Abstract
Background:
Helicobacter pylori eradication rates achieved with clarithromycin-based triple therapies are declining due to antibiotic resistance, but data regarding temporal changes in efficacy with these eradication therapies are scarce.
Objective:
To evaluate the efficacy of clarithromycin-based triple eradication regimens over time.
Design:
A comprehensive literature review and time-trend analysis.
Data sources and methods:
Bibliographies of recently published systematic literature reviews were searched and supplemented with a targeted literature review conducted using Medline and Embase databases and ProQuest from conception to May 2021. Studies reporting H. pylori eradication rates of clarithromycin-based triple therapies were included and temporal trends were estimated using a random-effects model.
Results:
Eradication rates for triple therapies containing proton pump inhibitors (PPIs), clarithromycin, and amoxicillin showed a significant decline over the past 23 years (p = 0.0315). However, this decline was not significant when eradication rates achieved with vonoprazan-based triple therapy were included (p = 0.3910).
Conclusion:
Vonoprazan-based triple therapy partially mitigated the decline in eradication rates seen with PPI-based triple therapy, likely due to more powerful acid suppression of vonoprazan.
Background
Helicobacter pylori (H. pylori) is the most common chronic bacterial infection, estimated to infect up to half of the world’s population, ranging from 20% to 50% in industrialized countries and up to 80% in developing countries. 1 Because of the established role of H. pylori in gastroduodenal disease, current clinical guidelines recommend eradication therapy for all patients diagnosed with active infection.2,3 Careful selection of first-line eradication therapy is important to guarantee high efficacy and limit the risk of antibiotic resistance. 3
Clarithromycin-based eradication therapies were successfully introduced in the 1990s 4 when their efficacy was at its peak and 7-day omeprazole–clarithromycin–amoxicillin treatment demonstrated an eradication rate of 94%. 5 Up until around the 2000s, reported clarithromycin resistance rates in H. pylori were generally at or below 10%. 6 However, between 2001 and 2014, clarithromycin resistance increased dramatically to over 20% in the United States, Europe, and Asia, with rates as high as 60% reported in some countries.6,7 In the United States, current resistance rates to clarithromycin, levofloxacin, and metronidazole are all above 30%.8,9 Infection with a clarithromycin-resistant strain of H. pylori has been associated with a sevenfold risk of treatment failure among patients who receive clarithromycin-containing regimens. 9 Published literature suggests that eradication rates achieved with available regimens, particularly those containing clarithromycin, are declining2,9,10; however, data regarding changes in the efficacy of clarithromycin-based eradication regimens over time are scarce.
Novel approaches are needed to mitigate the effects of antibiotic resistance and offer effective empiric eradication of H. pylori infection. Vonoprazan is a novel potassium-competitive acid blocker 11 that provides more potent and durable acid suppression than traditional proton pump inhibitors (PPIs). Recent trial data suggest that substituting vonoprazan for PPI in clarithromycin-based triple regimens improves eradication rates. 12 We aimed to evaluate the efficacy of clarithromycin-based triple regimens, including vonoprazan-based triple therapy, over time.
Methods
A comprehensive literature review via bibliographic searches of three key systematic literature reviews (Rokkas et al., 13 Xin et al., 14 and Therapeutics Initiative of the University of British Columbia 15 ) and a targeted literature review using index terms for H. pylori (Medical Subject Heading terms: ‘H. pylori’, ‘H pylori’, ‘Helicobacter pylori’ along with ‘eradication’) was performed. The full search strategy and methodology was previously described by Malfertheiner et al. 16 Databases searched from inception through the search date of May 2021 included Embase and MEDLINE via ProQuest. This review was not registered with PROSPERO; however, future systematic literature reviews will be registered with PROSPERO. A single reviewer assessed eligibility during title–abstract screening, and inclusion was determined via full-text screening according to PICOS criteria. Search results were further limited by interventions to identify prospective randomized studies reporting eradication rates of common triple therapies comprising either a PPI or vonoprazan, together with clarithromycin and amoxicillin.
From the included studies, treatment arms for clarithromycin-based triple therapies were extracted and a meta-analysis was conducted to estimate pooled H. pylori eradication rates with 95% confidence intervals based on a random-effects model. The meta-analysis was stratified by publication year, and between-group differences were tested using Cochrane’s Q-test.
A mixed-effects linear model was constructed with publication year as a predictor. A random intercept was included to account for within-study correlations between treatment arms from the same study. The coefficient ‘study year’ was used to evaluate the trend in H. pylori eradication rates over time. Additional analyses were conducted to include and exclude vonoprazan-based triple therapy.
Results
Overall, 67 study arms with clarithromycin-based triple therapies from 38 trials conducted in East Asian, South Asian, and Western countries including North America were used in the analysis (Figure 1, Table 1). The identified clarithromycin-based triple therapy regimens had the following acid suppression backbones: vonoprazan, rabeprazole, lansoprazole, esomeprazole, omeprazole, and mixed PPI.

Literature flow diagram.
Included studies.

AMX, amoxicillin; CLR, clarithromycin; ITT, intention to treat; PPI, proton pump inhibitor.
Before 2001, H. pylori eradication rates averaged around 83%. When considering all clarithromycin-based triple regimens in this study, the pooled H. pylori eradication rates over time were comparable to historic rates (82.00% post-2015; p = 0.3460), and differences between subgroups were not significant (Table 2).
Time stratified, pooled HP eradication rates.
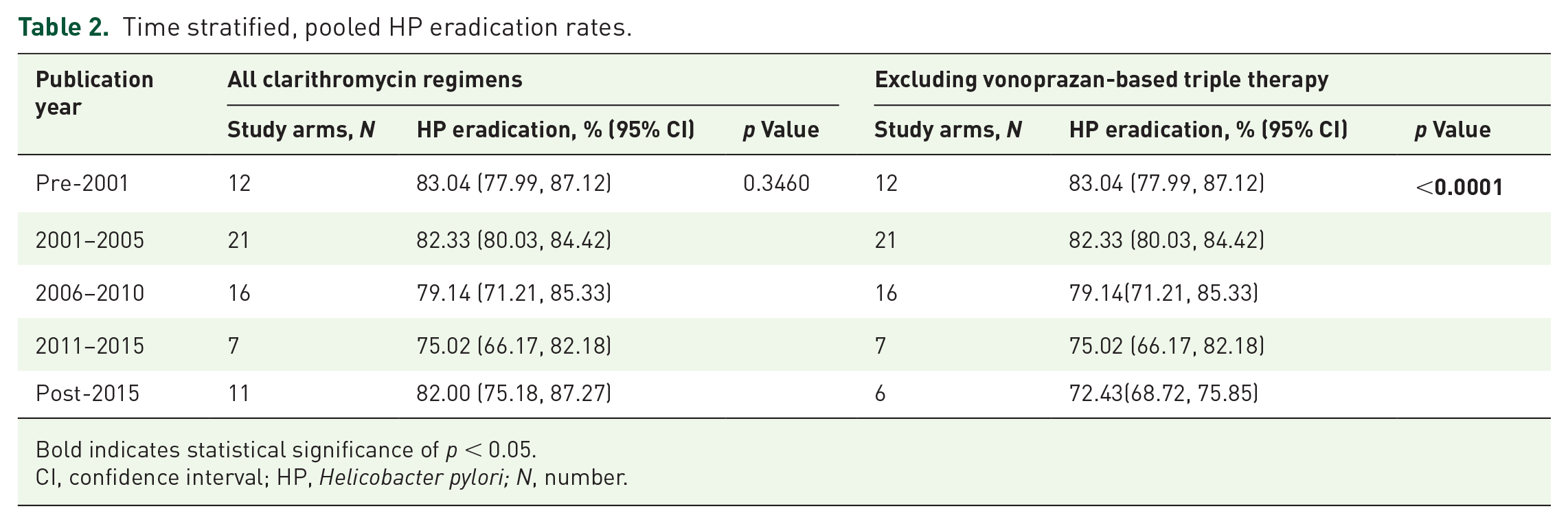
Bold indicates statistical significance of p < 0.05.
CI, confidence interval; HP, Helicobacter pylori; N, number.
Since eradication rates are known to be declining globally with PPI-based triple therapies, the impact of vonoprazan-based triple therapy on eradication rate trends was explored by analyzing the eradication rates over time of all clarithromycin-based triple regimens except vonoprazan-based triple therapy. After excluding vonoprazan-based triple therapy, pooled H. pylori eradication rates post-2015 for all other clarithromycin-containing triple therapies were significantly lower than historic rates (pre-2001: 83.04% versus post-2015: 72.43%; p < 0.0001). The results of the mixed-effects linear model (Figure 2) substantiated this negative trend. The negative time trend for eradication rates achieved with PPI triple therapies was statistically significant (p = 0.0315), yet once vonoprazan-based triple therapy was included, the trend was no longer significant (p = 0.3910).

HP eradication rates in clarithromycin-containing triple regimens over time. The annual coefficient represents the slope of eradication rate over time overall. The treatment coefficients represent the difference in eradication rate of the regimen of interest and the reference regimen (esomeprazole) at any point in time. Coefficients are provided in percentage scale. Calculated 2022 eradication rates are based on the year coefficient and treatment coefficient, extrapolated for 2022 based on the model. p Values represent the significance in the time trend in eradication rates (i.e. slope of the respective line produced in the linear model).
Overall, significantly lower H. pylori eradication rates were observed over time when assessing all clarithromycin-based treatments together (0.62% reduction per year; p = 0.0287). Vonoprazan-associated eradication rates were significantly higher than PPI-containing clarithromycin-based triple regimens, including the reference regimen, esomeprazole triple therapy (+14.81%; p < 0.0001) (Figure 2). When the results of this model were extrapolated to 2022 for each acid suppression backbone, vonoprazan was associated with an 84.20% eradication rate (i.e. no reduction from historical rate), whereas the PPIs all had estimated eradication rates below or equal to 70% (Figure 2).
Discussion
Lack of antibiotic stewardship, short treatment durations, differing usage and policies by geography, and increased, and often inappropriate, use of macrolides have all been possible contributors to increased clarithromycin resistance and decreased effectiveness of clarithromycin-based H. pylori eradication regimens.55,56 Guidance from both Europe and North America recommends against first-line empiric H. pylori treatment with clarithromycin containing PPI-based triple therapy because of antibiotic resistance and the risk of potential treatment failure.2,10 Nevertheless, a recent review of real-world evidence in North America suggests the PPI-based triple therapies are still commonly used. 57 Recent evidence suggests that triple therapy with the potassium-competitive acid blocker vonoprazan is an effective treatment for H. pylori, offering high rates of eradication, even among patients infected with clarithromycin-resistant H. pylori. 26
The results of this analysis confirmed that H. pylori eradication rates for PPI-based clarithromycin-based triple therapies have continued to decline over time. Inclusion of vonoprazan-based triple therapy partially mitigated the observed decline in eradication rates. Higher eradication rates observed with vonoprazan-based triple therapy 26 in comparison to the historical eradication rates of PPI-based triple regimens suggest that vonoprazan-based triple therapy may be less impacted by increasing clarithromycin resistance.
Vonoprazan acts on H+, K+-ATPase in parietal cells in an acid-independent manner, providing a rapid antisecretory effect that is maintained over 24 h. 58 Moreover, vonoprazan demonstrated superior acid suppression compared to lansoprazole, with a more rapid and sustained acid-inhibitory effect. 59 Acid suppression and maintenance of an intragastric pH between 6 and 8 is essential to optimal antibiotic action, particularly for amoxicillin, which is acid labile and requires bacterial replication for its antimicrobial effects.60,61 Thereby, the efficacy of vonoprazan-based triple therapy may be driven, at least in part, by potent acid suppression and the related effectiveness of amoxicillin, limiting the impact of clarithromycin resistance.
In a recent network meta-analysis which provided an indirect comparison of available H. pylori eradication regimens, vonoprazan-based triple therapy showed the highest relative efficacy, ranking higher according to surface under the cumulative rank than bismuth quadruple therapy and all PPI-based triple therapy regimens (regardless of individual PPI backbone). 16 Interestingly, vonoprazan dual therapy with amoxicillin ranked second only to vonoprazan-based triple therapy versus individual PPI triple therapies, providing further evidence that the more durable and potent acid suppression provided by vonoprazan may support maintained stability and optimal effectiveness of amoxicillin compared to PPIs.
The interpretation of the results of this temporal analysis is limited by the amount of available published evidence for vonoprazan-based triple therapy, since it has only recently become available in the United States. It was not feasible to correct for the heterogeneity of dosage and duration of treatment in multivariable models due to the limited amount of data. Among the studies analyzed, amoxicillin dosage was largely comparable between vonoprazan studies and other studies from 2015 onward.
Conclusion
Overall, the temporal decline in effectiveness of clarithromycin-based triple therapies may be mitigated by the advent of triple therapies with more potent acid suppression.
Footnotes
Acknowledgements
None.
