Abstract
Before the development of double-balloon enteroscopy (DBE), the standard management of small-bowel polyposis was surgical resection. This is an invasive procedure that could lead to short bowel syndrome. In the 21st century, several new enteroscopy techniques were distributed worldwide, including DBE, single-balloon enteroscopy, spiral enteroscopy, and motorized spiral enteroscopy. These devices enable the diagnoses and endoscopic interventions in the entire small bowel, even in patients with a history of laparotomy. In patients with Peutz–Jeghers syndrome (PJS), endoscopic ischemic polypectomy with clips or a detachable snare is the preferred method for managing pedunculated polyps because it is less likely to cause adverse events than conventional polypectomy. Although polyps in patients with PJS always recur, repeat endoscopic resection can reduce the total number and mean size of polyps in the long-term clinical course. Endoscopic reduction of small-bowel intussusception caused by PJS polyps can be successfully performed using DBE without surgery. A transparent hood is useful for securing a visual field during the treatment of small-bowel polyps, and minimal water exchange method is recommended to facilitate deep insertion. Familial adenomatous polyposis (FAP) is a genetic disorder that increases the risk of developing colorectal cancer. Because jejunal and ileal polyps in patients with FAP have the potential to develop into cancer via the adenoma–carcinoma sequence, periodical surveillance, and endoscopic resection are needed for them, not only polyps in the duodenum. In cases of multiple small-bowel polyps in patients with FAP, cold snare polypectomy without retrieval is an acceptable treatment option for polyps that are 10 mm or smaller in size. Additional good pieces of evidence are necessary to confirm these findings because this narrative review mostly includes retrospective observational studies from single center, case reports, and expert reviews.
Keywords
Introduction
Small-bowel tumor is comparatively rare, comprising <1% of all gastrointestinal (GI) tumors. 1 Before the development of double-balloon enteroscopy (DBE) or video capsule enteroscopy, the small bowel is difficult to be observed endoscopically due to its long length and poor fixation resulting in redundancy for push enteroscopy. 2 The endoscopic therapeutic strategy for small-bowel polyps is not as well established as that for gastric or colonic polyps. For general upper and lower GI endoscopies, cutting-edge devices for therapeutic intervention have been developed, and these devices enable endoscopic therapeutic interventions even in difficult circumstances, and they can help to reduce the need for surgical interventions. In the 21st century, DBE, single-balloon enteroscopy, spiral enteroscopy, and motorized spiral enteroscopy (MSE)3–6 were disseminated worldwide, and these endoscopies enable to establish the diagnoses and interventions in the entire small bowel. The development of small-bowel endoscopy has led to the widespread adoption of endoscopic polypectomy as a standard strategy for the treatment of small-bowel polyposis. This approach avoids the need for invasive surgical resection and reconstruction, which can be irreversible and sometimes lead to short bowel syndrome. Above all, multiple endoscopic polypectomies are almost required in patients with GI polyposis such as Peutz–Jeghers syndrome (PJS) and familial adenomatous polyposis (FAP). Patients with PJS often have a history of intestinal resection, as surgical resection was once the first-line treatment of small-bowel polyp impaction. Patients with FAP often have a history of total colectomy for preventing colorectal cancer. Even in patients with a history of laparotomy, DBE can generally be performed safely. However, these device-assisted endoscopy for the postoperative GI tract can induce severe adverse events in some cases.7–10 In this narrative review, we focus on endoscopic management and surveillance for small-bowel polyposis.
The strategy of endotherapy for PJS
PJS is an autosomal dominant genetic disorder that causes hamartomatous polyposis (Figure 1) throughout the GI tract except for the esophagus.11–15 The responsible gene for this hamartomatous syndrome is serine/threonine kinase 11, which encodes liver kinase B1 protein located on chromosome 19p13.3. Mucocutaneous pigmentation around the mouth and on the extremities may help in the diagnosis. Due to the high incidence of other organ malignancies, including lung, pancreas, breast, ovary, and testis, in patients with PJS compared to the general population, appropriate surveillance for these malignancies as well as the GI tract should be performed. 15 Hamartomatous polyps in the small bowel can be a cause of obscure GI bleeding, which is defined as bleeding that cannot be identified by conventional esophagogastroduodenoscopy or colonoscopy. Obscure GI bleeding is divided into overt and occult GI bleeding. Overt GI bleeding shows melena, hematemesis, tarry stool, and/or hematochezia, while occult GI bleeding shows persistent iron deficiency, anemia, and a positive fecal immunochemical test without overt bleeding. A large hamartomatous polyp can sometimes cause intestinal obstruction or intussusception due to retraction of the polyp head to the distal side, leading to acute abdomen and/or vomiting (Figure 2). Small-bowel polyps that are 15 mm or larger have a significant risk of causing intussusception, which occurs in the small bowel in 95% of cases. 16 Although most small-bowel polyps in PJS are benign, 30% of polyps >20 mm show adenoma or adenocarcinoma. 17 Therefore, prophylactic resection of polyps 15 mm or larger is recommended to avoid intussusception and small-bowel cancer. The Japanese Guidelines recommend the resection of polyps >15 mm (10 mm, if possible). 14 Even if polyps are 10 mm or smaller, they will eventually grow in the future and are recommended to be treated if the procedure time is acceptable. Although polyps in patients with PJS are determined to grow back despite repeat endoscopic removals, repeat endotherapy can reduce the total number of polyps and the mean size of polyps in the long-term clinical course. 18 In our institution’s policy, all polyps 5 mm or larger are endoscopically resected, even though the patient is asymptomatic. Endoscopic resection becomes difficult when the small polyp grows into a large polyp during the long observation period. Furthermore, as mentioned above, the benign nature of small polyp enables ‘resect and discard strategy’ (neither retrieval nor pathological examinations), which facilitates multiple polypectomies in the limited procedure time.

A hamartomatous polyp in a patient with Peutz–Jeghers syndrome.

Contrast-enhanced computed tomography showing an intussusception caused by a large polyp in a patient with Peutz–Jeghers syndrome: (a) coronal and (b) axial sections. The head of the polyp is indicated by the arrow.
Snare polypectomy with electrocauterization (hot snare polypectomy) or hot snare polypectomy with a submucosal injection (endoscopic mucosal resection, EMR) is widely used for small-bowel polypectomy in patients with PJS.17–21 Since use of electrocauterization can cause expansion of polypectomy defect 1 day after hot snare polypectomy or EMR, delayed bleeding or perforation can occur in the deep small bowel. Unlike the upper GI tract, the wall of small bowel is thinner than that of stomach that increases the risk of perforation. In the setting of delayed bleeding in the deep small bowel, hemostasis by device-assisted enteroscopy is more challenging than in the upper or lower GI tract. Although snare polypectomy without electrocauterization or submucosal injection (cold snare polypectomy, CSP) can reduce the risk of delayed bleeding and perforation, it is only indicated for sessile polyps that are 10 mm or smaller in diameter, which have little risk of intussusception and obstruction. Besides, most PJS polyps are pedunculated, which are difficult to resect without electrocauterization or can cause immediate massive bleeding. Considering the risk and benefit of multiple small-bowel polypectomies, we recommend the new technique called ‘endoscopic ischemic polypectomy (EIP)’ for removing PJS polyps.22,23 EIP is a technique that involves strangulating the polyp stalk using clips or a detachable snare without snare resection, resulting in ischemia and shedding of the polyp (Figures 3 and 4). 23 For clip applications to the stalk, a single clip strangulation is not always sufficient for changing polyp color to purple. Therefore, the ‘crossed-clip strangulation’ method should be used, in which the first clip is pushed using the distal attachment to be rotated by 90°, and then a second clip is applied beside the first clip to form a 90°-angle cross (Figure 5). 24 This strangulation method facilitates clip applications in crosswise manner, which enables complete ischemia. It is more effective than multiple clip applications from the same direction. This EIP technique enables the resection of many pedunculated polyps in one session and reduces adverse events such as perforation and delayed bleeding. 22 Although EIP may not be able to resect all polyps in one session, repeat EIP sessions have been shown to decrease the number and size of polyps. 23 In cases where PJS polyps show a malignancy-like surface pattern, EMR followed by polyp retrieval is recommended to evaluate the histopathology. The use of a retrieval net is the most reliable method for polyp retrieval in conventional polypectomy procedures. However, it can cause an impaction in the area with severe bending and adhesion of the small bowel. To avoid the impaction, foreign body removing forceps with five-arms is recommended in the deep small bowel.

Endoscopic ischemic polypectomy using a clip for a hamartomatous polyp in a patient with Peutz–Jeghers syndrome.
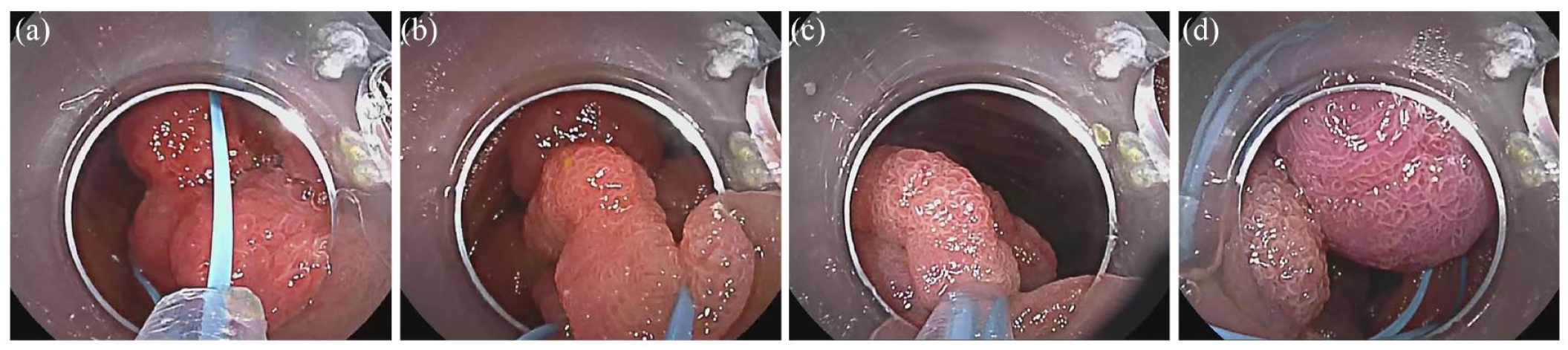
Endoscopic ischemic polypectomy using a detachable snare: (a) an open detachable snare catches the head of polyp in sight; (b) the detachable snare is moved to the stalk while closing; (c) complete closure of the detachable snare; (d) release of the detachable snare and the polyp becomes purple.

Crossed-clip strangulation method. (a) The first clip application to the stalk of the pedunculated polyp; (b) the transparent hood rotates the clip by 90°; (c) the second clip is being applied; (d) the completed crossed-clip strangulation makes the polyp purple.
Regardless of the polypectomy method, a transparent hood facilitates to secure the visual field and to identify the stalk of a pedunculated polyp by flipping the intestinal fold. The transparent hood must be adjusted and fixed to the tip of the endoscope with a length of 4 mm. The space inside the transparent hood allows safe scope operation with the clip open. Without the transparent hood, careless manipulation of the scope with the clip open may result in an iatrogenic intestinal perforation.
DBE can be used to reduce the intussusception due to PJS polyps, unless small-bowel necrosis has occurred. The emergency reduction by DBE can avoid intestinal resection. After the reduction by DBE, elective polypectomy by DBE should be performed.25,26 In the antegrade insertion, the tip of DBE is first advanced to the distal side beyond the intussusception area. The balloon at the tip of DBE is then inflated and gently retracted toward the oral side (Figure 6). In the retrograde insertion, the tip of DBE is advanced to just distal to the intussusception site. The balloon at the tip of DBE is inflated to wedge the small-bowel lumen, and then endoscopist elevates the luminal pressure by injecting water or contrast media. 25 MSE can also be used to reduce the intussusception due to a large PJS polyp. 27 However, we must also consider the risk and benefit of MSE to reduce the intussusception. To withdraw MSE, endoscopists must perform a continuous counterclockwise rotation movement. 6 In an emergency, such as perforation, MSE cannot be withdrawn as quickly as DBE, which can be rapidly withdrawn by deflating the balloons. Additionally, the counterclockwise rotation procedure of MSE after perforation may aggravate an iatrogenic hole, resulting in an enlarged resection area during the emergency operation. In the comparison study, MSE showed more frequent adverse events than DBE. 28 We believe that DBE is the preferred method for treating intussusception to avoid invasive surgical intervention. The problem of small-bowel resection is not only its invasiveness, but also the fact that it can make future management of PJS difficult due to peritoneal adhesions and short bowel syndrome. If the computed tomography shows obvious ischemia or necrosis of the small bowel, the indication of DBE must be carefully considered, as the insertion of the DBE itself has the potential to develop a wide-range perforation, resulting in a life-threatening situation. In such a situation, surgical resection must be positively considered, rather than adhering to endoscopic treatment.

Endoscopic reduction of intussusception. Antegrade DBE strategy: (a) the tip of the endoscope is inserted beyond the polyp and then the balloon is inflated; (b) the endoscope is carefully withdrawn while feeling subtle resistance. Retrograde DBE strategy: (c) the endoscope is advanced to just the anal side of the intussusception area, and then water injection is performed to increase the intraluminal pressure. The backflow is prevented by inflating the balloon at the tip of the endoscope; (d) endoscopic reduction is accomplished.
Undoubtedly, endotherapy cannot be started until the endoscope reaches the lesion. To accomplish deep enteroscopy, we recommend ‘water exchange method’ rather than carbon dioxide insufflation. 29 The method is applied to all patients with PJS during DBE in our facility. Additionally, water immersion technique also facilitates EIP by visualizing the stalk of a pedunculated polyp due to the effect of floating. On the other hand, the stalk of a very large polyp is sometimes more easily visualized and resected in the inflated lumen under carbon dioxide insufflation rather than water immersion. To perform water exchange method, we aspirate the residual gas in the small bowel and deliver the water by pushing the water supplying button without injecting water through the working channel. A large amount of water certainly elevates the intraluminal pressure, though the effect is less than that of carbon dioxide insufflation. The elevated intraluminal pressure decreases endoscopic maneuverability in the deep small bowel. The continuous pushing of water supplying button only provides a small amount of water that can be easily controlled by the endoscopist. This method was reported as ‘minimal water exchange method’ for colonoscopy.30,31 In terms of reaching a PJS polyp located in the deep small bowel, thin DBE (diagnostic type: EN-580XP; FUJIFILM, Tokyo, Japan) is more accessible than thick DBE (therapeutic type: EN-580T; FUJIFILM). 32 Although the detachable snare (HX-400U-30; Olympus, Tokyo, Japan) cannot be used in diagnostic type DBE (EN-580XP) due to its narrow working channel (2.2 mm), extracting its external sheath makes it thinner and it can be inserted into the narrow working channel. This idea enables EIP with a detachable snare, even using the diagnostic type DBE. 33
When bidirectional DBE is necessary, retrograde insertion is generally the first choice. If polypectomy is performed through the peroral route first, the resected and unretrieved polyp may impact on stenosis or anal side polyps, resulting in intestinal obstruction.
Although periodical small-bowel endoscopy every 1–3 years is recommended for patients with PJS,12–15 the maintenance DBE is modulated based on number and size of treated polyps in the last DBE session in our facility. Despite the lack of rigorous evidence, a 4- to 5-year interval is basically recommended in cases where the number of polyps resected is less than 10 and the size of polyps is <10 mm, even after a 3-year interval of DBE sessions. A 1-year interval is basically recommended in patients who have a total of more than 20 polyps resected or a maximum diameter of a polyp larger than 20 mm. Other patients who do not meet these criteria are recommended to undergo maintenance DBE every 2–3 years.
The success rate of total enteroscopy with bidirectional DBE is approximately 70% when total enteroscopy is attempted. 34 DBE cannot always be inserted in all part of the small bowel. Therefore, there are PJS polyps that are located in areas that bidirectional DBE cannot reach. In our experience, repeat DBE can reach approximately half of small-bowel polyps that were not reachable during the first bidirectional DBE session. Repeat DBE may be justified for the management of PJS polyps that are in the difficult-to-access location.
These strategies described above can be applied to treat small-bowel hamartomatous polyps derived from polyposis other than PJS, including juvenile polyposis syndrome and PTEN hamartoma tumor syndrome. However, the indication and follow-up strategy should be considered based on the characteristics of each disease and the patient’s condition.
The strategy of endotherapy for FAP
FAP is an autosomal dominant genetic disorder caused by a germline mutation of adenomatous polyposis coli gene, located on chromosome 5q21-q22.15,35,36 FAP is characterized by the development of multiple adenomatous polyps with high malignant potential in the GI tract, including colorectum, duodenum, and small bowel (Figure 7). The number of small-bowel polyps in patients with FAP is typically lower than the number of polyps in the colorectum or duodenum. FAP is classified into three subtypes based on the number of colorectal polyps: (1) profuse FAP shows a large number of colorectal adenomatous polyps, and normal colorectal mucosa cannot be observed; (2) sparse FAP shows multiple adenomatous polyps (>100), but normal colorectal mucosa can be visualized; and (3) attenuated FAP shows 10–100 adenomatous polyps. About half of the cases with profuse, sparse, and attenuated FAP develop colorectal cancer by the age of 40, 47, and 55, respectively. FAP also increases the risk of various malignancies other than those of the GI tract, including desmoid, thyroid, bone, and brain cancers, and therefore surveillance for other organs is important during follow-up for patients with FAP.15,35,36
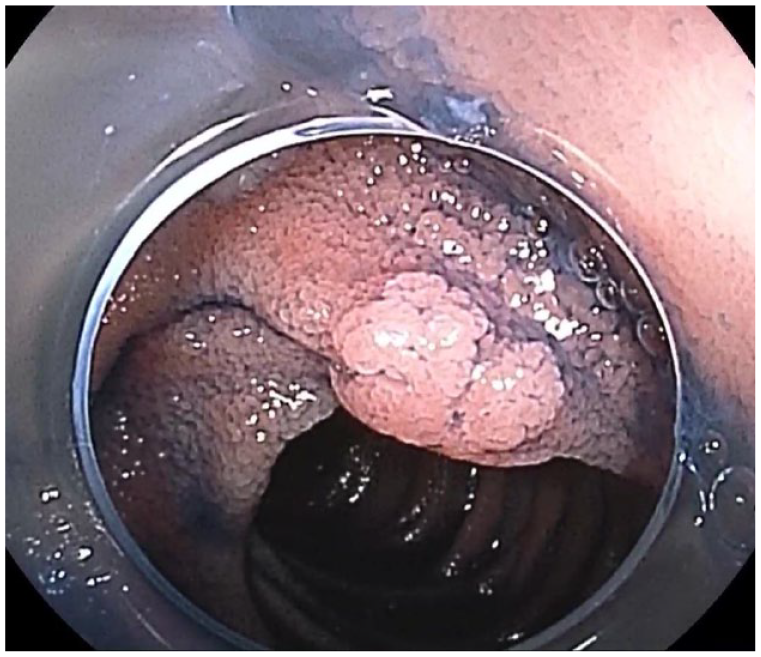
Endoscopic image of familial adenomatous polyposis of the small bowel observed by double-balloon enteroscopy-assisted chromoendoscopy using indigo carmine.
Periodical surveillance and endoscopic polypectomy of the duodenum are recommended.15,37,38 For the duodenal polyps in patients with FAP, intensive downstaging polypectomy, including CSP, cold forceps polypectomy, and underwater EMR enables down-staging of the Spigelman stage at 1-year follow-up endoscopy. 39 Although there are no definitive strategies to treat jejunal and ileal polyps in patients with FAP, these small-bowel polyps have the potential to develop into cancer via the adenoma–carcinoma sequence. 40 Therefore, we believe that jejunal and ileal polyps in patients with FAP should be resected endoscopically, not only polyps in the duodenum. Despite the lack of a robust evidence, periodical DBE surveillance is being conducted based on the criteria (Figure 8) in our facility. 41
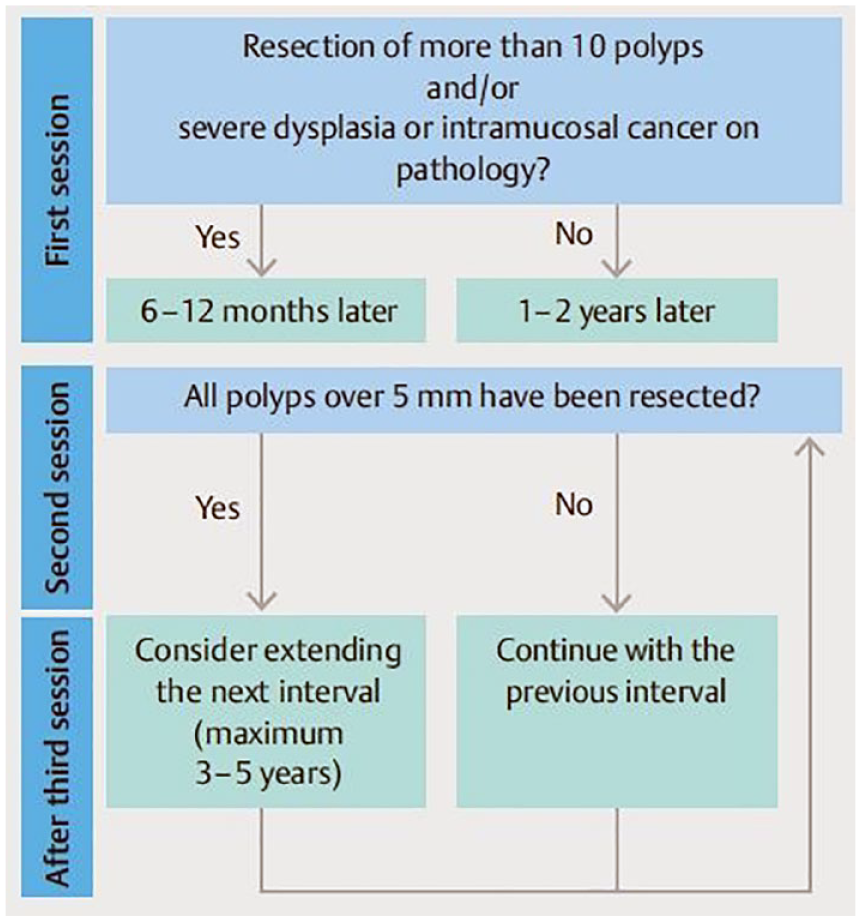
Follow-up strategy for familial adenomatous polyposis. Factors used to determine the interval to the next double-balloon enteroscopy session.
During endoscopic resection of small-bowel polyps in patients with FAP, a transparent hood is useful to secure a visual field, and minimal water exchange method facilitates deep insertion as well. We routinely perform indigo carmine dyeing 42 to improve polyp detection and use carbon dioxide insufflation during withdrawal. CSP is the first-choice endoscopic resection method for small-bowel polyps that are 10 mm or smaller in patients with FAP (Figure 9). For polyps >10 mm, hot snare polypectomy, EMR, or underwater EMR39,41,43 should be considered. Most of the polyps in patients with FAP is sessile, not pedunculated unlike polyps in patients with PJS, and therefore EIP is not generally indicated. Ablation therapy with argon plasma coagulation also facilitates multiple polypectomies.42,44,45 Discard strategy after CSP may be feasible for resecting polyps that are 10 mm or smaller due to the low potential of malignancy. For polyps >10 mm, EMR or underwater EMR with retrieval is recommended to secure negative horizontal and vertical margins. We previously reported that a jejunal large tubulovillous adenoma, 22 mm in diameter, was resected by DBE-assisted endoscopic submucosal dissection obtaining good maneuverability by fixing the scope at the redundant small bowel. 46 However, endoscopic submucosal dissection of the small bowel requires sophisticated techniques due to the thinner wall than that of the stomach. For adenoma involving the ampulla of Vater, we believe that endoscopic papillectomy (EP) is a standard strategy at the early stage. Electrocauterization may cause delayed bleeding or perforation by the extension of papillectomy defect, resulting in damages in the pancreas or biliary tract. The rate of en bloc resection with negative pathological margins of EP is 47–93%. 47 However, a high recurrence rate (58%) was reported, 48 and therefore periodical follow-up endoscopy after EP is necessary. If there is a recurrence after EP, endoscopic re-resection or cauterization with argon plasma coagulation is considered.

Cold snare polypectomy for a polyp of the small bowel in a patient with familial adenomatous polyposis: (a) snaring the polyp without submucosal injection or electrocauterization; (b) mucosal defect after the polypectomy.
The therapeutic strategies described above can be applied to resect small-bowel adenomas derived from small-bowel diseases other than FAP, including MUTYH-associated polyposis and Lynch syndrome-related polyps. Although surveillance endoscopy for FAP may be limited to the duodenum and colorectum in some cases, the malignant potential of jejunal and ileal adenomatous polyps is not also negligible. Therefore, periodical checkups are necessary in the entire small bowel.
Conclusion
There is no doubt that endoscopic polypectomy is a safe and useful procedure for the management of small-bowel polyps. In patients with PJS, EIP with clips or a detachable snare may be the first-line approach to reduce the risk of adverse events such as perforation and delayed bleeding, because the polyps are typically benign and pedunculated. In patients with FAP, CSP without polyp retrieval may be acceptable, especially in cases of multiple small polyps. The endoscopic management described in this review can be introduced easily in high-volume center that routinely performs small-bowel endoscopy. However, this narrative review mostly includes retrospective observational studies from single center, case reports, and expert reviews, due to the low prevalence of PJS/FAP. The evidence level is not high due to the study design. Additional rigorous evidence such as large randomized-controlled studies are necessary to confirm these findings.
