Abstract
Background:
Mildly elevated levels of transaminase and/or immunoglobulin G (IgG) are common in patients with primary biliary cholangitis (PBC). It is still unclear whether adding immunosuppressive therapy to ursodeoxycholic acid (UDCA) benefits those patients who are not fulfilling the diagnostic criteria of PBC with autoimmune hepatitis (AIH) features.
Objectives:
To assess the efficacy of adding immunosuppressive therapy to UDCA for patients with PBC and autoimmune phenomena but not fulfilling the diagnostic criteria of PBC with AIH features.
Design:
This is a retrospective–prospective cohort study in a tertiary medical center.
Methods:
Patients with PBC and autoimmune phenomena were defined by the elevation of IgG and/or transaminase but did not fulfill the diagnostic criteria of PBC with AIH features. We grouped these patients based on with and without add-on immunosuppressive therapy and balanced their baseline characteristics using inverse probability treatment weighting (IPTW).
Results:
A total of 652 patients with PBC and autoimmune phenomena were included, with a median follow-up of 4.08 years. After IPTW, the pseudo sample size in the add-on therapy and monotherapy groups was 558 and 655, respectively. After 1 year of observation, patients in the add-on therapy group had a higher biochemical response rate (normalization of transaminase and IgG levels) (49% versus 17%, p < 0.001). Furthermore, add-on therapy improved the transplant-free survival in the subgroup of patients with PBC and transaminase ⩾3 × upper limit of normal (ULN) or IgG ⩾1.3 × ULN (p = 0.033).
Conclusion:
Add-on immunosuppressive therapy may improve the normalization rates of transaminase and IgG levels in all patients with PBC and mildly elevated transaminase and IgG levels and the long-term outcomes in the subgroup of the patients with transaminase ⩾3 × ULN or IgG ⩾1.3 × ULN.
Plain language summary
Adding immunosuppressive therapy may enhance the normalization of ALT, AST and IgG levels in all PBC patients with mild elevation and improve long-term outcomes in those with more severe elevation of ALT, AST and IgG. These findings contribute to our understanding of treatment options for PBC patients with autoimmune phenomena.
Introduction
Primary biliary cholangitis (PBC) is a chronic autoimmune liver disease characterized by dramatically elevated serum levels of alkaline phosphatase (ALP) and immunoglobulin M (IgM), positive anti-mitochondrial autoantibodies (AMAs), and non-suppurative inflammation of the interlobular bile ducts.1,2 Autoimmune hepatitis (AIH) is another autoimmune liver disease characterized by marked elevation of serum levels of alanine aminotransferase (ALT), aspartate aminotransferase (AST), and immunoglobulin G (IgG), positive antinuclear antibody (ANA)/smooth muscle antibody (SMA), and interface hepatitis on histological examination. 3 The treatment of choice for typical PBC and AIH is ursodeoxycholic acid (UDCA) and immunosuppressive therapy, respectively.1–3
Some patients with PBC also have elevated levels of aminotransferase and/or IgG, positive ANA/SMA, and interface hepatitis either simultaneously or consecutively.4,5 PBC with AIH features (also known as PBC–AIH overlap syndrome) could be diagnosed if the patients fulfill the Paris criteria, 3 accounting for approximately 3–25% of PBC patients, depending on different diagnostic criteria, study populations, and sample sizes.6–10 A recent meta-analysis showed that combination therapy with UDCA and immunosuppressants may be superior to UDCA monotherapy in terms of biochemical improvement and transplant-free survival for these patients. 11
However, a Chinese study showed that a much higher proportion of patients with PBC have some autoimmune phenomena but do not fulfill the stringent criteria of PBC with AIH features. 12 It is also reported that this kind of atypical case may also benefit from immunosuppressive therapy.10,13,14 Some studies even proposed less stringent criteria for detecting milder forms of PBC with AIH features that may benefit from immunosuppressive therapy.10,13,15 However, over-diagnosing PBC with AIH features will expose the patients to unnecessary immunosuppressive therapy and side effects.16,17 Up to now, the efficacy and the criteria or timing for initiating immunosuppressive therapy for these atypical cases are still controversial issues.
Therefore, in the present study, we investigated the efficacy of add-on immunosuppressive therapy for PBC with autoimmune phenomena, which was defined as patients with PBC who had mildly elevated transaminase and/or IgG levels but did not fulfill the Paris criteria for PBC with AIH features.
Patients and methods
This retrospective–prospective cohort study included patients diagnosed with PBC at the Beijing Friendship Hospital, Capital Medical University, from 2000 to 2020 (retrospectively included patients between January 2000 and December 2017 and prospectively enrolled patients between December 2017 and December 2020). The diagnosis of PBC was based on the published criteria.1,2 The major inclusion criteria for the current study were patients with PBC and autoimmune phenomena, arbitrarily defined as patients with PBC and elevated IgG and/or transaminase levels (with or without histologically defined interface hepatitis) but did not fulfill the diagnostic criteria of PBC with AIH features. 3
The patients were excluded if they were (1) with concomitant drug-induced liver injury or viral hepatitis B or C; (2) with key baseline data missing; (3) without adequate UDCA therapy (13–15 mg/kg/day); (4) with irregular immunosuppressive therapy; (5) received immunosuppressants because of other autoimmune diseases; (6) received fibrates or obeticholic acid therapy; and (7) lost to follow-up or followed up shorter than 1 year.
The included patients were grouped by their treatment options: UDCA monotherapy and UDCA with add-on immunosuppressive therapy. The decision to use immunosuppressive therapy was at the discretion of the physicians after careful consideration of factors such as elevated levels of transaminases or IgG, liver pathology (e.g. interface hepatitis), suboptimal response to UDCA, and patients’ preferences. Then, the biochemical responses (normalization of serum levels of transaminase and IgG, Paris I criteria, Paris II criteria) and clinical outcomes (hepatic decompensation and liver transplantation/liver-related death) were compared between these two groups.
The study protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki (6th revision, 2008) and was approved by the institution’s human research committee. The ethics committee of the Beijing Friendship Hospital, Capital Medical University, reviewed and approved the study. All patients gave verbal consent that was granted by the ethical committee. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology statement. 18
Data collection and follow-up of the patients with PBC
We retrospectively collected data from the medical records of the patients with PBC between January 2000 and December 2017. Subsequently, starting in December 2017, we continuously monitored the previously enrolled patients and initiated regular follow-ups with newly enrolled patients with PBC every 6 months. Follow-up information was obtained by two specially trained doctors with telephone interviews and the review of medical records since 2017.
The following data were collected: age, sex, body mass index, AMA/AMA-M2, SMA, antibodies to soluble liver antigen/liver–pancreas, antibodies to kidney microsome-1 (anti-LKM1), immunoglobulin levels, HBsAg and antibody to hepatitis B core antigen (anti-HBc), complete blood count, blood biochemistry, abdominal ultrasound, liver histology, treatment options (UDCA monotherapy or UDCA with add-on immunosuppressive therapy), the start time, dose and treatment duration of UDCA and immunosuppressants, side effects of immunosuppressants, smoking and alcohol consumption, the occurrence time of hepatic decompensation, liver transplantation, and liver-related death.
Working definitions
For all patients, the first baseline was defined as the time point of UDCA therapy initiation. For patients receiving add-on therapy, we defined the second baseline as the time point of add-on therapy initiation; for patients continuing their UDCA monotherapy, we defined their second baseline as the approximate time point at which the add-on therapy group started immunosuppressants.
Biochemical response rates were calculated according to the Paris I, Paris II criteria, and normalization of ALT, AST, and IgG at the second baseline and 1 year after the second baseline. Cirrhosis was diagnosed based on the published guidelines. 19 Hepatic decompensation was defined as variceal bleeding, ascites, or hepatic encephalopathy. 20
Smoking habit was defined as smoking more than 100 cigarettes lifetime.21,22 Alcohol abuse was defined as an average alcohol consumption of >40 g/day for men or >20 g/day for women lasting longer than 5 years. 23 Past hepatitis B virus infection was defined as positive anti-HBc with negative HBsAg. PBC liver histology was categorized with the Ludwig staging system as early stage (I/II) and advanced stage (III/IV). 24
Statistical analyses
ALP, gamma-glutamyl transpeptidase (γ-GT), ALT, AST, globulin, IgG, IgM, total bilirubin (TBIL), and total bile acid (TBA) were described as a multiple of their respective upper limit of normal (ULN). Albumin and platelet count were described as a multiple of the lower limit of normal and absolute values. Categorical data were expressed as frequencies (percentages) and continuous data as median (interquartile range). To compare characteristics between the two groups, we used the Mann–Whitney U test for continuous variables and the chi-square test for categorical variables.
We conducted inverse probability treatment weighting (IPTW) to balance the baseline characteristics between the add-on therapy group and the UDCA monotherapy group. Propensity scores were calculated using multiple logistic regression adjusted for age, sex, cirrhosis, ALT, AST, IgG, and TBIL at the second baseline. Covariate balance before and after IPTW was assessed using the standardized mean difference. Weights based on propensity scores were recalculated for each subgroup analysis.
Kaplan–Meier curves with or without IPTW were generated to visually demonstrate the impact of add-on immunosuppressive therapy on clinical long-term outcomes. Group difference was calculated with the log-rank test. A two-sided p value <0.05 was considered statistically significant. All statistical analyses were performed with R version 4.2.2 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Clinical characteristics of patients with PBC and autoimmune phenomena at the first baseline
A total of 652 patients with PBC and autoimmune phenomena were identified by inclusion and exclusion criteria, including 71 patients with add-on immunosuppressive therapy and 581 with UDCA monotherapy (Figure 1).
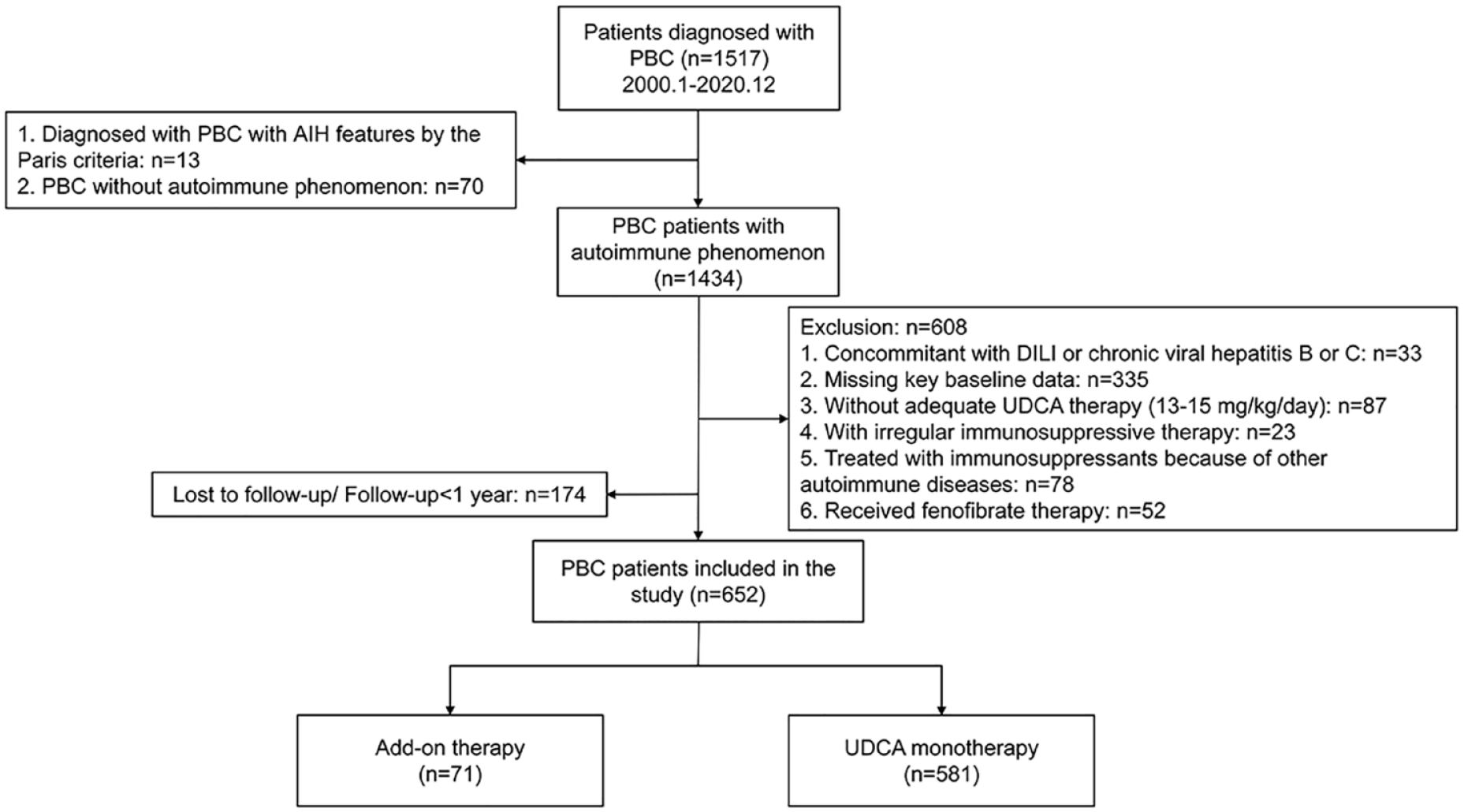
Flow chart of the study design.
Clinical characteristics of the patients at the first baseline (before UDCA therapy) are shown in Table 1. Compared with patients in the monotherapy group, patients in the add-on therapy group were younger and with a more advanced disease stage.
Clinical characteristics of PBC patients with autoimmune phenomena at the initiation of UDCA therapy.

Available in 497 patients.
Available in 351 patients.
Available in 284 patients.
Available in 405 patients.
Available in 164 patients.
ALB, albumin; ALP, alkaline phosphatase; ALT, alanine aminotransferase; AMA, anti-mitochondrial antibody; AMA-M2, anti-mitochondrial M2 antibody; anti-LKM1, antibodies to kidney microsome-1; anti-SLA/LP, antibodies to soluble liver antigen/liver–pancreas; AST, aspartate aminotransferase; BMI, body mass index; γ-GT, gamma-glutamyl transpeptidase; GLO, globulin; HBV, hepatitis B Virus; IgG, immunoglobulin G; IgM, immunoglobulin M; NA, not available; PBC, primary biliary cholangitis; PLT, platelet count; SMA, smooth muscle antibody; TBA, total bile acid; TBIL, total bilirubin; UDCA, ursodeoxycholic acid; ULN, upper limit of normal.
Clinical characteristics of patients with PBC and autoimmune phenomena at the second baseline
After a median of 6 months (interquartile range: 5.04–22.0) of UDCA monotherapy, 71 PBC patients received add-on immunosuppressive therapy (corticosteroids with or without immunosuppressants); therefore, we defined this time point as the second baseline. From the time of UDCA initiation to the second baseline, significant reductions were observed in ALT, AST, ALP, γ-GT, IgG, and IgM levels in both the add-on therapy group (n = 71) and monotherapy group (n = 581) (Table 2). Not surprisingly, at the second baseline, the biochemical response rates in the add-on therapy group were significantly lower than those in the UDCA monotherapy group (Supplemental Table S1, p < 0.05).
Characteristics of the PBC patients at the time of add-on therapy before and after IPTW.

ALB, albumin; ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; γ-GT, gamma-glutamyl transpeptidase; GLO, globulin; IgG, immunoglobulin G; IgM, immunoglobulin M; IPTW, inverse probability treatment weighting; LLN, lower limit of normal; PBC, primary biliary cholangitis; PLT, platelet count; SMD, standardized mean difference; TBA, total bile acid; TBIL, total bilirubin; UDCA, ursodeoxycholic acid; ULN, upper limit of normal.
At the second baseline, patients in the add-on therapy group were younger and in a more advanced disease stage compared with those in the monotherapy group, characterized by higher levels of ALT, AST, IgG, ALP, γ-GT, TBIL, and TBA (Table 2). The dosage and side effects of immunosuppressants in the add-on therapy group are shown in the Supplemental Material.
The liver histology examination was carried out in 44 patients in the add-on therapy group at the second baseline, with 36 of them having interface hepatitis, 13 having mild interface hepatitis, and 23 having moderate to severe interface hepatitis.
Biochemical response rates in patients with add-on immunosuppressive therapy versus those with UDCA monotherapy before and after IPTW
In the unweighted cohort, compared with patients with UDCA monotherapy, the patients with add-on therapy had higher response rates as defined by normalization of both ALT, AST, and IgG after 1 year of add-on therapy (Supplemental Table S2, p < 0.001). However, biochemical response rates, as defined by the Paris I or Paris II criteria, were similar between the two groups (Supplemental Table S2).
To increase the comparability between groups, patients in the add-on therapy group (n = 71) and patients in the monotherapy group (n = 581) were matched by IPTW, based on age, sex, cirrhosis, ALT, AST, IgG, and TBIL. After IPTW, the sample size in add-on therapy and monotherapy groups was 558 and 655, respectively. The sample size formed is based on the weight and the pseudo sample size generated by the actual sample. The two groups were well balanced at the second baseline covariates, with the p value >0.05 for all covariates, as shown in Table 2.
In the weighted cohort, the results were consistent with those from the unweighted analysis (Figure 2).
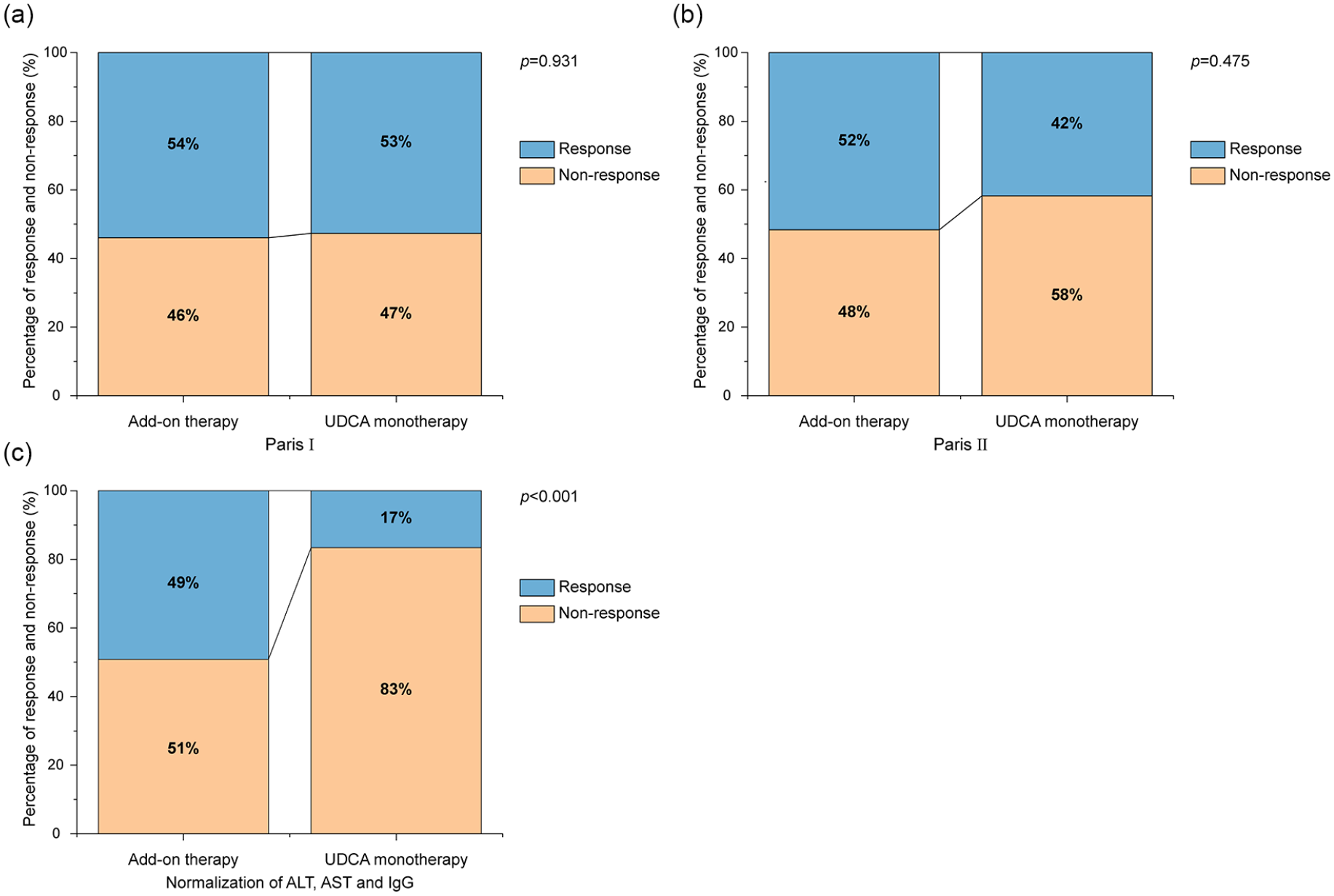
Percentage of response and non-response in PBC patients with add-on therapy or UDCA monotherapy after 1 year of add-on therapy in IPTW-weighted cohort.
Long-term outcomes of the patients with add-on immunosuppressive therapy versus those with UDCA monotherapy before and after IPTW
In the unweighted cohort, the median follow-up time in the add-on therapy group and the UDCA monotherapy group was 2.92 years (interquartile range: 1.58–4.92) and 4.17 years (interquartile range: 1.83–7.46), respectively. During follow-up, a total of 69 patients experienced liver transplantation or liver-related death, including 5 in the add-on therapy group and 64 in the UDCA monotherapy group. The transplant-free survival and cumulative incidence of decompensation were similar between the two groups [Supplemental Figure S1(a) and (b), p > 0.05] before IPTW, even after stratification by transaminase and IgG levels [Supplemental Figure S1(c) and (d), p > 0.05].
After IPTW, the median follow-up time in the add-on therapy group and the UDCA monotherapy group was 3.33 years (interquartile range: 1.10–4.97) and 4.17 years (interquartile range: 1.83–7.32), respectively. The transplant-free survival and cumulative incidence of decompensation were also comparable between the two groups [Figure 3(a) and (b), p > 0.05].
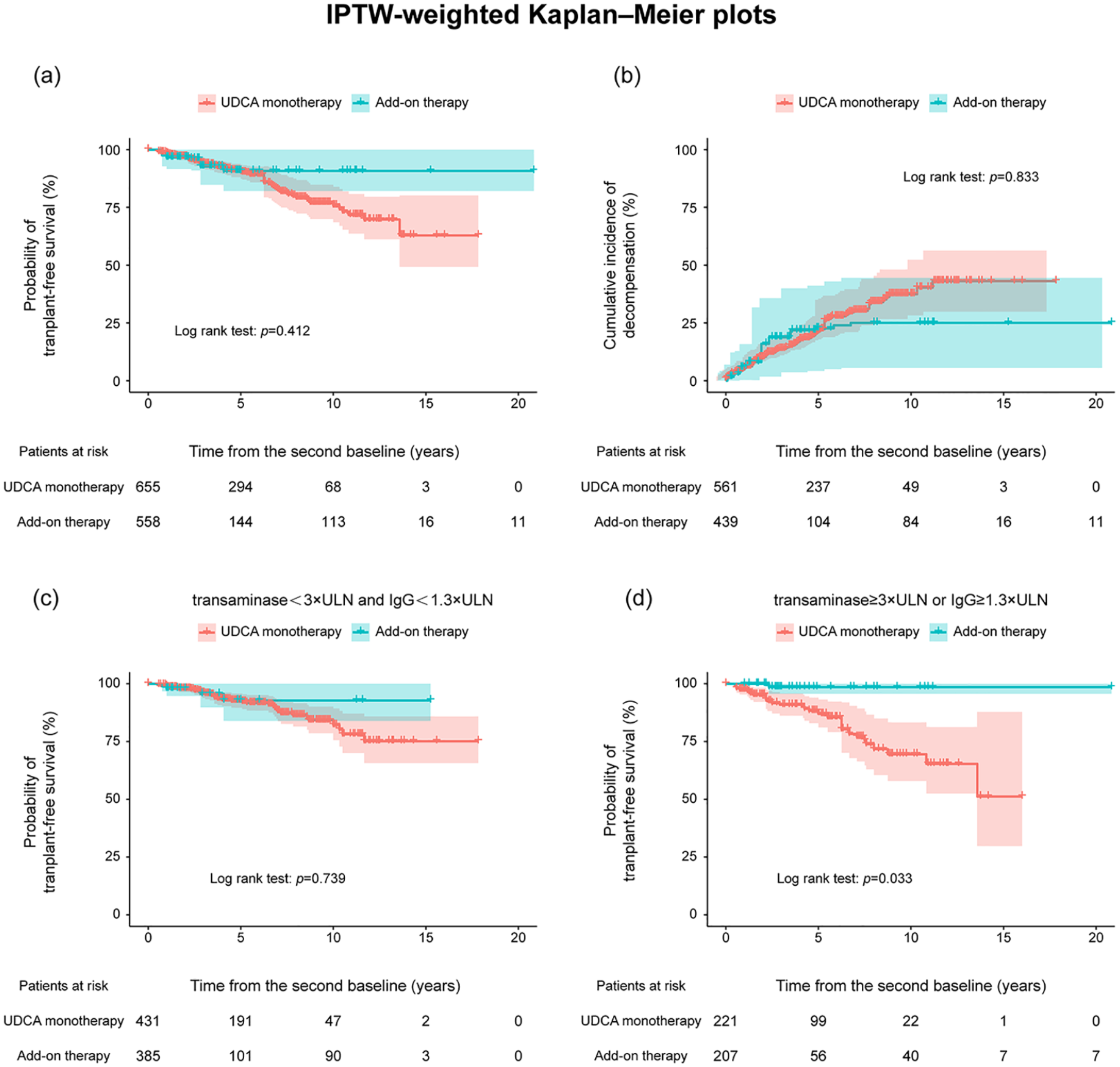
(a, b) IPTW-weighted Kaplan–Meier plots for transplant-free survival and cumulative incidence of decompensation in PBC patients with add-on therapy or UDCA monotherapy. (c, d) IPTW-weighted Kaplan–Meier plots for transplant-free survival in PBC patients with add-on therapy or UDCA monotherapy stratified according to the level of transaminase and IgG.
Then, we performed subgroup analysis based on the level of transaminase and IgG. In the patients with ALT (or AST) ⩾3 × ULN or IgG ⩾1.3 × ULN, 45 patients received add-on therapy, and 177 patients received UDCA monotherapy. After IPTW, the sample size in add-on therapy and monotherapy groups was 207 and 221, respectively. In this particular population, transplant-free survival was significantly improved in the add-on therapy group [Figure 3(d), p = 0.033]. By contrast, the transplant-free survival was similar between the two groups in patients with ALT and AST < 3 × ULN and IgG < 1.3 × ULN [Figure 3(c), p = 0.739]. The weighted Cox regression analysis also showed similar findings (Table 3), indicating that add-on therapy may reduce the incidence of liver transplantation or liver-related death in the subgroup of patients with ALT (or AST) ⩾ 3 × ULN or IgG ⩾ 1.3 × ULN.
Cox regression analysis of add-on therapy for liver transplantation or liver-related death in unadjusted and IPTW-adjusted population.

CI, confidence interval; HR, hazard ratio; IgG, immunoglobulin G; IPTW, inverse probability treatment weighting; ULN, upper limit of normal.
Discussion
In the current study, we found that for most patients with PBC and autoimmune phenomena (elevated IgG and/or transaminase), add-on immunosuppressive therapy may improve the normalization rate of transaminase and IgG levels and improve the long-term outcomes of patients with ALT (or AST) > 3 × ULN or IgG ⩾ 1.3 × ULN.
In our cohort of patients with PBC, the rate of PBC with AIH features defined by the Paris criteria was rare. Indeed, among 289 patients with PBC and a liver biopsy done at UDCA initiation, only 9 patients (3.1%) fulfilled the Paris criteria, which was similar to that reported by another Chinese study 8 but lower than that previously reported in Western countries (4.8–13.7%).7,9,25,26 This might be because Chinese patients with PBC usually have only mild elevation of transaminase and IgG levels, a lower SMA positive rate, and a lower proportion of patients who receive liver biopsy.8,27 However, our study observed a high proportion of PBC patients (1434/1504, 95.3%) with autoimmune phenomena (defined by the mild elevation of transaminase and/or IgG not fulfilling the criteria of PBC with AIH features), which is similar to another Chinese study. 12 Whether adding immunotherapy could benefit this kind of atypical cases is still a controversial issue.
We found that the patients with add-on therapy had higher response rates defined by normalization of ALT, AST, and IgG levels (p < 0.001) either before or after IPTW, which was consistent with a previous report. 28 In the unweighted cohort, the transplant-free survival was similar between the two groups despite the patients in the add-on therapy group having worse baseline biochemical variables, indicating that the add-on therapy may benefit the patients who had unfavorable baseline features. After IPTW, the demographics and disease severity of the two groups were well balanced (Table 2, p > 0.05). Importantly, we found that transplant-free survival was significantly improved by the add-on therapy in the subgroup of patients with ALT or AST ⩾3 × ULN or IgG ⩾ 1.3 × ULN (Figure 3). Therefore, our study provides further evidence to the latest PBC guidance by the Asia-Pacific Association for the Study of the Liver, which recommends that IgG ⩾ 1.3 × ULN instead of IgG ⩾ 2 × ULN is used as one of the criteria for the diagnosis of PBC with AIH feature in Asian patients. 29
Of note, out of the 45 patients with ALT (or AST) ⩾3 × ULN or IgG ⩾1.3 × ULN in the add-on therapy group, 30 received liver biopsy, and 17 of them showed moderate to severe interface hepatitis. This is in line with another report that in patients with PBC and median ALT, AST levels of 2–3 × ULN, and moderate to severe interface hepatitis, immunosuppressive therapy could improve both histological activity and fibrosis. 14 However, two studies reported that adding immunosuppressive therapy could not offer long-term benefits in patients with some features of AIH.28,30 One explanation would be that the patients included in these two studies had relatively lower ALT, AST, and IgG levels and only 30% of patients had moderate to severe interface hepatitis.28,30 Taken together, it seems that immunosuppressive therapy may improve the long-term outcomes only in a subgroup of PBC patients with relatively high levels of transaminase and IgG or histology activity, although not reaching the criteria of PBC with AIH features.
Lastly, we agree that second-line therapy (mainly including fibrates or obeticholic acid) would benefit those with suboptimal responses. However, the major indication for the second-line therapies is high levels of ALP and TBIL.31,32 However, our study focuses on patients with elevated levels of transaminases or IgG but did not meet the criteria for PBC with AIH features. We believe that these two strategies serve different purposes and may be complementary.
Our study had some limitations. First, this was a retrospective–prospective single-center study. We hope that the reasonable number of patients and sophisticated statistical analysis, including IPTW, could partially reduce the bias. Second, since not all patients underwent liver biopsy, we cannot completely exclude the possibility of PBC with AIH features in those without liver histology. Third, the relatively short follow-up duration may prevent an accurate evaluation of long-term clinical outcomes. However, after a median follow-up of 3.67 years, 119 patients (9.8%) have already reached the clinical endpoint (liver transplantation or liver-related death) in the adjusted cohort. So, we hope our results still shed some light on managing patients with PBC and some autoimmune phenomena.
Conclusion
In conclusion, add-on immunosuppressive therapy may improve the biochemical response in PBC patients with some autoimmune phenomena but did not fulfill the criteria for PBC with AIH features. Furthermore, add-on immunosuppressive therapy may improve the long-term outcomes of PBC patients with ALT (or AST) >3 × ULN or IgG ⩾1.3 × ULN. Prospective studies with larger sample sizes are justified to further validate our findings.
Supplemental Material
sj-doc-2-tag-10.1177_17562848231224840 – Supplemental material for Add-on immunosuppressive therapy may benefit selected patients with primary biliary cholangitis and autoimmune phenomena
Supplemental material, sj-doc-2-tag-10.1177_17562848231224840 for Add-on immunosuppressive therapy may benefit selected patients with primary biliary cholangitis and autoimmune phenomena by Mengqi Li, Sha Chen, Shuxiang Li, Tingting Lv, Buer Li, Shan Shan, Min Li, Na Zeng, Qianyi Wang, Yuanyuan Kong, Hong Ma, Xinyan Zhao, Xiaojuan Ou, Hong You, Weijia Duan and Jidong Jia in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-1-tag-10.1177_17562848231224840 – Supplemental material for Add-on immunosuppressive therapy may benefit selected patients with primary biliary cholangitis and autoimmune phenomena
Supplemental material, sj-docx-1-tag-10.1177_17562848231224840 for Add-on immunosuppressive therapy may benefit selected patients with primary biliary cholangitis and autoimmune phenomena by Mengqi Li, Sha Chen, Shuxiang Li, Tingting Lv, Buer Li, Shan Shan, Min Li, Na Zeng, Qianyi Wang, Yuanyuan Kong, Hong Ma, Xinyan Zhao, Xiaojuan Ou, Hong You, Weijia Duan and Jidong Jia in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-3-tag-10.1177_17562848231224840 – Supplemental material for Add-on immunosuppressive therapy may benefit selected patients with primary biliary cholangitis and autoimmune phenomena
Supplemental material, sj-tif-3-tag-10.1177_17562848231224840 for Add-on immunosuppressive therapy may benefit selected patients with primary biliary cholangitis and autoimmune phenomena by Mengqi Li, Sha Chen, Shuxiang Li, Tingting Lv, Buer Li, Shan Shan, Min Li, Na Zeng, Qianyi Wang, Yuanyuan Kong, Hong Ma, Xinyan Zhao, Xiaojuan Ou, Hong You, Weijia Duan and Jidong Jia in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
