Abstract
Background:
The growing numbers of early esophageal cancer (EEC) have increased the demand for endoscopic therapy.
Objectives:
To clarify the influential factors for the prognosis of patients with EEC receiving endoscopic surgery, and to construct a nomogram to evaluate the prognostic value of endoscopic therapy.
Design:
Prognostic analysis study.
Methods:
Clinical data of EEC patients who received endoscopic therapy between 2004 and 2015 were collected from the Surveillance, Epidemiology, and End Results database and used to construct the nomogram. The prognosis was analyzed by R language; the nomogram was constructed by Cox survival analysis; and the accuracy of the nomogram was verified by C index and the receiver operating characteristic (ROC) and calibration curves. X-Tile software was used to stratify the risk of patients.
Results:
Our study constructed the nomogram of the prognosis of patients with EEC treated by endoscopic surgery, including 1118 patients and 5 independent prognostic factors of esophageal cancer-specific survival. The C index and the area under the ROC curve (AUC) of the training and verification cohorts were all >0.75. The calibration curve also reflected the good consistency of the model in predicting survival. Significant difference in the risk of patients from different stratifications with the same T staging existed, and the model had a better C index than that of the T staging.
Conclusion:
Our study reports potential influential factors affecting the prognosis of EEC patients who received endoscopic therapy and establishes a reliable nomogram to predict the risk and prognosis, which has certain advantages compared with traditional TNM staging system.
Introduction
Esophageal cancer is one of the most common malignant tumors worldwide, including two major histological types of esophageal squamous cell carcinoma and esophageal adenocarcinoma. Esophageal cancer ranks third by incidence and fourth by cancer-related deaths in China. 1 In 2020, the estimated new cases of esophageal cancer reached 18,440 in the United States, among which there were about 16,170 estimated deaths. 2
For decades, the high mortality rate of esophageal cancer was thought to be closely related to the advanced stages at diagnosis. In recent years, there is a significantly increased rate of diagnosis at early stages owing to the enhanced monitoring of reflux esophagitis and Barrett’s esophagus as well as the improvement of health conscious of the public. Endoscopy plays an essential role in this process. 3 Emerging endoscopic techniques benefit endoscopy to play a vital role in treating esophageal cancer, and the growing numbers of early diagnoses have also increased the demand for endoscopic therapy.4,5 Nowadays, early esophageal cancer (EEC) can be treated by surgical resection, endoscopic resection, and adjuvant chemoradiotherapy. Surgical resection comprises total esophagectomy and subtotal esophagectomy, and endoscopic therapy includes endoscopic mucosal resection and endoscopic submucosal dissection. Compared with surgical treatment, endoscopic therapy shows obvious advantages in shortening the length of stay in the hospital, reducing the readmission rate, decreasing the prevalence of treatment-related complications, and improving the quality of life.6,7 However, it has a disadvantage in intervening local lesions and lymph nodes. Adjuvant chemoradiotherapy may have a potential to make up for this shortcoming, while its role remains controversial owing to the presence of side effects.
So far, no relevant prospective randomized controlled trials have been reported to provide a direct comparison between endoscopic therapy and surgical treatment. Some existing retrospective studies have compared the therapeutic efficacy, recurrence rate, and survival rate of endoscopic therapy and surgery in patients with EEC. According to some studies, endoscopic and surgical treatments had no obvious difference in improving the survival of patients, with the advantages of endoscopic therapy confirmed simultaneously.8–10 However, there are few studies analyzing the prognostic value of endoscopic therapy for EEC. Along with the wider application of endoscopic therapy in EEC, it is of great significance to clarify possible factors affecting the prognosis of EEC patients. The construction of a prognostic model for endoscopic therapy of EEC is helpful for clinicians to have a more intuitive understanding of the influence of various factors and to determine whether patients are suitable for endoscopic therapy. Moreover, the risk score calculated by the model can also provide evidence for evaluating the prognosis of patients after endoscopic therapy and developing individualized treatment for certain patients.
Our study analyzed the survival data collected from the Surveillance, Epidemiology, and End Results (SEER) database. The primary purpose of this study was to explore the influential factors that affect esophageal cancer-specific survival (ECSS) of patients with EEC who received endoscopic therapy and construct a nomogram of the prognosis model. Findings in our study are expected to provide reference for clinicians to evaluate the prognosis of patients with EEC after endoscopic therapy and assist clinical decision-making.
Methods
Data extraction and inclusion of target patients
Diagnosis and treatment data of cancer patients were collected from the SEER database (https://seer.cancer.gov/), a database widely used in clinical research. 11 Searching through the SEER database in 2022 by using SEER*STAT software (version 8.4.0), we downloaded the data of 1760 patients who had undergone endoscopic surgery and were diagnosed with first primary malignant tumor of esophageal cancer from 2004 to 2019.Eligible patient data were obtained based on the preset inclusion and exclusion criteria. The inclusion criteria were the following: Site recode: ICD-O-3/WHO 2008: esophagus; RX Summ-Surg Prim Site: 10-29; First malignant primary indicator: Yes; and Derived AJCC: T1N0M0. Indicators included age at diagnosis, gender, race recode, marital status, ICD-O-3 Hist/behave, primary site – labeled, grade, derived AJCC T, CS tumor size, radiation, and chemotherapy recodes. The exclusion criteria were patients with missing or unknown data of age, gender, race, histology, and TNM staging. Finally, 1118 patients who met the screening criteria were included in this study for further analysis. The selection flow of eligible patients is shown in Figure 1.
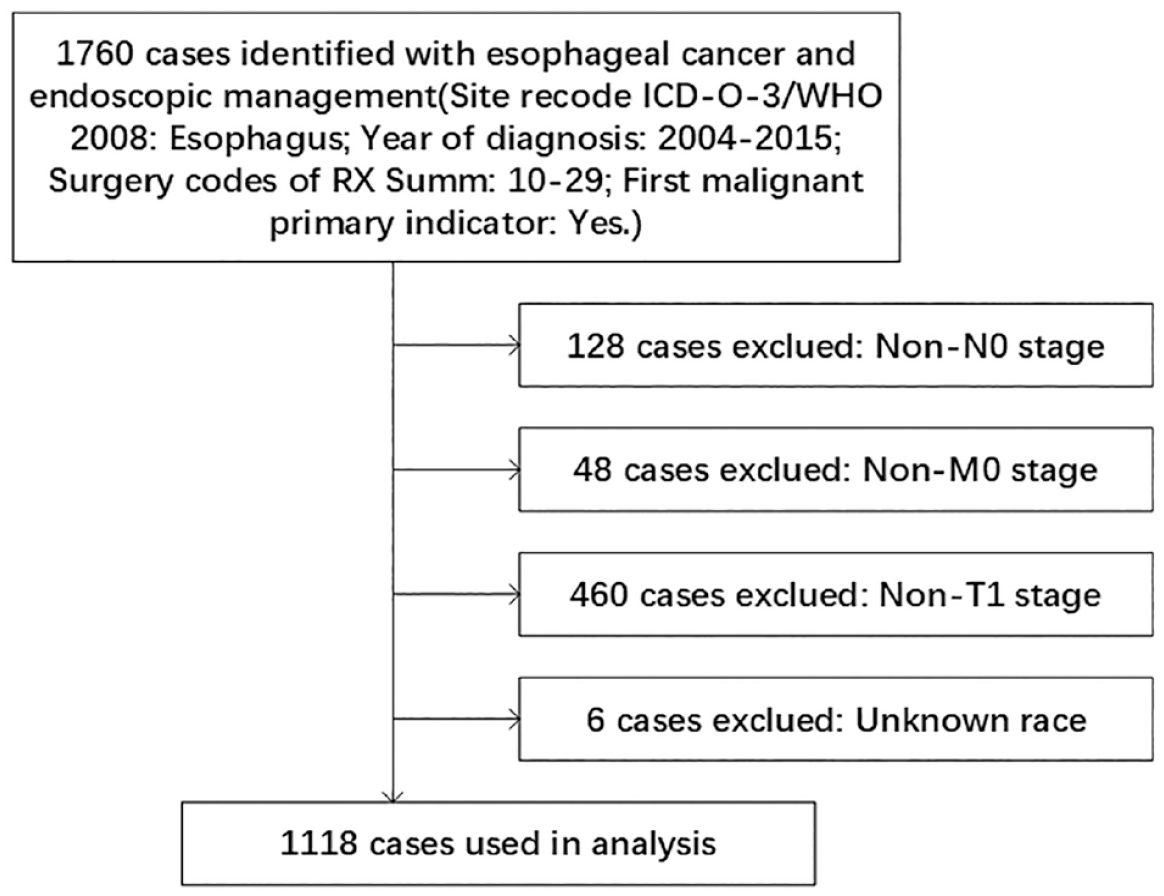
Flow diagram of selecting eligible patients from the SEER database between 2004 and 2019.
All data were de-identified, and exemption from the Medical Ethics Committee of Xiangya Hospital, Central South University was obtained. Informed consent was not required for this retrospective study. The reporting of this study conforms to The TRIPOD statement. 12
Software
The cut-off values of the risk index were calculated by X-Tile software (version 3.6.1), and all patients were divided into three subgroups: low risk, medium risk, and high risk. R language (version 4.0.2) (https://www.r-project.org/) was used for patient grouping, baseline analysis, Cox survival analysis, nomogram drawing and verification, and Kaplan-Meier curves drawing. 13 The R projects such as ‘rms’, ‘foreign’, ‘caret’, ‘survival’, ‘survival ROC’, and ‘plyr’ were used in the above analysis and plotting.
Survival analysis
All patients were randomly divided into training and verification cohorts with a ratio of 7:3 by R projects ‘tableone’. In addition to the comparison of baseline data, univariate Cox proportional hazards model analysis was carried out in the training cohort. Variables with statistically significant differences were included in the multivariate Cox proportional hazards model analysis to determine the independent prognostic factors of ECSS in EEC patients receiving endoscopic therapy. PASS 15 software was used to calculate the sample sizes needed for Cox regression analysis, and the pseudo variable method was used for the missing data. p < 0.05 was used to indicate the presence of statistically significant difference.
Establishment and verification of nomogram
The nomogram was drawn by using the factors with statistically significant differences in multivariate Cox analysis. The C index was calculated to test the accuracy of the model. 14 The receiver operating characteristic (ROC) curve and the area under the curve (AUC) were used to evaluate the performance of the model. The calibration curves were used to show the difference between the nomogram and the actual observation of the 3- and 5-year survival of patients, during which 1000 bootstrap resamples were used for evaluation. All validations involved the internal validation of the training cohort and the external validation of the verification cohort.
Stratification of risk index and survival prediction
The nomogram was used to calculate the risk score of each patient, and all patients were divided into three subgroups of low risk, medium risk, and high risk. 15 The Kaplan-Meier curve of each subgroup was plotted to analyze the survival difference of patients with different risk levels. The advantages and disadvantages of this risk model and T staging were compared through the Kaplan-Meier curve of different T stagings and the C index of the nomogram and T staging.
Results
Basic characteristics of enrolled patients
A total of 1118 patients were included according to the preset inclusion and exclusion criteria. There were 156 patients with endpoint event (14.0%). Enrolled patients were then randomly divided into the training cohort (n = 782) and the verification cohort (n = 336). Among them, the training cohort included 435 patients with T1a staging (55.6%) and 87 patients with T1b staging (11.1%), while the verification cohort included 174 patients with T1a staging (51.8%) and 39 patients with T1b staging (11.6%). The median survival and the interquartile range (IQR) of the two cohorts were 49 months (0–190 months, IQR: 67) and 50 months (0–181 months, IQR: 66), respectively. The major basis characteristics of patients in both cohorts were ages of 60–79 years, Caucasian, male, married, primary site of lower esophagus, adenocarcinoma, and T1a staging. The basic characteristics of the two cohorts are shown in Table 1. There was no difference in the baselines of patients from the two cohorts (all p > 0.05).
Patient characteristics in the training cohort and validation cohort.

Analysis of prognostic factors
In order to evaluate the influence of various factors on the prognosis of patients with esophageal cancer who received endoscopic therapy, univariate and multivariate Cox analyses were performed on ECSS of patients in the training cohort. According to the univariate analysis (Table 2), age, race, primary site, histology, grading, tumor size, T staging, radiation, and chemotherapy were considered to be related to the prognosis of patients (all p < 0.05), while no significant association of patients’ prognosis was found with gender and marital status (all p > 0.05). Then, factors with statistically significant differences in the univariate analysis were included in the multivariate analysis. As shown in Table 3, age, primary site, grading, tumor size, and T staging were independent prognostic factors of ECSS (all p < 0.05). Of these, higher age, overlapping lesion of the esophagus, higher grade, larger tumor, and T1b staging were associated with poor prognosis of patients, while no statistical difference was noticed in race, histology, radiation, and chemotherapy (all p > 0.05).
Univariate analysis using the Cox proportional hazards model in esophageal cancer patients.

95% CI, 95% confidence interval.
Multivariate analysis data of esophageal cancer patients using the Cox proportional hazards model.

95% CI, 95% confidence interval.
Establishment and verification of the nomogram
Factors with statistically significant differences in multivariate regression analysis were included in the establishment of the nomogram, including age, race, primary site, T staging, and tumor size. The vertical corresponding value of each item in the nomogram was the individual point of this item. All the scores of points were added together to get the total score, and the vertical corresponding value of the total score was the probability of 3- or 5-year survival of the enrolled patients. As shown in Figure 2, the contribution of all factors to the prognosis from large to small was ranked as follows: age, primary site, grade, T staging, and tumor size.

The survival nomogram of esophageal cancer patients undergoing endoscopic therapy. The sum of these numbers is located on the total scores axis, and a line is drawn downward to the survival axes to determine the likelihood of a 3- or 5-year ECSS.
In order to verify the accuracy of the constructed model, C index, ROC curve, AUC, and the calibration curve were all used to evaluate the nomogram. The C index of the model was 0.763 (95% CI: 0.717–0.809, p < 0.01) in the training cohort and 0.779 (95% CI: 0.706–0.853, p < 0.01) in the verification cohort. ROC curves of both cohorts were drawn simultaneously. Among them, the AUC values of 3- and 5-year survival were 0.786 and 0.766 in training cohort (Figure 3(a) and (b)), which were 0.769 and 0.768 in the verification cohort (Figure 3(c) and (d)), respectively. In addition, calibration curves were further drawn to determine the consistency between the 3- and 5-year ECSS predicted by the nomogram and the actual survival rate, as shown in Figure 4. Collectively, the above results support that the model may have good accuracy.

The ROC curve for the nomogram. (a) At 3-year ECSS in the training cohort; (b) at 5-year ECSS in the training cohort; (c) at 3-year ECSS in the validation cohort; and (d) at 5-year ECSS in the validation cohort.

The calibration curve for the nomogram. (a) At 3-year ECSS in the training cohort; (b) at 5-year ECSS in the training cohort; (c) at 3-year ECSS in the validation cohort; (d) at 5-year ECSS in the validation cohort.
Risk stratification
Based on the calculation of the total scores of all patients by the nomogram as the risk scores, the optimal cut-off values of the risk scores were analyzed by using the X-Tile software, as shown in Figure 5(a). Accordingly, enrolled patients were divided into three risk levels of low risk (0–104.8, 57.60%), medium risk (104.8–144.4, 31.48%), and high risk (>144.4, 10.91%), respectively. Figure 5(b) shows the Kaplan-Meier curve of patients with different risk levels, while Figure 5(c) to (e) are the Kaplan-Meier curves of patients with different risk levels in each T stage. Clearly, there were significant differences in the prognosis of patients with different risk levels in the same T stage (p < 0.01). In addition, we calculated the C index of the model using T staging to predict prognosis, which was 0.667 (95% CI: 0.620–0.714, p < 0.01) in the training cohort and 0.651 (95% CI: 0.577–0.725, p < 0.01) in the verification cohort. With respect to the above, the C index of the nomogram was better than that of T staging.

The cut-off values of risk scores and risk stratification for patients with different T stages. (a) The cut-off values of risk scores for all patients; (b) risk stratification for all patients; (c) patients with T1a stage; (d) patients with T1b stage; and (e) patients with T1NOS stage.
Discussion
Previous studies on the prognostic factors of esophageal cancer are carried out generally on patients undergoing surgery or neoadjuvant therapy. Few studies are conducted on patients treated with endoscopic therapy. 16 Nowadays, in addition to partial palliative treatment, most patients receiving endoscopic treatment are patients with EEC. For different treatment methods and patient groups, there may be significant difference in factors affecting prognosis and the influence of various factors. Age has always been considered one of the main factors affecting the prognosis of patients with esophageal cancer, which is also true in those receiving endoscopic therapy. It has been reported that there may be a deterioration in the prognostic outcome in patients with the increase of age. 17 Similarly, in our study, age was discovered to be an independent prognostic factor for ECSS, and it also had a great impact on ECSS among independent prognostic factors. Furthermore, studies have shown that tumor size is always a factor affecting the prognosis of esophageal cancer.18,19 Here in our study, we also reported a similar result. Meanwhile, it was also found that the primary site was also related to ECSS, and the overlapping lesion might predict a worse prognosis. An early study suggested that the overlapping lesion might be associated with the increased multicentric carcinogenic potential in the non-cancerous epithelium of the esophagus of patients with EEC. 20 Another study has found that overlapping esophageal lesions in patients with stage I esophageal cancer have adverse effects on the prognosis. 21 These results may indicate that for patients with overlapping lesions, there may be a higher risk of cancerization in the normal tissues of the esophagus, accompanied by a higher risk of recurrence after endoscopic therapy. Few previous studies have suggested that the overlapping lesion impacts the prognosis of patients undergoing surgery. 21 It may be attributed to less surgical resection of endoscopic therapy, and this difference can be more evident in patients who receive endoscopic therapy. Accordingly, the result related to the factor of overlapping lesion may indicate that endoscopic therapy is not suitable for patients with overlapping lesions. Meanwhile, the grade is a common prognostic factor for many cancers, and our results also revealed a better prognosis in patients with lower grades. It shall be noted that the subjects of study were patients with EEC, and hence patients enrolled were those with T1N0M0 stage. TNM staging has always been an important index to judge the prognosis of patients. 22 In our study, patients with T1a stage had a better prognosis than those with T1b stage. In addition, some studies have shown that a combined application of radiotherapy and chemotherapy can reduce postoperative metastasis risk in patients with EEC during endoscopic therapy, which can hence produce a better prognosis.23–25 However, no such result was observed in our study. It may be related to the small number of patients receiving radiotherapy and chemotherapy in the cohort, the large-proportion overlapping of patients who received both radiotherapy and chemotherapy, and the grouping of No chemotherapy and Unknown into one category in the database. 26 In addition, it can also be explained by the side effects of radiotherapy and chemotherapy. In addition to the risk factors mentioned above, there are other factors that have a certain impact on the prognosis of patients, such as lymphatic invasion, vascular invasion, and droplet infiltration.27–29 We did not add them to the study due to the limitations of the database, and we will pay attention to these factors in subsequent studies.
In this study, independent prognostic factors affecting ECSS were obtained by Cox regression analysis, and the nomogram was constructed with verification simultaneously. The nomogram can ensure the universality of the model by avoiding over-fitting. 26 The prediction ability of the model was further evaluated in our study by the C index, ROC curve, and calibration curve. Both C index and AUC values of the ROC curve have been reported to have the ability to assess the accuracy of the nomogram.30,31 The C indexes of both the training and verification cohorts were greater than 0.75, indicating the good differentiation ability of the model, while AUC values of both cohorts were greater than 0.75, also indicating the good prediction accuracy of the model. Furthermore, the calibration curve can judge the consistency between the actual risk and the predicted risk of the model. 32 In the results, the calibration curves of both cohorts also reflected a good consistency of the model in predicting survival.
As more attention on esophageal cancer is paid and with the popularity of endoscopic screening, there is an increasing elevation in the early diagnosis rate of this cancer. Moreover, endoscopic therapy has the advantages of minor trauma and high quality of life after operation, resulting in great demand for this therapy in the clinical setting. Therefore, it is essential to judge whether patients are suitable for endoscopic treatment and the prognostic outcome after treatment.
Endoscopic therapy is recommended for patients with high-grade intraepithelial neoplasia and mucosal carcinoma in the current S3 Guidelines, 33 ESMO Clinical Practice Guidelines, 34 and ACG Clinical Guidelines. 35 Meanwhile, it is also considered as an alternative to surgical treatment for patients with low-risk submucosal carcinoma (adenocarcinoma, T1bsm1, invasion depth of <500 μm, L0, V0, G1/2, no ulceration, and <20 mm in diameter), which, however, has a lower priority than that of surgical treatment.33–35 In this study, the prognosis model of EEC patients who received endoscopic therapy was constructed and the risk grade was classified. It may provide an intuitive understanding on the risk score and risk grade, and hence the risk of these patients receiving endoscopic treatment. The results showed that in each T stage, over 60% of patients had medium and low risk, and the corresponding 5-year survival probability was greater than 70% in the nomogram. It may reveal a good prognosis in these patients and indicate that these parts of patients are suitable for endoscopic therapy in different T stages. At the same time, the Kaplan-Meier curve of each risk grade was drawn in patients with different T stages. Consequently, in T1a, T1b, and T1NOS stages, difference in the prognosis of patients with different risk grades existed. Besides, the model showed a better C index than that of the T staging. All these results may support that relative to T staging, the model constructed in our study may have a superior role for doctors in determining the risk of endoscopic surgery and whether patients are suitable for endoscopic therapy.
At present, the pathological stage is still one of the primary factors for determining the prognosis of patients with EEC. For patients at the T1N0M0 stage receiving endoscopic therapy, as described in the current mainstream AJCC 8th edition staging system, 36 the pathological stage of patients is determined by T staging and grading mutually. With slight difference from the AJCC 8th edition, the S3 Guidelines 33 proposed that the stage of patients is determined by T staging merely. Our model has certain advantages over T staging, with the involvement of G grad. Therefore, the prognosis of patients receiving endoscopic therapy can be analyzed in a more diversified and targeted manner. Significantly, subsequent treatment and follow-up scheme can be formulated for patients according to the risk grade of patients and the evaluation of prognosis. Besides, it can facilitate active reexamination and follow-up for patients with higher risk and poor prognosis.
However, our research also has some limitations. First of all, our study was conducted as a retrospective study and hence there may be an inevitable selection bias, which may have a certain impact on the reliability of the study results. Secondly, there was a limited inclusion of prognostic factors in the study, with the failure of including some potential prognostic factors such as underlying diseases and economic status. Thirdly, there was a relatively smaller sample size in this study. Modeling based on larger sample size may enhance the accuracy and persuasiveness of our study to some extent.
In summary, findings in our study suggest that age, primary site, grading, tumor size, and T staging are independent prognostic factors for EEC patients receiving endoscopic therapy. The model constructed in our study is also verified with good accuracy based on the evaluation by both the training cohort and verification cohort. This model is also superior to TNM staging as confirmed by the evaluation of the prognosis of patients with different risk grades in each T stage. The model may benefit individualized evaluation clinically for patients with EEC, judge the risk of receiving endoscopic therapy, and predict corresponding prognosis, which is conducive to clinical decision-making.
Supplemental Material
sj-docx-1-tag-10.1177_17562848231170470 – Supplemental material for Prognostic analysis and nomogram establishment in patients with early esophageal cancer receiving endoscopic therapy: a population-based study
Supplemental material, sj-docx-1-tag-10.1177_17562848231170470 for Prognostic analysis and nomogram establishment in patients with early esophageal cancer receiving endoscopic therapy: a population-based study by Danping Sun, Jun Yi, Lingqi Gong, Yu Wu and Xiaowei Liu in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
The authors acknowledge the efforts of the Surveillance, Epidemiology, and End Results (SEER) Program tumor registries in the creation of the SEER database.
Declarations
Supplemental material
Supplemental material for this article is available online.
About prior publication
This work has been presented during Digestive Disease Week® (DDW) 2022. The abstract of this work was published in the online supplement of GIE: Gastrointestinal Endoscopy.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
