Abstract
Background:
While the detrimental impact of negative emotions on the clinical course of inflammatory bowel disease (IBD) and quality of life has been extensively investigated, evidence for a potential impact of positive emotions is scarce.
Objectives:
We aim to analyse contributing factors of positive affect and their predictive value for disease course in IBD patients.
Design:
In this retrospective cohort study, epidemiological, psychosocial and IBD disease characteristics of Swiss IBD cohort study patients were analysed longitudinally.
Methods:
Epidemiological, psychosocial and disease characteristics were extracted from the database of the Swiss IBD cohort study. Participants’ positive emotions were assessed cross-sectionally with the seven-item Marburg questionnaire (range 1–6) addressing positive affect in different aspects of daily life. Predictors of positive emotions were identified by linear regression. The quantitative longitudinal impact of positive emotions on the further disease course was analysed using a multivariable Cox proportional hazards model.
Results:
Among 702 IBD patients, those reporting more positive emotions were found to have significantly less intense medical treatment, less pain and fewer depressive symptoms (p < 0.05). A higher percentage of variability in positive emotions was explained by pain (36%) and depressive symptoms (13%) than by epidemiological characteristics (0.3%), or characteristics of IBD and its treatment (2.4%). Patients with higher levels of positive emotions (score > 3.5) experienced longer flare-free survival, also after adjusting for confounders (adjusted hazard ratio: 0.39, p < 0.05).
Conclusions:
The absence of pain and depressive symptoms were the strongest drivers for high positive affect. Higher scores of positive affect were associated with longer disease-free survival in IBD patients.
Introduction
Inflammatory bowel disease (IBD) is a group of multifactorial diseases resulting in a chronic relapsing inflammation of the gastrointestinal tract. The two main entities of IBD are Crohn’s disease (CD) and ulcerative colitis (UC). The prevalence of IBD is rising in industrialized countries, 1 therefore representing an increased burden on healthcare systems. 2 The treatment of IBD generally focuses on endpoints like clinical remission and endoscopic healing;3,4 however, the growing knowledge about the gut-brain axis and its bi-directional dimension, is increasingly considered in the disease concept and new target strategies.4–6 Apart from the burden of the chronic disease on mental health of IBD patients, there is evidence for an impact of stress levels on subsequent disease outcomes.7,8 Research focuses mainly on quality of life (QoL), 9 negative affect, psychological stress, depression, or anxiety.10–12
The gut-brain axis has been investigated in animal models. 13 In rodents, experimental colitis can lead to sickness, social withdrawal, loss of motivation, fatigue, mediated by the afferent vagal nerve and immune activation in the central nervous system with cytokines and nitric oxide pathways.14–16 On the other hand, manipulation of mental state by induction of experimental depression was able to aggravate experimental colitis for which antidepressants showed protective effects.17,18 These data suggest an intimate connection between gut inflammation and altered mental state but neither gut inflammation nor the complex human mental state is likely faithfully reflected in animal models.
The mental state of patients can be assessed with a variety of tools, some of them specifically validated for the use in IBD. Often applied are the well-established Intestinal Bowel Disease Questionnaire (IBDQ), 19 the short-form (36) health survey (SF-36) 20 and the hospital anxiety and depression scale (HADS-A and -D, respectively). 21 Studies show that diagnosis and progression of IBD have a significant impact on mental health.22,23 Therefore, screening for negative emotions can identify patients with a more severe disease course, who potentially benefit from more intense anti-inflammatory treatment or psychiatric intervention.
Besides negative affect, individuals also experience positive affect, an umbrella term used to subsume feelings like happiness, contentment and joy. 24 Positive and negative affect are associated with different personality traits25,26 and are not just opposites of each other. 27 Emotional wellbeing, another often used term in the same context, is defined as the experience of frequent positive affect and infrequent negative affect 28 and is one of the central dimensions of health and quality of life. 29 In a systematic review, Diener et al. concluded that higher levels of subjective wellbeing improve health and longevity. 30 A study addressing positive and negative emotions in patients with cardiovascular disease (CVD) found a modulating effect of high positive affect on subsequent CVD-related events. 31
Considering these robust biological and health-related effects of positive emotions,30,32 we hypothesize that positive affect might be a contributor to the mental state, interacting with intestinal inflammation via the gut-brain axis. So far, in contrast to negative affect, data regarding the role of positive affect in patients with IBD has only been investigated in small studies 33 and a systematic evaluation in a large patient cohort is missing. The Marburg questionnaire on habitual health findings,34,35 from now on referred to as Marburg questionnaire, is an easy-to-use tool which addresses the positive affectivity of patients 36 (e.g. presence of positive feelings and contentment in different areas of daily life 34 ). For the retrospective study at hand, we used the Marburg questionnaire to examine habitual positive affect in IBD patients and to assess its potential influence on the subsequent IBD disease course.
Material and methods
Study population
The Swiss IBD cohort study (SIBDC) has been collecting data of IBD patients in Switzerland since 2006. The design of SIBDC cohort has been explained elsewhere in detail;37,38 in brief, its aim is to improve the understanding of pathogenesis, trigger factors for flares, as well as the consequences of CD and UC on patients’ physical, psychological and social status using the gathered data of a large cohort.37,38 At a first assessment, the data of newly included patients is collected retrospectively, subsequently it is updated yearly with patient- and physician-based questionnaires. All patient data used are de-identified and cannot be followed back to an individual patient.
In the context of a separate analysis in the SIBDC 39 a questionnaire focussing on pain and its influence on QoL was sent to all patients at that time point included in the SIBDC cohort in 2013.
For our retrospective analysis, data from 2064 IBD patients with 948 responses to the German Pain Questionnaire (GPQ) were available. For inclusion into our study population, patients required at least one regular follow-up after receipt of the questionnaire and completeness of responses related to the Marburg questionnaire.
Measures
The questions sent to all SIBDC patients were based on the validated questionnaire ‘Deutscher Schmerzfragebogen (DSF)’, a GPQ, 36 which focuses on a biomedical-psycho-social pain model. The GPQ also includes the Marburg questionnaire on habitual health findings, addressing the patient’s positive affect, such as the ability to enjoy life, experience joy and cosiness, feel contentment with work performance and physical condition and ability to fulfil daily requirements. 35 It consists of seven questions rated on a six-point Likert scale (1–6; 1 representing low positive affect) and its focus lies on the emotions experienced in the 2 weeks prior to assessment. 34 The questions are averaged for the final score, resulting in scores from one to six. A maximum of one missing item can be replaced with the mean of the other six items. Validation of the questionnaire showed an internal consistency with Cronbach’s α of 0.91. 34 An English version of the questionnaire (author’s translation, not used for data acquisition) is shown in Supplemental Table 1, as well as the German original and the French version used for data acquisition (Supplemental Tables 2 and 3). For the calculation of flare recurrence, positive affect was used as a nominal value and high value was defined before analysis as a score higher than 3.5 (middle of the scale). 3.5 is the lower bound for a patient being on average at least slightly happy; no other thresholds were analysed.
We extracted demographic information such as sex, age, education, ethnicity, marital status, education, smoking40,41 and drinking habits from the SIBDC database. Clinical information included diagnosis, age at diagnosis, past therapies and treatments (biologicals, steroids, surgeries), and extraintestinal manifestations. Information concerning disease activity (Crohn’s disease activity index, CDAI 42 ; Modified Truelove and Witts Severity Index, MTWAI 43 ) and psychosocial information (HADS, 21 SF36, 20 IBDQ 19 ) were extracted from the last regular assessment before the completion of the Marburg questionnaire.
First occurrence of a flare was defined following the methods used in previous studies.44–46 We defined a flare as the first occurrence of one or more of the following events after filling the questionnaire: high disease activity (MTWAI > 10 or CDAI > 150), physician-reported flare events or worsening of the disease (assessed by the physician and recorded in the SIBDC data base), fistulas and stenosis, anal fissure, abscess, any type of IBD surgery and initiation of treatment with steroids or biologics (tumour necrosis factor (TNF) – inhibitor or vedolizumab). No patients were treated with ustekinumab, as the data export was in 2015. Patients who were already having a flare at the point in time of the questionnaire were excluded from the analysis. If the last regular assessment was more than 12 months prior to the questionnaire, data was not considered. In subgroup analyses we tested for significant differences in several subgroups such as diagnosis (CD or UC), patient sex, severity of the disease (measured with the CDAI and MTWAI) and the level of pain described by the patient (CDAI/MTWAI derived). Also taken into account were the pain and disability metrics of the GPQ.
Statistical analysis
We first compared responders of the pain questionnaire to non-responders with t-tests for continuous data and chi-square tests for categorical data. The distribution of the reported wellbeing of our cohort was compared to that of other studies, by assessing the means and 95% confidence intervals. 47 We built four multivariable linear models, each including only variables from a specific covariable grouping (epidemiological, IBD-related, pain-related and psychosocial characteristics) to visualize the relative importance of the covariates in explaining the variation of patient-reported wellbeing. For final model selection, we generated a Lasso regression model 48 considering all covariates, and those covariates with estimated non-zero coefficients were presented as the selected model. We computed relative importance of the covariates in each of the five models with proportional marginal variance decomposition. 49 Each model was assessed for multi-collinearity via calculation of variance inflation factors (VIF) for each covariate. 50 Corrected p-values are presented as derived through the use of the lasso projection method (de-sparsified Lasso), which uses bootstrap samples to assess uncertainty surrounding parameter estimates.51,52
To assess the relationship between flare occurrence and patient wellbeing, we computed the time-to-flare or end of follow-up for each patient starting at the date entered on the wellbeing questionnaire. A flare was defined as the physician-reported flare in the SIBDC database. Any patient not having a flare by our censoring date of 7 October 2015 or who dropped out was counted as censored at those time points. Differences in survival between the high- and low-wellbeing groups were first assessed with Kaplan–Meier estimation 53 and a log-rank test. A multivariable Cox proportional hazards model was fit to model the hazard of flare controlling for relevant confounders. For this analysis specifically, mean-value imputation was done for two of the independent variables (IBDQ and GPQ disability score). Analyses were carried out in R version 3.5.2 54 using the packages glmmTMB and relaimpo.55,56
This study was conducted following the principles outlined by the STROBE cohort guidelines. 57
Results
Study population
A total of 948 patients of the SIBDC filled out the questionnaire; of those a final population of 400 CD and 302 UC patients met our study inclusion criteria. Characteristics of responders versus non-responders to the Marburg questionnaire are shown in Supplemental Table 4. Table 1 gives an overview of the study population.
Study population characteristics. Variables are broken into four groups based on a general grouping.
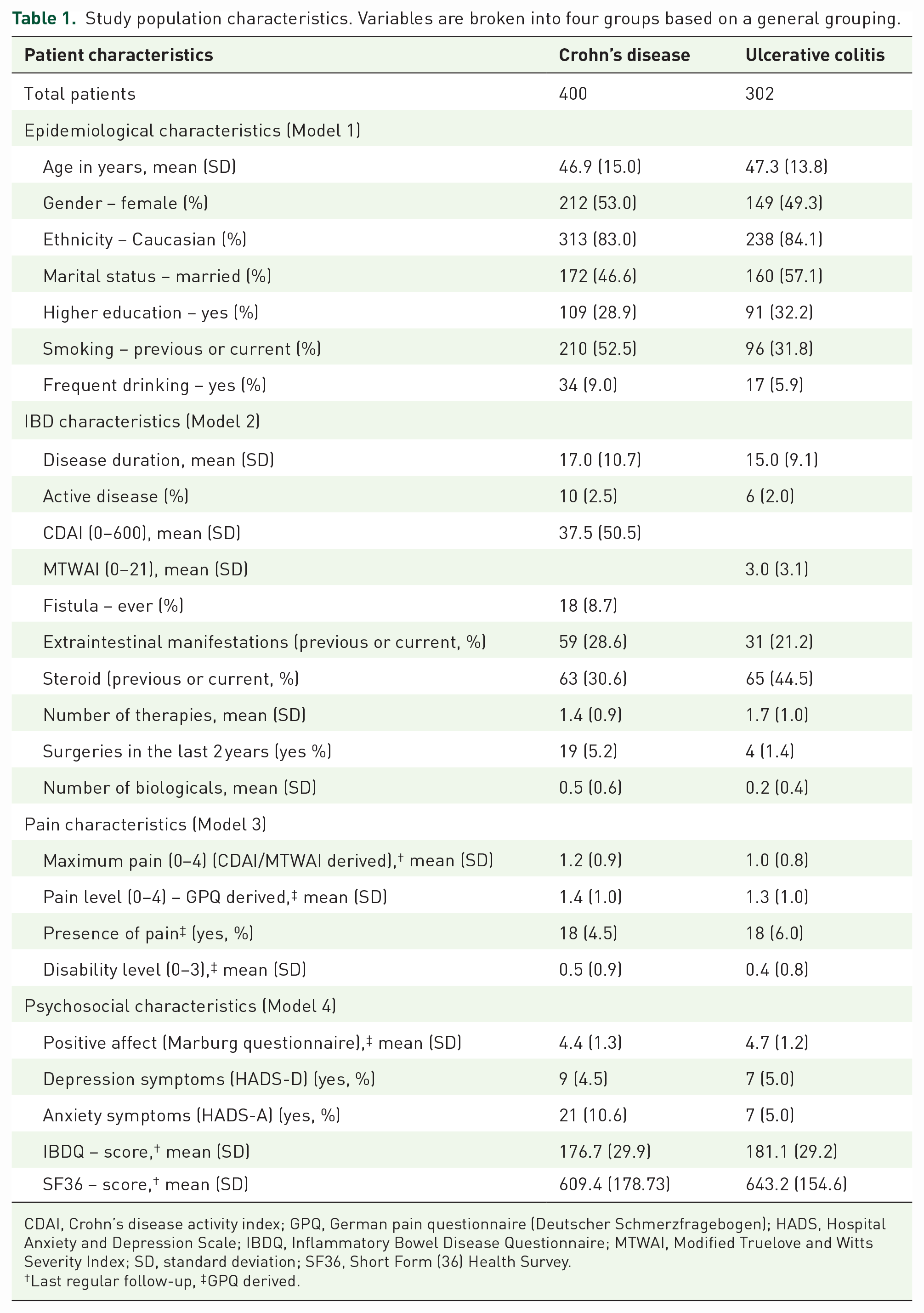
CDAI, Crohn’s disease activity index; GPQ, German pain questionnaire (Deutscher Schmerzfragebogen); HADS, Hospital Anxiety and Depression Scale; IBDQ, Inflammatory Bowel Disease Questionnaire; MTWAI, Modified Truelove and Witts Severity Index; SD, standard deviation; SF36, Short Form (36) Health Survey.
Last regular follow-up, ‡GPQ derived.
Positive affect in IBD
In our cohort, the average Marburg score was 4.65 for UC and 4.42 for CD (Figure 1). We first conducted a univariate regression analysis to identify important factors contributing to positive affectivity (Supplemental Table 5). Relevant predictors were inactive disease, no extraintestinal manifestations (EIM), lower levels of pain and no symptoms of depression.

Positive affect in Swiss IBD cohort patients. Yellow and blue scatter plots: distribution and median value of positive affect (as assessed by the Marburg questionnaire) for CD and UC patients from the SIBDCS.
To simultaneously test for the effects of several parameters, we used multivariable linear regression analyses. Due to the high number of predictors we created four separate models for epidemiological, IBD-related, pain-related, or psychosocial characteristics, respectively (Table 2). Most associations observed in the univariate analyses were confirmed. Considering epidemiological features (Model 1), younger age [Slope: −0.009, 95% CI (−0.016, −0.0025)], Swiss citizenship [Slope: 0.29, 95% CI (0.033, 0.54)], higher education [Slope: 0.24, 95% CI (0.033, 0.45)], and no smoking [Slope: −0.29, 95% CI (−0.48, −0.098)] predicted higher values of positive affectivity. Regarding IBD characteristics (Model 2), inactive disease [Slope: −1.3, 95% CI (−2, −0.72)], absence of EIM [Slope: −0.53, 95% CI (−0.74, −0.33)] and treatment characteristics [few past or current steroids (Slope: −0.4, 95% CI: (−0.62, −0.17)) and no/ low number of biologics (Slope: −0.39, 95% CI (−0.56, −0.21) in the history] were associated with high values. The pain and psychosocial model, showed various descriptors of pain [Slope: 0.79, 95% CI (0.37, 1.2)], depression and anxiety [Slope: −0.16, 95% CI (−0.19, −0.14)] as well as the IBDQ [Slope: 0.024, 95% CI (0.021, 0.027)] and SF6 scores to be main influencers of positive affectivity scores (Models 3 and 4). Assessment of VIF for each model suggested that multicolinearity was not a large issue in our models.
Reduced model for positive affectivity.
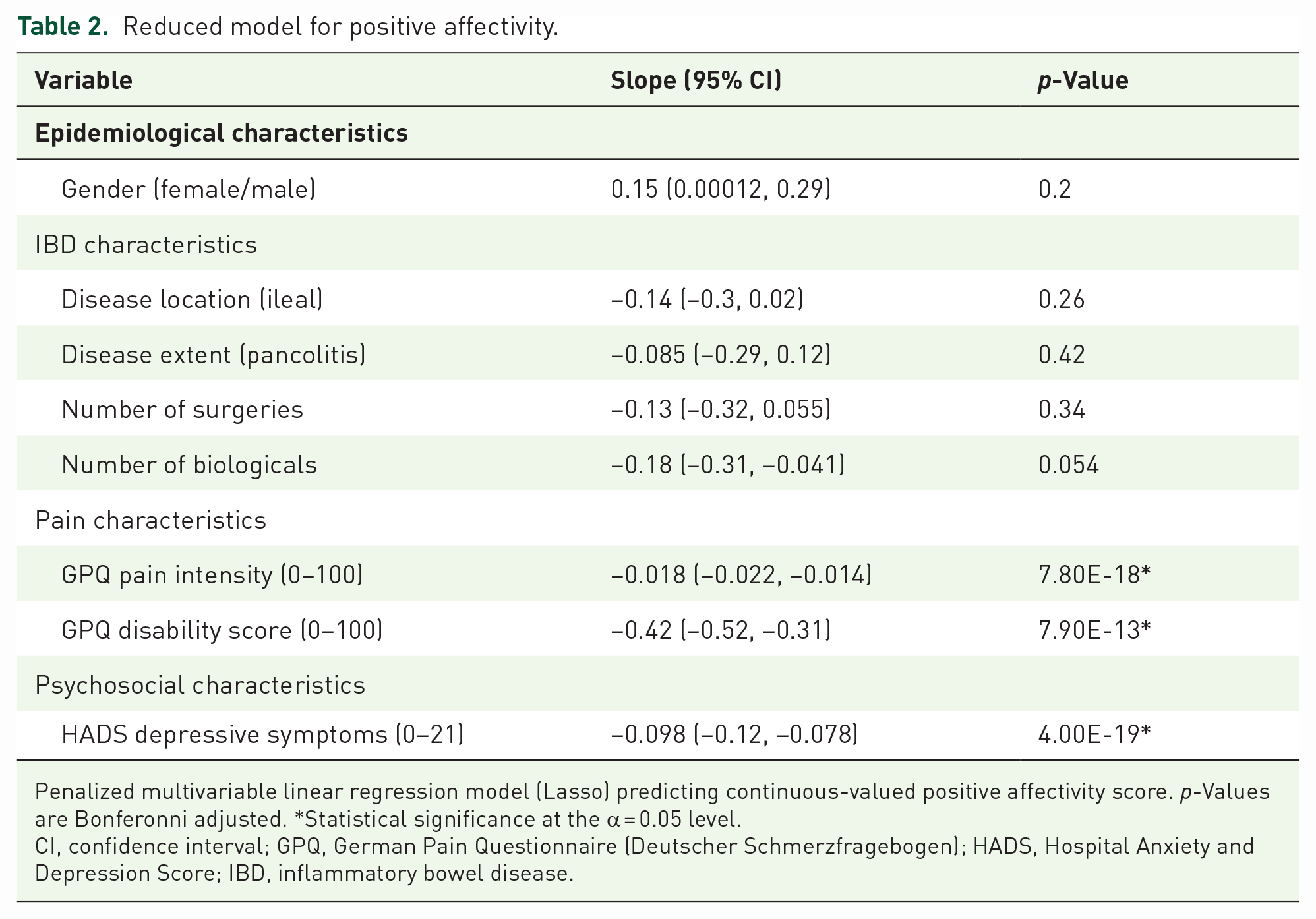
Penalized multivariable linear regression model (Lasso) predicting continuous-valued positive affectivity score. p-Values are Bonferonni adjusted. *Statistical significance at the α = 0.05 level.
CI, confidence interval; GPQ, German Pain Questionnaire (Deutscher Schmerzfragebogen); HADS, Hospital Anxiety and Depression Score; IBD, inflammatory bowel disease.
To determine the most relevant parameters in a comprehensive simplified model, we constructed a multivariable lasso-penalized regression model. This model included all covariates and selected the predictors that were found to have non-zero coefficients. We found that no/low number of biologicals [Slope: −0.18, 95% CI (−0.31, −0.041)], low pain intensity [Slope: −0.018, 95% CI (−0.022, −0.014)] and pain disability [Slope: −0.42, 95% CI (−0.52, −0.31)], both according to the GPQ) and the absence of depressive symptoms [Slope: −0.098, 95% CI (−0.12, −0.078)] to significantly predict higher scores of positive affect (Table 2).
We analysed how much of the variability in positive affect (as assessed with proportional marginal variance decomposition, see Methods) could be explained by our multivariate models of positive affect (Models 1–4 and the fitted model). The epidemiological model and the model with IBD characteristics (parameters only present in IBD patients) explained only 4% and 10% of the variability (Figure 2(a) and (b)). In contrast, the explanatory power of the pain model and the psychosocial model were larger, explaining 46% and 43% of the variability (Figure 2(c) and (d)). The predictive power of the fitted model with selected characteristics from all models was highest (52% of variability, Figure 3). In this model, critical drivers for low positive affect were the pain intensity and pain disability as well as the presence of symptoms of depression.

Relative importance of variables predicting positive affect. Four multivariable linear regression models are fit predicting positive affect as a continuous variable. Relative importance is calculated with proportional marginal variance decomposition. Relative importance of model variables within each model are shown with area proportional to importance: (a) epidemiological model, (b) IBD characteristics, (c) pain model and (d) psychological model.

Relative importance of variables in Lasso-penalized model for positive affect. A lasso-penalized regression is fit predicting positive affect as a continuous variable. Relative importance is calculated with proportional marginal variance decomposition. Relative importance of model variables within each model are shown with area proportional to importance.
Flare-free survival
We aimed to test, whether positive affect was a protective factor regarding future IBD flares. In our study, a flare was defined as a new occurrence of one or more of the following events after assessment of positive affect: high disease activity (MTWAI > 10 or CDAI > 150), physician-reported flare, fistulas and stenosis, anal fissure, abscess, any type of IBD surgery and initiation of intake of steroids or biologicals.44–46 When patients were stratified by quantile of reported positive affectivity, less flare events were found in groups with higher levels of positive affectivity (Supplemental Table 6). We tested whether high scores of positive affect (Marburg score >3.5) were associated with patient flare-free survival with Kaplan Meier curves and the log-rank test and found that patients with high scores of positive affect experienced a longer flare-free follow-up period (p = 0.0027, Figure 4). This association remained significant after adjustment when we assessed hazard of IBD flare in a Cox proportional hazards model corrected for relevant confounders (HR = 0.39, p = 0.0048, Table 3).

Kaplan–Meier curves for flare-free survival. Positive affect is split into two categories (high/low) around the value of 3.5. Red curve: High positive affect (>3.5). Blue curve: Low positive affect (⩽ 3.5). Shaded regions are 95% CI for the Kaplan–Meier curves. ‘p’ is the unadjusted log-rank test p-value. ‘adjusted p’ is the unadjusted p-value for positive affect from a multivariable Cox proportional hazards model.
Multivariable Cox proportional hazards model predicting flare-free survival.

Age in years has been scaled as (X/10) for convenience in this table. *Statistical significance at the α = 0.05 level.
CI, confidence interval; CDAI, Crohn’s disease activity index; GPQ, German pain questionnaire (Deutscher Schmerzfragebogen); HADS, Hospital Anxiety and Depression Scale; HR, hazard ratio; IBDQ, Inflammatory Bowel Disease Questionnaire; MTWAI, Modified Truelove and Witts Severity Index; SF36, Short Form (36) Health Survey.
Discussion
In our study on the impact of positive affect on the disease course of IBD patients we were able to identify two IBD-relevant patterns: higher scores were associated with less pain, and absence of symptoms of anxiety and depression. Further, higher values of positive affect were associated with longer flare-free survival.
In previous non-IBD studies, emotional wellbeing has been associated with positive health outcomes, including longevity30,32,59 and several mechanisms have been advanced: (i) positive affect improved the coping with stress: In a British study with healthy volunteers, objective markers of stress such as heart rate, saliva cortisol and fibrinogen stress response were elevated in the least happiest quintile compared to the happiest quintile. 60 (ii) positive emotions also affected the immune system and inflammation. 30 For instance, positive affectivity predicted resistance to experimental rhinovirus and influenza virus infection. 61 (iii) subjective positive affect has been robustly associated with physical activity and protected against obesity, heavy drinking, smoking and risk-taking behaviour. 30
In IBD, our findings are in line with a gut-brain axis, mediating the interaction of mental state with intestinal inflammation. 13 Previous studies established an increased risk for IBD recurrence and a more aggressive disease course in patients with symptoms of anxiety and depression.6,44–46,62–64 Our study extends this work to positive affect. High values seem to be not just the absence of depression or anxiety since robust effects of positive affect were observed even after correcting for negative emotions in our multivariable Cox proportional hazard model. Therefore, negative affect (HADS) and positive affect (Marburg questionnaire) may constitute independent components of an overall IBD-relevant mental state factor, interacting with the gut. Since intestinal inflammation can trigger symptoms of depression and anxiety 6 and recurrence of disease activity was associated with lower scores of positive affect in our study, a bidirectional association between IBD activity and mental state via a gut-brain axis seems likely. 7
In our study, the absence of various descriptors for pain were the strongest predictors for high values of reported positive affect. This is not surprising since negative effects of pain on QoL have been previously described,39,65 parallel to an intimate relationship of pain with depression. 66 Unfortunately, approximately half of IBD patients (49% in UC and 55% in CD) reported pain as a long-standing problem. 39 This emphasizes the necessity for physicians to prioritize the treatment of pain, anxiety and depression for optimal patient care.
The strongest IBD-related variable predicting higher values of positive affect was the absence of EIM, followed by descriptors for treatment, low disease activity and extent of disease. However, IBD characteristics explained a lower fraction of the variance of positive affect compared to pain or psychiatric symptoms. This might be due to adaptation of the IBD patients. Epidemiological research has shown that positive affect is not influenced by life events more than 3 months ago. 67 This indicates temporal stability of positive affect and the presence of an individual set point which will only be transiently affected by positive or negative events.32,67
Our study suggests the Marburg questionnaire as a simple psychological assessment tool in IBD patients. All items relate to positive aspects of normal daily life and none of the questions imply psychiatric disease. Therefore, patients might respond more acceptingly since fear of stigmatization due to psychiatric co-morbidity is not relevant. Interventions to directly increase positive emotions have been developed 68 ; however, exercises in mindfulness can also strengthen wellbeing. 69 In IBD patients, mindfulness-based interventions were able to reduce stress and depressive symptoms and improve QoL,70,71 improve biomarkers of IBD such as faecal calprotectin and c-reactitive protein (CRP) 72 but no direct effect on physical symptoms or frequency and severity of flares could be demonstrated.70,71
Our approach has several strengths and limitations. Using the extensive dataset of the well-characterized SIBDC patients, we could control for a great variety of confounders and were able to leverage the longitudinal nature of the SIBDC cohort design. Further, our study sheds light on the role of positive affect in IBD which has been understudied so far.
Limitations include the composition of our cohort since SIBDC tends to be biased towards larger centres and patients with more severe disease. Also, the questionnaire assessing positive emotions was filled out only once and the response rate was below 50%. As responders to the questionnaire showed longer disease duration, higher age, higher socioeconomic status and were less anxious and depressed; results could be biased by those differences. Due to limited data in this retrospective study, we were not able to use purely objective measures for intestinal inflammation, such as CRP, calprotectin, or endoscopic measures. We were also not able to control for the effect of negative affect (apart from symptoms of depression and anxiety), since a global mood scale has not been used in the SIBDC. Further, other relevant confounders might have been missed. Finally, since our study is non-interventional, direct and indirect effects of positive emotions cannot be distinguished.
In conclusion, in IBD patients, high values of positive emotions are strongly associated with the absence of depressive symptoms and pain. High scores were associated with longer flare-free episodes in our IBD cohort, independent from psychiatric symptoms and positive emotions might be an important compound of the mental state, interacting with intestinal inflammation via the mental state variable.
Supplemental Material
sj-docx-1-tag-10.1177_17562848231179335 – Supplemental material for Because I’m happy – positive affect and its predictive value for future disease activity in patients with inflammatory bowel diseases: a retrospective cohort study
Supplemental material, sj-docx-1-tag-10.1177_17562848231179335 for Because I’m happy – positive affect and its predictive value for future disease activity in patients with inflammatory bowel diseases: a retrospective cohort study by Brian M. Lang, Martina Ledergerber, Sebastian Bruno Ulrich Jordi, Niklas Krupka, Luc Biedermann, Philipp Schreiner, Pascal Juillerat, Jacqueline Wyss, Stephan R. Vavricka, Jonas Zeitz, Roland von Känel, Gerhard Rogler, Niko Beerenwinkel and Benjamin Misselwitz in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
