD and GC. (d) Forest plot for studies assessing the association of OLGIM II versus 0–I stages with the risk of HGD and GC.
OLGA and OLGIM score system and the risk of HGD and GC
Patients with OLGA and OLGIM stages III–IV also had a significantly higher risk of developing the composite secondary outcome of HGD and GC (RR 26.85, 95% CI 4.99–144.60 and RR 11.06, 95% CI 6.33–19.29, respectively; Figures 2(c) and 3(c)). Furthermore, OLGA and OLGIM stage II were associated with higher risk of HGD and GC compared with OLGA and OLGIM stages 0–I (RR 12.76, 95% CI 3.73–43.60 and RR 8.15, 95% CI 3.52–18.87), respectively (Figures 2(d) and 3(d)).
Sensitivity analyses
When we pooled data of cohorts from countries with high incidences of GC the effect remained high in OLGA and OLGIM stages III–IV stages (RR 27.14, 95% CI 7.77–94.85, and RR 14.75, 95% CI 5.45–39.89, respectively; Figures S1 and S2). Computing RD did not change the magnitude of the results. OLGA stages III–IV are associated with an increase in the absolute risk of GC of 4% (Figure S3); and OLGIM stages III–IV with an increase in the absolute risk of GC of 5% (Figure S4), contrasted with OLGA and OLGIM stages 0–II, respectively. The exclusion of one study that included mostly patients with AIG decreased the heterogeneity when evaluating the predictive capacity of the OLGA system, but aggregated results remained similar (Figures S5 and S6).
Discussion
Main findings
Our systematic review and meta-analysis, including only prospective cohorts, revealed that OLGA and OLGIM III–IV stages are strongly associated with an increased risk of developing GC. Moreover, a higher risk of HGD and GC was found in patients with OLGIM II, compared to OLGIM 0–I. None of the studies were categorized as having a high risk of bias according to the NOS assessment.
Although a significant increase in the risk of GC was not found in patients in the OLGA II stage when compared with OLGA 0–I, a significant increase in the secondary composite outcome of HGD and GC was evident. Including only studies from countries with a high incidence of GC, the effect was the same. By excluding the study by Esposito et al., in which they mainly recruited patients with AIG, the risk of developing GC was higher in OLGA and OLGIM stages III–IV. Patients with OLGA and OLGIM III–IV stages also had a significantly increased risk of developing the composite outcome of HGD and GC.
What is known in the literature about the research question?
Hp infection is the main risk factor for developing GC. 26 In more than 90% of GC cases, there is a well-established causal relationship with chronic Hp infection. 27 A multistep carcinogenic process driven by chronic inflammation related to Hp infection can lead to CAG with or without IM. Several previous studies have shown a higher incidence of GC in patients with CAG and IM.28,29 The risk of progression of CAG to GC ranges from 0.1% to 0.3%, depending on severity and extension.20,30
Thiruvengadam et al. 31 found using a microsimulation model that universal 5-year endoscopic surveillance of IM reduced the incidence and mortality of GC. However, the possibility of having tools to identify patients with a higher risk of GC could facilitate the implementation of strategies that allow the early diagnosis of this neoplasia, which would lead to improving the prognosis in these patients.
OLGA and OLGIM classifications are histological systems proposed to evaluate the risk of GC according to the extension and severity of atrophic or intestinal metaplastic changes.7,14 A previously published systematic review and meta-analysis, which primarily included case-control studies, reported an association between advanced stages (III/IV) of the OLGA and OLGIM staging system and an increased risk of GC. 8 Only two cohort studies were included in this review, and most of them had small samples.
Den Hollander et al. 20 described that the proportion of patients who presented neoplastic progression in a low-risk population was similar between OLGIM 0–II and OLGIM III–IV. In contrast, in our meta-analysis of pooled data, a significant risk of GC was found in the group of patients with OLGA and OLGIM III–IV stages. Den Hollander et al. suggested that the use of a combination of histological methods and pepsinogen ratio could be of additional value in identifying the population at highest risk of GC development. A low pepsinogen I serum level, a low pepsinogen I/II ratio, or both, are proposed indicators of CAG and IM,32,33 which are defined as precancerous lesions for GC. However, in a previous systematic review on the significance of pepsinogen as a biomarker of GC, it only exhibited a moderate diagnostic value for identifying GC 34 and lacks of proper validation in Western countries.
It is essential to determine an efficient surveillance endoscopy interval based on the risk of cancer development while simultaneously reducing the number of endoscopy-related adverse effects and costs. The ESGE and the AGA recommend that patients with OLGA and OLGIM III–IV stages should be followed up with a high-quality endoscopy every 3 years.9,10 Nevertheless, optimal surveillance interval has not yet been determined, and additional risk factors should be considered such as quality of baseline endoscopy, family history of GC, presence of incomplete IM, persistent Hp infection, or smoking history.9,10 In these specific cases, a shorter follow-up interval could be beneficial. Furthermore, a recent retrospective study from 15 centers in Japan described that current Hp infection and a surveillance interval of >1.5 years were independent risk factors for detecting pT1b or deeper GC. 35
Although the risk of progression to GC is greater in OLGA and OLGIM III–IV stage, it is suggested that intermediate stages (OLGA and OLGIM II) may also be at significant risk, especially if there is incomplete metaplasia and/or family history of cancer. 9 Lee et al. 11 showed that OLGIM II also carries a significant risk of dysplasia and CG in follow-up. The median time to appearance of HGD and GC in the OLGIM III–IV group was 22.7 months (range 12.7–44.8 months) and in the OLGIM II group, it was 50.7 months (range 28.4–73.3). Although in this group of patients, the progression time was longer than the more advanced stages, they suggested that patients with OLGIM II should be included in the endoscopic follow-up, but not as exhaustively as that proposed in high-risk patients. Latorre et al. 23 found similar findings, with a median time for the appearance of HGD and GC of 35 months (range 18–37 months) in OLGIM III–IV and 70.5 months (range 67–74 months) in OLGIM II. Likewise, when evaluating the time of progression using the OLGA score system, he found a median time for the appearance of HGD and GC of 33 months (range 18–38 months) in OLGA III–IV and 70.5 months (range 67–74 months) in OLGA II. 23
In a retrospective follow-up study, Wu et al. 36 also challenged the notion of OLGIM II as a low-risk stage for GC, particularly in patients with persistent Hp infection. They found that among eight patients who progressed to GC, three had been classified as OLGIM II and had experienced failed Hp eradication, suggesting that bacterial persistence may accelerate disease progression. A recent case-control study, including 196 early GC patients and 196 matched controls, found that OLGA and OLGIM stage II are more prevalent among cases than controls. 37 Notably, OLGIM II was associated with a more than fourfold increased risk of early GC (odds ratio (OR) 4.11; 95% CI 2.51–6.74) compared to OLGIM 0–I. Regarding Hp status, the study found that past infection (OR 1.87; 95% CI 1.16–3.02) posed a greater risk for early GC than current infection, suggesting that the long-term effects of Hp-induced damage persist even after bacterial clearance. Moreover, in a previous case-control study, Marcos et al., 38 found that OLGIM stage I–II was associated with more risk of dysplasia and early GC (despite in lower risk than OLGIM III–IV), and OLGA stage I–II was not.
In our review, a higher risk of GC was found in patients with OLGIM stage II, compared to those in OLGIM stage 0–I. An increased risk of cancer was found in patients classified as OLGA II compared to OLGA 0–I, but without reaching a significant difference. However, an increased risk of HGD and GC was found within OLGA stage II. In a previous study, Rugge et al. 39 found two cases of cancer in patients classified as OLGIM stage II, both coexisting with OLGA stage III gastritis. They proposed that by focusing on IM alone, OLGIM could be less sensitive in identifying high-risk patients, resulting in the down-staging of patients who should be offered follow-up. Rugge et al. 39 proposed that OLGIM staging system may underestimate the true risk in some patients with atrophy but without IM. Meanwhile, OLGIM system is characterized by higher interpathologist agreement and has proven to be simpler when compared with OLGA.7,40 Den Hollander et al. 20 found that when using the OLGA system, the four patients who developed HGD/GC had a stage 0–I on the initial endoscopic examination, so they would not have been included in the follow-up. Therefore, considering the performance of each of these classifications, the use of both classifications should be recommended to more accurately characterize the risk of GC.
Although the majority of patients in the initial stages of the OLGA and OGIM systems will not benefit from endoscopic surveillance, it is important to highlight that there may be progression or regression of the stages.11,21,23 While it is not clearly known what factors may determine the change in stage, some variables such as family history of GC and smoking have been proposed as factors that increase the risk of progression. 41 Lee et al. 11 found that the pepsinogen ratio could contribute as a significant predictor of progression from OLGIM I to OLGIM II–IV. Furthermore, cases of incomplete IM, despite being limited to the antrum, have also been found to be related to a higher risk of GC. 21
AIG is a chronic inflammatory condition that results in the loss of the parietal cell mass, resulting in the appearance of atrophy and metaplasia. There is some evidence suggesting that AIG patients, particularly those with extensive atrophy, are at increased risk for developing GC.42–44 Observational studies showed that the risk of GC is higher within the first year of diagnosis; therefore, some societies recommend an upper endoscopy after making a diagnosis of AIG.9,10 However, Rugge et al. 45 found that AIG, in the absence of Hp infection, does not appear to be associated with an increased risk of developing GC. Jeong et al. 46 suggested that patients with AIG exhibit a more benign metaplastic phenotype and lack the proliferative metaplastic lineages associated with an increased risk of adenocarcinoma. Still, AIG patients have an increased risk of progression of neuroendocrine proliferations.45,46
Esposito et al. 22 found a low performance of the OLGA/OLGIM systems in predicting cancer development. This finding is probably because most of their cohort were patients with AIG. OLGA and OLGIM systems were developed for assessing GC risk in patients with CAG, in which both the antrum and the corpus mucosa are involved. In contrast, AIG is characterized by corpus-dominant advanced atrophy, which may not be fully reflected in the OLGA/OLGIM system, as it was designed to assess the topographical distribution of atrophy and is not optimized for conditions predominantly affecting the oxyntic mucosa. 15 Although our analysis included a study primarily composed of patients with AIG, 22 the overall findings consistently demonstrated that higher OLGA/OLGIM stages were associated with an increased risk of GC. This is likely due to the relatively small sample size of the study by Esposito et al. compared to the larger cohorts included in the analysis. Moreover, when excluding this study, the risk of GC development was higher in OLGA and OLGIM III–IV stages.
Jun et al. 47 point out that performing at least one endoscopy could reduce the risk of death from GC and that the greater the number of endoscopies in one’s life, the greater this effect. Furthermore, Yasuda et al. 35 found that a shorter follow-up time interval (<1.5 years) is associated with a greater possibility of diagnosing pT1a GC and a better prognosis. In our review, the majority of GC were found in stages I and II, being managed endoscopically or surgically. Probably, if these patients diagnosed with GC had not been included in a follow-up cohort, it would not have been possible to find them in the initial stages, so we could suggest that the implementation of an endoscopic surveillance program gives us the possibility of diagnosing a gastric neoplasia in early stages.
What our study adds to the literature
To the best of our knowledge, no meta-analysis has exclusively focused on evaluating the risk of GC development only including prospective cohort studies. Unlike case-control studies, which are prone to selection bias and retrospective data collection limitations, prospective cohort studies provide stronger evidence by ensuring that OLGA/OLGIM staging was assessed before GC development. This design enhances the validity of the temporal relationship between precancerous staging and cancer risk, making our findings more robust for clinical application. We found that there is a greater risk of developing HGD and GC in patients with advanced stages of OLGA and OLGIM systems. Even in the intermediate stages of OLGA and OLGIM systems (OLGA and OLGIM II), an increased risk of developing HGD and GC can be found. These findings should help us establish closer follow-up in patients selected as being at high risk of developing GC. The implementation of OLGA and OLGIM systems in healthcare will permit prioritizing high-risk populations, allowing for targeted surveillance and early intervention.
Limitations
This review has some limitations to consider. First, all included studies were rated as having a moderate risk of bias. Therefore, our results should be interpreted with caution. This limitation highlights the need for future high-quality, large-scale prospective cohort studies that incorporate comprehensive confounder adjustment to strengthen the evidence on the predictive value of the OLGA and OLGIM staging systems for GC risk assessment. Second, some of the studies included patients diagnosed with AIG. However, the small number of patients included with this diagnosis limited its impact on the overall effect of the OLGA/OLGIM stages. Moreover, when doing a sensitivity analysis and removing the study made up mostly of patients with AIG, the risk of developing GC was higher in the advanced stages of OLGA and OLGIM. Third, the Hp infection status was not considered as a risk factor for developing GC. However, a recent study suggested that there are likely host genetic or epigenetic factors related to the progression to GC beyond Hp infection. 48 Future prospective cohorts that incorporate detailed Hp status assessments at baseline and during follow-up are suggested, including eradication status and reinfection rates. This approach would help clarify the role of Hp in modulating the progression of gastric precancerous lesions within the OLGA and OLGIM staging frameworks. Fourth, subtype analysis of IM was not considered. A previous study pointed to the presence of incomplete IM as an additional risk factor for GC. 49 Only two studies provided information on the subtype of metaplasia, so an analysis of this variable as a risk factor could not be performed. Fifth, although we conducted a systematic search, there may be unpublished studies that could be overlooked.
Conclusion
Our systematic review and meta-analysis provide a comprehensive assessment of the prognostic value of the OLGA and OLGIM staging systems in predicting the risk of GC and its precursors. Our findings indicate that higher stages in both OLGA and OLGIM systems are significantly associated with an increased risk of developing GC. Also highlights the substantial risk associated with intermediate stages (II) in both OLGA and OLGIM classifications, which, although lower than stages III–IV, still demonstrate a significant likelihood of progression to more severe conditions. The OLGA and OLGIM systems are probably not suitable for assessing GC risk in patients with AIG.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251325461 – Supplemental material for OLGA and OLGIM staging systems on the risk assessment of gastric cancer: a systematic review and meta-analysis of prospective cohorts
Supplemental material, sj-docx-1-tag-10.1177_17562848251325461 for OLGA and OLGIM staging systems on the risk assessment of gastric cancer: a systematic review and meta-analysis of prospective cohorts by Harold Benites-Goñi, Dacio Cabrera-Hinojosa, Gonzalo Latorre, Adrian V. Hernandez, Hugo Uchima and Arnoldo Riquelme in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848251325461 – Supplemental material for OLGA and OLGIM staging systems on the risk assessment of gastric cancer: a systematic review and meta-analysis of prospective cohorts
Supplemental material, sj-docx-2-tag-10.1177_17562848251325461 for OLGA and OLGIM staging systems on the risk assessment of gastric cancer: a systematic review and meta-analysis of prospective cohorts by Harold Benites-Goñi, Dacio Cabrera-Hinojosa, Gonzalo Latorre, Adrian V. Hernandez, Hugo Uchima and Arnoldo Riquelme in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
