Abstract
Background:
Efficacy of eradication regimens in Helicobacter pylori (Hp) infection is commonly reported with proton pump inhibitors (PPIs). In patients with corpus atrophic gastritis, characterized by impaired acid secretion, PPI treatment is questionable.
Objectives:
The current study aimed to assess in clinical practice the tolerability and eradication rate of modified eradication regimens without PPI as first-line treatment in patients with histologically Hp-positive corpus atrophic gastritis.
Design:
Real-life longitudinal observational study.
Methods:
Overall, 76 patients (77.6% females, age 58.5 (26–88) years) with histologically Hp-positive corpus atrophic gastritis were consecutively diagnosed (2001–2022). First-line eradication treatment was prescribed without PPIs: concomitant or sequential amoxicillin-based therapy (ABT) until 2016 (n = 30), then single-pill bismuth treatment (SPBT; n = 46). Treatment adherence and adverse events were clinically evaluated and treatment efficacy was assessed by histopathology (updated Sydney system) at 6 ± 3 months after treatment.
Results:
Only mild adverse events not requiring medical treatment were observed in four patients treated with SPBT without PPIs (vomiting, self-limiting diarrhoea, nausea, abdominal discomfort) and in two treated with ABT without PPIs (vomiting and abdominal discomfort). Overall, 71/76 (93.4%) corpus atrophic gastritis patients completed the treatment: 43/46 (93.5%) SPBT without PPIs and 28/30 (93.3%) ABT without PPIs. Successful cure of Hp was observed in 64/71 patients: overall eradication rate 90.1%, 95%CI 69.4%–115.1%. 42/43 corpus atrophic gastritis patients treated with SPBT without PPIs were successfully cured against 22/28 of those treated with ABT without PPIs. The eradication rate was higher for SPBT than ABT: 97.7%, 95%CI 70.4%–132.0% vs 78.6%, 95%CI 49.2%–118.9%, p = 0.013.
Conclusion:
In clinical practice, Hp cure can be achieved without PPIs as first-line treatment in about 90% of patients with corpus atrophic gastritis.
Plain language summary
The common treatment against Helicobacter pylori (Hp) infection, the bug causing gastritis, peptic ulcer, and gastric cancer, is a combination of antibiotics and drugs blocking gastric acid secretion, called proton pump inhibitors (PPIs). Corpus atrophic gastritis, is a particular type of gastritis at increased risk for gastric cancer, in which the gland producing gastric acid are damaged and therefore the use of PPIs does not make sense. It is unknown, whether treatment against Hp using only antibiotics without PPIs works in the same manner. The current research aimed to understand in clinical practice whether in patients with corpus atrophic gastritis and Hp infection, never treated before, the treatment against Hp with antibiotics but without using PPIs, is well tolerated and successful to eradicate the bacterium. This study involved 76 patients with histological Hp infection and corpus atrophic gastritis (ascertained by gastroscopy and biopsies): 30 patients were treated with amoxillin together with another antibiotic, and 46 patients with a single-pill bismuth treatment (SPBT) available since 2016. All treatments were prescribed without using PPIs. At 6 ± 3 months after treatment, adverse events and treatment efficacy, i.e. whether Hp disappeared on gastric biopsies taken during gastroscopy, was checked. What this study found was that adverse events occurred in only 6 patients, all mild and not requiring treatment. Of the 76 patients, 71 completed the treatment. The eradication of the bacterium was yielded in 64 (90.1%) patients. The SPBT worked better compared to the amoxicillin treatment as more patients in the first were successfully cured from the infection: 97.7% against 78.6%. This research shows that in clinical practice in patients with corpus atrophic gastritis, the cure of Hp infection can be achieved without.
Keywords
Introduction
Helicobacter pylori infection is the main cause of chronic gastritis and gastric cancer; around 90% of gastric cancer cases can be attributed to the infection.1,2 H. pylori eradication should be considered as the main therapeutic strategy to reduce the progression towards gastric precancerous conditions, such as atrophy, intestinal metaplasia and cancerous lesions.1–3 Proposed treatment regimens are mainly bismuth based on Western guidelines and less commonly sequential or hybrid regimens.2,4–6 Randomized clinical trials showed that classical bismuth quadruple therapies combining bismuth with a proton pump inhibitor (PPI), metronidazole and tetracycline are more effective than triple therapies.7–9 Interest in this therapy grew and the difficulties related to the multi-pill administration have been elegantly overcome with the commercialization of a single-pill-bismuth-based therapy (SPBT) that showed high efficacy in clinical trials and clinical practice.2,9 However, the results of these trials were mainly obtained from patients with chronic superficial, non-atrophic H. pylori gastritis, not considering the peculiar condition of corpus atrophic gastritis. This is a chronic inflammatory condition characterized by partial or complete loss of the gastric corpus glands, replaced by pseudo-pyloric or intestinal metaplasia, or fibrosis, and consequent impairment of the gastric acid secretion. 10 For this reason, in corpus atrophic gastritis, the use of PPIs is questionable 11 and it has even been reported to be harmful increasing the risk of gastric cancer. 12 Concurrently, the efficacy of the common treatment regimens against H. pylori with two or more antibiotics without associating PPIs in corpus atrophic gastritis may not be equivalent to that observed associating PPIs in patients with H. pylori gastritis and preserved gastric acid secretion. Still, data on this issue are completely lacking. Thus, the current study aimed to assess the tolerability and the eradication rate of modified eradication regimens without using PPIs in patients with corpus atrophic gastritis as first-line treatment of H. pylori infection.
Materials and methods
Study design and population
This article was drafted according to the strengthening of the reporting of observational studies in epidemiology guidelines to ensure the quality of reporting. 13
As shown in Figure 1, in the current real-life longitudinal observational study, between 2001 and 2022, in our teaching hospital, corpus atrophic gastritis was histologically diagnosed on gastric biopsies obtained during gastroscopy in 721 consecutive adult (>18 years) patients (67.9% females, median age 62 years, range 22–85 years), presenting more commonly with anaemia or dyspepsia, less frequently with autoimmune thyroid disease or family history of gastric cancer. Patients with corpus atrophic gastritis were considered eligible for inclusion when they were aged at least 18 years, had H. pylori infection diagnosed by a histopathological assessment performed on gastric biopsies taken during gastroscopy, complete gastric biopsy sampling protocol according to the updated Sydney system, 14 and complete clinical, endoscopic and histopathological data of gastric biopsies, and were naïve for H. pylori eradication treatment. Patients were included irrespective of the presence or absence of concomitant antral superficial or atrophic gastritis. Patients were excluded when they were H. pylori negative or had a diagnosis of oesophageal and/or gastric neoplastic lesions at baseline gastroscopy or partial or total gastrectomy.

Flowchart of included study population of patients with corpus atrophic gastritis.
At the time of diagnosis of corpus atrophic gastritis, H. pylori infection was assessed by histology in 146 patients. H. pylori infection was considered present when the following two criteria were fulfilled: the presence of the bacterium at Giemsa stain and the presence of neutrophil granulocytes at histology, as previously reported. 15
According to our clinical protocol, all 146 patients underwent a baseline gastroscopy with biopsies according to the updated Sydney system 14 in the gastrointestinal endoscopy unit and a subsequent clinical evaluation in the outpatient gastroenterology clinics of our teaching hospital during which a clinical interview based on a structured questionnaire was performed and eradication therapy was prescribed. H. pylori infection was treated with a concomitant or sequential amoxicillin-based therapy (ABT) regimen until 2016; since this year, SPBT became available in Italy and was used as first-line treatment (see section ‘Eradication therapy regimens’). All treatment regimens were prescribed without PPIs and eventual PPI treatment before diagnosis of corpus atrophic gastritis was systematically withdrawn. A second clinical evaluation and a second gastroscopy, following the same protocol as reported below, were scheduled 6 ± 3 months from the end of the eradication therapy to assess treatment adherence, adverse events and treatment efficacy (Figure 2). The absence of H. pylori and neutrophil granulocytes at histology was considered to be consistent with successful eradication. 14 Unfortunately, 70 patients did not undergo the second gastroscopy for histological assessment of the efficacy of eradication treatment (65 were lost at follow-up and 5 underwent total gastrectomy for gastric neoplasms) and were thus excluded. The clinical and biochemical features of the finally included 76 corpus atrophic gastritis patients with histological assessment of the efficacy of eradication treatment are summarized in Table 1.

Clinical management protocol.
Baseline characteristics of the 76 corpus atrophic gastritis patients with histologically assessed Helicobacter pylori infection.

Data were expressed as number/total (%) or median (range).
To assess the tolerability and the eradication rate of the modified eradication regimens without PPIs in patients with corpus atrophic gastritis, the number and the type of adverse events and the number and proportion of patients successfully cured of infection were evaluated in all patients who completed treatment and underwent the second histological assessment.
To assess whether eradication rates of treatment regimens without PPIs were non-inferior to those with PPIs, we compared the rates obtained in corpus atrophic gastritis patients with a group of 49 patients with histologically H. pylori-positive chronic superficial non-atrophic gastritis, treated with the same eradication regimens, but with PPIs. In these patients, analogously to patients with corpus atrophic gastritis, treatment efficacy was assessed by histology on biopsies taken during a second gastroscopy performed after 6 ± 3 months from the end of the eradication therapy. In our centre, we routinely propose histological assessment of treatment efficacy in patients at higher risk for gastric cancer, that is positive family history of gastric cancer or corpus-involving H. pylori positive superficial gastritis. In detail, this group was selected amongst n = 693 consecutive patients undergoing first-time gastroscopy with biopsies for dyspepsia, anaemia, reflux symptoms and/or family history of gastric cancer between 2016 and 2022; n = 553 patients were excluded because they were H. pylori negative; n = 140 patients were diagnosed with histologically H. pylori-positive chronic superficial non-atrophic gastritis and treated with eradication regimens with PPIs; n = 91 of them were not considered because they did not adhere to undergo a second gastroscopy to assess treatment efficacy or were lost at follow-up, and, therefore, 49 patients (49% females, median age 56, range 22–81 years) with histological H. pylori-positive chronic superficial non-atrophic gastritis and histological assessment of treatment efficacy were included for analysis.
Written informed consent was provided by all participants, and approval of the local ethical committee was achieved (No. 7022/2020). Patients’ details have been anonymized in such a way that the original identifying characteristics have been removed and, therefore, they cannot be linked back to any specific person, even if combined with other information sources. For that reason, patients’ consent for publication does not apply to this study.
Eradication therapy regimens
As detailed in Table 2, H. pylori infection was treated with concomitant or sequential ABT regimens until 2016; since this year, when SPBT was placed on the market in Italy, this treatment was prescribed as the first line. To increase compliance to treatment, in contrast to the regimen of three pills four times daily for 10 days according to the SPBT technical datasheet, we prescribed a modified eradication therapy regimen of three pills three times daily for 13 days after meals. In all patients with H. pylori-positive corpus atrophic gastritis, antibiotics were prescribed without PPIs. Table 2 gives an overview of prescribed eradication treatment regimens including dosage and administration mode.
Eradication treatment regimens prescribed in patients with corpus atrophic gastritis with histologically assessed Helicobacter pylori infection without using proton pump inhibitors.

In the 49 patients with H. pylori-positive chronic superficial non-atrophic gastritis, antibiotics were prescribed together with PPIs, such as pantoprazole 40 mg or esomeprazole 40 mg, both prescribed once a day, 30 min before breakfast. In 40 of them, SPBT with PPI in 9 ABT regimens with PPIs were prescribed.
Endoscopic procedures
Gastroscopies were performed by fully trained gastroenterologists and endoscopists. All patients underwent pharyngeal anaesthesia (xylocaine spray puffs) and conscious intravenous sedation (midazolam 3–5 mg). Gastroscopies were performed using white light mode and since 2015, when it became available in our centre, using electronic chromoendoscopy (narrow band imaging). Biopsies were collected according to the updated Sydney system 14 : two biopsies from the antrum, one from the incisura angularis and two from the corpus, and sent for histopathological assessment in different vials.
Histopathological assessment
Histopathological assessment was performed by an expert pathologist of upper gastrointestinal pathology. The histopathological report was redacted according to the criteria of the updated Sydney system. 14 The presence of atrophy was graded on a four-grade scale: absence of replacement (score 0), replacement to a mild (score 1), moderate (score 3) or severe degree (score 3). Based on a morphological-histopathological assessment, intestinal metaplasia was defined as the substitution of normal gastric glands with intestinalized glands. Pseudopyloric metaplasia was defined as the replacement of the oxyntic glands resembling the mucosa-secreting cell-lined glands normally present in the antral region. H. pylori infection was considered present when the bacterium was retrieved at Giemsa stain and an inflammatory infiltrate of neutrophil granulocytes was present at histological evaluation, as previously reported. 15
Assessment of cure of H. pylori infection
The efficacy of eradication treatment was assessed by histology of gastric biopsies obtained during gastroscopy after 6 ± 3 months from the end of the antibiotic therapy. Treatment was defined as successful when H. pylori and neutrophil granulocytes infiltrate could not be retrieved anymore at any of the gastric biopsies. 15
Adherence to treatment and safety
The adherence was evaluated in all patients after the end of the follow-up with a second clinical evaluation. The adherence was defined as adequate if the patient had completed at least 90% of the prescribed therapy. The adverse events experienced by the patients during the eradication therapy were defined depending on the intensity of symptoms as evaluated by the physician during the second clinical evaluation: mild (not interfering with the daily routine), moderate (affecting daily routine), intense/severe (prohibiting normal daily routine) and serious (causing major problem health, hospitalization and/or requiring intervention to prevent permanent damage).
Statistical analyses
Data were expressed as median (range) and/or number/total (%).
To assess the tolerability and eradication rate of the modified eradication regimens without PPI in patients with corpus atrophic gastritis, the number and type of adverse events and the number and proportion of patients successfully cured of infection in all patients who completed the treatment and underwent the second histological assessment were evaluated.
Eventual differences in eradication rates and adverse events in different eradication regimens were analysed using Chi-square test or Fisher exact test, as appropriate. Two-tailed p values < 0.05 were considered statistically significant. Statistical analyses were performed by using MedCalc Statistical Software version 22.013 (MedCalc Software, Ostend, Belgium; http://www.medcalc.org; 2023).
Results
A total of 76 corpus atrophic gastritis patients (77.6% females, median age 58.5, range 26–88 years) were treated with modified eradication regimens without using PPIs. The modified SPBT regimen was prescribed in 46 (60.5%) patients, and the modified ABT regimen in the remaining 30 (39.5%) patients.
Tolerability of eradication regimens
Overall, no serious, severe or moderate adverse events were reported. In patients who underwent the modified SPBT without PPIs, four mild adverse events were recorded in four patients: one patient reported an episode of vomiting, another two episodes of self-limiting diarrhoea, one nausea and one abdominal discomfort. In patients undergoing the modified ABT without PPIs, two mild adverse events were recorded in two patients: one patient had one episode of vomiting and one patient reported abdominal discomfort. None of these adverse events required medical treatment. The low number of recorded adverse events did not allow statistical comparison between different treatment regimens groups.
Eradication rates in patients with corpus atrophic gastritis
Overall, 71 (93.4%) of the 76 treated corpus atrophic gastritis patients completed the modified eradication regimens. In detail, 43 of 46 (93.5%) patients completed the modified SPBT without PPIs, and 28 of 30 (93.3%) completed the ABT without PPIs. Successful cure of H. pylori infection was observed in 64 of the 71 patients who completed treatment and, thus, the overall eradication rate of the modified regimens without PPIs in corpus atrophic gastritis patients was 90.1% (95%CI 69.4%–115.1%).
As shown in Table 3, amongst patients treated with SPBT without PPIs, 42 of the 43 corpus atrophic gastritis patients who completed the treatment were successfully cured against 22 of the 28 who completed the treatment with ABT without PPIs. The eradication rate was significantly higher in patients who underwent the modified SPBT compared to the modified ABT: 97.7%, 95%CI 70.4%–132.0%, versus 78.6%, 95%CI 0.49.2–118.9%, p = 0.013. No difference in age between treatment groups was observed.
Comparison between the eradication rates of the modified eradication treatment regimens without PPI in patients with Helicobacter pylori-positive corpus atrophic gastritis and with PPI in patients with H. pylori-positive chronic superficial non-atrophic gastritis.
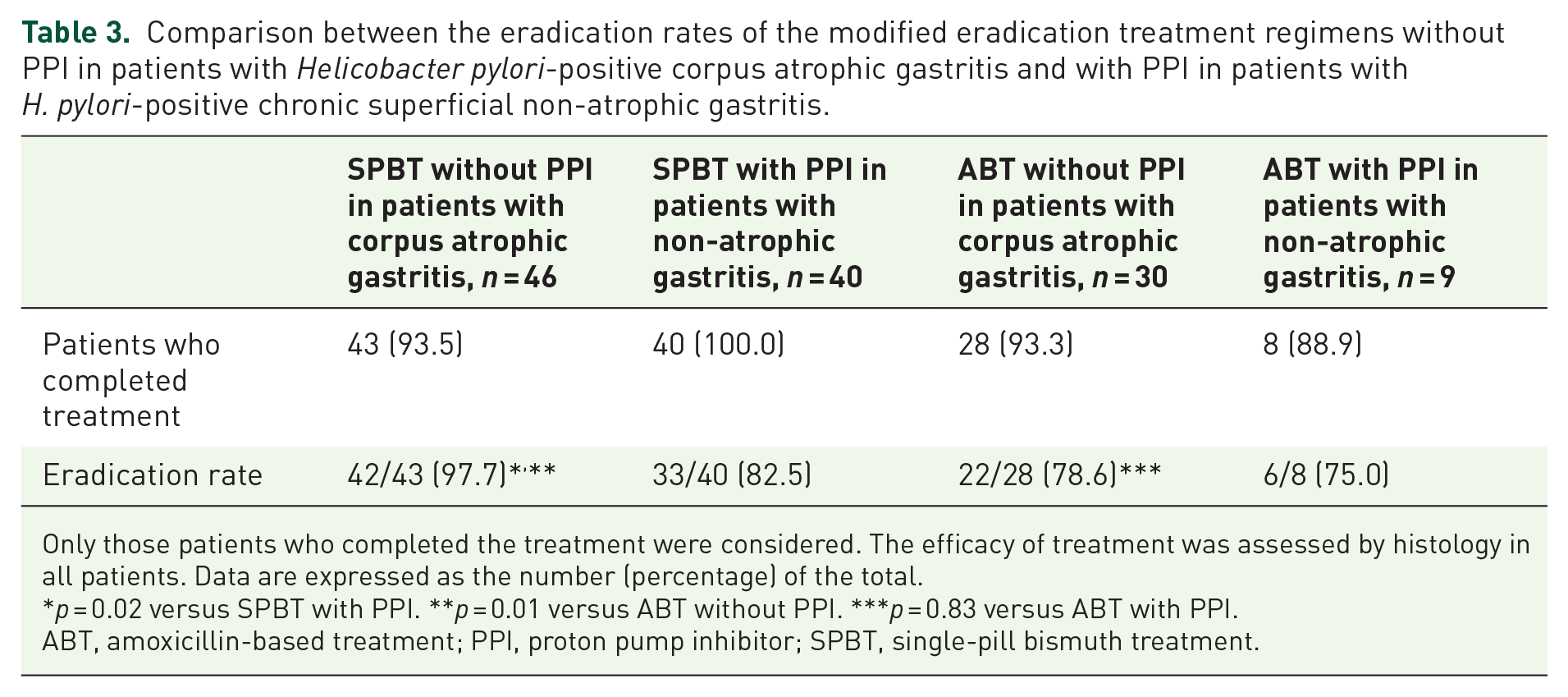
Only those patients who completed the treatment were considered. The efficacy of treatment was assessed by histology in all patients. Data are expressed as the number (percentage) of the total.
p = 0.02 versus SPBT with PPI. **p = 0.01 versus ABT without PPI. ***p = 0.83 versus ABT with PPI.
ABT, amoxicillin-based treatment; PPI, proton pump inhibitor; SPBT, single-pill bismuth treatment.
Eradication rates of modified treatment regimens without PPIs compared to those with PPIs
To better understand the performance (non-inferiority) of the modified treatment regimens without PPIs, we compared the obtained eradication rates in patients with corpus atrophic gastritis to those of 49 patients with H. pylori-positive superficial non-atrophic gastritis treated with the same treatment regimens (SPBT, n = 40 and ABT, n = 9), but with PPIs.
As detailed in Table 3, all 40 patients with H. pylori-positive superficial non-atrophic gastritis treated with the modified SPBT with PPI completed the treatment, and 33 of them achieved the cure of infection; thus, the eradication rate was 82.5% (95%CI 56.8%–115.9%). Of the nine patients with H. pylori-positive superficial non-atrophic gastritis treated with ABT with PPIs, eight completed the treatment and six of them were successfully cured of infection, thus resulting in an eradication rate of 75% (95%CI 27.5%–163.2%).
The eradication rate of SPBT without PPI was non-inferior but even higher compared to that of SPBT with PPI (97.7% vs 82.5%, p = 0.02); also, the eradication rate of ABT without PPIs was non-inferior to that with PPIs (78.6% vs 75%, p = 0.83). No difference in age between treatment groups was observed.
Discussion
To our knowledge, this is the first real-life longitudinal observational study reporting data on the tolerability and efficacy of eradication treatment regimens in patients with histologically H. pylori-positive corpus atrophic gastritis treated with SPBT and ABT, but without PPIs.
H. pylori infection, by far the most common agent causing chronic gastritis, is recognized as being a type 1 human carcinogen, 16 and its eradication is recommended, particularly when the infection has spread to the corpus mucosa increasing the risk of gastric cancer.1–4 Corpus atrophic gastritis is a well-known precancerous condition affecting the oxyntic acid-secreting mucosa, thus leading to impaired gastric acid secretion.10,16
Harmful effects of long-term PPI treatment concerning increased gastric cancer risk have been reported in several population-based studies.17–21 Even if the use of a 2-week course of PPI treatment associated with eradication treatment cannot be considered a long-term use, caution may be warranted in patients with corpus atrophic gastritis, per se at increased risk of gastric cancer. In patients with autoimmune corpus-restricted atrophic gastritis who were on PPI use before diagnosis, a positive association with subsequent development of gastric neoplastic lesions was shown (OR 9.6, 95%CI 2.3–40.3). 12
In addition, it should be taken into consideration that from a pharmacokinetic point of view, PPIs are administered as pro-drugs and acidic pH is needed for their activation 22 : after the acid space-dependent concentration of the PPIs at the luminal surface of the mucosal proton pump, that is about 1000-fold of that in the blood, the subsequent essential step is the low pH-dependent conversion from the accumulated prodrug to the activated drug that is a highly reactive cationic thiophilic reagent; in other words, protonation of these compounds and thus the presence of gastric acid secretion is required for their activation to form disulphides with cysteines of the H+, K+-ATPase to make them effective; hence, PPIs are recommended to be taken about 30 min before breakfast to ensure that proton pumps are active when peak concentrations of the PPIs are present in the blood. 22 Consequently, due to the oxyntic damage-related increased intragastric pH, in patients with corpus atrophic gastritis PPI treatment is not indicated and should be definitely withdrawn at the moment of diagnosis.
H. pylori treatment regimens have been tested in clinical trials including mainly patients with superficial non-atrophic gastritis and, up to now, no data are available on the treatment efficacy of the commonly used eradication treatment regimens without using a concomitant PPI treatment. The current real-life longitudinal observational study showed that modified ABT and SPBT eradication regimens without associating PPIs are well tolerated and able to achieve successful cure of infection in about 90% of patients with corpus atrophic gastritis. Importantly, the eradication rates of the modified treatment regimens without PPIs observed in patients with corpus atrophic gastritis were not inferior to the commonly used treatment regimens with PPIs in patients with H. pylori-positive superficial non-atrophic gastritis. Hence, eradication regimens against H. pylori infection in corpus atrophic gastritis avoiding the prescription of PPIs are feasible and efficacious, exceeding the ‘borderline acceptable’ eradication rates in clinical practice being between 85% and 89%. 23
PPIs are considered essential in the eradication treatment of H. pylori, enhancing the antibiotics’ bioavailability and effectiveness, and this benefit related to the increased intragastric pH is considered even higher with double than with single doses daily with classic amoxicillin- and clarithromycin- or metronidazole-based triple therapies; conversely, this ‘supporter’ role of PPIs is not well settled in quadruple treatments.24–26 A recent study based on the ‘European Registry on Helicobacter pylori management’ (Hp-EuReg) 27 evaluated the role of dosage of PPIs in the effectiveness of H. pylori eradication treatments and concluded that one of the variables most strongly influencing the H. pylori treatment effectiveness were the prescribed PPIs dose; however, according to their data, the use of high-dose PPIs in 14-day triple therapy and 10-or 14-day quadruple concomitant therapy can be recommended, while standard-dose PPIs seem to be sufficient in 10-day bismuth quadruple therapies, showing a less strong role of PPIs in this latter regimen.
In the current study, in corpus atrophic gastritis patients, SPBT without PPIs association was highly efficacious (eradication 97.7%) outperforming ABT without PPIs (78.6%, p = 0.01). The finding that bismuth-based regimens can achieve higher eradication rates has been previously observed in regimens with PPIs,2,7–9 and the current study provides for the first time data that this seems to be true also without using PPIs. The high (>90%) eradication rates with bismuth-based regimens may be explained by the fact that bismuth salts themselves have a negative effect on H. pylori infection, as they can exert direct bactericidal effect on H. pylori in different ways: by forming complexes in the bacterial wall and periplasmic space, by inhibiting different enzymes, ATP synthesis and adherence of the bacteria to the gastric mucosa, albeit these mechanisms of anti-bacterial action are not yet fully understood.28,29 In addition, synergic action between bismuth salts and antibiotics was observed, and, up to date, no resistance to bismuth has been reported; hence, the use of this component may even prevent the development of acquired antibiotic resistance of H. pylori 29 ; both metronidazole and clarithromycin resistances can be overcome by associating bismuth to the eradication regimen,28,29 making bismuth an indispensable component of the first-line treatment. Unfortunately, bismuth salts have become commercially unavailable in Italy, except for the formulation of SPBT.
PPIs are one of the most commonly prescribed and used drugs and their overprescription has been associated with economic burden, adverse events and polypharmacy. Thus, PPI deprescribing is felt by many medical communities as an important strategy for reducing costs, risks and, not ultimately, ‘pill burden’.30–32 Impaired gastric acid secretion may be associated with other conditions than corpus atrophic gastritis, such as previous gastric surgery for peptic ulcer or gastric cancer (partial gastrectomy Billroth I or II) or bariatric surgery (sleeve gastrectomy or Roux-en-Y gastric bypass) due to resection of acid-producing corpus mucosa or gastrin-producing antral mucosa; analogously to patients with corpus atrophic gastritis, also in these patients the use of PPIs not indicated. 33 In the case of H. pylori positivity in patients with one of these conditions, eradication regimens without concomitant PPI treatment might be taken into consideration, albeit studies in these specific clinical settings are needed.
Limitations
We are aware of some limits of the current study: (i) the long period of time (21 years) over which this longitudinal observational study was conducted may represent a possible source of bias. However, the clinical management protocols and diagnostic methods during the study period were substantially the same as all patients were diagnosed based on histological assessment of gastric biopsies obtained during gastroscopy according to the updated Sydney system and also the efficacy of eradication treatment was assessed by histology following the same standards. Since 2015, gastroscopes equipped with narrow-band imaging have been available which might have increased diagnostic reliability of gastritis and Hp infection, but eradication rates did not differ over the years. (ii) The first-line treatment protocol changed with the availability of SPBT in 2016 in Italy, making the ABT less attractive due to the easier prescription and patients’ use and adherence of the single pill treatment; (iii) this study was not designed as a clinical trial to assess treatment efficacy; nevertheless, it reflects treatment efficacy of current treatment regimens derived from real-life clinical practice; and (iv) patients were lost at follow-up or were not willed to undergo a second gastroscopy for assessment of efficacy of treatment (48%). This was a necessary criterion for inclusion in the current study, because in corpus atrophic gastritis, non-invasive tests for H. pylori may not be reliable due to increased intragastric pH 34 to ascertain the cure of the infection, which is crucial in patients at higher risk for gastric cancer.
Conclusion
In conclusion, the current study shows that in clinical practice, the cure of H. pylori infection can be achieved without concomitant PPIs in about 90% of patients with corpus atrophic gastritis who do not have an indication to suppress acid secretion due to their disease-related acid secretion impairment. In this specific clinical setting, a bismuth-based regimen (SPBT) without PPI association was highly effective and well tolerated.
