Abstract
Background:
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic has complicated the management of inflammatory bowel diseases (IBD).
Objectives:
This study aimed to assess the efficacy of different anti-SARS-CoV-2 vaccines under different treatments in IBD patients and identify predictive factors associated with lower serological response, including anti-tumor necrosis factor (anti-TNF) drug levels.
Design:
A prospective, double-center study of IBD patients was conducted following messenger ribonucleotide acid (mRNA) and non-mRNA anti-SARS-CoV-2 vaccination.
Methods:
Healthy control (HC) patients were enrolled to reduce bias. Baseline and control samples were obtained 14 days after the second dose to assess the impact of conventional and biological treatments. Clinical and biochemical activity, serological response level, and anti-TNF drug levels were measured.
Results:
This study included 199 IBD (mean age, 40.9 ± 12.72 years) and 77 HC participants (mean age, 50.3 ± 12.36 years). Most patients (76.9%) and all HCs received mRNA vaccines. Half of the IBD patients were on biological treatment (anti-TNF 68.7%). Biological and thiopurine combined immunomodulation and biological treatment were associated with lower serological response (p < 0.001), and mRNA vaccination promoted better antibody levels (p < 0.001). Higher adalimumab levels caused lower serological response (p = 0.006). W8 persistence of anti-SARS-CoV-2 level was equal in IBD and HC groups. Vaccination did not aggravate clinical disease activity (p = 0.65).
Conclusion:
Anti-SARS-CoV-2 vaccination is considerably efficacious in IBD patients, with mRNA vaccines promoting better antibody levels. The negative impact of combined biological treatment, especially with high adalimumab drug levels, on serological response to vaccination should be considered. Although midterm durability of vaccination is encouraging, more data are needed to expand the existing understanding on this issue.
Plain Language Summary
Adjustment of COVID-19 vaccination to adalimumab trough level is considerable due to the reduced serological response. mRNA vaccination should be preferred in case of IBD patients with an equal durability of anti-SARS-CoV-2 level of subjects and healthy control participants.
Introduction
The Coronavirus Disease 2019 (COVID-19) pandemic caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has raised issues in the management of inflammatory bowel diseases (IBD), namely ulcerative colitis (UC) and Crohn’s disease (CD). 1 Concerns regarding the outcome of COVID-19 in relation to IBD or an immunocompromised state emerged first.2,3,4 A significant proportion of IBD patients are considered to be immunosuppressed. Patients receiving >20 mg of corticosteroids, thiopurines, or biologics clearly belong to this group, although malnourished patients can also be considered to have a higher risk for infections.5,6 While clinical trials investigating the efficacy of vaccines against SARS-CoV-2 exclude immunosuppressed patients, all professional organizations recommend all types of COVID-19 vaccinations for patients with IBD.7,8,9 A population-based vaccination program involving the adenovirus vector vaccine (Sputnik®, Gamaleya Research Institute of Epidemiology and Microbiology) and inactivated virus vaccine (Sinopharm®, Sinopharm’s Beijing Institute of Biological Products) started relatively early in Hungary, with messenger ribonucleotide acid (mRNA) vaccines following a little later. However, while most gastroenterologists advised patients to receive mRNA-based vaccination, a portion of IBD patients received a vector vaccine or inactivated virus vaccine at the early stages of the national vaccination program. 10 There was some uncertainty regarding the efficacy of these vaccines among both patients and treating physicians, considering that the European Medical Agency had yet to approve their administration when they were first available.
Recently published meta-analyses have investigated the effectiveness, safety, and durability of anti-SARS-CoV-2 vaccination among IBD patients. Accordingly, Bhurwal et al. reported a cumulative seroconversion rate of 96.8% after the second vaccine dose. 11 Moreover, they found no significant differences in serological response according to the ongoing treatment. The same study, which collected the results of 21 articles mostly on mRNA- and vector-type vaccines, found an adverse event rate of 0.09%, most of which were mild. Jena et al. analyzed available literature regarding the durability of anti-SARS-CoV-2 vaccination in IBD patients in addition to the efficacy. 12 After synthesizing data from 46 studies with more than 9000 IBD patients, they found an overall seroconversion rate of 96% after complete vaccination based on around 31 eligible articles. Subgroup analysis according to vaccine type showed a 96–98% and 78–90% seroconversion rate in cases receiving mRNA and vector vaccines. Based on nine enrolled studies, they concluded that titers decrease 4 weeks after anti-SARS-CoV-2 vaccination.
Although the large body of clinical data published to date have answered many questions, to our knowledge, few data exist on the relationship between serum levels of biological therapy (BT) and the rate of seroconversion. Data regarding non-mRNA vaccinations among immunosuppressed IBD patients are lacking, with no direct comparison having been performed. Moreover, it remains unknown whether any predictors of immune response might influence vaccination strategy in this cohort. Patients are often hesitant regarding vaccine administration owing to fears, based on perceived or real information or experience, that the vaccine will result in disease flare-ups. Finally, given that only a few studies have used adequate controls, comparative data on the durability and persistence of responses between immunosuppressed patients and healthy controls (HCs) have been lacking.
Aims
We conducted a double-center, prospective cohort study to compare the level of the seroconversion after vaccination with different types of COVID-19 vaccines in IBD patients. Our primary objective was to investigate the serological response and persistence in the levels of specific anti-SARS-CoV-2 S antibodies after the administration of different SARS-CoV-2 vaccines in IBD patients receiving various therapeutic agents and compared them to those in HCs. Our secondary objective was to identify clinical and biochemical predictors associated with inadequate serological response. Furthermore, we aimed to assess the effects of vaccination on disease activity. Our tertiary objective was to investigate serum anti-TNF levels and serological response rates in IBD patients following the administration of different vaccines against SARS-CoV-2.
Materials and methods
Study design and settings
We conducted a double-center, prospective cohort study between March 2021 and February 2022. at the University of Szeged and Semmelweis University, Hungary. Patients who satisfied the inclusion/exclusion criteria were divided into four groups according to their treatment. A distinction was made between those receiving combined biologic and immunosuppressive therapy (COMB), those receiving biologic (BT) or immunosuppressant monotherapy [azathioprine (AZA)] and those who did not receive either biologic or immunosuppressive therapy (NONE). Eligible patients were requested to visit for blood sampling within 24 h after their first vaccination. Patients received the second dose of the vaccine at intervals specified in the vaccine’s label. Serological testing of each subject was performed 14 days after their second vaccination. The reporting of this study conforms to the STROBE statement. 13
Participants
Consecutive patients diagnosed with IBD who visited the outpatient department were enrolled in this study. Healthy subjects included in the H-UNCOVER randomized trial were used as the control group. 14 Participants with a history of COVID infection and those <18 years of age were excluded: blood tests were performed at the scheduled clinical control, during which a serological test was performed to rule out previous asymptomatic COVID infection. Participation was voluntary, and data were collected anonymously. Subjects consented to the use of data only for scientific purposes.
Data source and measurements
Demographic and clinical data were obtained at baseline. Sex, age at inclusion, type of IBD, ongoing treatment, disease classification (according to Montreal definitions 15 ), and disease activity were recorded. Clinical activity was assessed using the Crohn’s Disease Activity Index (CDAI 16 ) and pMayo score (pMayo 17 ) in UC patients. Biochemical activity was assessed using C-reactive protein (CRP) measurements. Extended disease was defined as bowel involvement proximal to the rectum in cases with UC and as at least two involved segments in cases with CD. The type of vaccine was registered, and serum infliximab or serum adalimumab concentrations were determined at this time point.
The anti-SARS-CoV-2 S (S) antibody levels were measured using the Elecsys Anti-SARS-CoV-2 Spike Antibody Immunoassay® (Roche®, Basel, Switzerland), with the cutoff value set at 0,8 U/mL according to the manufacturer’s protocol. The assay had a sensitivity of > 99.5% for confirming SARS-CoV-2 infection on the 14th day following polymerase chain reaction (PCR) as per the product’s label.
Serum infliximab (IFX; #Ridascreen IXF Monitoring®, R-Biopharm®, Darmstadt, Germany) and adalimumab (ADA; #Ridascreen ADM Monitoring®, R-Biopharm®, Darmstadt, Germany) concentrations were determined using the ELISA method as per the manufacturer’s protocol (R-Biopharm®, Darmstadt, Germany). The sensitivity of the IFX and ADA assays was <1 ng/mL, respectively. The intra- and inter-assay coefficients of variation for both assays were <15%.
Additional data were obtained by conducting two sub-analyses at weeks 4 and 8 following the second anti-SARS-CoV-2 vaccination in one of our tertiary referral centers (University of Szeged). We assessed persistence of serological response and the effect of vaccination on disease activity during the follow-up period. Anti-SARS-CoV-2 S antibody levels were measured at baseline (before vaccination) and 4 and 8 weeks after the second vaccination. In this sub-analysis, clinical activity was interpreted using the Physician’s Global Assessment (PGA) score. 18 Clinical and biochemical activities were compared at baseline and again 8 weeks after the second dose.
Statistical analysis
Statistical analysis was performed via IBM SPSS software (IBM SPSS Statistics for Windows, Version 26.0, IBM Corp., Armonk, NY, USA). Normality was tested using visual interpretations. Descriptive statistics were interpreted as mean ± standard deviation of the mean (SD) for continuous variables and count + percentages for categorical variables. After checking assumptions, the Welch test or Mann–Whitney test and Kruskal–Wallis test were applied to compare groups described with continuous variables. Significance values had been adjusted using the Bonferroni correction for multiple tests. On the other hand, groups described with categorical variables were compared using the chi-squared test and Fisher’s exact test. A p value of <0.05 indicated statistical significance. To reduce bias, propensity score matching (using age, sex, and type of vaccine as variables) was used to select HC patients.
To examine predictive factors associated with serological response, linear regression models were constructed using age, BT, vaccine type, disease type, concomitant corticosteroid treatment, disease duration, extended disease, and clinical and biochemical activities as variables. Linear regression models were constructed to assess the relationship between anti-TNF drug levels and serological response.
To measure serological persistence, the Welch test was used based on ln + 1 values of anti-SARS-CoV-2 S antibody levels.
Results
Baseline characteristics
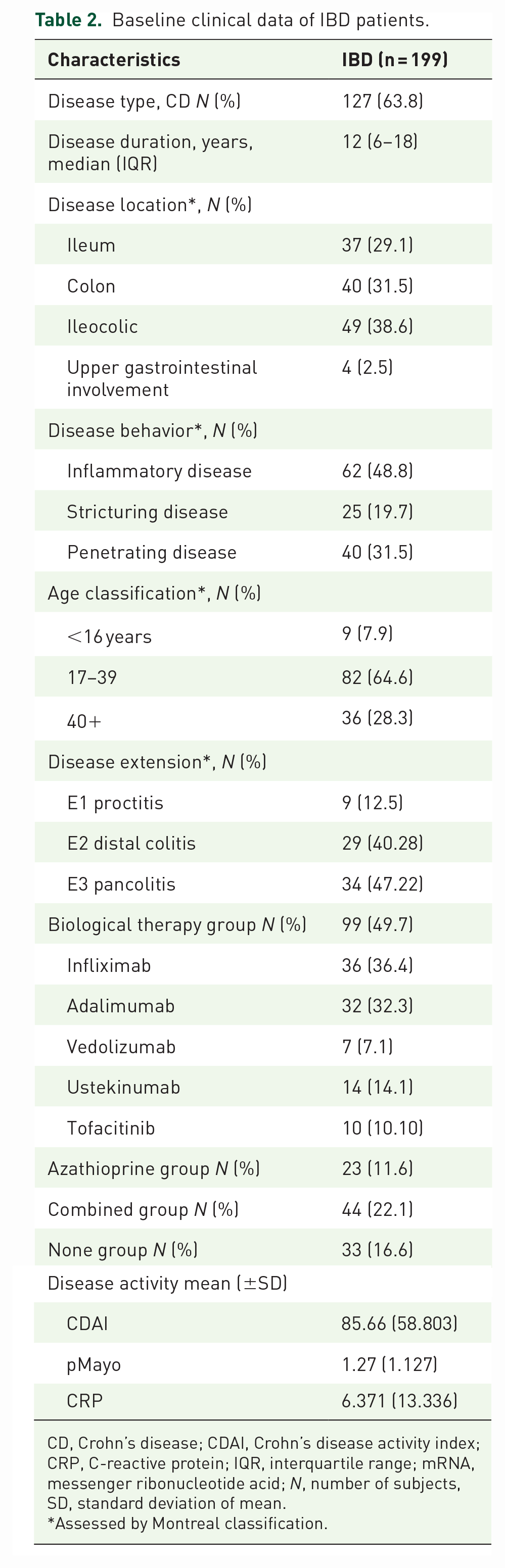
This study included 199 IBD patients (male/female ratio 95/104, mean age 40.9 ± 12.72 years). More patients had CD than UC (n = 127, 63.8% versus n = 72, 36.2%). Moreover, propensity score matching from a database including 105 patients was used to select 77 HCs. The HCs were older than IBD patients (50.3 ± 12.36 versus 40.94 ± 12.72 years; p < 0.001). Most of the patients received mRNA-type vaccines (n = 153, 76.9%), whereas 46 patients (23.1%) received non-mRNA vaccines. Most of the HC participants received mRNA-type vaccines (n = 66, 85.7%). Baseline demographic data and type of vaccinations are shown in Tables 1 and 2.
Baseline demographic data of IBD patients.

SD, standard deviation of mean; N, number of subjects; mRNA, messenger ribonucleotide acid.
Baseline clinical data of IBD patients.
CD, Crohn’s disease; CDAI, Crohn’s disease activity index; CRP, C-reactive protein; IQR, interquartile range; mRNA, messenger ribonucleotide acid; N, number of subjects, SD, standard deviation of mean.
Assessed by Montreal classification.
In total, 63.8% of the patients had CD, with most cases having ileocolonic localization and inflammatory phenotype (38.6% and 48.8%, respectively). Almost half of the UC patients had pancolitis (47.22%). Moreover, 49.7% of the patients were in the BT group, and more than two-thirds of them were on anti-TNF therapy (68.7%). In total, 11.6% of the patients received azathioprine as monotherapy (AZA group), 22.1% received it in combination with biological agents (COMB group), and 16.6% received neither biologics nor azathioprine (NONE group). Based on the clinical activity indexes, most of the CD patients were in clinical remission (mean CDAI 85.66 ± 58.8), whereas UC patients showed remission to mild disease activity (mean pMayo 1.27 ± 1.3; Table 2.).
Serological response to vaccination across different groups
Following all-type and mRNA vaccinations, anti-SARS-CoV-2 S antibody levels were significantly higher in the NONE group (p < 0.001); however, no significant difference between the groups was observed among cases receiving non-mRNA vaccination (p = 0.447). Further details are available in Table 3.
Serological response in different groups.

AZA, azathioprine; BT, biological therapy; COMB, biological therapy and azathioprine combination; HC, healthy control; mRNA, messenger ribonucleotide acid; n, number of subjects; SD, standard deviation of mean.
Anti-SARS-CoV-2 S antibody titers in patients showed a decreasing trend in the following order of treatment: AZA, HC, BT, and combined biologic and immunosuppressive therapy (mean values of mRNA vaccination subgroup: NONE group: 8179 U/mL, AZA group: 4880 U/mL, HC group: 1931 U/mL, BT group: 1861 U/mL, COMB: 1624.5 U/mL; p < 0.001). Anti-SARS-CoV-2 S antibody levels were significantly higher in the NONE group compared to the BT group (p = 0.003), COMB (p < 0.001), and HC (p < 0.001). Post hoc analysis was performed to determine the effect of vedolizumab (VDZ) and ustekinumab (UST) compared to the NONE group in serological response, which revealed no significant differences (p = 0.698). Analysis of TOFA versus NONE group was not performed due to the limiting effect of the size of TOFA group. Other comparisons did not show any significance. Table 3 and Figure 1 provide further data regarding the serological response to vaccination.

Comparison of anti-SARS-CoV-2 S antibody levels following mRNA vaccinations between groups. Anti-SARS-CoV-2 S antibody levels of the NONE group were significantly higher compared to the BT (p = 0.003), COMB (p < 0.001), and HC groups (p < 0.001). No significant difference was observed between the NONE and AZA groups (p = 0.99).
mRNA vaccination (p < 0.05) promoted better serological reponse compared to non-mRNA vaccination (p = 0.571) in all cases except the vedolizumab treatment group.
According to our model, mRNA vaccines were associated with higher serological response (B = −0.523; p < 0.001). In addition, age had a negative impact on anti-SARS-CoV-2 S antibody levels (B = −0.169; p = 0.014), and biological treatment was associated with lower serological response (B = −0.163; p = 0.016). Clinical and biochemical (CRP and lymphocyte count) activities or disease type did not influence anti-SARS-CoV-2 S antibody levels according to the same model. Concomitant corticosteroid usage, disease duration, and disease extent had no significant impact on serological response (B = −0.130, p = 0.074; B = −0.102, p = 0.205; B = 0.017, p = 0.813). Coupling data are shown in Tables 3–4. Model details with selected variables are available in Table 5.
Anti-SARS-CoV-2 (S) levels according to the type of the vaccine across different treatments.

BT, biological therapy; AZA, azathioprine; COMB, biological therapy and azathioprine combination; HC, healthy control; n, number of subjects; SD, standard deviation of mean.
Linear regression model to assess higher serological response. Model summary: R = 0.627, R2 = 0.393, F = 12.779, and p < 0.001.

B, standardized coefficients beta; CD, Crohn’s disease; CI, Confidence interval; CRP, C-reactive protein; UC, ulcerative colitis.
Serological response and anti-TNF serum level
80.0% of patients receiving ADA and 76.7% of the IFX group have been vaccinated with mRNA vaccination. No difference was observed in the distribution of patients between vaccinations groups (χ2 = 0.12; p = 0.73); therefore, we assessed the impact of the serum IFX and ADA levels on anti-SARS-CoV-2 S antibody titers.
Accordingly, we found no significant correlation between serum IFX levels and serological response (B = 0.332; p = 0.078). However, higher ADA levels were associated with lower anti-SARS-CoV-2 S antibody levels (B = −0.404; p = 0.006). Significant data are summarized in Table 6. Figure 2 shows the connection between adalimumab drug level and serological response.
Linear regression model to assess serological response influence of ADA level. Model summary: n = 45, R = 0.404, R2 = 0.163, F = 8.395, and p < 0.001.

B, standardized coefficients beta; CI, confidence interval.

Linear regression model regarding ln logarythm of adalimumab trough level and serological response. Higher adalimumab level was associated with reduced serological response to anti-SARS-CoV-2 vaccination (B = −0.404, p = 0.006).
Persistence of SARS-CoV-2 S antibody levels following mRNA vaccination
Based on the results of our single center sub-analysis, follow-up data of 100 participants were collected (IBD n = 61, HC n = 39) after mRNA vaccination. Age was statistically similar in both groups (p = 0.53). No significant difference was observed between the IBD and HC groups either before the second dose (p = 0.091) or at weeks 4 (p = 0.084) and 8 (p = 0.953) after the second dose of the vaccine. Coupling data are detailed in Table 7 and Figure 3.
Anti-SARS-CoV-2 levels during follow-up among IBD and HC participants who received mRNA vaccines.

HC, healthy control; SD, standard deviation of mean.
Comparisons of groups based on ln + 1 values.

Persistence of anti-SARS-CoV-2 S antibody levels during follow-up period in IBD patients after mRNA vaccination. No significant difference was observed before (p = 0.091), after 4 weeks (p = 0.084), and after 8 weeks (p = 0.953) of the second vaccine dose.
Impact of anti-SARS-CoV-2 vaccination on disease activity
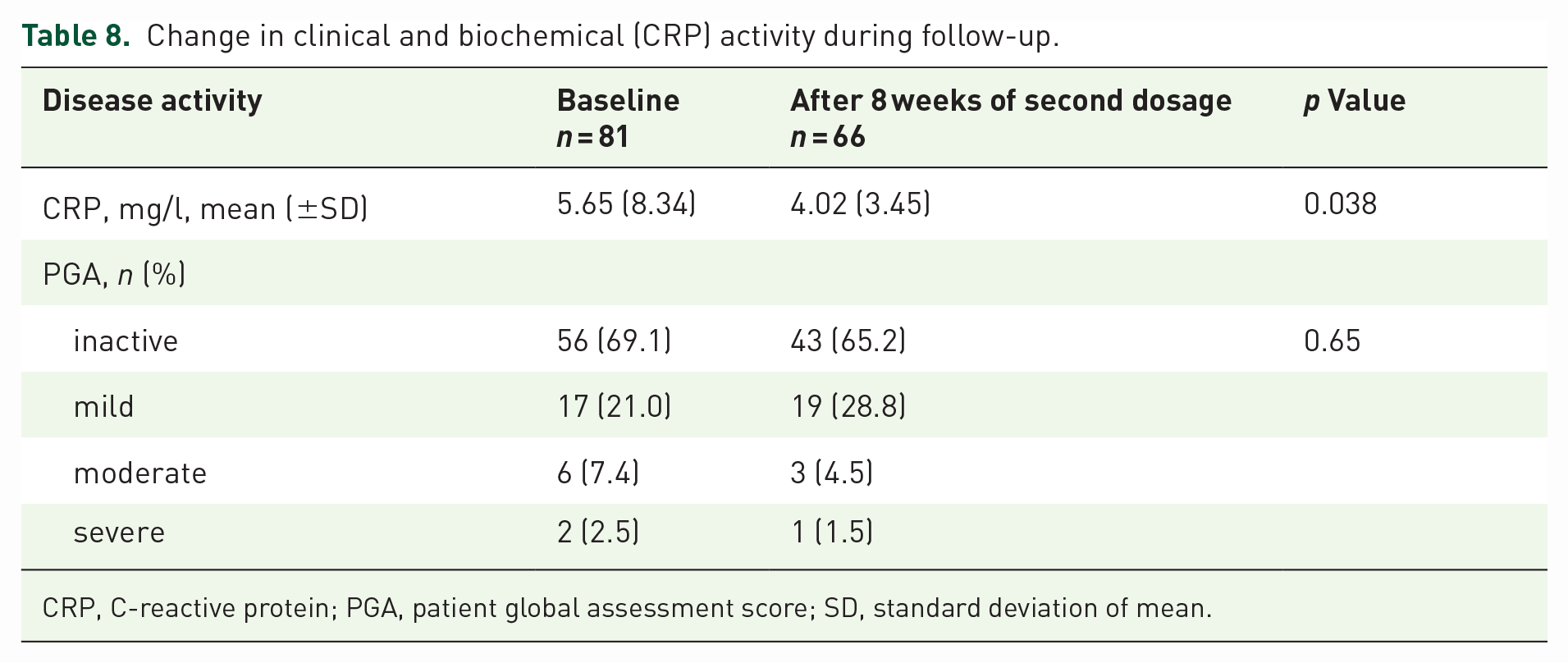
Follow-up data for 81 and 66 IBD participants were analyzed at baseline and 8 weeks after the second dose of anti-SARS-CoV-2 vaccination. CRP levels, a marker of biochemical activity, significantly decreased from a mean baseline level of 5.65 ± 8.34 mg/L to a mean level of 4.02 ± 3.45 mg/L at week 8 after the second vaccine dose (p = 0.038). No significant difference in clinical disease activity was observed between baseline and follow-up measurements (0.43 ± 0.74 and 0.41 ± 0.61; p = 0.65). Related data are summarized in Table 8.
Change in clinical and biochemical (CRP) activity during follow-up.
CRP, C-reactive protein; PGA, patient global assessment score; SD, standard deviation of mean.
Discussion
The current study focused on the serological response after SARS-CoV-2 immunization considering following issues. Contradictory and limited data have been available among immunocompromised patients after anti-SARS-CoV-2 immunization. To our knowledge, our prospective cohort analysis has been the first unique study to compare different types of vaccines (mRNA and non-mRNA) and biological and/or immunosuppressive treatment on serological response in a well-defined cohort.
Our double-center, prospective cohort study analyzed the data of 199 IBD and 77 HC participants. Based on Hungarian IBD recommendations, most patients received mRNA-type anti-SARS-CoV-2 vaccines; however, to compare our findings to internationally existing data, we also analyzed the non-mRNA vaccines. In accordance with clinical practice guidelines, anti-TNF therapy was the most common biological treatment among our enrolled patients.19,20
Our study confirmed that the mRNA vaccines promoted superior seroconversion levels than did non-mRNA vaccines among immunocompromised IBD patients. Comparing non-mRNA vaccinations, no difference was observed between attenuated adenovirus vaccines and inactivated whole virion vaccines, according to an Indian prospective cohort study. 21 However, data are still lacking regarding non-mRNA vaccinations in IBD populations. Among IBD patients, ongoing biological, and/or immunomodulatory treatment definitely resulted in lower antibody response. Although higher ADA levels were associated with lower serological response, no significant difference was observed in cases receiving IFX. The durability of anti-SARS-CoV-2 S antibody levels did not differ between the IBD and HC groups 8 weeks after the administration of mRNA vaccines. Our results suggest no causal relationship between disease flares and immunization based on clinical and biochemical parameters.
Several studies have analyzed the possible effects of biologic treatments on seroconversion in IBD patients due to both infection and vaccination. A multicenter prospective observational cohort study in the United Kingdom (CLARITY) had shown that IFX significantly attenuated seroprevalence, seroconversion, and the magnitude of anti-SARS-CoV-2 antibody reactivity after SARS-CoV-2 infection, especially in the combination therapy group, compared to vedolizumab. 22 A retrospective study in the same population showed that the negative effects of ADA on serological response were similar to those of IFX. In cases with undetectable TNF inhibitor levels, the seropositivity rate was comparable to vedolizumab. 23
However, more data have emerged in the past few months. After recruiting consecutive IBD patients, the CLARITY study investigated the immunogenicity to BNT162b2 and ChAdOx1 nCoV-19. Unsurprisingly, IFX therapy promoted a lower serologic response not only after infection but also after a single vaccine dose, especially in those receiving combination treatment, compared to vedolizumab monotherapy. This effect was blunted after the second vaccine dose. 22
Another prospective multicenter study from Israel found that although two doses of BNT162b2 seroconverted all IBD patients, TNF inhibitor therapy resulted in significantly lower anti-SARS-CoV-2 S Ig antibody levels. Older age also independently showed a negative association with anti-SARS-CoV-2 S Ig antibody levels. 24
In our cohort, both biological treatment and combined therapy were associated with lower serological response compared to AZA and patients without ongoing treatment, however significant differences were not proved during VDZ and UST treatment. Although, the low number of patients in VDZ/UST groups should be enhanced during interpretation of the results. The difference between ongoing treatments was more prominent in participants receiving mRNA-type vaccines. Our post hoc analysis showed similarity of serological response between ustekinumab/vedolizumab and the NONE group which highlights the dissimilarity of different biological agents. However, interpretation of data is limited by low sample sizes in each treatment groups.
Notably, the serological response was higher in the NONE group compared to the HC group. A possible explanation for this phenomenon could be the significantly higher age in the HC group, in accordance with the study mentioned above. 24 This relation highlights the potential role of age regarding serological response.
The VARIATON study investigated the effects of mRNA (BNT162b2, CX-024414) and vector (ChAdOx1 nCoV-19, Ad26.CoV2.S) vaccines in IBD patients. 25 SARS-CoV-2 S antibody levels were significantly higher after two doses of mRNA vaccines than after administration of vector vaccines. Interestingly IBD itself proved to have a negative impact on anti-spike protein IgG levels. Anti-TNF, anti-IL 12/23 therapy, and Janus kinase (JAK) inhibitors were associated with significantly lower median SARS-CoV-2 levels compared to patients receiving 5-aminosalicylates (5-ASA), immunomodulators, or steroids or no medication. Older age and TNF inhibitor therapy were independent negative confounding factors in the IBD group. No significant difference was observed between TNF inhibitor monotherapy and combination therapy.
Results from a single tertiary IBD center that compared the effects of two doses of mRNA BNT162b2 (Comirnaty; Pfizer-BioNTech, USA), mRNA CX-024414 (Spikevax; Moderna, Cambridge, Massachusetts, USA), or vector ChAdOx1 nCoV-19 (Vaxzevria; AstraZeneca, UK) vaccines on serological response showed that neither biological monotherapy (IFX, ADA, vedolizumab, ustekinumab) nor trough levels were associated with lower SARS-CoV-2 IgG antibody levels. In contrast, variables, such as older age or the combination of biological and immunosuppressive treatment, were identified as negative confounding factors. The lowest antibody levels were found in patients receiving TNF inhibitor and concomitant immunosuppressive treatment (azathioprine/methotrexate). The vector vaccine Vaxzevria was unable to promote seroconversion in 2.2% of IBD patients and induced significantly lower levels of antibodies either in IBD patients or the control group compared to mRNA vaccines. 26
Our data showed that mRNA vaccines were superior to non-mRNA types in all groups, excluding vedolizumab treatment. However, the low number of patients receiving VDZ precluded us from drawing significant conclusions. In line with existing international data, our study confirmed the negative effects of older age, combined biological treatment and non-mRNA vaccines on serological response.26,27 Based on our results, we therefore highlight the importance of treatment over disease activity on antibody response.
A study by Edelman–Klapper revealed no correlation between anti-TNF drug levels and serological response. 24 Our data showed that higher ADA serum levels had a negative effect on anti-SARS-CoV-2 S antibody levels; however, no correlation was observed in cases who received IFX treatment, consistent with the previous study. Our possible hypothesis for this discordance is that the dosage regimen during ADA therapy provides relatively stable drug levels in contrast to IFX, which promotes alternating serum levels. Study protocol did not allow to standardize the time of the sampling of the drug levels due to the real-world setting.
In the current study, we revealed that anti-SARS-CoV-2 S antibody levels persisted for up to 8 weeks after the second dose of the mRNA vaccine. We found no difference between IBD and HC participants during the follow-up period, in contrast to the data published in a few existing studies.28,25 Our analysis revealed that vaccination had no significant impact on clinical disease activity based on PGA. Although a statically significant decrease in biochemical activity was observed during follow-up, no clinically significant decrease was noted. A multicenter study by Lev-Tzion et al. showed similar exacerbation rates after vaccination between vaccinated and non-vaccinated IBD patients. 29
The strength of the current analysis is our double-center, prospective setting with a relatively high number of enrolled patients. Only a few studies have examined the possible correlation between anti-TNF drug levels and serological response. Multivariable analysis has allowed us to review multiple connections. Furthermore, during the study period, Hungary was characterized as one of the countries with highest COVID-19 incidence rates both in Europe and the world, resulting in ingenuous and objective patient selection and enrolment. Notably, only mRNA vaccinations were available in most of the European countries during this period; thus, studies only reported on such vaccines.
The pandemic situation overruled some viewpoints on scientific methodology, resulting in certain limitations in the current study. Testing of serological and therapeutic drug levels in anti-TNF-treated patients was performed at the day of the first vaccination according to the Hungarian immunization protocol, regardless of the treatment cycle. Separated analysis of VDZ, UST, and TOFA groups were not performed due to the low number of patients and potentially misleading results. Biochemical activity was measured by CRP due to its excessive availability; however, fecal calprotectin could provide more accurate data. Almost three times more patients received mRNA vaccines compared to those who received non-mRNA vaccines, which could potentially distort the interpretation of our findings. The proportion of patients enrolled in the study subgroups differed, reflecting the financial protocols in Hungary. Propensity score matching could result in statistical bias during HC selection process.
Based on our double-center, prospective study, anti-SARS-CoV-2 vaccination has considerable efficacy in IBD patients, with mRNA-type vaccines being superior to non-mRNA vaccines. The negative impact of combined biological treatment, especially with high adalimumab drug levels, on serological response to vaccination should be considered with adjustment of vaccination to adalimumab trough level. Midterm durability of vaccination is encouraging; however, more data are needed to expand our existing data in the field of this issue.
Supplemental Material
sj-docx-1-tag-10.1177_17562848231183529 – Supplemental material for Nationwide experiences with trough levels, durability, and disease activity among inflammatory bowel disease patients following COVID-19 vaccination
Supplemental material, sj-docx-1-tag-10.1177_17562848231183529 for Nationwide experiences with trough levels, durability, and disease activity among inflammatory bowel disease patients following COVID-19 vaccination by Tamás Resál, Péter Bacsur, Miklós Horváth, Kata Szántó, Mariann Rutka, Anita Bálint, Anna Fábián, Renáta Bor, Zoltán Szepes, János Fekete, Klaudia Farkas, Pál Miheller and Tamás Molnár in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
Writing support was provided by Enago and funded by Klebelsberg Library, University of Szeged. The authors are grateful to Csilla Tóth-Káli, Gabriella Pócsik, Brigitta Csorba, and Krisztina Ördög for study support (Department of Medicine, University of Szeged).
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
