Abstract
Background:
Transarterial radioembolization (TARE) is increasingly used in patients with hepatocellular carcinoma (HCC). This treatment can induce or impair portal hypertension, leading to hepatic decompensation. TARE also promotes changes in liver and spleen volumes that may modify therapeutic decisions and outcomes after therapy.
Objectives:
We aimed to investigate the impact of TARE on the incidence of decompensation events and its predictive factors.
Design:
In all, 63 consecutive patients treated with TARE between February 2012 and December 2018 were retrospectively included.
Methods:
We assessed clinical (including Barcelona Clinic Liver Cancer stage, portal hypertension assessment, and liver decompensation), laboratory parameters, and liver and spleen volumes before and 6 and 12 weeks after treatment. A multivariate analysis was performed.
Results:
In total, 18 out of 63 (28.6%) patients had liver decompensation (ascites, variceal bleeding, jaundice, or encephalopathy) within the first 3 months after therapy, not associated with tumor progression. Clinically significant portal hypertension (CSPH) and bilobar treatment independently predicted the development of liver decompensation after TARE. A significant volume increase in the non-treated hemi-liver was observed only in patients with unilobar treatment (median volume increase of 20.2% in patients with right lobe TARE; p = 0.007), especially in those without CSPH. Spleen volume also increased after TARE (median volume increase of 16.1%; p = 0.0001) and was associated with worsening liver function scores and decreased platelet count.
Conclusion:
Bilobar TARE and CSPH may be associated with an increased risk of liver decompensation in patients with intermediate or advanced HCC. A careful assessment considering these variables before therapy may optimize candidate selection and improve treatment planning.
Introduction
Transarterial radioembolization (TARE) is an internal radiation therapy, consisting of the infusion of microspheres loaded with a radioisotope, mainly Yttrium-90, into the tumor-supplying arteries.
TARE is used for the treatment of advanced hepatocellular carcinoma (HCC) and as a bridging therapy to more radical options, as it promotes ipsilateral atrophy and potentially contralateral hypertrophy, sometimes allowing salvage surgery in initially non-resectable tumors. 1 TARE has been recently included in the Barcelona Clinic Liver Cancer (BCLC) staging and treatment strategy in the setting of early-stage HCC (BCLC A) when first-line options (ablation, surgery, or liver transplant) are not feasible and tumor burden is limited to a single nodule of less than 8 cm2; it has been also and accepted by FDA for HCC treatment. 3
Although TARE is generally safe, new-onset or aggravation of portal hypertension (PH) has been described. 4 Recent studies have also shown that liver impairment may also occur, especially when the treated volume is large. 5 Previous studies addressing the characteristics and mechanisms of liver impairment after TARE have included a heterogeneous population.4,6,7 Thus, risk factors associated with liver impairment and its prognostic impact remain to be completely understood, making its evaluation an active research area. 8 The presence of clinically significant portal hypertension (CSPH), defined by a hepatic venous pressure gradient (HVPG) ⩾10 mmHg, has been associated with poor outcomes in patients with cirrhosis, including an increased risk of decompensation after liver resection.9,10 On the other hand, several studies have addressed the evolution of splanchnic volumes after TARE.7,11 Although some reports have described the influence of ‘silent cirrhosis’ in this context, 11 the specific influence of CSPH has not been fully evaluated.
Therefore, the aim of this study was to investigate the impact of TARE on liver function and in the development of decompensating events in patients with intermediate and advanced HCC, specifically evaluating the implication of CSPH and the extension of therapy. In addition, the study aimed to evaluate the influence of CSPH on liver volume changes after TARE.
Methods
Patients
In all, 73 consecutive patients with HCC treated by TARE between January 2012 and December 2018 were evaluated for inclusion in this retrospective study. Diagnosis was performed using computed tomography (CT) in cirrhotic patients, or histology in non-cirrhotic patients. 12 Four patients were considered potential liver transplant candidates; six of them were lost to follow-up after TARE and were not included in the study. The final study population was comprised of 63 patients.
Supplemental Table 1 shows the indications and contraindications of TARE for HCC in our group.
The local Institutional Review Board approved the study protocol (17 December 2018).
TARE procedure
TARE was performed as described elsewhere. 13 Briefly, technetium-99m macroaggregated albumin was infused into the tumor-feeding arteries. Post-infusion planar scintigraphy and abdominal SPECT/CT (Single Photon Emission Computed Tomography with Computed Tomography) allow the assessment of extrahepatic shunt, treatment planning, and dose calculations. Then, Yttrium-90-loaded microspheres were selectively infused into the tumor-feeding branches from the hepatic artery. Both glass spheres (TheraSphere®; Boston Scientific, Marlborough, MA 01752, USA; n = 38) and resin spheres (SIR-Spheres®; SIRTex; n = 25) were used according to the tumor size and vascular anatomy: glass spheres were used in case of lesions ⩽3 cm in largest diameter, or when higher activity was required, and resin spheres were reserved for larger lesions with complex vascularity, necessitating fluoroscopic guidance during infusion due to the potential risk of treatment migration. A reduced 90Yttrium activity was used in extensive bilobar treatments when the lung absorbed dose exceeded 30 Gy to prevent complications and reduce lung exposure. Patients with bilobar involvement were treated in two sessions separated by 4–8 weeks, and the day of the first session was considered as day 0. Post-treatment dosimetry was performed on the Y-90-PET/CT in the 36 patients which was available using the 3D voxel-based dosimetry software (MIM SurePlan LiverY90-MIM, MIM Software INC, Cleveland OH 44122, USA).
Definitions
Patient information was collected at baseline (within 1 month before treatment) and at 6 and 12 weeks after TARE. Patients were followed up at least bi-monthly until death or liver transplantation.
The tumor stage was assessed following the BCLC system. 2 Cirrhosis was diagnosed based on unequivocal clinical, histological, or radiological findings. CSPH was diagnosed when at least one of the following criteria was present: (i) HVPG ⩾10 mmHg; (ii) collateral circulation in imaging procedures or endoscopic examination; or (iii) platelet–spleen ratio <909.14,15
When performed, HVPG was calculated as in clinical practice by subtracting the free hepatic venous pressure from the wedged hepatic venous pressure (a pressure-sensitive catheter was positioned at the distal portion of the hepatic vein to measure free hepatic venous pressure, and then the balloon catheter was inflated to occlude the outflow and measure wedged hepatic venous pressure).
Laboratory values and their variations were assessed together with albumin–bilirubin (ALBI), a Model for End-Stage Liver Disease (MELD), and Child-Pugh. Functional status was assessed by the Eastern Cooperative Oncology Group scale. 16
Hepatic decompensation was defined as the development of ascites detected at clinical examination, 15 hepatic encephalopathy, 17 portal hypertensive bleeding, or jaundice.17,18 Considering that in randomized trials of TARE, progression-free survival was between 16 and 24 weeks, we considered early decompensation when appeared within the first 12 weeks.19,20
A CT scan was performed at baseline, and at 6 and 12 weeks after TARE. Images were analyzed using the Philips Portal-IntelliSpace-V8 Workstation. Liver and spleen volumes were measured as previously described with the Liver Analysis Application. 21 The software initially performed automated segmentation of the entire liver, and margins were subsequently verified and adjusted if necessary. The total volume of the liver was measured, and to determine the individual volumes of each lobe, segments I to III were isolated from segments IV to VIII according to the Couinaud classification (left and right lobes, respectively). In a straightforward automated process, the spleen was also segmented to determine its volume.
Statistical analysis
Continuous variables were expressed as means with standard deviation (SD) or medians and interquartile range (IQR). Categorical variables are shown as proportions and compared with the chi-squared test. Changes in liver and spleen volumes, biochemical variables, and liver function scores between baseline and weeks 6/12 were analyzed using ANOVA with Bonferroni corrections for multiple comparisons. The Kaplan–Meier estimator was used for survival analysis. The correlation between quantitative variables was explored by Pearson’s test. To investigate potential factors associated with early decompensation, univariate and multivariate logistic regression analyses were performed. The backward stepwise method was used, setting p < 0.05 and p > 0.10 values as the criteria for the inclusion or exclusion of the variables, respectively.
Statistical analyses were performed using SPSS 21.0 (IBM SPSS Statistics; IBM-Corp., Armonk, NY, USA). Figures were created using GraphPad Prism 9.0.0 (GraphPad Software, San Diego, CA, USA) and R-software 3.6.3 (R Foundation for Statistical Computing, Vienna, Austria). All reported p values are two-sided. The level of statistical significance was set at p < 0.05.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology statement. 22
Results
Patient characteristics
Baseline characteristics are summarized in Table 1. The study cohort mostly comprised male patients (89%) with a median age of 68 years (57–77). Most of them had cirrhosis (76.2%) with intermediate (50.8%) or advanced HCC (33.3%). All patients were clinically compensated at the time of TARE [9/63 (14.3%) have had previous decompensation, mainly mild ascites]. In all, 59 patients were Child-Pugh A (93.6%), of which 31 were ALBI 1 (52.5%) and 28 were ALBI 2 (47.5%). Child-Pugh B patients (6.4%) had minor laboratory alterations or adequately controlled ascites. In total, 37 patients (58.7%) had undergone previous treatments (transarterial chemoembolization = 25; ablation = 8; surgery = 4).
Baseline characteristics.

ALBI score, albumin–bilirubin score; BCLC, Barcelona Clinic Liver Cancer; CSPH, clinically significant portal hypertension; ECOG, Eastern Cooperative Oncology Group; HCV, hepatitis C virus; IQR, interquartile range; MELD, Model for End-Stage Liver Disease; NAFLD, non-alcoholic fatty liver disease.
Median progression-free survival and overall survival after TARE were 5 (3–12) and 18 (10–29.5) months. One, 3- and 5-year survival rates were 75%, 33%, and 18%, respectively (Figure 1).
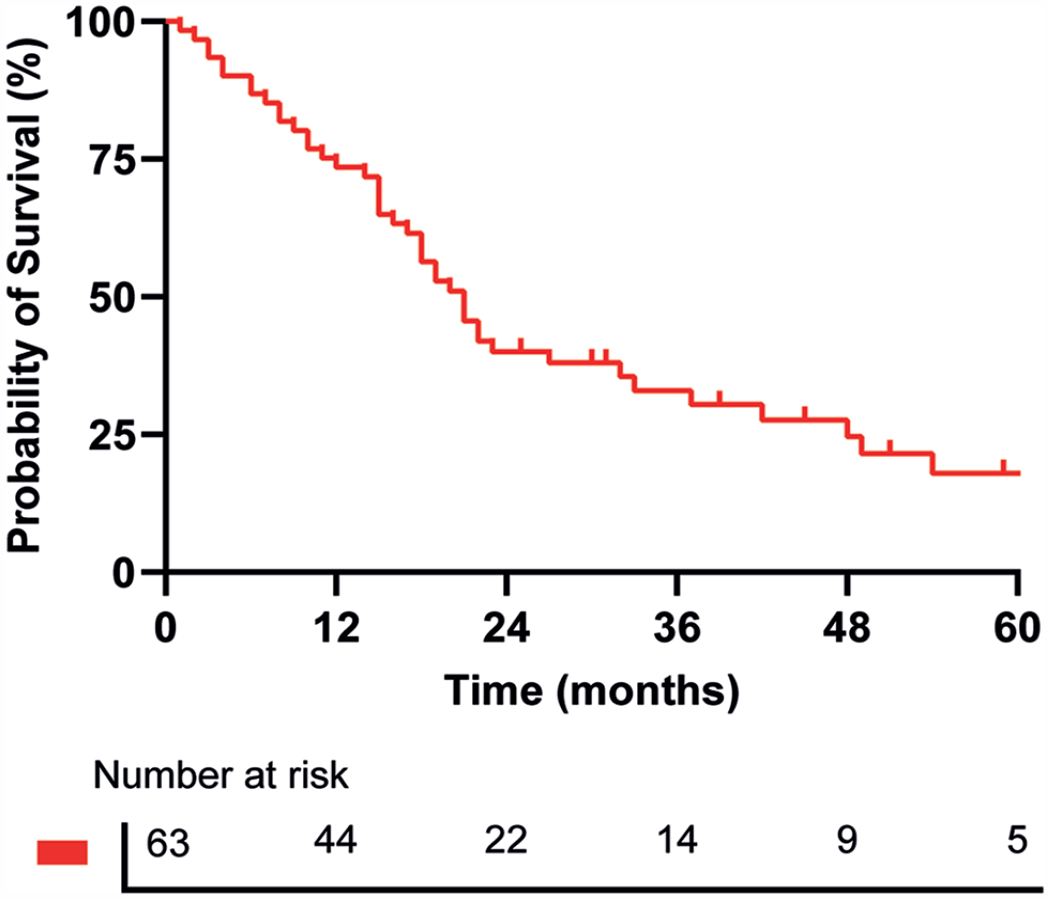
Overall survival after treatment whole cohort (n = 63).
Treatment characteristics
Treatment was segmental (1–2 segments), lobar (>2 segments, same lobe), or bilobar (>2 segments, both lobes). Technical aspects are shown in Table 2(a), and dosimetry data in Table 2(b).
Treatment technical features.

Dosimetry data were available in the most recently treated patients (2016–2018): 27 with right lobar TR, 8 with bilobar TR, and 1 with left lobar TR.
GBq, gigabecquerel; Gy, grays, IQR, interquartile range; TR, transarterial radioembolization.
Clinical course
Decompensation rates were 15.9% (10/63) and 28.6% (18/63) at 6 and 12 weeks. At week 6, nine patients had developed ascites and one patient had encephalopathy. At week 12, 11 patients had shown ascites, 6 hepatic encephalopathy, and 1 variceal bleeding. None of them had intrahepatic tumor progression according to mRECIST. When presented, liver decompensation was persistent or required maintenance treatment (as diuretic dose adjustments). Patients with early decompensation showed a significant decrease in 1-year survival rates (42% versus 84%, p = 0.019).
There was a mild but significant deterioration in bilirubin, INR, albumin, and platelets (Table 3; Figure 2), with no association between the extent of treatment and main laboratory values (Supplemental Table 2).
Evolution of the laboratory parameters during follow-up.

dL, deciliter; g, gram; INR, international normalized ratio; mg, milligram; ns, nonsignificant; SD, standard deviation; µL, microliter.

Platelet count and spleen volume evolution within 12 weeks after treatment (overall cohort: 27 patients with right lobar SIRT, 17 patients with bilobar SIRT, 6 patients with left lobe treatment). Platelet count at week 12 (p = 0.081) and spleen volume at week 12 (p = 0.000).
Nine (15.5%) and five patients (8.6%) with Child-Pugh class-A at baseline progressed to classes B and C, respectively, at week 12. The ALBI score significantly worsened (50% grade 1 and 50% grade 2 at baseline versus 31% grade 1, 41% grade 2, and 28% grade 3 at week 12; p < 0.0001). Nine patients (15.5%) with grade 1 at baseline progressed to grade 2 and 10 patients (15.9%) with grade 2 at baseline progressed to grade 3 at week 12. Therefore, 19 of 63 patients (30.2%) had ALBI score deterioration at week 12. Finally, a worsening in MELD score was also observed (6.3 [0, 3] at baseline and 6.6 [0, 9] at week 12; p = 0,053) (Figure 3). 23
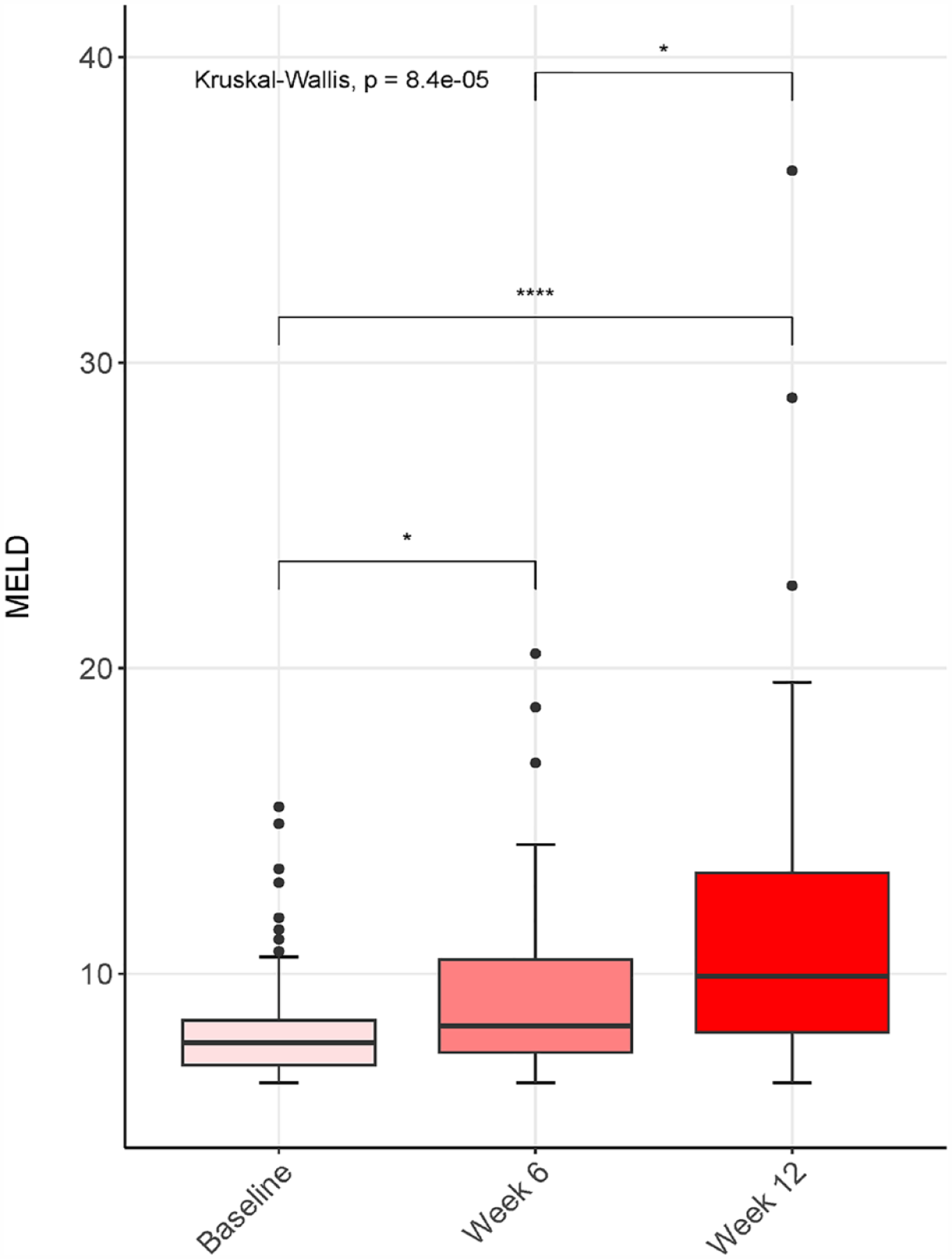
MELD score evolution during follow-up (overall cohort). Median MELD at baseline was 6.3 [0, 3], and median MELD at 12 weeks was 6.6 [0, 9]; p = 0.053.
Factors associated with early decompensation
Uni- and multivariate analyses showed that CSPH and bilobar therapy were independently associated with early decompensation after TARE (Table 4). To evaluate whether the extension of radioembolization could modify the relationship between CSPH and outcomes, an interaction term (CSPH × volume of parenchyma treated) was included in the model, showing a lack of significance (p = 0.483). Therefore, the TARE extension did not modify the relationship between CSPH and the outcomes.
Univariate and multivariate logistic regression analyses of variables associated with the development of early liver decompensation after treatment.

ALBI score, albumin–bilirubin score; BCLC, Barcelona Clinic Liver Cancer; CI, confidence interval; CSPH, clinically significant portal hypertension; MELD, Model for End-Stage Liver Disease; OR, odds ratio.
Variables shaded in blue indicate statistically significant results in the univariate analyses, while variables shaded in red indicate statistically significant results in the multivariate analyses.
Early decompensation was more frequent in patients with CSPH [16/33 (48.5%) versus 3/30 (10%); p < 0.001]. In addition, it occurred in 10/43 (23.3%) patients who received lobar therapy, as compared to 9/20 patients with bilobar TARE (45%), p = 0.08. Furthermore, the increase in MELD score was greater in patients with CSPH [+3.92 (5.74) points versus +1.22 (2.58) points; p = 0.041] and in those who received bilobar treatment [+4.32 (6.01) points versus +1.61 (3.36) points; p = 0.050)]. Supplemental Figure 1 illustrates this concept.
Finally, no patient without CSPH undergoing segmental or unilobar TARE developed early decompensation, as opposed to 7/11 (63.6%) patients with CSPH and bilobar treatment.
Dosimetry was only available for 36 patients, precluding its inclusion in the multivariate model. No significant correlation was found between dosimetry of non-tumoral tissue and the 12-week evolution of MELD (Pearson’s coefficient −0.012; p = 0.951), ALBI (−0.101; p = 0.569), platelets (0.007; p = 0.971), or spleen enlargement (−0.070; p = 0.698). Nevertheless, decompensation rates were reduced in patients with dosimetry compared to those without: 19.4% (7 out of 36 patients) versus 40.7% (11 out of 27 patients). These differences approached statistical significance (p = 0.064). We provide a comparison of patients with and without dosimetry in Supplemental Table 3.
Changes in splanchnic volumes
The evolution of visceral volumes was evaluated in 44 patients who underwent right lobar (n = 27) and bilobar TARE (n = 17). Patients with right-sided TARE experienced ipsilateral atrophy [18.5% (−7.8–32.0)] with left lobe hypertrophy [20.2% (3.2–38.2)], while the total liver volume remained unchanged (Table 5). Patients with CSPH had a lower left lobe increase [8.0% (−3.2–30.2) versus 25.6% (17.9–66.1); p = 0.05]. No correlation between the dose delivered to the non-tumoral parenchyma and the rate of left lobe enlargement in those 25 patients with right TARE and available dosimetry (Pearson’s coefficient 0.225; p = 0.290). However, patients who received more than 80 Gy in the non-tumoral tissue showed greater left lobe hypertrophy (62.1 ± 86% versus 27.4 ± 55.4%). The small number of patients who received more than 80 Gy (n = 4) precluded an accurate statistical comparison.
CT scan volumetric changes during follow-up.

mL, milliliter; SD, standard deviation.
Patients who received bilobar TARE developed liver atrophy [median volume loss: 4.7% (0–10.3)], with no significant changes in each individual lobe (Table 5). CSPH was not associated with the degree of atrophy in patients with right lobar TARE (right lobe volume loss: 12.3% versus 15.1%; p = 0.973) nor those with bilobar treatment (global atrophy: 2.1% versus 7%; p = 0.244).
A marked spleen enlargement was observed, especially after bilobar TARE [median increase after bilobar and right lobe treatment: 34.0% (0.5–74.9) versus12.5% (2.1–35.3), respectively; p < 0.001] (Figure 2; Table 5).
Finally, 6 of 32 patients (19%) without portosystemic collaterals at baseline developed ex novo collateral circulation.
Adverse events
Four complications occurred: radiation-induced cholecystitis (grade 3 adverse event) and renal failure (grade 2), both of which resolved; radiation-induced esophagitis in a patient with previous abdominal surgery and radioembolization-induced liver disease leading to death in both patients (grade 5). 24 It should be noted that both deceased patients underwent bilobar TARE.
Discussion
TARE is a consolidated therapeutic option for HCC. Importantly, this tumor occurs in patients with chronic liver disease in 90% of cases, most of them with cirrhosis 2 ; therefore, anticipating potential negative effects of radioembolization on liver function is critical. Although cirrhosis and Child-Pugh score are well-known risk factors, 25 the predisposing factors for further liver damage after TARE are not fully understood. Thus, the study aimed to investigate the impact of TARE on liver function and its predisposing factors in patients with intermediate or advanced HCC and underlying liver disease.
The study shows that TARE may affect liver function: 28.6% of previously compensated patients experienced at least one decompensating event within 12 weeks, associated with a significant worsening of ALBI, MELD, platelet count, and a marked increase in splenic volume. Finally, up to 19% of patients without collateral circulation at baseline, developed ex novo collaterals. The results are consistent with recent data 26 and show that non-selective TARE has a non-negligible impact on liver function in this population, in line with previous data showing a high rate of liver decompensation (31.6%) in the short term in a similar population. 27
Concerning HCC screening, the ultrasound-based surveillance strategy has limited sensitivity for detecting small lesions, and more than 50% of early lesions are not detected. 28 On the other hand, promising results have been suggested by using abbreviated magnetic resonance imaging protocols, specifically when hepatobiliary-specific contrast agents are applied; this strategy could be especially useful in metabolic-associated fatty liver disease, a rapidly growing cause of HCC, which mainly affects obese or very obese patients. 29 Consequently, it is likely that the proportion of early and very early-stage HCC diagnoses will increase in the future, aiming the use of less invasive treatments. Given the favorable results observed with TARE in the early stage setting, it would be reasonable to expect an increasing utilization of this treatment in such a setting, becoming necessary for a thorough assessment of the risk of liver decompensation.
In our study, decompensation severely worsens prognosis in patients with HCC regardless of tumor progression, as shown by the 1-year survival decrease observed in decompensated patients. Furthermore, clinically evident decompensation limits access to new lines of treatment. As commented in previous studies, we cannot rule out that the observed alterations were not partially secondary to the natural evolution of the tumor or underlying cirrhosis. 30 To minimize this problem, we focused our study on a time frame in which deterioration for the above-mentioned reasons is unexpected.
Identifying predisposing factors of decompensation after treatment is crucial in the selection of candidates for TARE. The study provides two variables independently associated with post-TARE decompensation. CSPH, which increases the risk of decompensation in cirrhosis, 9 and it is also a strong predictor of a worse prognosis after surgery. 10 Thus, it is biologically plausible that CSPH resulted in an increased risk of decompensation after TARE. Although previous studies have indirectly evaluated the impact of PH on TARE outcomes,19,26,27,30,31 this study clearly states that CSPH is strongly related to the risk of decompensation, suggesting that its diagnosis may help in patient’s candidacy for TARE. Second, the extension of parenchyma exposed to radiation, and presumably its distribution, increases the risk of decompensation. In fact, as tumor burden was similar regardless of unilobar or bilobar, it is reasonable to assume that bilobar treatment implies a greater exposure of the non-tumoral parenchyma, leading to a higher degree of liver injury. Several studies suggest that a non-selective approach is associated with a higher risk of decompensation, 19 and that sequential lobar therapy may improve short-term toxicity compared with a bilobar single session. 32 Considering the described impact of CSPH, this entity should be specially considered when planning bilobar TARE.
Although dosimetric data could not be included in the multivariate analysis, there was no significant correlation between the dose absorbed by non-tumor tissue and the evolution of variables associated with liver function and PH, such as MELD, ALBI, platelets, and splenomegaly. Interestingly, the logistic regression model did not identify the type of microsphere as an independent predictor of outcome.
Importantly, it should be recognized that the decompensation rate was lower in patients with available dosimetry than in those without dosimetry. While there were no major differences apart from more advanced stages in the non-dosimetry group, albeit speculative, this might be attributed to the extension of treatment.
It should be emphasized that the observed outcomes after TARE, and the possible influence of CSPH, may be similar to other local or systemic therapies. In fact, there are no prospective data on the impact of systemic therapies on the natural history of cirrhosis. This is also true regarding adverse events during therapy that are commonly observed in patients with advanced HCC, independently of the therapeutic modality.
Another important result of the study relates to liver volume changes. Patients with right lobar TARE developed marked atrophy of the treated lobe and left lobar hypertrophy, while the total liver volume remained unchanged. By contrast, bilobar therapy caused homogeneous liver atrophy of almost 5%, as described in previous studies. 33 Interestingly, we observed less contralateral hypertrophy in patients with CSPH, suggesting a possible association between the pattern of contralateral lobe hypertrophy and the severity of PH. Although splenomegaly, thrombocytopenia, and Child-Pugh score have been previously linked to lower contralateral hypertrophy after TARE,34,35 we provided here information about the influence of CSPH on this ‘atrophy/hypertrophy complex’. This might be useful to estimate the risk of early decompensation and predict future liver remnant hypertrophy when TARE is used as a bridge therapy. Remarkably, four patients in the cohort were able to undergo successful right hepatectomy after TARE.
By contrast, no correlation between the dose received by the non-tumoral parenchyma and the degree of left lobe enlargement in 25 patients with available dosimetry undergoing right lobar TARE was found. However, we observed, as suggested, 36 that the amount of radiation dose received by non-tumoral parenchyma influences contralateral hypertrophy: patients with a non-tumoral parenchymal dose greater than 80 Gy underwent a higher left lobe increase, although the small sample size precluded accurate comparisons. Whether there is a minimum dose that should be delivered to the non-tumoral liver to trigger contralateral hypertrophy, and the optimal particle distribution for this purpose, is an important issue that warrants further research.37,38
The study has some limitations. Its retrospective nature and the relatively small sample size make necessary confirmatory studies. However, the findings derived from a homogeneous and well-characterized cohort, which includes all patients consecutively treated by TARE during 2012–2018 at a reference center, counteracting this limitation. Second, direct HVPG measurement was not available in all patients. However, the diagnosis of CSPH was strongly established by HVPG measurement (11 patients), the existence of esophageal or gastric varices (22 patients), abdominal imaging showing collateral circulation (5 patients), the presence of advanced fibrosis or cirrhosis (9 patients), or the platelet–spleen ratio (16 patients). It should be also emphasized that considering the characteristics of the study cohort, our results apply only to patients with intermediate and advanced HCC disease, which are the target population of ongoing clinical trials combining systemic therapy and TARE.
The inclusion of patients with portal vein thrombosis may confound toxicity assessment. In addition, portal vein thrombosis may also alter the ‘atrophy/hypertrophy’ complex in cases with contralateral or main trunk occlusion. However, we decided to maintain them in the cohort trying to reproduce clinical practice and reinforce the external validity of our results. Finally, a complete evaluation of tumor and non-tumor absorbed dose is lacking, which precluded its inclusion in the regression models. Although partial information related to this issue suggesting the validity of the results was provided, the real impact of radiation dose on the reported outcomes cannot be completely ascertained.
In conclusion, bilobar TARE and TARE in patients with intermediate and advanced HCC with CSPH may have an increased risk of liver decompensation.19,26,27,30,31 Assessing the presence of CSPH in the evaluation of patients eligible for radioembolization could help to estimate the risk of short- and medium-term decompensation after TARE and could help to predict the pattern of contralateral lobe hypertrophy when TARE is used as a bridge therapy.
Supplemental Material
sj-docx-1-tag-10.1177_17562848231206995 – Supplemental material for Portal hypertension increases the risk of hepatic decompensation after 90Yttrium radioembolization in patients with hepatocellular carcinoma: a cohort study
Supplemental material, sj-docx-1-tag-10.1177_17562848231206995 for Portal hypertension increases the risk of hepatic decompensation after 90Yttrium radioembolization in patients with hepatocellular carcinoma: a cohort study by Laura Carrión, Ana Clemente-Sánchez, Laura Márquez-Pérez, Javier Orcajo-Rincón, Amanda Rotger, Enrique Ramón-Botella, Manuel González-Leyte, Miguel Echenagusía-Boyra, Arturo Luis Colón, Laura Reguera-Berenguer, Rafael Bañares, Diego Rincón and Ana Matilla-Peña in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848231206995 – Supplemental material for Portal hypertension increases the risk of hepatic decompensation after 90Yttrium radioembolization in patients with hepatocellular carcinoma: a cohort study
Supplemental material, sj-docx-2-tag-10.1177_17562848231206995 for Portal hypertension increases the risk of hepatic decompensation after 90Yttrium radioembolization in patients with hepatocellular carcinoma: a cohort study by Laura Carrión, Ana Clemente-Sánchez, Laura Márquez-Pérez, Javier Orcajo-Rincón, Amanda Rotger, Enrique Ramón-Botella, Manuel González-Leyte, Miguel Echenagusía-Boyra, Arturo Luis Colón, Laura Reguera-Berenguer, Rafael Bañares, Diego Rincón and Ana Matilla-Peña in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-3-tag-10.1177_17562848231206995 – Supplemental material for Portal hypertension increases the risk of hepatic decompensation after 90Yttrium radioembolization in patients with hepatocellular carcinoma: a cohort study
Supplemental material, sj-docx-3-tag-10.1177_17562848231206995 for Portal hypertension increases the risk of hepatic decompensation after 90Yttrium radioembolization in patients with hepatocellular carcinoma: a cohort study by Laura Carrión, Ana Clemente-Sánchez, Laura Márquez-Pérez, Javier Orcajo-Rincón, Amanda Rotger, Enrique Ramón-Botella, Manuel González-Leyte, Miguel Echenagusía-Boyra, Arturo Luis Colón, Laura Reguera-Berenguer, Rafael Bañares, Diego Rincón and Ana Matilla-Peña in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-4-tag-10.1177_17562848231206995 – Supplemental material for Portal hypertension increases the risk of hepatic decompensation after 90Yttrium radioembolization in patients with hepatocellular carcinoma: a cohort study
Supplemental material, sj-tif-4-tag-10.1177_17562848231206995 for Portal hypertension increases the risk of hepatic decompensation after 90Yttrium radioembolization in patients with hepatocellular carcinoma: a cohort study by Laura Carrión, Ana Clemente-Sánchez, Laura Márquez-Pérez, Javier Orcajo-Rincón, Amanda Rotger, Enrique Ramón-Botella, Manuel González-Leyte, Miguel Echenagusía-Boyra, Arturo Luis Colón, Laura Reguera-Berenguer, Rafael Bañares, Diego Rincón and Ana Matilla-Peña in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
