Abstract
Background:
The Coronavirus Disease 2019 (COVID-19) pandemic poses a massive crisis to global public health. Gastrointestinal (GI) symptoms are increasingly reported in COVID-19. The characteristics of laboratory findings of COVID-19 are critical for clinical diagnosis and treatment.
Objectives:
The study aimed to summarize laboratory features in COVID-19 with GI symptoms and non-GI symptoms.
Design:
This study was a systematic review and meta-analysis. Electronic literature searches were conducted for studies that included patients infected COVID-19 with GI symptoms and non-GI symptoms. GI symptoms included diarrhea, abdominal pain, nausea and vomiting, and anorexia. This study used a random-effects model to assess pooled data.
Data sources and methods:
We systematically searched PubMed, Embase, Cochrane, Web of Science for studies through 31 October 2021, with no language restrictions. We used the following search terms: ‘COVID-19’ OR ‘2019-nCoV’ OR ‘SARS-CoV-2’ OR ‘coronavirus 2019’ OR ‘severe acute respiratory syndrome coronavirus 2’ OR ‘coronavirus’ OR ‘novel coronavirus’ OR ‘nCoV’ AND ‘gastrointestinal symptoms’ OR ‘digestive symptoms’ AND ‘clinical feature’ OR ‘clinical characteristics.’ Data mostly originated from Chinese and American studies.
Results:
Of 796 identified studies, 14 were eligible and were included in our analysis (N = 8396 participants). Meta-analysis showed that GI symptoms group had an elevated alanine aminotransferase (ALT) [pooled mean difference (MD), 4.5 U/L; 95% confidence interval, [0.45, 8.55]; p = 0.03; I2 = 87%]. No publication bias was detected by Begg’s and Egger’s regression test (p = 0.130). COVID-19 with the GI symptoms also showed a trend toward decreased white blood cell count, lymphopenia, neutrophilia, thrombocytopenia and elevated total bilirubin.
Conclusion:
GI symptoms are common in COVID-19. No significant differences were found in most laboratory indicators except elevated ALT.
Registration:
CRD42020209039 (PROSPERO).
Introduction
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) that causes COVID-19 has rapidly spread to more than 200 countries, with close to 43 million confirmed cases in the United States alone (https://www.coronavirus.gov). Although respiratory symptoms are the most common and usually the earliest symptom, as the number of human infections with SARS-CoV-2 increased, the number of COVID-19 patients with gastrointestinal (GI) symptoms as the first manifestation were increasing.1–4 From the present investigations, we find that there is inconsistency with the clinical outcome of COVID-19 patients with GI symptoms. Some studies show that GI symptoms are very frequent in patients with COVID-19 and may not be associated with a poor prognosis.5,6 In contrast, other studies reported COVID-19 with GI symptoms were associated with poor clinical outcomes.1,7 In addition, some studies have reported laboratory test variations about COVID-19 patients with GI symptoms. In terms of hematologic observations, lymphopenia is the relatively common white blood cell (WBC) derangement. 8 A portion of COVID-19 affected persons with GI symptoms develop liver biochemistry abnormalities. 9 Procalcitonin (PCT) is a sensitive and specific indicator of bacterial infection. Therefore, patients with viral infections do not show significant elevated levels of PCT. 10 There are also some studies that report post-infection functional gastrointestinal disorders following COVID-19. These patients also experience abdominal pain, diarrhea, nausea and vomiting, anorexia, and other GI symptoms. However, these symptoms usually appear months after infection. 11 With increased numbers of COVID-19 patients with GI symptoms now published, it has been possible to summarize laboratory data about COVID-19 patients with GI symptoms. The purpose of this study is to summarize and compare the laboratory indicators of COVID-19 with GI symptoms to help clinicians effectively deal with the disease.
Methods
Search strategy
In this systematic review and meta-analysis, we searched PubMed, Embase, Web of Science databases, and Cochrane Library through 31 October 2021, with no language restrictions. We used the following search terms: ‘COVID-19’ OR ‘2019-nCoV’ OR ‘SARS-CoV-2’ OR ‘coronavirus 2019’ OR ‘severe acute respiratory syndrome coronavirus 2’ OR ‘coronavirus’ OR ‘novel coronavirus’ OR ‘nCoV’ AND ‘gastrointestinal symptoms’ OR ‘digestive symptoms’ AND ‘clinical feature’ OR ‘clinical characteristics’. This meta-analysis followed the preferred reporting items for systematic reviews and meta-analyses reporting guidelines. This systematic review and meta-analysis have been registered with PROSPERO (CRD42020209039). The protocol can be accessed at: https://www.crd.york.ac.uk/prospero/.
Inclusion and exclusion criteria
Articles and clinical studies that met the following inclusion criteria were eligible for this meta-analysis: (1) studies performed in adult, human subjects; (2) studies that provided laboratory parameters between GI symptoms and non-GI symptom groups; (3) studies where laboratory data were recorded at admission.
Studies were excluded if they (1) were reviews, original researches with <20 samples, case reports, letters, comments; (2) were about pregnant women; (3) lacked proper controls; (4) did not provide raw data to perform a quantitative meta-analysis; (5) did not have an available manuscript; (6) were duplicates; (7) were ongoing.
Data extraction
Two authors (J Shi and YC Song) independently searched for articles and extracted data according to these eligibility criteria. The disagreement was discussed with another author (R Zhao) and subsequently resolved via consensus. We have incorporated the name of the first author, year, country, study design, sample size, age and gender. As for the literature quality evaluation, we will use the Newcastle–Ottawa Scale (NOS) and a rating scale for non-randomized observational studies adapted from Duckitt and Harrington. We considered a study awarded seven or more stars as a high-quality study for the NOS. We excluded any study that did not score a total score <7 out of 10 maximal points.12,13 The two criteria must be met simultaneously. Quality assessment was completed independently by two reviewers.
Statistical analysis
We performed this meta-analysis with RevMan Software (Version 5.3, Cochrane Collaboration, London, UK), Stata (version 12.1) to assess publication bias, and GraphPad Prism 8 for plotting. We extracted the mean value, the standard deviation (SD), and the number of patients. If the study provided medians and interquartile ranges, we estimated the means and SD described by Hozo et al. 14 The 95% CI and weighted mean difference or standardized mean difference were used as measures to determine whether laboratory parameters differed between COVID-19 patients with the GI symptoms and non-GI symptoms groups. The percentage of I2 < 25%, 50%, and >75% indicated low, moderate, and high heterogeneity. To get a more conservative estimate of the 95% CI, we selected a random-effects model. The Egger’s test was applied to evaluate for potential publication bias. Sensitivity analysis was carried out by omitting one study at a time. Subgroup analysis of alanine aminotransferase (ALT) by country was performed.
Results
A total of 14 studies were ultimately selected for complete data extraction. After removing duplicate studies, 796 remained. Seven hundred seventy studies were excluded based on title and abstract. Finally, 26 potentially relevant publications were obtained. Of these 26 studies, 5 were excluded as they were review articles,7,10,15–17 2 were excluded as study subjects were children,18,19 2 studies were excluded because they were unrelated,20,21 1 was excluded for being case series with fewer than 20 patients, 1 1 study was excluded for lack of available data, 22 and 1 article was excluded due to high risk of selection bias (the inclusion criteria was mostly health-care workers). 23 Considering the apparent selection bias that would result, the study eliminated some laboratory data with health-care workers who were infected. 24 Thus, 14 studies,8,24–36 including 8396 patients with COVID-19, met the inclusion criteria. A flowchart of inclusion and exclusion process is shown in Figure 1. Characteristics of the included studies are shown in Table 1. The results of the included study quality evaluation are shown in Table 2. Most studies were from China, except for two from the United States, and one from Europe.

Study selection.
Characteristics of included studies.

NR: no report.
The quality assessment of included studies.

Selection of participants, bComparability of groups, cOutcomes definition, dAscertainment, eSample size, fStudy design.
A study can be awarded a maximum of 1 star for each numbered item within the selection and exposure categories, and a maximum of 2 stars in the comparability category. In total, quality scores range from 0 to 9 stars, and a study with a score of ≥6 is defined as a high-quality study. On the basis of the NOS, all 3 studies included in the meta-analysis were of high quality.
Hematology
There was a trend toward decreased WBC in COVID-19 patients with GI symptoms (14 studies, n = 8396, pooled MD, −0.10; 95% CI [−0.36, 0.15]; p = 0.42), see Figure 2(b). There was a trend toward association of GI/non-GI group with decreased platelet count (11 studies, n = 4250, pooled MD, −2.99; 95% CI [−6.49, 0.52]; p = 0.09), see Figure 2(a), lymphocytes (13 studies, n = 7816, pooled MD, −0.06; 95% CI [−0.13, 0.01]; p = 0.07, Figure 2(b)), and neutrophil count (7 studies, n = 3297, pooled MD, −0.15; 95% CI [–0.33, 0.04]; p = 0.11, Figure 2(b)). However, this did not reach statistical significance.

Results of meta-analysis comparing lab abnormalities in COVID-19 patients with GI symptoms versus non-GI symptoms (a and b). (c–f, and j) Illustrate neutrophil count, WBC count, lymphocytes count, PLT, and AST having a downward trend. (g–i) Illustrate TBil, LDH, and CRP having an upward trend. However, the difference between the two groups was not statistically significant.
Biochemical
We found that ALT (11 studies, n = 4341, pooled MD, 4.5; 95% CI [0.45, 8.55]; p = 0.03; I2 = 87%) were high in COVID-19 patients with GI symptoms (Figure 3(a)). Lactate dehydrogenase (8 studies, n = 4268, pooled MD, 0.1; 95% CI [−0.15, 0.35]; p = 0.44, Figure 2(b)), and total bilirubin (TBil; 8 studies, n = 2719, pooled MD, 0.03; 95% CI [−0.06, 0.12]; p = 0.51, Figure 2(b)) appeared to be increased among GI group. Conversely, aspartate aminotransferase (AST) (11 studies, n = 4341, pooled MD, −0.49; 95% CI [−3.56, 2.58]; p = 0.76), CKMB (7 studies, n = 5088, pooled MD, −0.74; 95% CI [−5.17, 3.69]; p = 0.74), creatinine (10 studies, n = 4023, pooled MD, −0.13; 95% CI [−3.38, 1.32]; p = 0.39), and urea (4 studies, n = 1271, pooled MD, −0.05; 95% CI [−0.73, 0.64]; p = 0.90) showed a downward trend. However, except for ALT, the remaining biochemical blood indices did not achieve statistical significance.

Forest plot of ALT in COVID-19 patients with gastrointestinal (GI) symptoms versus non-GI symptoms (a, all included studies; c, included studies from China), Figure 3b and 3d illustrated ALT had an upward trend (b, all included studies; d, included studies from China).
Inflammatory biomarkers
Reactive protein showed a significant increase (12 studies, n = 7988, pooled MD, 0.08; 95% CI [−0.10, 0.27]; p = 0.37) in GI group compared with that in non-GI group (Figure 2B). The other laboratory data are shown in Figure 2(a) and (b).
Sensitivity analysis and publication bias
Heterogeneity in the study results was high, with I2 = 87% for ALT. By removing one study at a time, we conducted a sensitivity analysis to assess the influence of each included study on the pooled MD. The studies by Luo et al. and Zhou et al. were eliminated. I2 for ALT decreased from 87 to 45%. The result of ALT sensitivity analysis is shown in Figure 4. It suggested that these two studies were the main source of heterogeneity24,32 Begg’s and Egger’s regression tests showed a low probability of publication bias in our study (p = 0.130).

Sensitivity analysis of ALT.
Subgroup analysis
Table 3 shows the results of subgroup analyses. To assess the stability of the primary estimates’ outcomes and to identify the potential resources of heterogeneity, we performed subgroup analyses. WBC, lymphocyte count, and CRP subgroup were further divided into two subgroups based on different countries. The association between decreased WBC was stronger in United States (pooled MD, −0.68; 95% CI [−0.95, −0.41]; p < 0.00001; I2 = 0%) compared to that in China (pooled MD, 0.05; 95% CI [−0.13, −0.22]; p = 0.60). United States had a low WBC count in COVID-19 patients with GI symptoms. Conversely, WBC showed an upward trend in China. Lymphocyte count had shown a declining trend in United States and China. But the difference was statistically significant only in China (pooled MD, −0.08; 95% CI [−0.15, −0.01]; p = 0.02; I2 = 85%). CRP levels of the two groups had statistical significance in United States (pooled MD, −0.1; 95% CI [−0.18, −0.02]; p = 0.02; I2 = 0%). We also made a subgroup analysis for ALT by different countries, ALT subgroup in China still had statistical significance (pooled MD, 5.36; 95% CI [0.59, 10.13]; p = 0.03; I2 = 89%).
Results of subgroup analyses about WBC, Lymphocyte count, CRP, and ALT.
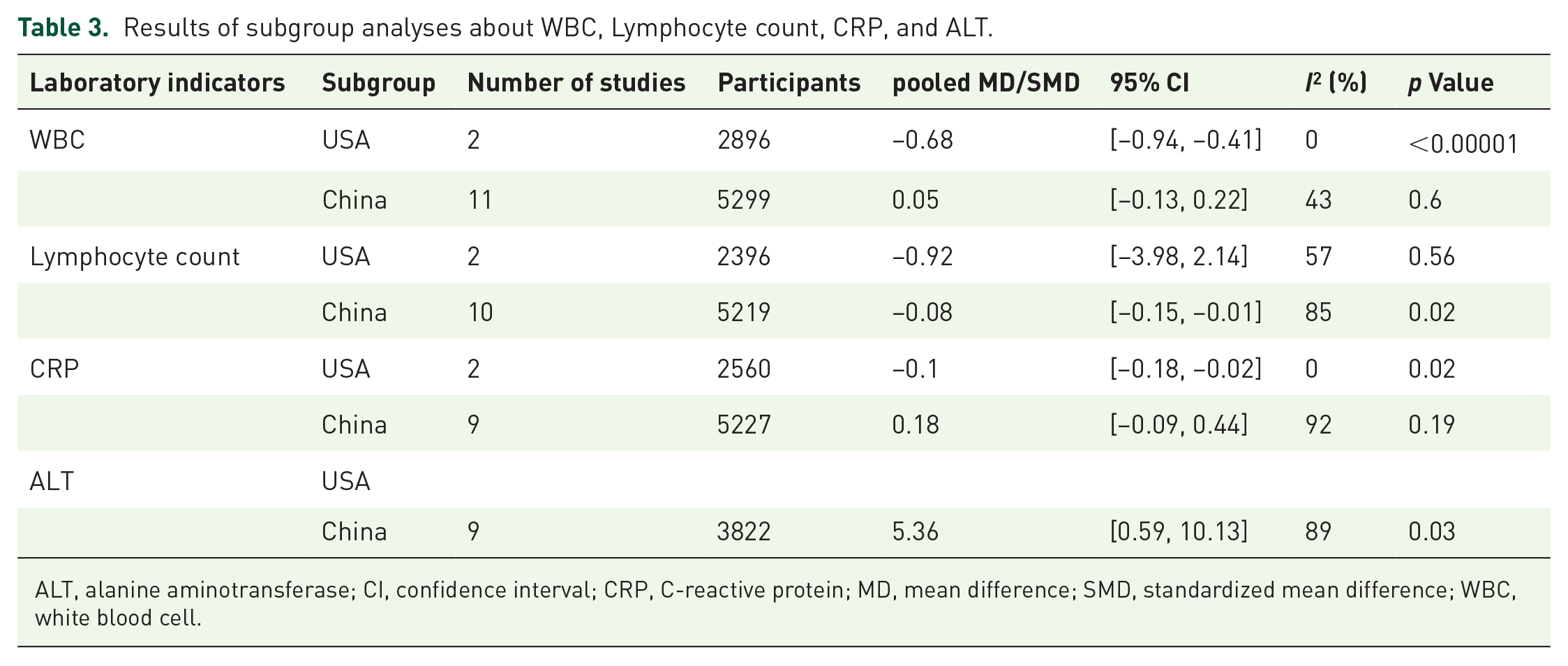
ALT, alanine aminotransferase; CI, confidence interval; CRP, C-reactive protein; MD, mean difference; SMD, standardized mean difference; WBC, white blood cell.
Discussion
Facing the huge threat of COVID-19 to human health, more attention should be paid to laboratory evaluation and early prediction of patient’s conditions. As the studies of COVID-19 with GI symptoms gradually increases, the characteristics of laboratory examination results of COVID-19 with GI symptoms were reported, but the discrepancies were observed between these reports in different countries.7,35 In this study, we systematically compare the differences in laboratory tests between COVID-19 patients with GI symptoms and non-GI symptoms. We found that ALT was increased in patients in the GI group. ALT and AST are used to monitor liver function. ALT is mainly distributed in the liver and monitors hepatocellular status; AST is mainly present in hepatocytes’ mitochondria and reflects mitochondrial problems. 37 In our meta-analysis of 14 studies, the ALT was increased in patients with GI symptoms. It showed that the liver was injured by SARS-CoV2. Besides, while there was a trend toward an increase in TBil, statistical significance was not achieved. Currently, the mechanism between liver injury and COVID-19 infection was mostly speculative. As in SARS and MERS, the primary functional receptor in coronavirus is ACE2.38,39 Studies indicated that ACE2 was expressed in multiple tissues or organs,40,41 such as the type 2 alveolar cells of the lungs, cholangiocytes of the liver, intestinal enterocytes in the gastrointestinal tract. So these tissues and organs can be damaged, which may cause changes in the corresponding laboratory indices. Also, patients with GI symptoms tended to have decreased WBC, lymphocytes, and elevated CRP, although this was not statistically significant. Interestingly, the laboratory indicators of WBC, lymphocytes count, and CRP were inconsistent between patient from China and United States. The patients with COVID-19 with GI symptoms show decreased WBC, lymphocytes count, and CRP in the United States. However, the patients with COVID-19 with GI symptoms show increased WBC and CRP, decreased lymphocyte counts in China. This discordance in findings may be due to differences in different races and the small size of prior studies.
To explore the source of heterogeneity, we performed sensitivity analysis and subgroup analysis based on the country of the studies for ALT. We found heterogeneity was mainly derived from these two studies by sensitivity analysis.24,31,32 These two studies were similar to most of the included studies. For instance, they were based on whether they had GI symptoms. However, the study of Zhou et al. included medical staff and non-medical staff, and the representation of patient populations was skewed. The study of Luo et al. had unclearly defined GI symptoms. The results of ethnic subgroup analysis showed that the heterogeneity did not decrease for ALT. For this reason, we do not consider that ethnic differences may be one of the sources of heterogeneity for this research. We deleted the study of Wei et al. and the clinical data of medical staff from Zhou et al.23,24 These clinical data are removed, as most of the infected staff were young health-care workers without comorbidities. The average age of patients from Wei et al. (37 years) and Zhou et al. (36 years) was significantly lower than that of other studies. In addition, the meantime is less delay from onset to admission.
This meta-analysis has several potential limitations. First, most of the studies were retrospective. The individual studies were at high risk of bias. Second, our findings were primarily drawn from Chinese and American findings. Hence, additional studies are needed to confirm these findings in other study populations. Third, since the selected studies could not provide the laboratory data for children, we did not include children in this meta-analysis. This aspect of research needs to be supplemented and verified by more data in the future.
In conclusion, COVID-19 patients with GI symptoms are not uncommon. This study provides the latest laboratory information on GI symptoms of COVID-19 patients. Compared with patients with non-GI symptoms, those with GI symptoms had an elevated ALT and no significant differences in other laboratory indicators. However, the data were obtained mainly from Chinese and American studies. More studies based on other countries are needed.
