Abstract
Background:
Patients with inflammatory bowel disease (IBD) are often accompanied by a more significant burden of depression or anxiety, and approximately one-third are prescribed antidepressants. However, previous studies on the efficacy of antidepressants in IBD have shown inconsistent results.
Objectives:
To evaluate the effect of antidepressants on depression, anxiety, disease activity, and quality of life (QoL) in IBD patients.
Design:
A systematic review and meta-analysis.
Methods:
We searched MEDLINE via Ovid, EMBASE via Ovid, the Cochrane Library, CINAHL, PsycINFO, Chinese CBM Database, China National Knowledge Infrastructure, VIP, and Wanfang Database from inception to 13th July 2022 without language restrictions.
Results:
In all, 13 studies containing 884 individuals were included. Compared with the control group, antidepressants were superior in reducing depression scores [standardized mean difference (SMD) = −0.791; 95% confidence interval (CI): −1.009 to −0.572; p < 0.001], anxiety scores (SMD = −0.877; 95% CI: −1.203 to −0.552; p < 0.001), and disease activity scores (SMD = −0.323; 95% CI: −0.500 to −0.145; p < 0.001). Antidepressants had a positive effect in reaching clinical remission [risk ratio (RR) = 1.383; 95% CI: 1.176–1.626; p < 0.001]. Higher physical QoL (SMD = 0.578; 95% CI: 0.025–1.130; p = 0.040), social QoL (SMD = 0.626; 95% CI: 0.073–1.180; p = 0.027), and Inflammatory Bowel Disease Questionnaire (SMD = 1.111; 95% CI: 0.710–1.512; p < 0.001) were found in the experimental group. No significant differences were observed in clinical response (RR = 1.014; 95% CI: 0.847–1.214; p = 0.881), psychological QoL (SMD = 0.399; 95% CI: −0.147 to 0.944; p = 0.152), and environmental QoL (SMD = 0.211; 95% CI: −0.331 to 0.753; p = 0.446).
Conclusion:
Antidepressants are effective for ameliorating depression, anxiety, disease activity, and QoL in IBD patients. Due to most studies having a small sample size, further well-designed studies are required.
Introduction
Inflammatory bowel disease (IBD) is a chronic disorder that comprises ulcerative colitis (UC) and Crohn’s disease (CD), the former affecting only the colon, and the latter can involve any part of the gastrointestinal tract. 1 IBD is a lifelong disease characterized by repeated remissions and relapses with no cure. The clinical manifestations of IBD are diverse, and typical symptoms include abdominal pain, weight loss, diarrhea, etc. 2 IBD has a significant impact on the quality of life (QoL) of patients. A systematic review confirmed that compared with healthy individuals, the QoL of patients with IBD was poorer. 3 Further studies found that the QoL of IBD was worse during active disease. 4 In addition, IBD treated with steroids had lower QoL than that of non-users,5,6 which was because steroids use was associated with increased disease activity, 7 and side effects of steroids could affect QoL, such as moon face, acne, weight gain, etc. 8 A recent review reported the overall prevalence of IBD surpasses 0.3%. 9 In the United States, the prevalence of IBD in children and adults in 2016 is 77 and 478.4 per 100,000 people, respectively. 10 The prevalence of IBD is estimated to be 970 per 100,000 population in the United Kingdom and 725 per 100,000 population in Canada.11,12 In South Korea, the prevalence of UC and CD between 2009 and 2016 has increased from 41.4 and 16.0 to 66.0 and 29.6 per 100,000 population, respectively. 13
IBD is often accompanied by psychiatric disorders encompassing depression and anxiety.14,15 The incidence of depression is significantly higher in IBD patients than in non-IBD patients. 16 Two recent reviews revealed the pooled prevalence of depression in IBD was 21.2% 17 and 25.6%, 18 and the pooled prevalence of anxiety was 35.1% 17 and 32.1%, 18 respectively. Depression or anxiety symptoms negatively correlate with the QoL in patients with IBD.19,20 Meanwhile, IBD with depression or anxiety has a significantly higher disease burden, with some studies reporting higher rates of steroid or biologic use, hospitalization, surgery, relapse, and use of emergency resources in such populations.21–23 Alexakis et al. 24 conducted a systematic review and meta-analysis and found no association between comorbid depression and worsening IBD disease course. However, a recent meta-analysis on this subject, which included more original studies with longer follow-up, suggested that symptoms of depression or anxiety were significantly related to worsening IBD disease course. 25
Approximately 10–30% of patients with IBD have been treated with antidepressants. 26 However, there are no standard strategies for IBD patients with depression or anxiety. Most studies have shown that IBD benefits from antidepressants, whereas a small randomized controlled trial (RCT) found no benefit of fluoxetine on the condition or mental health of IBD patients. 27 A recent review by Mikocka-Walus in 2019 exploring the efficacy of antidepressants in patients with IBD failed to reach definitive conclusions due to the small sample size and the inclusion of non-RCTs (NRCT). 28 In this study, we synthesized new data to systematically investigate the effect of antidepressants on depression, anxiety, disease activity, and QoL in IBD population.
Methods
Search strategy
We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines to develop our search strategy. 29 Searches of MEDLINE via Ovid, EMBASE via Ovid, the Cochrane Library, CINAHL, PsycINFO, Chinese CBM Database, China National Knowledge Infrastructure, VIP, and Wanfang Database from inception to 13th July 2022 were conducted, using the following terms: inflammatory bowel disease, Crohn’s disease, ulcerative colitis, depression, depressive disorder, anxiety, anxiety disorder, and antidepressants. To get more original research, we did not restrict languages. Search strategies in every online database are introduced in the Supplemental Material. This study was not registered on PROSPERO, but we will complete the corresponding registration in future studies.
Inclusion and exclusion criteria
The inclusion criteria were as follows: (1) all published RCTs; (2) individuals were diagnosed with IBD, including CD and UC, according to standard practice; (3) antidepressants, including selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), norepinephrine reuptake inhibitors, norepinephrine–dopamine reuptake inhibitors, noradrenergic and specific serotonergic antidepressants, serotonin antagonist and reuptake inhibitors, monoamine oxidase Inhibitor, tricyclics, and other unclassified antidepressants, were used in the intervention group; (4) outcomes were continuous outcomes of depression, anxiety, disease activity, QoL or dichotomous outcomes of clinical remission or clinical response.
The exclusion criteria were as follows: (1) animal experiments; (2) observational studies, retrospective studies, case reports, letters, reviews, and meta-analysis; (3) Chinese medicine treatment for IBD; and (4) no full text, or insufficient data for calculation.
Selection studies
Two investigators independently screened studies that met pre-determined inclusion and exclusion criteria. Discrepancies were resolved by discussion, and if required, the third author was asked to arbitrate.
Data extraction
Two reviewers conducted data extractions and extracted the following data if mentioned in the original literature: (1) basic information: first author name, publication year, country, sample size, interventions of treatment and control groups, trial duration; (2) demographic characteristics: age, gender, type of IBD, course of illness, and severity; (3) results: psychological assessment (depression and anxiety), disease activity (disease activity score, laboratory test results), clinical efficacy (clinical remission and clinical response defined by the respective article authors), and QoL. For each trial, we extracted data at the final time of follow-up. For missing or unclear information, we contacted the investigators to obtain relevant information.
Quality assessment
The risk of bias for each study was assessed by two independent authors according to the Cochrane risk of bias tool, covering six domains. 30 Each item was categorized as ‘Low risk,’ ‘High risk’, or ‘Unclear risk’. Any disagreement was discussed, reaching a consensus.
Statistical analysis
All analyses were performed using RevMan 5.4 and STATA. 17.0. Standardized mean difference (SMD) and 95% confidence interval (CI) were used to characterize continuous outcomes. Risk ratio (RR) and 95% CI were used to represent discontinuous outcomes. We calculated Cochrane chi2 and I2 to evaluate the heterogeneity across studies. A p value of less than 0.10 was defined as significant heterogeneity. Thresholds for the interpretation of the I2 statistic are as follows: 0–40%, indicating that heterogeneity may not be important; 30–60% may represent moderate heterogeneity; 50–90% may represent substantial heterogeneity; and 75–100%, representing considerable heterogeneity. 31 If p < 0.10 or I2 > 40%, we considered the heterogeneity significant and conducted a meta-analysis using a random-effects model. The fixed-effect model was applied if p > 0.10 or I2 < 40%. We generated a funnel plot to test publication bias and performed Begg’s and Egger’s tests. A sensitivity analysis was executed to check the robustness of the summarized results. In addition, subgroup analysis and meta-regression were conducted to explore the possible source of heterogeneity, including mean age, gender ratio, type of antidepressants, and length of treatment.
Results
Literature search
In all, 1556 potentially eligible studies were identified. After removing duplicates, we scanned the titles and abstracts of 1171 articles. Articles excluded were systematic reviews, non-RCTs, not IBD or UC or CD related, or without antidepressant treatment. In all, 46 papers were read in full. We contacted the authors for articles with insufficient data, and only Liang 32 responded. In total, 33 papers were excluded for different reasons. In all, 18 studies included combination therapy without separate data on antidepressant efficacy, five were not RCTs, four lacked appropriate control measures, three were clinical trials with overlapping data from published literature, and three did not explore the effect of antidepressants on depression or anxiety symptoms (Figure 1). References of qualified articles were also read in full. No additional citations were added. In all, 13 articles were finally included.27,32–43
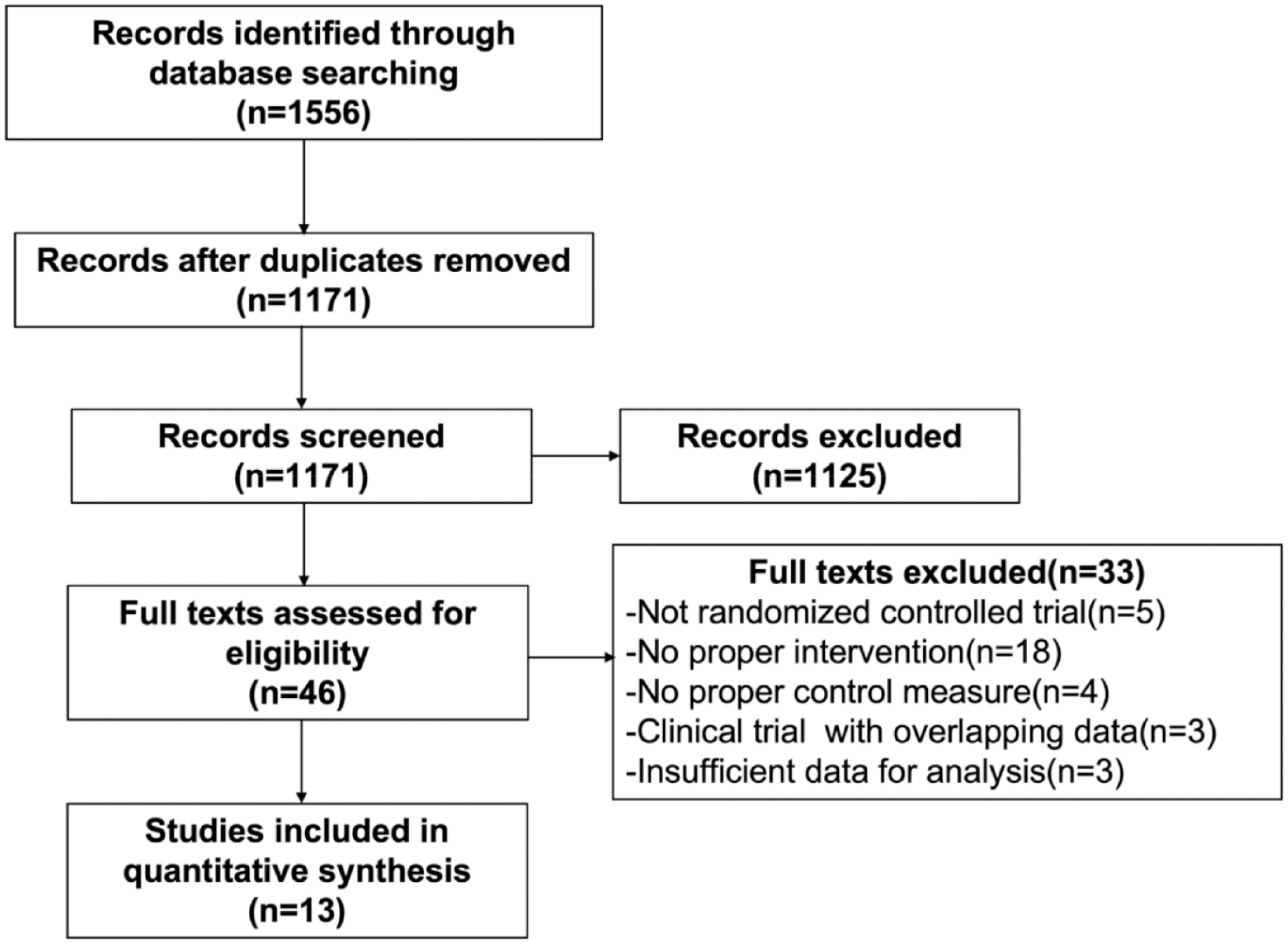
Flow diagram of study selection.
Study characteristics
A total of 884 individuals were recruited. There were 446 participants in the experimental group and 438 in the control group. The sample sizes ranged from 18 to 126. One study was from Australia 27 and one from Iran, 42 and the others were from China.32–41,43 All eligible articles reported the mean age and sex ratio. Most RCTs enrolled patients with UC,33–41,43 only Mikocka-Walus et al. 27 included patients with CD, and Liang et al. 32 and Daghaghzadeh et al. 42 recruited patients with UC or CD. The mental health of the patients recruited in each study was disparate, with six trials only recruiting IBD with depression,33,35,37–39,41 while four enrolled IBD with depression or anxiety,32,34,40,43 and three studies had no requirements on the psychological states of IBD patients included.27,36,42 Follow-up durations ranged from 2 weeks to 12 months. Interventions differed between studies. Sertraline was used in six experiments,33,35,37–39,41 paroxetine was used in four experiments,34,36,40,43 and venlafaxine, 32 fluoxetine, 27 and duloxetine 42 were used in one experiment each.
Assessment tools for depression and anxiety varied among trials. Five studies used Hamilton Depression Scale to measure the severity of depression,34,36,38,40,43 five utilized the Self-Rating Depression Scale,33,35,37,39,41 and three used Hospital Anxiety and Depression Scale-Depression (HADS-D).27,32,42 Meanwhile, the Hamilton Anxiety Scale was employed to assess the severity of anxiety in four RCTs,34,36,40,43 and HADS-Anxiety was applied in three trials.27,32,42 Regarding the secondary outcome measures, seven articles reported disease activity scores, of which four used the Ulcerative Colitis Activity Index,35,37,38,41 one used the Lichtiger Colitis Activity Index, 42 one used the Clinical Activity Index, 43 and one paper used CDAI and Mayo score, respectively. 32 Other metrics for disease activity assessment involved erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), tumor necrosis factor alpha (TNF-α), etc. The definition of clinical remission and clinical response was inconsistent. Clinical remission was used to examine clinical efficacy in 10 experiments,27,33–41 and 9 of them also reported clinical response.33–41 The Inflammatory Bowel Disease Questionnaire (IBDQ), physical QoL, psychological QoL, social QoL, and environmental QoL were applied to measure the QoL. Characteristics of included trials are displayed in Table 1.
Characteristics of the included studies.

IBD, inflammatory bowel disease; HADS-D, depression subscale of the Hospital Anxiety and Depression Scale; HADS-A, anxiety subscale of the Hospital Anxiety and Depression Scale; ST, standard therapy; HASD, Hospital Anxiety and Depression Scale; CDAI, Crohn’s Disease Activity Index; ESR, erythrocyte sedimentation rate; CRP, C-reactive protein; TNF-a, tumor necrosis factor α; IL-10, interleukin 10; IBDQ, Inflammatory Bowel Disease Questionnaire; UC, ulcerative colitis; NA, no available; SDS, Self-rating depression scale; HAMD-17, 17-Item Hamilton Rating Scale for Depression; HAMA, Hamilton Anxiety Scale; UCAI, ulcerative colitis activity index; HAMD, Hamilton Depression Scale; CD, Crohn’s disease; QOL, quality of life; LCAI, Lichtiger Colitis Activity Index; DAI, disease activity index; CAI, clinical activity index.
Risk of bias
The appraisal results of the risk of bias for included trials are presented in Figures 2 and 3.

Risk of bias graph: review authors’ judgments about each risk of bias item presented as percentages across all included studies.

Risk of bias summary: review authors’ judgments about each risk of bias item for each included study.
Primary outcomes
Depression
All RCTs completed assessments of depression. Since I2 = 56.9% (p = 0.006), we reported random-effect models. Significant reductions in depressive scores were observed with the use of antidepressants in IBD patients (Figure 4(a); SMD = −0.791; 95% CI: −1.009 to −0.572; p < 0.001). The funnel plot showed no publication bias (Figure 4(b)). The result was consistent with Begg’s (p = 0.669) and Egger’s tests (p = 0.554). A sensitivity analysis was performed by excluding included articles one by one, indicating that the result was consistent and without fluctuation (Figure 4(c)). Subgroup analyses based on mean age, sex ratio, type of antidepressants, and length of treatment found that the intervention did not affect depression in the SNRI subgroup and that heterogeneity between studies could be explained by the length of treatment (Table 2). The meta-regression also demonstrated that the length of treatment was associated with heterogeneity (Table 3).
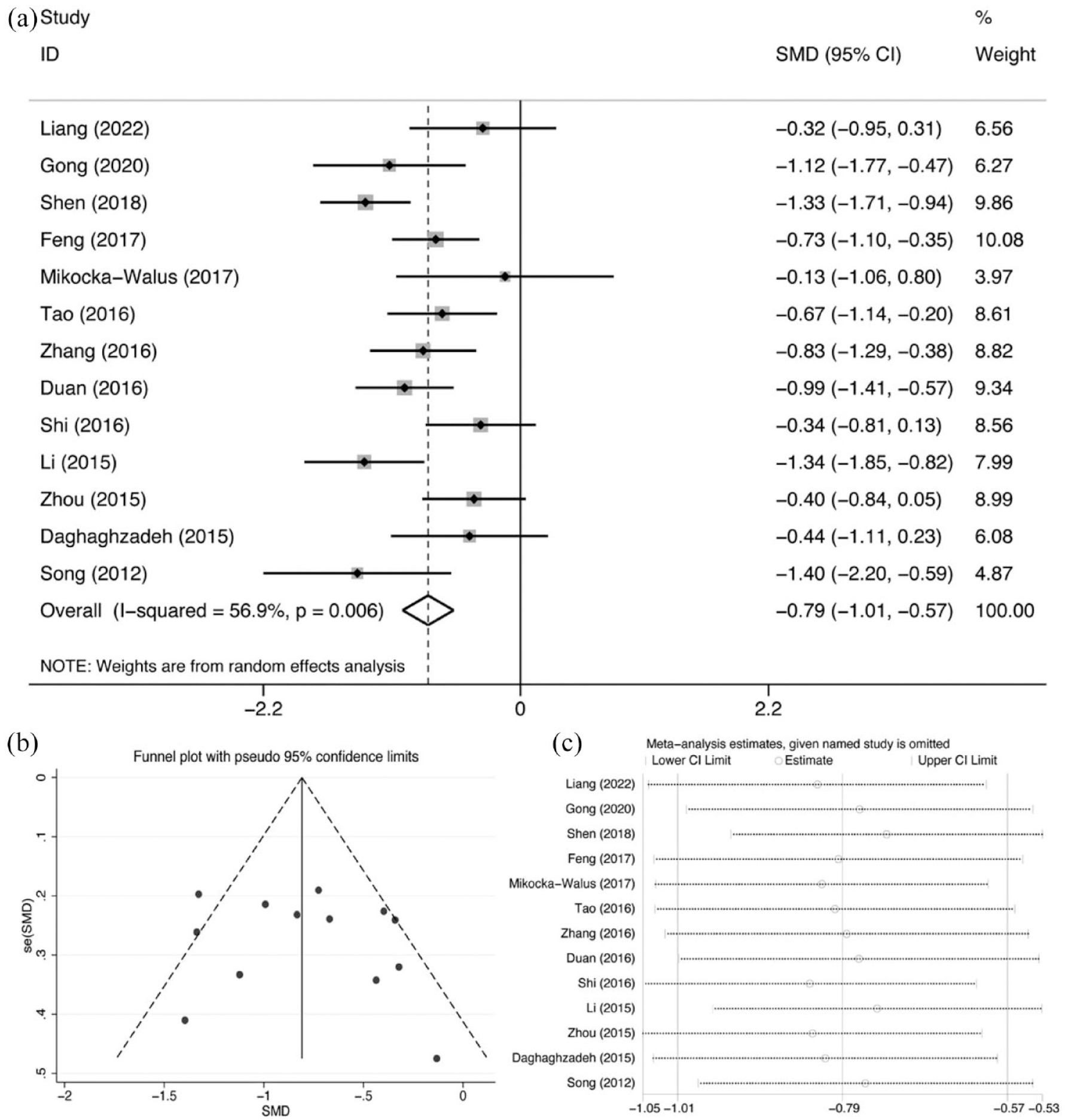
Comparison of depression symptoms: (a) forest plot, (b) funnel plots, and (c) sensitivity analysis.
Subgroup analysis for depression symptoms.

CI, confidence interval; N, number of trials; SMD, standardized mean difference;SNRI, serotonin-norepinephrine reuptake inhibitors; SSRI, selective serotonin reuptake inhibitor.
Meta-regression analysis for depression symptoms.
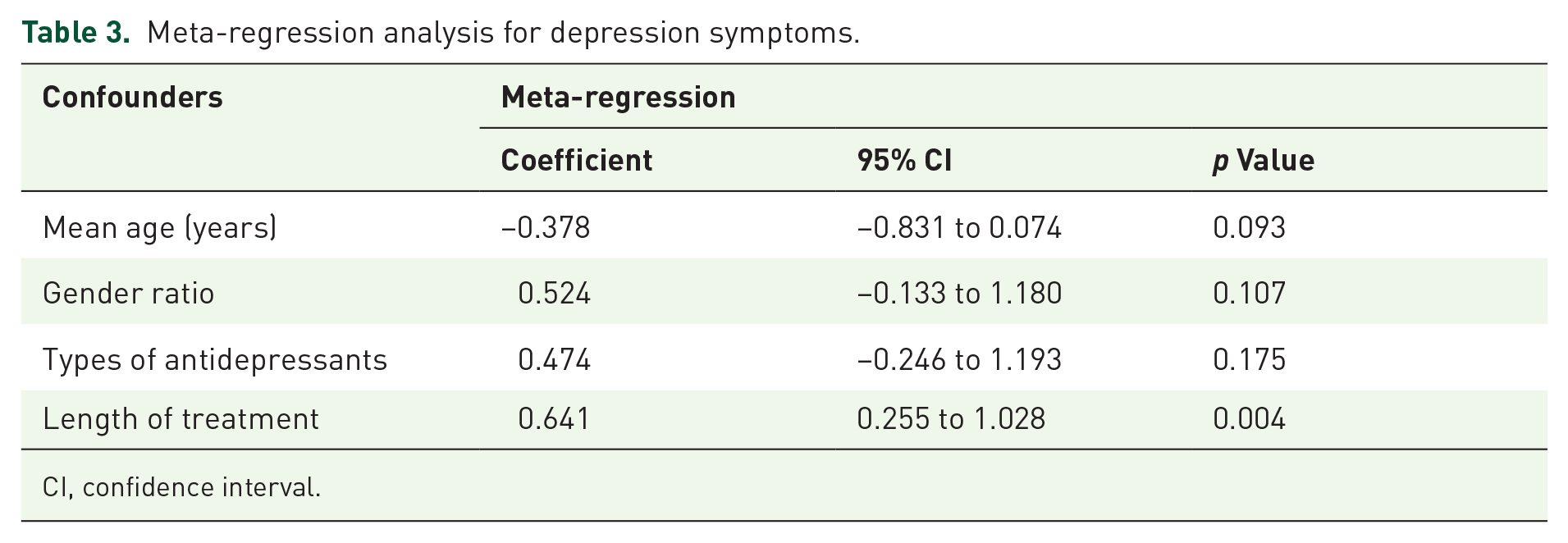
CI, confidence interval.
Anxiety
Seven studies discussed the impact of antidepressants in reducing anxiety scores in patients with IBD. We pooled data from 395 participants using a random-effect model (Figure 5(a); I2 = 53.6%; p = 0.044), indicating that antidepressants did ease anxiety symptoms (Figure 5(a); SMD = −0.877; 95% CI: −1.203 to −0.552; p < 0.001). Publication bias and meta-regression were not assessed due to fewer than 10 articles.44,45 Sensitivity analysis confirmed the robustness of the results (Figure 5(b)). Subgroup analysis indicated that the length of treatment might be a contributing factor to heterogeneity. There was no evidence that interventions differed in efficacy across subgroups of mean age and type of antidepressant (Table 4).

Comparison of anxiety symptoms: (a) forest plot and (b) sensitivity analysis.
Subgroup analysis for anxiety symptoms.

CI, confidence interval; N, number of trials; SMD, standardized mean difference; SNRI, serotonin-norepinephrine reuptake inhibitors; SSRI, selective serotonin reuptake inhibitor.
Secondary outcomes
Disease activity scores
Eight studies assessed the ability of antidepressants on disease activity using correlation scales. Antidepressants could significantly alleviate the severity of the disease (Figure 6(a); SMD = −0.323; 95% CI: −0.500 to −0.145; p < 0.001), with low heterogeneity between studies (Figure 6(a); I2 = 22.7%; p = 0.241). Sensitivity analyses were conducted, and no obvious changes were observed after each study was sequentially omitted (Figure 6(b)). We noted no significant difference in disease activity scores in the subgroup with a mean age > 40 (Supplemental Table 1).
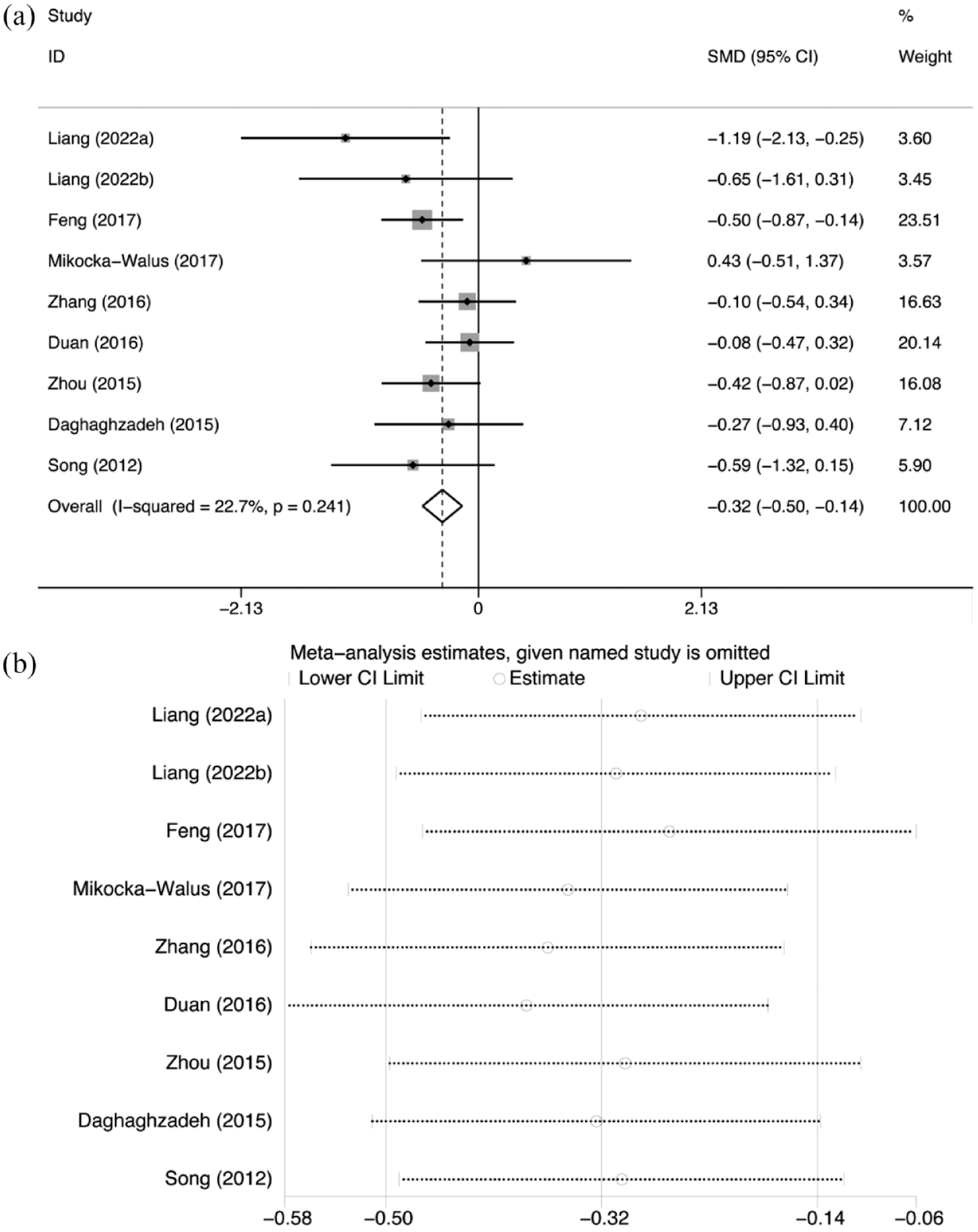
Comparison of disease activity scores: (a) forest plot and (b) sensitivity analysis.
Clinical remission
Ten RCTs reported data for the effect of antidepressants on clinical remission. The pooled results revealed that antidepressants were effective in the induction of clinical remission (Figure 7(a); RR = 1.383; 95% CI: 1.176–1.626; p < 0.001; I2 = 34.7%). Visual inspection of the funnel plot appears to be asymmetric (Figure 7(b)). However, Begg’s (p = 0.592) and Egger’s (p = 0.270) tests demonstrated that publication bias was not significant. Sensitivity analysis showed that the results were stable (Figure 7(c)). Subgroup analysis according to mean age, length of treatment, and gender ratio showed no effect of antidepressants on clinical remission in subgroups with a gender ratio of ⩽1 or treatment duration of ⩽8 weeks (Supplemental Table 2). Regression analysis also did not hint at the source of heterogeneity (Supplemental Table 3).

Comparison of clinical remission: (a) forest plot, (b) funnel plots, and (c) sensitivity analysis.
Clinical response
We pooled data from nine manuscripts and found no benefit from the intervention in inducing a clinical response (Figure 8(a); RR = 1.014; 95% CI: 0.847−1.214; p = 0.881; I2 = 0%). Sensitivity analysis confirmed the stability of the results (Figure 8(b)). In terms of clinical response, none of the subgroup analyses indicated an advantage of the intervention (Supplemental Table 4).
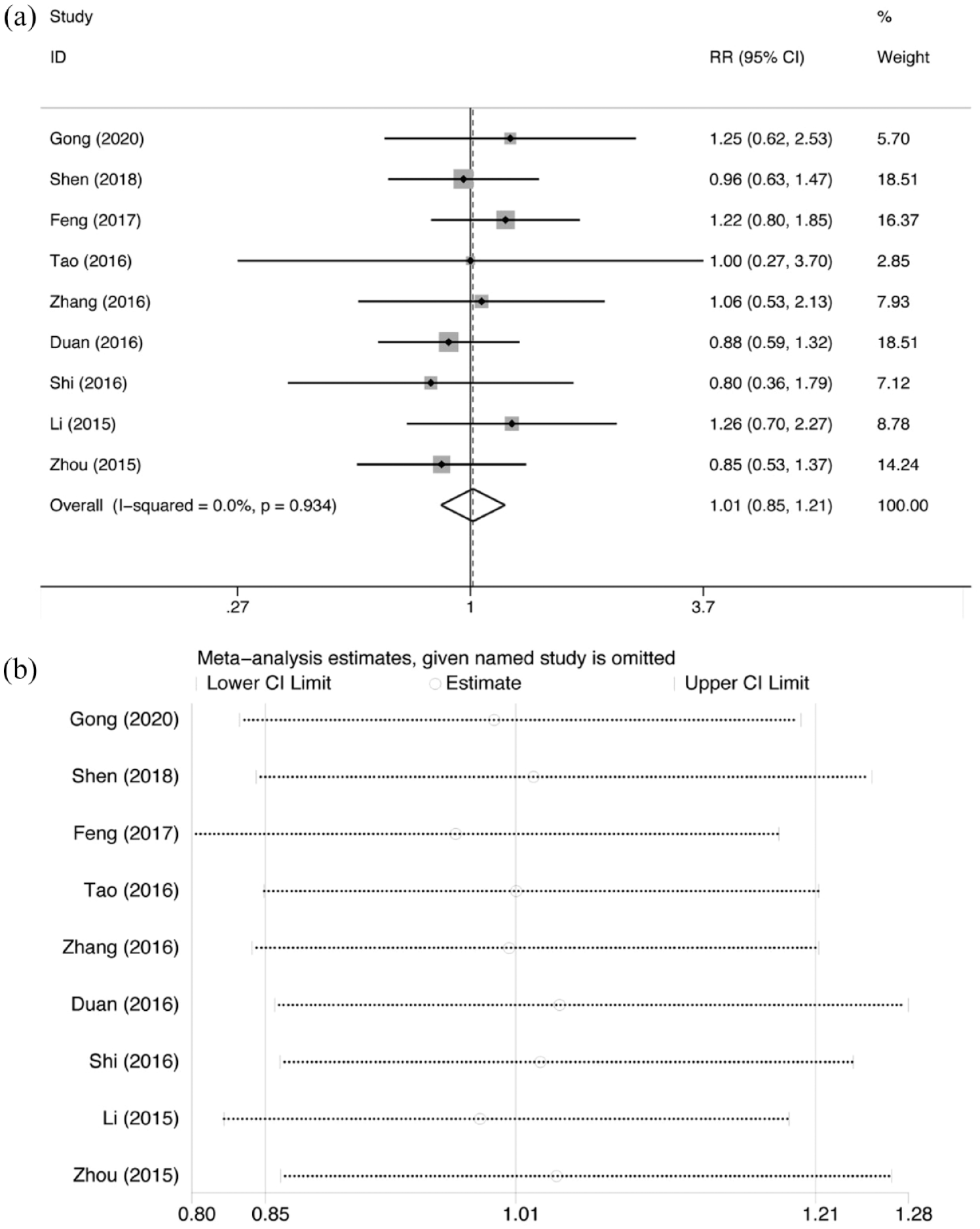
Comparison of clinical response: (a) forest plot and (b) sensitivity analysis.
Quality of life
Two articles documented data on physical QoL, psychological QoL, social QoL, and environmental QoL. We only observed benefits in physical QoL (Figure 9(a); SMD = 0.578; 95% CI: 0.025–1.130; p = 0.040; I2 = 0%), and social QoL scores (Figure 9(c); SMD = 0.626; 95% CI: 0.073–1.180; p = 0.027; I2 = 0%). No significant change in psychological QoL (Figure 9(b); SMD = 0.399; 95% CI: −0.147 to 0.944; p = 0.152; I2 = 0%) and environmental QoL (Figure 9(d); SMD = 0.211; 95% CI: −0.331 to 0.753; p = 0.446; I2 = 0%) scores.

Comparison of QoL: (a) physical QoL, (b) psychological QoL, (c) social QoL, (d) environmental QoL, and (e) IBDQ.
Two other articles used the IBDQ as a questionnaire to assess health-related QoL in IBD patients. Compared with the control group, the IBDQ score of the experimental group was significantly higher (Figure 9(e); SMD = 1.111; 95% CI: 0.710–1.512; p < 0.001; I2 = 28.2%).
All secondary outcomes were analyzed using a fixed-effects model. Some other secondary outcomes, such as ESR, CRP, etc., were mentioned in only one study, thus, we did not perform a meta-analysis.
Discussion
The results suggested that using antidepressants in IBD patients could mitigate depression, anxiety, and disease activity, and improve physical QoL, social QoL and IBDQ scores. As for clinical efficacy, antidepressants were effective only in the induction of clinical remission not in clinical response. Given that clinical remission has far higher heterogeneity than clinical response, this finding needs to be interpreted with caution.
Sensitivity analysis proved the robustness and stability of our results, although significant heterogeneity was observed when analyzing changes in depression and anxiety scores. Further subgroup analysis suggested that intervention duration might be a source of heterogeneity. The WFSBP guidelines recommend 2–4 weeks to evaluate the early efficacy of antidepressants, and at least 8–10 weeks to achieve maximum symptom reduction. 46 However, a retrospective population-based study in the United Kingdom found that 34% of IBD discontinue antidepressants within 28 days, which may be because of poor tolerability. 47 We, therefore, selected the minimum treatment duration, 8 weeks, as the basis for the subgroup analyses. Antidepressants were effective in both subgroups, it was interesting to note that compared with IBD patients treated for >8 weeks, patients treated for ⩽8 weeks benefited more from antidepressants in alleviating depression and anxiety symptoms. This result might arise from some studies with treatment duration >8 weeks that did not recruit IBD with depression or anxiety. Subgroup analyses showed that SNRI improved depressive symptoms in IBD patients, but the difference was not significant. Considering that the sample size of the subgroup was small may not be able to check out the difference between the control and experimental groups. No significant improvement in disease activity scores was observed between the experimental and control groups, with a mean age >40. Neither sex ratio ⩽1 nor treatment duration ⩽8 weeks of IBD had clinical remission benefits from antidepressant use. In addition to the small sample size, these subgroups were highly heterogeneous, and we also needed to interpret these results with caution.
A previous systemic review and meta-analysis, limited by sample size and the inclusion of NRCTs, had cautiously recommended that antidepressants improve depressive and anxiety symptoms in IBD patients. 28 In this paper, all the studies included were RCTs, and the quality of evidence may be higher. In addition, meta-analysis is a statistical method that can combine results from different but thematically similar studies. In addition to the relevant English databases, the Chinese databases were also searched in this paper, and articles in Chinese that met inclusion and exclusion criteria were included. Furthermore, every effort was made to contact the authors of potentially eligible research for which data of interest were not available in the published manuscript, and in the end, only the authors of one study published in 2022 responded. 32 We included a total of 13 eligible articles, nine more than included in the Cochrane review, and four times more people than included in the Cochrane review. 28 Finally, subgroup analysis was performed to explore the potential sources of heterogeneity, the efficacy of antidepressants for IBD with different clinical features, and the effect of different antidepressants in IBD. This meta-analysis is helpful to standardize the use of antidepressants in patients with IBD.
Antidepressants work by compensating for the reduction or absence of neurotransmitters. 48 Previous studies have found lower levels of serotonin and norepinephrine in the inflamed mucosa of patients with IBD than in healthy controls.49,50 It is hypothesized that the SSRI- and SNRI-induced increase in serotonin and norepinephrine is the underlying reason for the efficacy of antidepressants in IBD. Moreover, a growing number of articles have reported that antidepressants have anti-inflammatory properties.51–53 In animal models of IBD, antidepressants reduced the serum concentrations of interleukin 1β and TNF, attenuated intestinal myeloperoxidase levels, and improved intestinal inflammation,54,55 possibly through modulation of nuclear factor-κB and nitric oxide (NO) signaling. 56
This review also has some limitations. First, some studies we included may have enrolled patients with IBD who had previously used antidepressants or who had other psychiatric conditions. Considering that antidepressants have a significant time-course relationship 57 and can also improve symptoms of other psychiatric disorders,58–60 our findings may be exaggerated. Second, most of the studies were conducted in Asia, which means that our results may not be generalizable to all IBD populations. Third, the volume of the available literature is small, and the power to assess the effectiveness of antidepressants in the IBD population may be insufficient. Fourth, the evidence quality was low, with most of the literature did not specify the implementation of randomization and blinding. This limits the persuasiveness of our results. Ultimately, IBD is a recurrent disease, and we think it is essential to explore the long-term effects of antidepressants in IBD patients. Masaki Kato et al. found that in patients with severe depression, antidepressant treatment of <6 months after remission was associated with increased risk of relapse of depression. 61 Unfortunately, because none of the included studies provided data on follow-up after antidepressant discontinuation, we cannot make a judgment.
In the future, high-quality, large-sample, multicenter RCTs with adequate follow-up periods are needed to understand the effects of antidepressants on individuals with IBD. More objective inflammatory indicators, like CRP and ESR, are also required.
Conclusion
Our study demonstrated that antidepressants improved outcomes in IBD patients, including comorbidities of depression and anxiety, disease activity, clinical remission, physical and social QoL, and IBDQ. However, antidepressants did not show advantages in terms of clinical response, psychological QoL, and environmental QoL. In conclusion, antidepressants are a potential adjunctive therapy for IBD. Considering that most of these studies have a very small sample size (<100 in most) and the low quality of some studies, high-quality studies are warranted in the future.
Supplemental Material
sj-docx-1-tag-10.1177_17562848231155022 – Supplemental material for Effect of antidepressants on psychological comorbidities, disease activity, and quality of life in inflammatory bowel disease: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tag-10.1177_17562848231155022 for Effect of antidepressants on psychological comorbidities, disease activity, and quality of life in inflammatory bowel disease: a systematic review and meta-analysis by Liangfang Wang, Chang Liang, Pingrun Chen, Yubin Cao and Yan Zhang in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
The authors are grateful to Sichuan University for access to MEDLINE, EMBASE, Chinese CBM Database, China National Knowledge Infrastructure, VIP, and Wanfang Database.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
