Abstract
Pancreatic fluid collection often occurs as a local complication of acute pancreatitis, and drainage is indicated in symptomatic patients. The drainage may be surgical, percutaneous, or endoscopic ultrasound (EUS) guided. In symptomatic collections older than 4 weeks and localized in the upper abdomen, EUS-guided drainage is the first choice of treatment. Lumen-apposing metal stents are useful in cases of walled-off necrosis, facilitating access to the cavity; however, they do not reduce the number of necrosectomy sessions required. In most pancreatic pseudocysts requiring drainage, plastic stents remain the first choice of treatment. This review aimed to summarize the principles and techniques of step-up therapy of pancreatic fluid collections, including preprocedural and postprocedural assessment and practical approaches of drainage and necrosectomy, making available evidence more accessible to endoscopists aiming to train for this procedure. Successful and safe EUS drainage connotes early recognition and treatment of complications and the presence of a multidisciplinary team for optimal patient management. However, the best time for necrosectomy, modality of drainage method (lumen-apposing metal stents or plastic stents), and duration of antibiotherapy are still under evaluation.
Plain language summary
This review summarizes the current knowledge on the indications, techniques, outcomes, complications, and treatment-associated EUS drainage of pancreatic fluid collections. Special attention is paid to the practical approach in necrosectomy and postprocedural antibiotherapy, discontinuation of proton pump therapy, and timing of stent removal.
Keywords
Introduction
Natural history of pancreatic fluid collections
According to the Atlanta classification, 1 only 30% of acute fluid collections after interstitial edematous pancreatitis (within 4 weeks from onset) will develop pancreatic pseudocysts (PPs) more than 4 weeks from onset 2 ; however, only 10% will need drainage. 3 Approximately 50% of acute fluid collections after necrotizing pancreatitis (within 4 weeks) will develop walled-off necrotic collections (WON), with 21–63% requiring further interventions. 4 The reason is that among asymptomatic necrosis, the content is liquefied in 28–35% of cases and the size decreases, especially in extra-pancreatic WON or WON without disconnected pancreatic duct, without the need for further necrosectomy.5–6
According to a recent meta-analysis, 7 early interventions (before 4 weeks) for necrotizing pancreatitis were associated with higher mortality, the same rate of adverse events, and clinical success compared to delayed interventions; however, another meta-analysis reported similar outcomes for early or delayed intervention, but longer hospital stay for early interventions. 8 Asymptomatic pancreatic fluid collections (PFCs) are not indicated for drainage, regardless of their size, because of the risk of complications associated with all the drainage methods. At present, the indications for drainage are as follows9,10:
- Pressure symptoms, such as pain, gastroduodenal/common bile duct obstruction, appetite loss, or portal hypertension.
- Complications: fistulas in the peritoneum or upper gastrointestinal tract and infections (representing approximately 26% of new admissions). 11
PFCs above the mesocolon, including ‘the lesser sac’ and the region close to the hilum of the spleen and left kidney, are amenable to transgastric drainage. Any extension into the left paracolic space, below the level of the left kidney hilum, would require further percutaneous drainage. PFCs originating from the head and isthmus of the pancreas, located in the lesser sac and subhepatic space, should be drained via the transduodenal route; however, care should be paid to the proximity of the peritoneum.
According to standard recommendations, PFCs should not be drained sooner than 4 weeks from the initial episode of acute pancreatitis to allow the collections to wall off.9,10 In case of infected necrosis, a randomized controlled trial found that the early drainage after diagnosis (using catheter drainage and percutaneous necrosectomy), compared delayed drainage until the development of walled-off necrosis, resulted in similar mortality (13% versus 10%). 12 In addition, the number of interventions (4.4 in the early drainage group and 2.6 in the postponed group) and incidence of adverse events were comparable. 12 In the postponed group, conservative treatment was possible in 39% of patients, showing that postponement of drainage is more appropriate for the management of PFC. 12 Another prospective trial on patients with endoscopic treatment demonstrated the same superiority in the postponed group compared to the early treatment group (mortality 13% versus 4%, rescue open therapy 7% versus 1%). 13
Materials and methods
Medical literature on the technical performance of endoscopic ultrasound (EUS) drainage is rich, demonstrating the rapid advancement of this method. This narrative review focuses on existing knowledge regarding the choice of EUS-guided pseudocyst and walled-off pancreatic drainage, preprocedural and postprocedural assessment and procedural tips, principles and procedural tips for drainage and necrosectomy, clinical outcomes, adverse events, and training for these procedures.
Preprocedural assessment
Necrosis inside PFCs is usually assessed using magnetic resonance imaging (MRI), which is a preferred method to computed tomography (CT) because it can assess ductal changes and necrosis. 10 EUS can be used for the same purpose, as it was comparable to MRI in diagnosing necrosis in one study; 14 however, a protocol for three-dimensional quantification of necrosis remains lacking. The percentage of solid components assessed by EUS had fair or poor interobserver agreement but moderately influenced the decision to perform direct endoscopic necrosectomy (DEN) and substantially influenced the timing of DEN. 15
Therapeutic EUS is associated with a high risk of bleeding. P2Y12 receptor antagonists (clopidogrel, prasugrel, and ticagrelor) should be temporarily discontinued in case of high-risk conditions (coronary artery stents); aspirin should be continued if the patient has drug-eluting coronary stent inserted for <6–12 months or a bare metal coronary stent inserted >1 month. In addition, direct oral anticoagulants (dabigatran, rivaroxaban, apixaban, and edoxaban) should be discontinued 3 days prior to the procedure and restarted 2–3 days after the procedure. 16 Warfarin should be discontinued 5 days prior to the procedure in patients with high-risk conditions (prosthetic metal valve in aortic/mitral position, atrial fibrillation, and prosthetic valve/mitral stenosis/previous stroke or transient ischemic attack, or previous venous thromboembolism on anticoagulation). 16 In this conditions, low-molecular weight heparin (LMWH) should be started 2 days after the discontinuation of warfarin, except on the day of the EUS procedure, and the combination warfarin-LMWH should be restarted the evening on the day of the procedure, with subsequent discontinuation of LMWH depending on the international normalized ratio (INR) level. 16
In low-risk patients (xenograft heart prosthesis, atrial fibrillation without high-risk factors, or >3 months after venous thromboembolism), P2Y12 receptor antagonists should be discontinued 7 days before the procedure and restarted 1–2 days after the procedure; aspirin should be continued; warfarin should be stopped for 5 days without LMWH replacement until INR < 1.5 and restarted the evening after the procedure, and INR checked 1 week later. 16
Current state of knowledge regarding EUS-guided pseudocyst drainage
EUS or other drainage methods?
There exists three drainage methods: surgical, percutaneous, EUS-guided or transpapillary. Surgical drainage is the first effective therapeutic modality for the management of PP; however, the laparoscopic approach is preferred over open surgery because of the lower rates of incisional hernia and new-onset diabetes. 17
A randomized study of PPs comparing the EUS approach using plastic stents (PSs) with surgical laparoscopy showed similar efficiency despite the small number of patients in each group. 18 The current guidelines do not recommend the use of percutaneous drainage for pseudocysts. The transpapillary approach is reserved for pseudocysts communicating with the main pancreatic duct, located in the head of the pancreas and <5 cm.17–19 Routine transpapillary drainage added no benefit to PFC treatment, except that there was a disruption of the pancreatic duct. 20
Conventional endoscopy-guided drainage is no longer used because of the risk of bleeding in the gastroduodenal wall. 19 The EUS approach is accepted as the first choice of a minimally invasive procedure for the drainage of PPs, and it is associated with good outcomes and acceptable adverse event rates. PSs are recommended for drainage instead of lumen-apposing metal stents because of their lower costs and similar efficiency. 20 Treatment success was defined as the complete resolution or decrease in size of the pseudocyst to ⩽2 cm on CT, in addition to the clinical resolution of symptoms. 19
Procedural tips
EUS-guided drainage of PPs is performed using a linear therapeutic echoendoscope, with the patient in the lateral decubitus position and under general anesthesia or deep sedation; CO2 insufflation and fluoroscopy are compulsory. 21 It is important to carefully choose the puncture site, with a stable scope position, preferably in a short position with the pseudocyst in the middle of the screen. The distance between the gastroduodenal and collection walls should not exceed 10 mm. Blood vessels in the gastroduodenal wall within 1 cm of the puncture site should be avoided to prevent possible bleeding during subsequent dilatations.
The drainage is performed in four steps (Table 1) as follows:
Puncture of the PP wall.
Dilatation of the puncture site using a cystostoma or via balloon dilatation.
The insertion of two guidewires is preferable.
Insertion of the stents.
Technical procedural considerations in the drainage of PFCs.

DEN, direct endoscopic necrosectomy; LAMS, lumen-apposing metal stent; PFCs, pancreatic fluid collections; WON, walled-off necrosis.
The puncture site should avoid the cardia due to its proximity to the pleura and pylorus, possibility of direct access of food into the pseudocyst cavity, and the presence of significant vessels at this gastric level.
Unilocular PFCs are drained through one tract of cystostomy (‘single gate technique’). 22 The drainage with PS is a multistep approach, which involves passing one guidewire through a 19G fine-needle aspiration needle, dilating with a balloon, inserting a second guidewire through an endoscopic retrograde cholangiopancreatography (ERCP) canula, and placing the two PS (‘cold technique’). Another technique directly targets the PFC with a cystotome attached to a pure cut of 100–120 W to create the pseudo-cystostomy (‘hot technique’), followed by the passage of two guidewires, radial expansion balloon dilatation to 12–15 mm in diameter, and placement of two pigtail stents. As a technical tip, dilatation of the created pseudo-cystostomy is important, especially when the pseudocyst wall is fibrous and the cysto-gastrostoma created is insufficient for the placement of the second PS. In addition, the use of a combined-cut coagulation current should be avoided because of the prolonged time required to access the pseudocyst cavity. 23
If the cystotome cannot penetrate the cystic wall with the usual power settings, the power of the pure cut needs to be increased to 150 W or the Endo-cut I setting should be used. The guidewire should never force into the needle because its outer coating may peel. In such situations, the needle and guidewire should be withdrawn together. Antibiotic therapy with cephalosporins or imipenem should be administered for 3–5 days peri-procedurally, 21 although no medical support exists for prolonged use in cases of non-infected PC.
The PS may remain in place until the imaging resolution of PP or indefinitely in cases of disconnected duct syndrome or pancreatic duct obstruction by stones or strictures. 10
Current state of knowledge on EUS drainage of walled-off necrosis
EUS or surgery?
Endoscopic and surgical laparoscopic WON drainages have been compared in few studies. The TENSION trial compared EUS drainage (with PS) of infected WON and surgical laparoscopic drainage, demonstrating that the rates of major complications at 6 months were 43% and 45%, respectively (RR: 0.97; 95% CI: 0.62–1.51, p = 0.88) and the mortality rates at 6-month follow-up were 18% and 13%, respectively (RR = 1.38; 95% CI: 0.53–3.59, p = 0.50); the rate of pancreatic fistula was 5% in the EUS group and 32% in the surgical group (RR = 0.15; 95% CI: 0.04–0.62, p = 0.0011). 24
Among the 98 patients included in this study, 27 patients from each group were followed up for 7 years (Ex-TENSION trial). 25 The rate of fistula was lower in the EUS group than in the surgical group (8% versus 34%; RR, 0.23; 95% CI: 0.08–0.83) (8% versus 34%; RR = 0.93; 95% CI: 0.65–1.32; p = 0.688) and the need for reinterventions was lower (7% versus 24% RR, 0.29; 95% CI, 0.09–0.99); however, pancreatic insufficiency and quality of life were not different. 26
Another randomized control study comparing laparoscopic drainage with EUS drainage with lumen-apposing metal stents (LAMS) or PSs tailored to the amount of necrosis (PSs were indicated for WON with <30% of necrosis) demonstrated that a resolution of WON within 4 weeks without re-intervention for secondary infection was observed in 80% of cases in the laparoscopy group and in 75% of cases in endoscopic group, without significant differences (p = 0.89); however, median hospital stay was shorter in the endoscopic group than in the laparoscopy group [4 (4–8) versus 6 days (5–9); p = 0.03], 25 consolidating the belief that EUS drainage is the first-choice procedure for WON drainage.
EUS or percutaneous drainage?
According to the American Gastroenterological Association, percutaneous drainage of pancreatic necrosis should be considered in patients with infected or symptomatic acute necrotic collections in the early acute period (<2 weeks), and in those with WON but are too ill to undergo endoscopic or surgical interventions. 9
Other indications of percutaneous drainage are as follows: as an adjunct to endoscopic drainage for WON with deep extension into the paracolic gutters or as a salvage therapy after endoscopic or surgical debridement with residual necrosis burden. 9 The comparison of EUS-drained WON with percutaneous method-drained WON followed by video-assisted retroperitoneal debridement demonstrated that EUS was associated with higher clinical success (91% versus 81%) and faster organ failure resolution than percutaneous drainage 27 ; in addition, EUS drainage was associated with lower mortality and complications rates (4% versus 19%, p = 0.0012 and 7% versus 22%, p = 0.005, respectively). 6 This demonstrates that EUS remains the first choice for PFCs located in the upper abdomen. However, the two methods are complementary in cases in which the PFC extends to the lower abdomen.
Percutaneous or surgical drainage?
Open necrosectomy, compared with percutaneous drainage followed, if necessary, by minimally invasive retroperitoneal necrosectomy, is associated with higher new-onset multiple-organ failure (40% versus 12%, p = 0.002) but with the same mortality rate (19% versus 16%, p = 0.70). 17 As a result, open debridement may be best performed in patients with a large burden of necrosis diffusely distributed throughout the abdomen. 9
Principles of EUS drainage of WON
The main outcome of a step-up therapy is the reduction in the PFC to below 2–3 cm in diameter with minimal complications. 24
The therapy respects the 3D principles of delays, drains, and debris. If drainage alone fails, it is followed by step-up therapy such as DEN, an additional drainage site (endoscopic or percutaneous), or surgical intervention. In a series of 81 patients with WON, the step-up therapy was performed in 63% of patients, and the negative predictors of clinical success were collection size >10 cm (OR = 6.94), paracolic extension (OR = 3.79), and >30% of necrosis (OR = 7.1). 28
The European guidelines recommend switching from dual antiplatelet therapy to aspirin monotherapy and prophylactic antibiotics before considering therapeutic EUS 29 and management with antiplatelet and anticoagulant agents is similar to that described above in the case of PP.
Both CT and/or MRI are recommended to assess the necrotic content of WON, which influences the choice of drainage stents. 21
The choice of stent type and size depends on PFC characteristics (size and content) and the endoscopist’s expertise, which have both advantages and disadvantages (Table 2).
Advantages and disadvantages in using lumen-apposing metal and PSs in the drainage of PFCs.
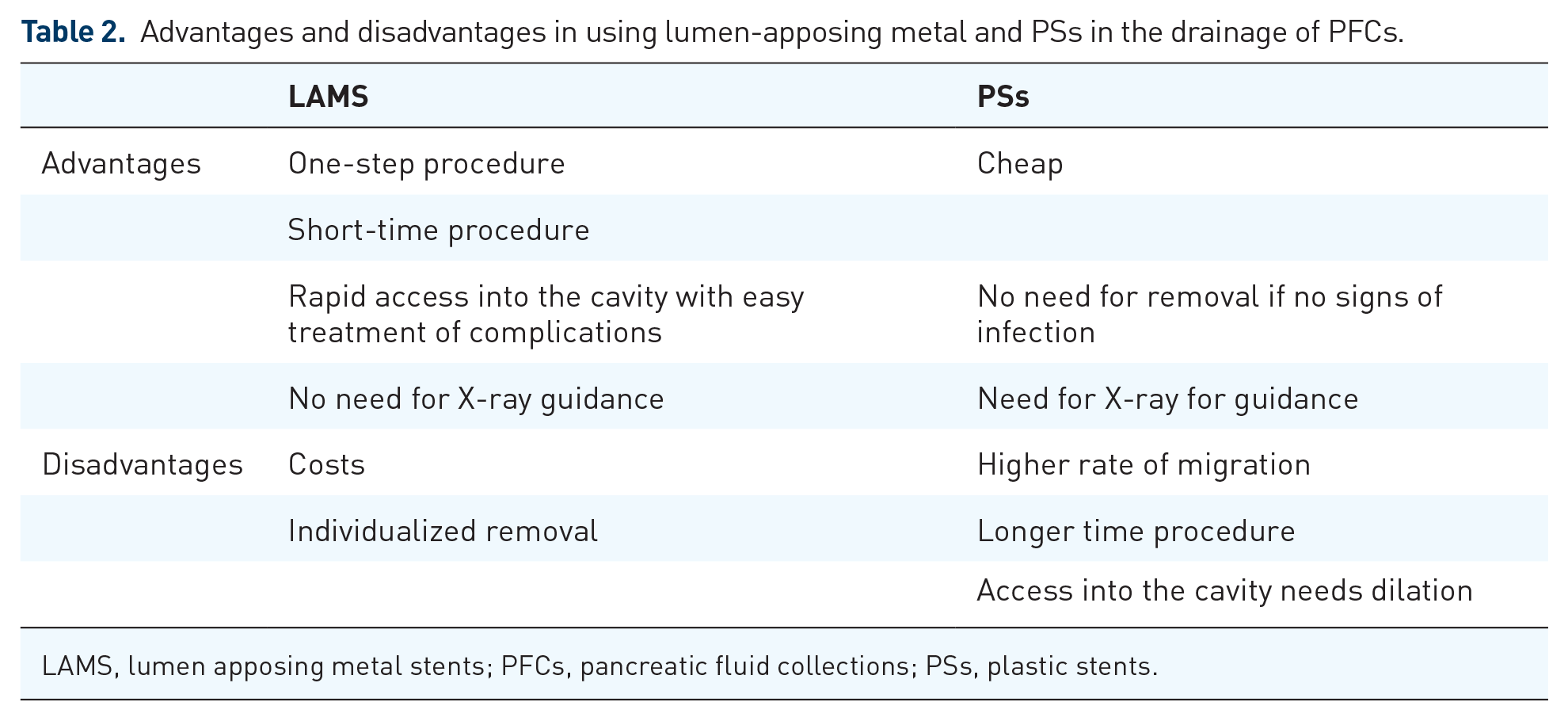
LAMS, lumen apposing metal stents; PFCs, pancreatic fluid collections; PSs, plastic stents.
LAMS provide a higher lumen-apposing force that can hold two non-adhering lumens together, with an anti-migration effect and larger diameter (Table 3).
Technical characteristics of different metal stents available for endoscopic ultrasound-guided pancreatic fluid collection drainage.

Electrocautery-enhanced lumen-apposing metal stents.
Biflanged metallic stents without electrocautery.
Different manufacturers have provided 6- to 20-mm-diameter LAMS 29 ; however, the 15-mm-diameter LAMS has been extensively used for EUS drainage, whereas the 20-mm-diameter LAMS showed similar clinical success (92.2% versus 91.7%), with lower number of DEN sessions, 30 and they were considered useful for WON with solid debris over one-third of the PFC. 22 The development of longer (15 mm) LAMS allowed the approach of the PFC located at 10–14 mm distance from the gut wall, which can be safely drained by EUS. 31
Tubular metallic stents are available in the market, but their use requires a multistep procedure, similar to PS, and are not recommended owing to their high side effects (14%). 32
Procedural tips for drainage
The EUS-guided drainage of WON is similar to the drainage of PPs, with the same principles applicable for the choice of puncture site, followed by the ‘cold’ or ‘hot techniques for placing the PSs or metallic stents’ (Tables 1 and 2). With the development of the electrocautery-enhanced LAMS, its one-step ‘freehand’ insertion is preferred, 29 as it does not require guidewires or fluoroscopy. With the power cut setting at 100–120 W, the LAMS is advanced through the gut wall into the WON cavity, and touching the distal wall of the WON with electrocautery should be avoided. In cases of a significant amount of necrosis with a low liquid component, the water-filling technique through a nasocystic catheter could be helpful. Some authors recommend filling with a contrast and then saline to create a 3 cm diameter anechoic structure to facilitate LAMS deployment. 33 The NAGI™ (Taewoong) and SPAXUS™ (Taewoong) stents are deployed by simple retraction of a catheter sheath, whereas the deployment of the AXIOS stent is characterized by a Luer lock mechanism that allows independent deployment of distal and proximal stent anchors. The NAGI stent has no electrocautery enhancement, and access to the WON should be achieved using other devices, thus requiring multiple steps, as in the case of PS.
After opening the distal flange, the entire device should pulled back until the bell shape becomes asymmetrical to create an apposition to the wall of the target organ. If the distal flange opening is not seen properly under EUS owing to a significant amount of necrosis, movement back and forth to observe the movement inside the WON or the use of X-rays for confirmation may be helpful. It is important to place the LAMS with the elevator in an upward position to allow the proximal flange to open properly into the working channel. The echoendoscope is torqued clockwise and withdrawn to release the proximal flange into the gastric/duodenal lumen. Another possibility is to open the proximal flange under endoscopic vision; however, this technique is less preferred because of the risk of dislodgement. The operator should wait until the stent is opened and then remove the electrocautery of the LAMS to avoid accidental removal of the delivered stent.
Dilatation of the LAMS should be avoided because of the risk of accidental dislodgement. There are reports of irrigation of the WON through an ERCP cannula passed through the scope or a nasocystic drain until the content becomes homogeneous anechoic (200–250 mL of normal saline every 4–6 h).
In complex WONs with multiple compartments, large (> 12 cm) WONs, or suboptimal response to single transluminal gateway drainage, the WONs could be drained using the multiple transluminal gateway known as the ‘multigate technique’ treatment. 10 This treatment gateway consists of one tract to flush normal saline solution via a nasocystic catheter and one or more tracts with a PS for drainage of necrotic contents (clinical success 91.7–97.8%).34,35 Other techniques used LAMS plus PS (Orlando protocol) 29 or two LAMS for the drainage of the necrotic content. 36
The integrated approach in EUS drainage of PFC, known as Orlando protocol, tailored the use of LAMS or PS for EUS drainage according to PFC characteristics: PS for pseudocysts and LAMS for WON. 22 Another study used PS in PP or WON with <30% of pancreatic necrosis and LAMS for WON with >30% necrosis. 28
Furthermore, in the Orlando protocol, LAMS was used in cases of disconnected pancreatic duct syndrome (DPDS) with WON of 6–10 cm, and replaced with a PS after 3–4 weeks. 22 In case of DPDS and WON > 10 cm, the modified multigate technique was used; the technique comprised the placement of LAMS at the level of the body/antrum to facilitate access for DEN, associating two other PSs placed proximally (proximal body or cardia), corresponding to the body/tail of the pancreas. In 3–4 weeks of DEN, the LAMS was removed and the PSs were left in place indefinitely to avoid PFC recurrence. 22 Any PFC extending to the lower part of the abdomen needs dual drainage by EUS and percutaneous techniques. 28
Principles of DEN
DEN represents a therapeutic option in patients with large amounts of infected necrosis who do not respond to drainage alone, and patients presenting with fever, bacteremia, high inflammatory markers, clinical deterioration, and/or remnant fluid in the PFC cavity.
DEN is easier to perform with a therapeutic gastroscope through LAMS; however, it has been performed in WON drained with PS by removing the PS, followed by 15 mm balloon dilation of the transmural tract to facilitate the insertion of the gastroscope into the WON. LAMS ensures large-diameter gastroduodenostomy, allowing for one or multiple sessions of DEN. However, the number of DEN is not reduced when a LAMS is used, as demonstrated in 53 patients with infected WON treated with LAMS (median of 2.5 DEN, need for DEN in 64%) compared to a historical group of 51 patients from the TENSION trial, 24 where the WON was treated with PSs (median of 2.5 DEN, need for DEN in 53%). 37
This new powered endoscopic debridement (PED) system is expected to reduce the number of DEN sessions. It has been used in 30 patients, demonstrating 70% removal of necrosis on Day 21 after index procedure, 66% removal of necrosis per session, fewer interventions (maximum of four), and lower hospital duration compared to conventional DEN 38 ; however, no randomized controlled study exists.
Procedural tips for DEN
Devices used during DEN
Balloon dilatation should be performed to facilitate easy access to the gastroscope inside the WON. Mechanical removal of necrosis can be performed using different devices such as polypectomy snares, rat-tooth forceps, tripod forceps, Dormia baskets, Roth baskets, and Necrolit (MedItalia Inc.). These devices should be opened beyond the stent inside the WON cavity to avoid dislodgement of the LAMS (Table 1). Owing to the risk of bleeding from small vessels, endoscopists should avoid the removal of necrosis adherent to the wall and remnant pink pancreatic tissue.
DEN is time-consuming because the devices slip on the necrotic surface. Forceps can remove only small amounts of necrosis at once, thus requiring multiple on–off movements inside the WON, whereas the baskets and snares cut through the necrosis but cannot be maneuvered easily inside the cavity and cannot always grasp the necrotic fragment. An over-the-scope grasper [OTSC Xcavator (Ovesco)] attached to the tip of the endoscope can remove up to 1 cm3 of necrotic tissue and discharge it into the stomach. The disadvantage is the need for complete withdrawal of the gastroscope because of the necrosis adhering to the tip of the scope; this maneuver was needed in half of the patients 39 and it is therefore recommended to wet the external part of the gastroscope. This device was used in 37 DEN cases, but difficulties were encountered in transduodenal insertion into the WON cavity. The median number of pieces removed was eight, and the median time for removal was 32 min. 39
The EndoRotor PED system (Interscope, Inc., Northbridge, MA, USA) is motorized (1000–1700 rotations/min) and provides simultaneous suction cutting and debriding of tissue using negative pressure (50–550 mmHg). It is recommended for >75% of necrosis inside WON but it can be used with an angulation of 120°. Some side effects, such as pneumoperitoneum and bleeding, were observed, and there was a technical limitation due to the angulation of the device over 160°. 38
Use of H2O2 as necrolytic agent
Initially, the necrolytic agent used for intermittent irrigation at 8-h intervals was 20 mL of 3% H2O2, followed 10 min later with 100 mL of saline solution through a nasocystic tube 40 ; however, more recently, the necrolytic agent was used sprayed directly over the necrotic material (10 –1000 mL of 0.3 –1.5 % hydrogen peroxide). 41
A retrospective study reported that adding hydrogen peroxide during DEN improved clinical success, with similar side effect rates compared to DEN without hydrogen peroxide. 40 However, this agent cannot dissolve the necrosis, and the removal remains mechanical, with large variability depending on the endoscopist’s technique and how much time is allocated for the DEN session. Moreover, side effects, such as perforation, have been reported. 42
A meta-analysis including 7 studies and 186 patients obtained good outcomes, with 19.3% side effects, including 7.9% bleeding, 11.3% stent migration, 5.4% perforation, and 2.9% pulmonary events, which were higher than the results of other meta-analyses on PFC drainage. 43
Additional use of PSs over LAMS
Three small retrospective trials have assessed the efficacy of double pig tail stents (DPS) placement through cautery-enhanced LAMS at the initial endoscopic session in decreasing adverse events or the need for reintervention. These trials demonstrated that there was no additional value in terms of clinical success when double PSs were used, 44 but the results for the adverse event rates were conflicting.45,46
The outcomes from one randomized controlled trial NCT03049215 are expected.
One retrospective study including PFC with viscous solid debris treated with PSs and PS plus nasocystic catheter demonstrated a complete resolution at 12 months (58% versus 79%), but the difference was not significant. 47 Further randomized studies were not published, perhaps because of the low quality of life of patients after such drainage related to saline flush (60–200 mL) through the nasal drain every 2 h during the first 48 h postoperatively and every 4–6 h the following days. 48 Reports on the local administration of antibiotics (imipenem) were done in small retrospective series, showing reduced need for DEN and low mortality in cases of infected necrosis; however, further studies are needed. 49 The results of such studies should be interpreted with caution, taking into consideration the transgastric/duodenal route of the nasocystic catheter. Continuous irrigation with streptokinase through a percutaneous catheter lowers the need for necrosectomy, bleeding, and mortality compared to saline infusion 50 or H2O2, 51 but no studies exist in the case of DEN.
Timing of DEN
DEN in immediate settings at the time of LAMS placement and in delayed settings (1 week after the LAMS placement) showed higher stent dislodgements in the immediate group, although the total number of DEN sessions was lower (3.1 versus 3.9), with the same clinical success and overall adverse events. 52 We usually perform DEN at a minimum of 48 h after LAMS placement, and we attempt to gain complete access to the WON cavity with the devices used during DEN to avoid accidental removal of the LAMS. This ‘scheduled DEN’ after 72 h appears to be associated with only 1.3% migration rate 53 ; however, the best time for the commencement of DEN needs further research.
Interval between DENs
The interval between DEN sessions depends on the WON infection, patient tolerability to long sedation (which could be 30–50 min), and the availability of an endoscopy suite for such a long procedure. The interval should vary from 48 h to 1 week.
Predicting DEN outcome
The presence of necrosis diminished the clinical success of EUS drainage with PSs (93.5% in case of PP versus 63.2% in WON, OR = 7.6) 54 and the content of necrosis >30% was associated with an increased probability of DEN (OR = 6.7).28,55 A multilocular WON or one with a sponge sign on CT and remaining necrosis after EUS drainage ⩾35% were independent predictive factors of a step-up approach. 56
In addition, obesity and poor medical health (American Society of Anesthesiology class III) predicted necrosectomy failure with the use of PSs.57,58
When the solid necrotic content is over 50% (Figure 1(a) and (b)), the number of DEN sessions increases to 2 or 3,42,59 and this represents a negative factor for predicting the success of DEN. 60 In addition, pancreatic volume necrosis >620 cm3 was strongly associated with re-admission and reintervention in a retrospective study. 61 Negative prediction model of LAMS success in WON includes paracolic gutter extension and increased Acute Physiology and Chronic Health Evaluation II score.28,60
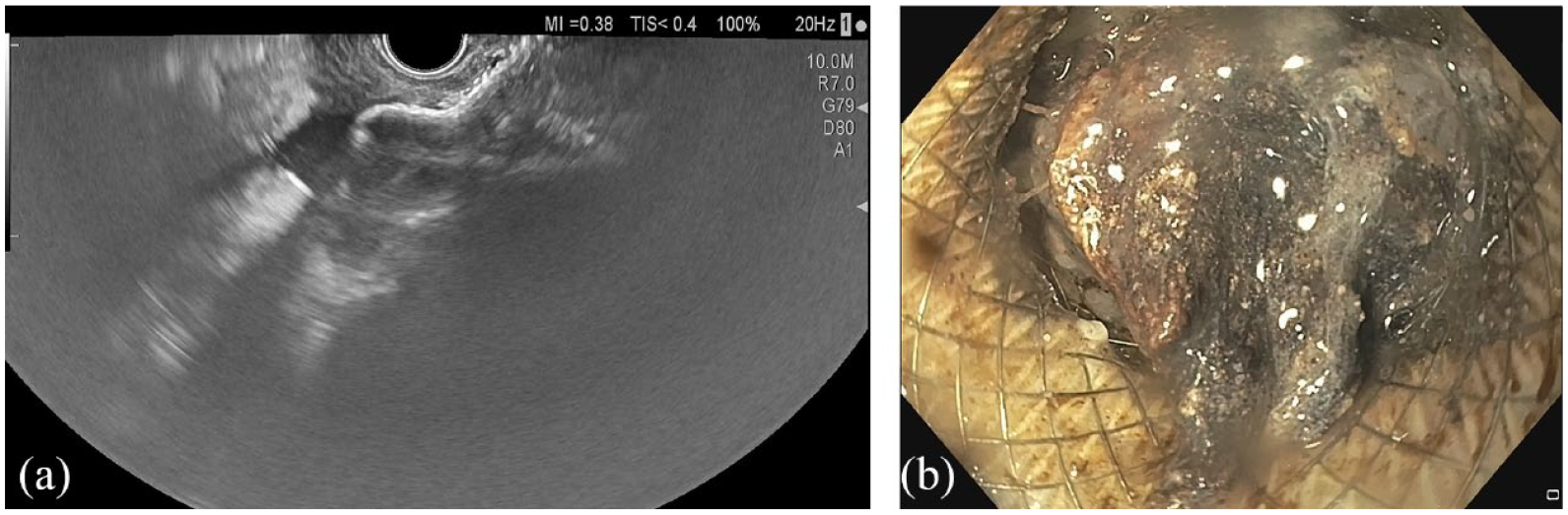
(a) EUS aspect of walled-off pancreatic necrosis with the lumen-apposing metal stent system before stent deployment. (b) Endoscopic aspect of necrosis obstructing the lumen-apposing metal stent placed for the drainage of WON.
Postprocedural approach
Stopping proton pump inhibitors (PPI) therapy
In a retrospective study of 272 patients with DEN using LAMS, the use of PPI increased the number of DEN sessions compared to the number of sessions in non-PPI patients (4.6 versus 3.2) and increased the rate of LAMS obstruction (20% versus 9.5%), but the clinical outcomes were similar, suggesting that continuous PPI use results in higher rates of early stent occlusion. 62
Antibiotic treatment
A retrospective study reported bacterial and fungal infections (73% and 49%, respectively) of PFCs drained endoscopically. 63 In non-infected PFCs, antibiotics are administered as prophylaxis periprocedurally for 3–5 days, 29 but prolonged therapy is not beneficial. 64 Infected necrosis is associated with 30% mortality 17 and the use of antibiotherapy is recommended, but its duration is not properly specified. It is recommended to choose an antibiotic that can penetrate the pancreatic tissue (carbapenems, quinolones, metronidazole) without the addition of an antifungal routine. 9 In one randomized controlled study comparing infected PFCs to sterile PFCs treated by EUS drainage, 48 there was no difference in long-term clinical success (20-month follow-up) and complications rate; therefore, the usefulness of antibiotics in sterile PFCs drained by EUS requires further elucidation.
Removal time of LAMS
The recommended mean time for LAMS removal is 4 weeks 10 ; however, in a retrospective study, LAMS removal <3 weeks (median 21 days) compared to LAMS removal >3 weeks (median 35 days) was similar in the rate of adverse events (52% versus 48%, p = 0.67). 44 Early removal can prevent bleeding, especially when there are surrounding collateral vessels. 65
However, a longer maintenance of LAMS is possible if the patient continues to have large amounts of necrosis at 6 weeks despite DEN. In such situations, consider replacing the LAMS with a new stent to minimize the risks of bleeding and embedding because of the possible deterioration of the polymer of the prolonged stent. 65 The individualized removal of the stent can be performed by follow-up at Days 7 and 30 after the index procedure via questioning the resolution of symptoms, inflammatory laboratory tests (CRP, leukocytes), and transabdominal ultrasound. 25 Usually, the stent is removed when the cavity is <3 cm in diameter on imaging, 42 although other cutoff values for the remnant cavity have been reported (Table 4). Special care should be paid to the nutritional status of the patient.
Comparison of outcomes of different stents used for the drainage of PFCs.
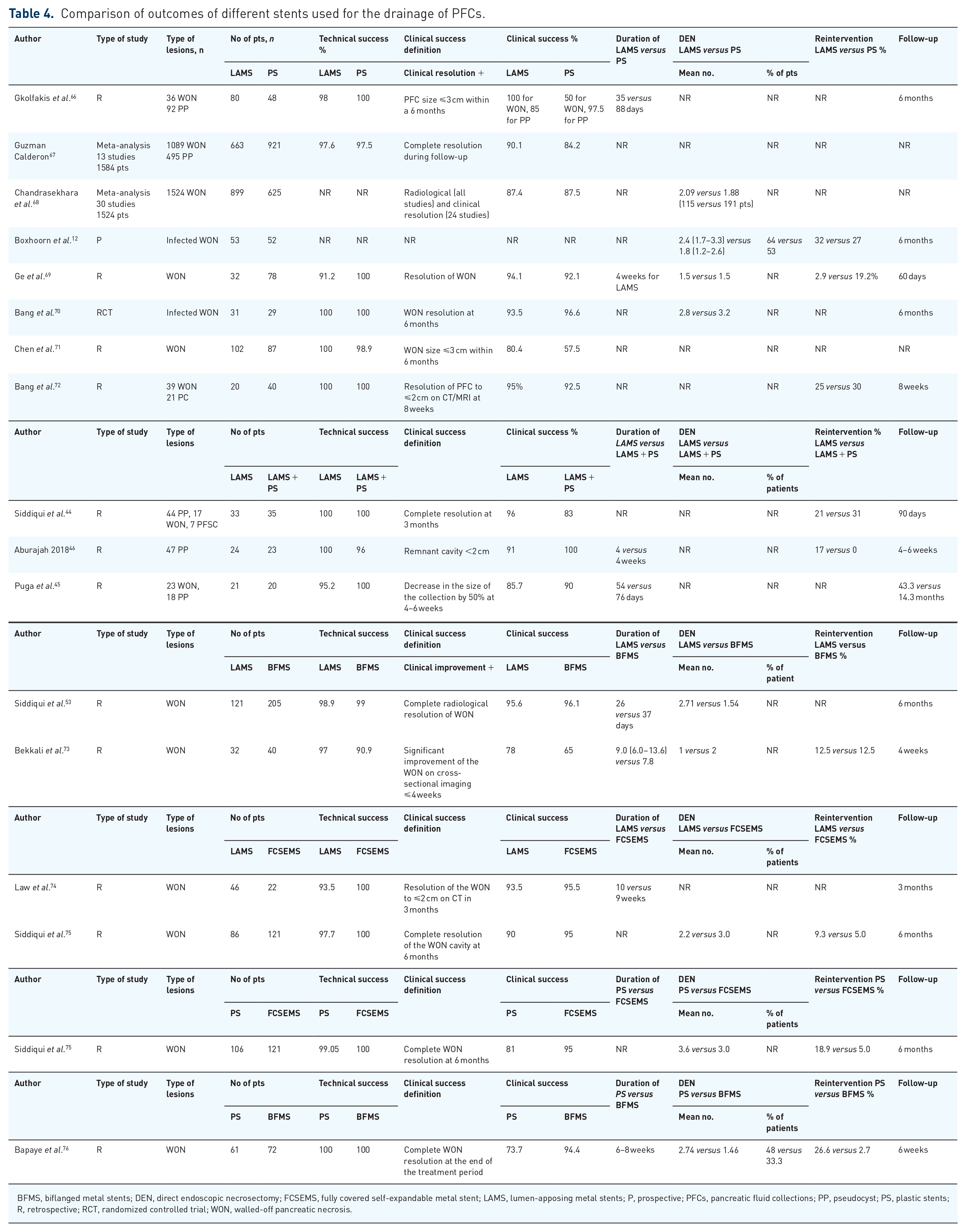
BFMS, biflanged metal stents; DEN, direct endoscopic necrosectomy; FCSEMS, fully covered self-expandable metal stent; LAMS, lumen-apposing metal stents; P, prospective; PFCs, pancreatic fluid collections; PP, pseudocyst; PS, plastic stents; R, retrospective; RCT, randomized controlled trial; WON, walled-off pancreatic necrosis.
Clinical outcomes of EUS drainages
Treatment success was defined as a reduction in PFC size to 2–3 cm or less on follow-up CT, in addition to symptom resolution at 6 months 29 (Table 4). One meta-analysis comprising 13 studies and 1584 patients showed that the clinical success rate of EUS-guided drainage for PP was 97.2% with LAMS and 90.8% with PSs, whereas for WON, the clinical success rate was 89.9% with LAMS and 83.1% with PSs, without significant difference. 67
Attempts at drainage with biflanged metallic stents showed similar outcomes to LAMS; meanwhile, the use of fully covered self-expandable metal stents was abandoned owing to the risk of stent migration.
PFC recurs in 9–25% of cases,77,78 and it is explained by the communication with the pancreatic duct, known as DPDS, with an isolated body and neck of the pancreas that remain functional and drain their secretions into the PFC or produce external pancreatic fistula. The imaging diagnosis of this pathology is difficult, based on MRI or ERCP criteria. 79 In addition, its management is complex because the low-output external fistula (<200 ml/day) closes spontaneously within 3 months 80 ; and although it can be treated conservatively, symptomatic cases require EUS drainage with/without transpapillary drainage or surgery.
One meta-analysis (30 studies, 1355 patients) demonstrated that endoscopic transmural drainage of disconnected pancreatic duct was superior to transpapillary drainage (success rate, 90.6% versus 58.5%), and the success rate was comparable between endoscopic and surgical methods. 81
Another meta-analysis comprising 5 studies and 941 patients with EUS drainage of PFC showed that the imaging PFC resolution and symptoms were similar in patients with or without DPDS (OR = 0.77). 82 The recurrence of PFC and rate of PS use were higher in DPDS (OR = 6.72 and 15.9, respectively), 82 especially within the first 2 years after drainage. 83
Contrary to these findings, a randomized controlled trial that included 104 patients with LAMS-drained WON drained compared the results of an additional PS with those of no stenting. After removal of LAMS at 4 weeks, PFC recurrence was not significantly different at 12 months between cases with or without additional PS (13.5% versus 25%, p = 0.14), whereas the recurrences after LAMS removal were mostly asymptomatic, requiring few re-interventions (5.7% versus 7.6%). 77
Further research is necessary to confirm the usefulness of an additional PS. This study had several limitations; it was considered underpowered and had a short follow-up duration (maximum of 12 months), 84 with other studies having a median of 22–34.4 months to assess recurrence.83,85
In rare cases, when ERCP is unsuccessful, the pancreatic duct can be drained by EUS in an antegrade, transmural manner or with the use of the rendezvous technique. This technique is complex and should be attempted only in high-volume expert centers. 29
The remnant pancreatic tissue above the disrupted pancreatic duct might predict PFC recurrence, although this was not investigated in the aforementioned studies. 86 Long-term PSs appear safe, with 3.5% local complications and 3.5% migration. 87
Costs
In the TENSION trial, the mean costs for treatment (since randomization to 6 months follow-up) with EUS drainage using PS and step-up approach were €60,228; for the surgical step-up approach, the cost was €73,883. 24 Drainage with PP with PS proved to be more cost-efficient than that with LAMS, 20 thus serving as the indication for PP drainage in the Asian guideline. 21 In infected WONs, although the initial costs of endoscopic drainage were higher when LAMS were used, the total healthcare costs (including percutaneous drainage need and intensive care costs) of EUS drainage with PSs exceeded the costs of drainage with LAMS, sustaining the positive impact of LAMS for daily practice in such cases. 37
Adverse events
The rate of adverse events in the last published meta-analysis was similar between LAMS- and PS-based drainage of PP (7.1 versus 15%), but was lower in LAMS-based WON drainage than in PS-based WON drainage (13.1 versus 19.2%). 67 The rate of LAMS adverse events was 24% in a large multicenter study; they occurred in 25 days (range: 6–30) with 6.3% severe adverse events, and the lack of balloon dilatation was associated with higher rate of adverse events. 88
Obstruction followed by infection occurs in 3.2–3.8% of EUS drainage with LAMS cases and in 5.2–6.1% of EUS drainage with PS cases.67,68 Antibiotic therapy and DEN are recommended, as described above.
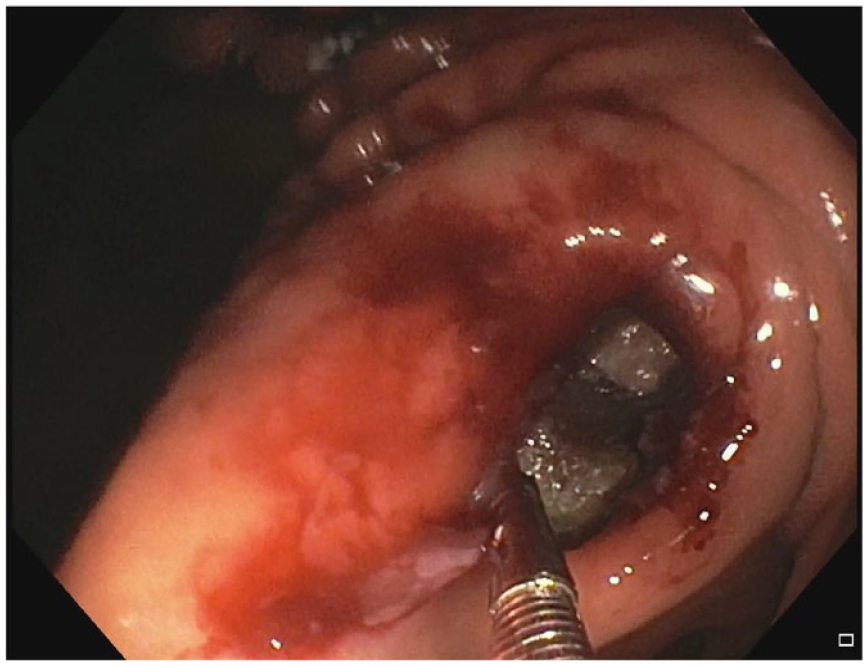
Bleeding occurs in 2.5–5% of EUS drainages with LAMS and in 3.6–4.6% of EUS drainages with PS, according to two meta-analyses,67,68 although there are studies reporting higher rates of 15.4–17%.12,89 Bleeding can occur early in the first week after placement of LAMS, an occurrence that can be prevented by carefully checking the site of gastroduodenostomy and avoiding the parietal vessels within 1 cm of the puncture site. Management consists of removing food or necrotic tissue as much as possible to visualize the bleeding point, followed by spray coagulation or gold probe hemostasis. In case of hemostasis failure, the solution is arterial embolization, but open surgery should be avoided owing to the high risk of death. 42 Delayed bleeding usually occurs within 3–4 weeks after the procedure, resulting from erosion of the small vessels around the PFC wall during the collapse of the cyst cavity (Figure 2). 90

Endoscopic view of the parietal bleeding inside the WON after complete necrosectomy.
Individualized timing of LAMS removal would be helpful, as described above, 65 especially when an intracavity vessel is identified. 91 Cirrhosis did not represent a risk factor for bleeding in a large retrospective series 91 (Figure 3(a) and (b)).

(a) Endoscopic view through a lumen-apposing metal stent of a retroperitoneal vessel inside the WON. (b) Endoscopic view through a lumen-apposing metal stent of a retroperitoneal vessels inside the pancreatic fluid collection.
Perforation with pneumoperitoneum has been reported in 0.5% of cases with LAMS-based drainage and 1.1% of cases with PS-based drainage.67,77 Treatment should be conservative, with the percutaneous drainage of CO2 pneumoperitoneum when it is immediately recognized; however, a multidisciplinary approach with surgical intervention may sometimes be necessary. Therefore, drainage of the PFC located >1 cm from the gastroduodenal wall should be avoided, especially when PS drainage is used.
Buried LAMS due to gastric mucosal overgrowth can occur in up to 4% of patients but it is usually managed conservatively by endoscopic extraction (Figure 4). 11
Endoscopic view of a buried lumen-apposing metal stent placed for the drainage of a WON.
In cases of accidental dislodgements of stents (more frequently during DEN), which can occur in 0.9–5.9% of EUS drainages with LAMS and in 6.1–6.8% of EUS drainages with PS,67,68 there is no risk of intestinal obstruction. If the remnant cavity is >3 cm in diameter, the placement of a second LAMS or PS should be considered. There are reports of reusing the same deployed stent by grasping the LAMS with biopsy forceps and retrieving the proximal flange into the therapeutic channel, followed by repositioning it into the PFC cavity.92,93
Prediction of adverse events
In a multicenter retrospective study, the authors assessed the factors predicting possible side effects, such as the injury of pancreatic duct (leak with OR = 2.51, complete disruption OR = 2.61), perigastric varices (OR = 2.90), pseudoaneurysm (OR = 2.99), multigate technique, (OR = 3.00), and percutaneous drainage (OR = 2.81). 94 In case of cirrhosis, one retrospective study showed lower clinical success and higher rate of complications, related mainly to DEN. 95
EUS drainage of PFC training
EUS drainage of the PFC is the first step in therapeutic EUS. The first requirement for EUS drainage is that trainees have diagnostic EUS competence. 96 Training should be performed in centers with multidisciplinary support for PFC drainage, including interventional radiologists, surgeons, and anesthesiologists, to prevent and manage complications. The trainees should have high success rate of EUS tissue acquisition (>90%) and ability to puncture a 5 mm lesion before commencing PFC drainage. 97 Another concept is that knowledge of X-ray and guidewire manipulation 96 is helpful, although this is not needed when the LAMS are used and ERCP skills are beneficial, but not essential to the endoscopist learning EUS-guided drainage. 21 The skills in EUS-guided PFC drainage are best acquired through observation, followed by hands-on training in the porcine model and execution of the procedure in patients. 21 The European Society of Gastrointestinal Endoscopy (ESGE) guideline recommends 10–25 cases for proving proficiency 96 and the EUS Asian Group 5–10 cases of plastic stent pseudocyst drainage under supervision. 21
Conclusions
EUS drainage of the PFC requires careful patient selection and complex preprocedural assessment, including MRI.
Although LAMS does not reduce the number of DEN sessions, it is useful for WON because it facilitates access to the cavity.
PS remains the main recommendation for PP drainages.
Careful, step-by-step procedures should be performed in every case to avoid complications. Take time to complete the procedure.
EUS drainage is associated with a non-negligible rate of complications, and a multidisciplinary team may be required for optimal management.
Closely follow-up the patient.
Further research is needed to determine the best time for necrosectomy, perform double drainage with lumen-apposing metal stents and plastic stents, and determine the best duration of antibiotherapy.
