Abstract
Background:
Low bioavailability steroids, including beclomethasone dipropionate (BDP) and budesonide MMX, have been developed to ensure colonic targeting and low systemic activity than systematic corticosteroids in treating patients with ulcerative colitis (UC).
Objectives:
This systematic review and meta-analysis evaluated the efficacy and safety of BDP and budesonide MMX® compared with 5-aminosalicylic acid (5-ASAs) or placebo, in patients with mild-to-moderate UC.
Design:
Systematic review and meta-analysis
Methods:
We searched MEDLINE, EMBASE, and the Cochrane central register of controlled trials from inception to December 2021. We included all available randomized controlled trials (RCTs) comparing oral BDP or budesonide MMX with 5-ASAs or with placebo in induction of remission of mild-to-moderate UC. Odds ratios (ORs) and 95% confidence intervals (CIs) were calculated.
Results:
We identified two RCTs comparing BDP 5 mg with 5-ASA, one RCTs comparing BDP 10 mg with 5-ASA, two RCTs BDP 5 mg versus placebo, one RCT BDP 10 mg versus placebo, two RCTs budesonide MMX 9 mg versus 5-ASA, and six RCTs budesonide MMX 9 mg versus placebo. In terms of achieving clinical remission or improvement, BDP 5 mg, BDP 10 mg, and budesonide MMX 9 mg were more effective than placebo (OR 2.36, 95% CI 1.37–4.08; OR 2.23, 95% CI 1.02–4.87; and OR 2.03, 95% CI 1.45–2.85, respectively). The drugs were also more effective than placebo in achieving endoscopic remission. Regarding the comparisons with 5-ASA, we found no differences between 5-ASA and BDP 5 mg or BDP 10 mg or budesonide MMX 9 mg in achieving clinical remission or improvement (OR 0.90, 95% CI 0.51–1.57; OR 1.54, 95% CI 0.42–5.64; and OR 1.17, 95% CI 0.82–1.66). However, 5-ASA was more effective than budesonide MMX 9 mg in achieving histological remission (OR 0.33, 95% CI 0.16–0.70). Overall, all the drugs were safe and well tolerated.
Conclusion:
Low bioavailability steroids were more effective than placebo in achieving clinical remission, clinical and endoscopic remission, and histological remission. No differences were found between 5-ASA and BDP or budesonide MMX. Surely, more RCTs, also comparing BDP and budesonide MMX, are mandatory to confirm or not these results.
Keywords
Introduction
Ulcerative colitis (UC), a chronic inflammatory bowel disease, is characterized by a continuous mucosal intestinal inflammation commencing in the rectum and extending proximally for a variable extent.1,2 Patients with UC could experience intermittent flares of disease activity, treated with medical therapy,1,2 and their quality of life could get affected.3,4 The treatment of active UC is generally guided by the severity, extension, relapse frequency, disease course, response to previous medications, and extraintestinal manifestations. 5
Current guidelines and recent meta-analyses recommend 5-aminosalicylic acids (5-ASAs) and low bioavailability steroids as first-line treatment for the induction of remission in patients with mild-to-moderate UC.5,6 Oral corticosteroids were first used 60 years ago, and the first trial demonstrating their efficacy in the treatment of UC was conducted in the 1950s. 7 However, the use of glucocorticosteroid drugs is limited by the frequent and, in same cases, severe adverse events such as metabolic, dermatological, gastrointestinal, musculoskeletal and central nervous effects, hypertension, hypothalamic–pituitary–adrenal axis suppression and infections. 8 Thus, low bioavailability steroids with fewer and less severe side effects have been developed.9–11
Particularly, beclomethasone dipropionate (BDP) has anti-inflammatory effects in patients with UC, demonstrated in several trials,9,10 with low systematic bioavailability characteristics and with a predominantly colonic action. Budesonide, another topically acting corticosteroid, 11 is commercialized with three different formulations: two of them including a controlled-ileal release capsule and a pH-dependent capsule, which release the drug in the distal small intestine and right colon and mainly used in patients with Crohn’s disease, and a budesonide with a multi-matrix technology (budesonide MMX) releasing the drug throughout the entire colon. 11
Although the widespread use of these different low bioavailability steroids in mild-to-moderate UC, evidences on comparative effects between them and 5-ASAs in these patients are limited. Therefore, we performed a systematic review with meta-analysis evaluating the efficacy and safety of BDP and budesonide MMX compared with 5-ASAs or placebo, in patients with mild-to-moderate UC.
Methods
Search strategy and study selection
A search of the medical literature was conducted using MEDLINE (1946 to the 31 December 2022), EMBASE and EMBASE classic (1947 to the 31 December 2022), and the Cochrane central register of controlled trials (December 2022). Randomized controlled trials (RCTs) examining the efficacy and safety of BDP and budesonide MMX® compared with 5-ASAs or placebo, in adult patients (>90% of participants over the age of 16 years) with mild-to-moderate UC, were eligible for inclusion (Box 1).
Eligibility criteria.

Trials using BDP 5 or 10 mg, budesonide MMX 9 mg, and any dose of 5-ASAs were considered eligible. Studies had to report an assessment of achievement of remission in patients with mild-to-moderate UC at the last time point of assessment in the trial. Trials had to report one or more of the following endpoints: a composite of clinical and endoscopic remission; clinical remission or improvement; endoscopic remission; or histological remission. We planned to contact first and senior authors of the studies to provide additional information on trials, where required. Ethical approval for this evidence synthesis was not required.
Studies were identified with the terms ulcerative colitis or colitis (both as medical subject headings and as free-text terms). These were combined using the set operator AND with studies identified with the terms: mesalamine, mesalazine, aminosalicylic, 5-ASA, 5ASA, 5-aminosalicylic$, 5-aminosalicylate$, 5aminosalicylic$, 5aminosalicylate$, beclomethasone dipropionate, BDP, budesonide, and budesonide-MMX. There were no language restrictions. We screened the titles and abstracts of all citations identified by our search for potential suitability and retrieved those that appeared relevant to examine them in more detail. We performed a recursive search, using the bibliographies of all eligible articles. We translated foreign language articles, where required. If a study appeared potentially eligible, but did not report the data required, we planned to contact authors to obtain the Supplemental Material. We performed eligibility assessment independently. This was done by two investigators (B.B. and I.M.), using predesigned eligibility forms. We resolved any disagreements by consensus and measured the degree of agreement with a kappa statistic. The study protocol was not published in the PROSPERO international prospective register of systematic reviews. Ethical approval for this evidence synthesis was not required.
Outcome assessment
The primary outcomes assessed were the efficacy of BDP and budesonide MMX® compared with 5-ASAs or placebo, in terms of achieving clinical, endoscopic, and histological remission in patients with mild-to-moderate UC. Secondary outcomes included adverse events occurring due to therapy, including total numbers of adverse events, and adverse events leading to study withdrawal.
Data extraction
Data were extracted independently by two investigators (B.B. and I.M.) onto a Microsoft Excel spreadsheet (XP professional edition; Microsoft, Redmond, WA, USA) as dichotomous outcomes. Any disagreements were resolved by consensus. We extracted the following clinical data for each trial, where available: number of centers, country of origin, distribution of UC, endpoints used to define remission, dosage, route, schedule of the drug used, duration of therapy, and number of individuals incurring each (or any) of the adverse events of interest. Where individual trials used more than one endpoint to define remission, we extracted data separately for each of the endpoints reported. An analysis of treatment effect was performed on an intention-to-treat basis, considering dropouts and missing data as treatment failures.
Quality assessment and risk of bias
We used the Cochrane Risk of Bias tool to assess the quality of studies. 12 Two investigators (B.B. and I.M.) assessed study quality independently, with disagreements resolved by discussion. For all RCTs, we recorded the method used to generate the randomization schedule and conceal treatment allocation, whether participants, personnel, and outcome assessments were blinded, whether there was evidence of incomplete patient outcome data, and whether there was evidence of selective reporting of patient outcomes.
Data synthesis and statistical analysis
We pooled the proportion of patients randomized to placebo or active drug achieving remission. We used a random-effects model to pool data to provide a conservative estimate of the frequency of adverse events, according to the methodology of DerSimonian and Laird. 13 We assessed heterogeneity between studies using the I2 statistic, which ranges between 0% and 100%. We considered values of 25%–49%, 50%–74%, and ⩾75% to represent low, moderate, and high levels of heterogeneity, respectively. 14 We used StatsDirect version 3.2.7 (StatsDirect Ltd, Sale, Cheshire, England) to generate Forest plots of pooled prevalence and pooled odds ratio (OR) with 95% confidence intervals (CIs).
Results
The search strategy generated 1890 citations, 27 articles of which we retrieved for further assessment as they appeared to be relevant. In total, 8 of these articles, reporting 10 RCTs, fulfilled the eligibility criteria (Figure 1 and Supplemental Table 1).10,15–21 Out of them, two RCTs compared BDP 5 mg with 5-ASA,15,16 one compared BDP 10 mg with 5-ASA, 15 two RCTs compared BDP 5 mg with placebo,10,17 one compared BDP 10 mg with placebo, 17 two RCTs compared budesonide MMX 9 mg with 5-ASA,18,21 and six RCTs reported in four articles compared budesonide MMX 9 mg with placebo.18–21 Agreement between investigators for assessment of study eligibility was excellent (kappa statistic = 0.85). Characteristics of all included studies are reported in Table 1. Risk of bias for all included trials is reported in Table 2.
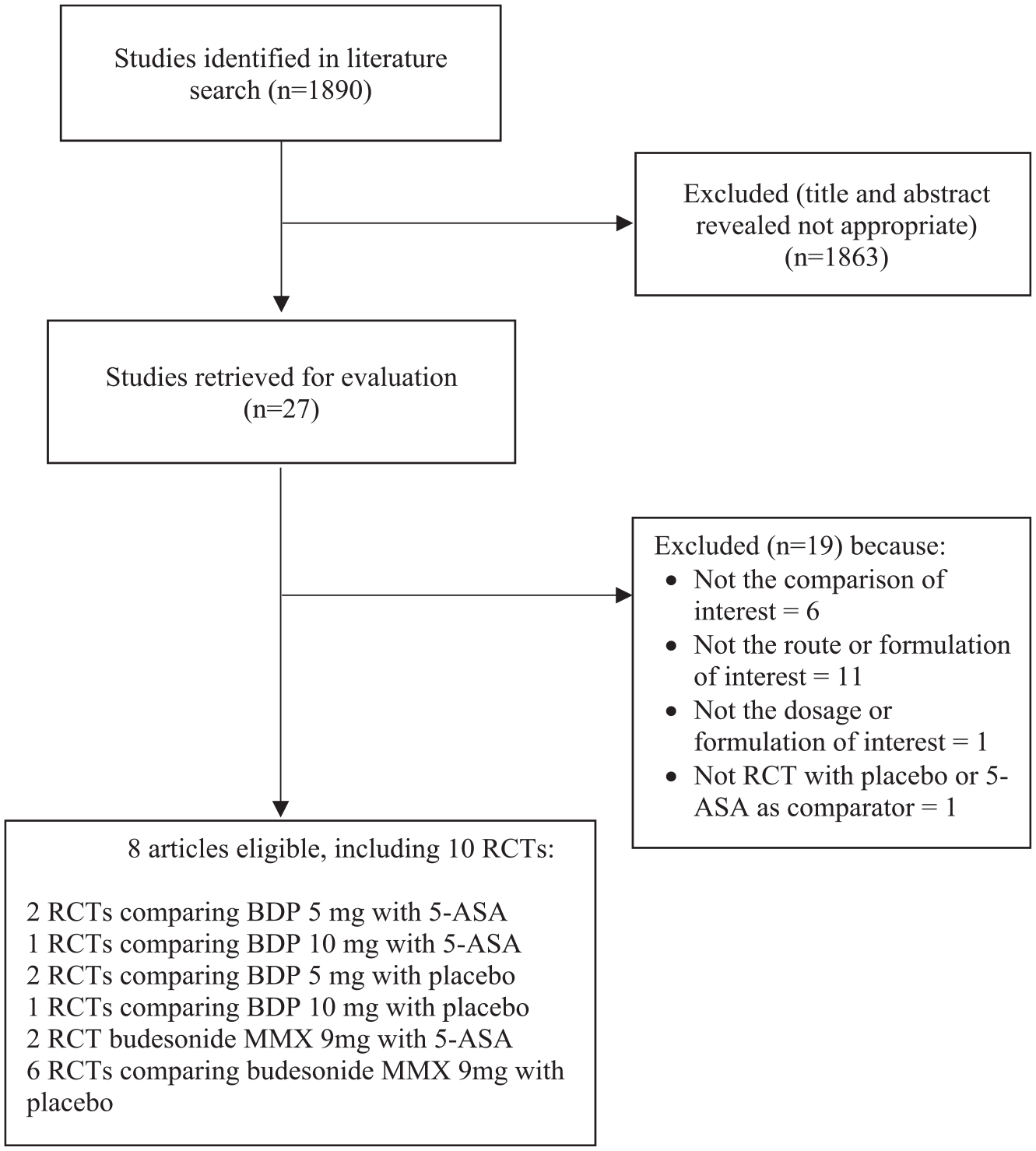
Flow diagram of assessment of studies identified in the network meta-analysis.
Characteristics of the studies included in the meta-analysis.

5-ASA, 5-aminosalicylic acid; BDP, beclomethasone; UC, ulcerative colitis; o.d., once daily; t.i.d., three times daily.
Risk of bias.

BDP versus placebo
In terms of clinical remission or improvement, both BDP 5 mg10,17 and BDP 10 mg 17 were more effective than placebo (OR 2.36, 95% CI 1.37–4.08, I2 = 0%, p = 0.37; OR 2.23, 95% CI 1.02–4.87, respectively) (Figure 2).

Forest plots of RCTs of oral BDP 5 or 10 mg versus placebo in inducing clinical remission or improvement, endoscopic remission, and histological remission.
Regarding endoscopic remission, as a separated outcome, one trial compared BDP 5 mg 10 with placebo demonstrating the superiority of the intervention arm (OR 2.70, 95% CI 1.28–5.67) (Figure 2). In the same study, no differences between BDP 5 mg and placebo were found in obtaining histological remission (OR 2.30, 95% CI 0.95–5.52) (Figure 2). 10
Budesonide MMX versus placebo
Budesonide MMX 9 mg was more effective than placebo in achieving clinical remission or improvement (OR 2.03, 95% CI 1.44–2.84, I2 = 51.9%, p = 0.06) in six RCTs reported in four articles (Figure 3).18–21 Three of these studies evaluated the efficacy of budesonide MMX 9 mg in achieving clinical and endoscopic remission, as a combined outcome, versus placebo (OR 2.65, 95% CI 1.52–4.63, I2 = 29.2%, p = 0.24) (Figure 3).18–20 Budesonide MMX 9 mg was superior to placebo also in achieving histological remission compared to placebo in five RCTs reported in four articles (OR 1.54, 95% CI 0.85–2.78, I2 = 55.8%, p = 0.06) (Figure 3).18–21 Four RCTs included in three articles evaluated endoscopic remission, as a separated outcome, finding a superiority over placebo of budesonide MMX 9 mg (OR 1.57, 95% CI 1.20–2.04, I2 = 0.0%, p = 0.94) (Figure 3).18,20,21

Forest plots of RCTs of oral budesonide MMX 9 mg versus placebo in inducing clinical remission or improvement, clinical and endoscopic remission, endoscopic remission, and histological remission.
BDP versus 5-ASA
In terms of achieving clinical remission or improvement, no differences were found between 5-ASA and BDP 5 mg (OR 0.90, 95% CI 0.51–1.57, I2 = 0%, p = 0.37) in two trials (Figure 4).15,16 One trial evaluated clinical remission or improvement in patients treated with BDP 10 mg compared to those treated with 5-ASA, founding no superiority of BDP 10 mg over 5-ASA (OR 1.54, 95% CI 0.42–5.64) (Figure 4). None RCT evaluated efficacy of BDP 5 or BDP 10 mg in obtaining endoscopic remission compared to 5-ASA. While one study 16 found no difference in achieving histological remission between BDP 5 mg and 5-ASA (OR 1.17, 95% CI 0.61–2.26) (Figure 4).

Forest plots of RCTs of oral BDP 5 or 10 mg versus 5-ASA in inducing clinical remission or improvement, histological remission.
Budesonide MMX versus 5-ASA
Two RCTs evaluated the efficacy of budesonide MMX 9 mg compared to 5-ASA.18,21 No differences were found in achieving clinical remission or improvement and endoscopic remission between budesonide MMX and 5-ASA in two trials18,21 or in clinical and endoscopic remission as a combined outcome in one trial 18 (OR 1.17, 95% CI 0.81–1.66, I2 = 0%, p = 0.75; OR 1.42, 95% CI 0.99–2.05, I2 = 0%, p = 0.96; OR 1.56, 95% CI 0.77–3.18, respectively) (Figure 5). In the same trials, however, 5-ASA was more effective than budesonide MMX 9 mg in achieving histological remission (OR 0.33, 95% CI 0.11–0.95, I2 = 0%, p = 0.99) (Figure 5).18,21

Forest plots of RCTs of oral budesonide MMX 9 mg versus 5-ASA in inducing clinical remission or improvement, clinical and endoscopic remission, endoscopic remission, and histological remission.
Safety
None of the active treatments were more likely to lead to adverse events, compared with placebo (OR 0.51, 95% CI 0.21–1.24 for BDP 5 mg10,17; and OR 1.15, 95% CI 0.87–1.53 for budesonide MMX 9 mg18–21). Likewise, both BDP 5 mg15,17 and budesonide MMX18,21 were as safe as 5-ASA (OR 1.10, 95% CI 0.07–17.87 and OR 0.79, 95% CI 0.57–1.13, respectively).
Finally, any of the intervention drugs led to adverse events causing withdrawals compared to placebo (OR 0.15, 95% CI 0.03–0.68 for BDP 5 mg 10 ; OR 0.95, 95% CI 0.64–1.42 for budesonide MMX 9 mg18–21). Moreover, there were no significant differences in withdrawals due to adverse events between budesonide MMX 9 mg and 5-ASA (OR 1.08, 95% CI 0.62–1.87).18,21
Discussion
In this systematic review with meta-analysis, we evaluated the efficacy and safety of BDP and budesonide MMX compared with 5-ASAs or placebo, in patients with mild to moderate UC. We found that, in terms of clinical remission or improvement, both BDP 5 mg and BDP 10 mg were more effective than placebo. In addition, BDP 5 mg was also more effective than placebo in achieving endoscopic remission, but not in obtaining histological remission. Budesonide MMX 9 mg was more effective than placebo in achieving clinical remission or improvement. Finally, it demonstrated superiority over placebo in achieving clinical and endoscopic remission as a combined outcome and endoscopic remission as a separate outcome, but not in achieving histological remission.
Regarding the comparisons with 5-ASA, we found no differences between BDP, 5 or 10 mg, and 5-ASA in achieving clinical remission or improvement or histological remission. Likewise, no differences were found between budesonide MMX and 5-ASA in achieving clinical remission or improvement, clinical and endoscopic remission, and endoscopic remission as a separate outcome. However, 5-ASA was more effective than budesonide MMX 9 mg in achieving histological remission. Overall, BDP, budesonide MMX 9 mg, and 5-ASA were safe and well tolerated.
We used rigorous methodology with two reviewers who independently performed the literature search, eligibility assessment and data extraction, with any discrepancies resolved by consensus. We included only RCTs excluding prospective or retrospective observational studies. To limit the risk of publication bias, we did not impose restrictions by language or year of publication and made attempts to identify all trials to obtain data which strengthened our meta-analysis.
Our study presents some limitations. The small number of the included trials limited our conclusions. In addition, only 4 out of 10 included RCTs were at low risk of bias. The comparison between BDP, budesonide MMX and 5-ASA was mainly limited to the induction phase because of the short follow-up times of our included studies (less than 8 weeks). Also, the criteria used to define the disease activity varied in each study. Moreover, there was a lack of uniformity of drug dosage and treatment duration among the various trials using 5-ASA and BDP; however, the same BDP dosage (5 or 10 mg) was used in all trials we chose to compare. Finally, in some included trials, BDP or budesonide MMX was administered in 5-ASA refractory patients, and one included trial was a comparison of BDP and placebo as add-on therapy to high-dose 5-ASA.
A previous network meta-analysis by Bonovas et al. 22 compared budesonide MMX or 5-ASA against placebo, or against each other, or different dosing strategies in patients with mild-to-moderate UC. The authors found that budesonide MMX (OR = 2.68; 1.75–4.10), 5-ASA > 2.4 g/day (OR = 2.75; 1.94–3.90), and 5-ASA 1.6–2.4 g/day (OR = 2.17; 1.55–3.05) showed higher efficacy than placebo. However, none of the comparisons of budesonide MMX versus 5-ASA > 2.4 g/day and 5-ASA 1.6–2.4 g/day was statistically significant. Moreover, serious adverse events occurrence was not shown to be statistically significantly different between budesonide MMX, 5-ASA > 2.4 g/day, 5-ASA 1.6–2.4 g/day, and placebo. 22
A recent network meta-analysis comparing oral sulfasalazine, 5-ASA [low dose (<2 g/day), standard dose (2–3 g/day), or high dose (>3 g/day)], controlled ileal-release budesonide or budesonide MMX, alone or in combination with rectal 5-ASA therapy, and compared to each other or placebo in patients with UC, demonstrated that budesonide MMX was not more effective than combined oral and rectal 5-ASA or high-dose mesalamine and has inferior tolerability. 23 Another meta-analysis by Manguso et al. 24 published in 2016 and including five RCTs showed that BDP 5 mg was superior to 5-ASA in achieving clinical remission or clinical improvement considered separately, albeit the authors included one trial comparing BDP versus 5-ASA as add-on therapy with prednisone (OR 1.30, 95% CI 0.76–2.23 and OR 1.41, 95% CI 1.03–1.93, respectively).
However, in clinical practice, if a patient does not respond to induction treatment with 5-ASA, oral steroids are usually the next step. Papi et al., 25 in a study administering oral BDP 10 mg/day for 4 weeks followed by a 4-week administration of 5 mg/day in 64 mild-to-moderate UC patients with a previously 5-ASA treatment failure, found a remission rate of 75% with most patients achieving 1-year maintenance of remission with no need for further steroid treatment. 25 These data support the crucial role of oral BDP as an alternative therapy to systemic steroids in patients with a mild-to-moderate flare of the disease that is not responsive to 5-ASA. Moreover, these evidences suggest that further larger randomized studies comparing low bioavailability steroids with 5-ASA are needed.
Therefore, our findings confirm what has already been shown by the previous scientific literature: BDP and budesonide MMX are effective therapies in patients with mild-to-moderate UC compared to placebo, however, the comparative analyses did not demonstrate the superiority of these drugs over 5-ASA. The review of the literature showed that there are not RCTs comparing BDP and budesonide MMX therapies with each other. Moreover, very little research on these drugs in UC has been conducted so far; therefore, this also makes difficult building network meta-analysis with direct and indirect comparisons. Further randomized controlled double-blind trials comparing the two drugs each other are necessary to clarify the exact role of these treatments in patients with mild-to-moderate UC.
Supplemental Material
sj-docx-1-tag-10.1177_17562848231188549 – Supplemental material for Efficacy and safety of oral beclomethasone dipropionate and budesonide MMX versus 5-aminosalicylates or placebo in ulcerative colitis: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tag-10.1177_17562848231188549 for Efficacy and safety of oral beclomethasone dipropionate and budesonide MMX versus 5-aminosalicylates or placebo in ulcerative colitis: a systematic review and meta-analysis by Brigida Barberio, Ilaria Marsilio, Andrea Buda, Luisa Bertin, Gianluca Semprucci, Annalisa Zanini, Martina Crepaldi, Fabiana Zingone and Edoardo Savarino in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
