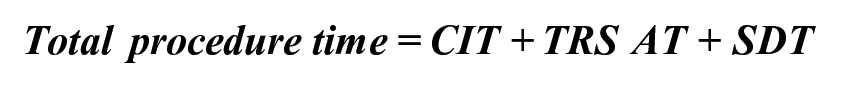Abstract
Background:
The endoscopic submucosal dissection (ESD) is a technically demanding and time-consuming procedure, with an increased risk of adverse events compared to standard endoscopic resection techniques. The main difficulties are related to the instability of the operating field and to the loss of traction. We aimed to evaluate in a pilot trial a new endoscopic platform [tissue retractor system (TRS); ORISE, Boston scientific Co., Marlborough, MA, USA], designed to stabilize the intraluminal space, and to provide tissue retraction and counter traction.
Method:
We prospectively enrolled all consecutive patients who underwent an ESD for sigmoid/rectal lesions. The primary outcome was the rate of technical feasibility. Further technical aspects such as en-bloc and R0 resection rate, number of graspers used, circumferential incision time, TRS assemblage time, submucosal dissection time, and submucosal dissection speed were provided. Clinical outcomes (recurrence rate and adverse events) were recorded as well.
Results:
In all, 10 patients (M/F 4/6, age: 70.4 ± 11.0 years old) were enrolled. Eight out of 10 lesions were located in the rectum. Average lesion size was 31.2 ± 2.7 mm, and mean lesion area was 1628.88 ± 205.3 mm2. The two sigmoid lesions were removed through standard ESD, because the platform assemblage failed after several attempts. All rectal lesions were removed in an en-bloc fashion. R0 resection was achieved in 7/8 (87.5%) patients in an average procedure time of 60.5 ± 23.3 min. None of the patients developed neither intraprocedural nor postprocedural adverse events.
Conclusion:
TRS-assisted ESD is a feasible option when used in the rectum, with promising result in terms of efficacy and safety outcomes. Nevertheless, our pilot study underlines few technical limitations of the present platform that need to be overcome before the system could be widely and routinely used.
Background
Colorectal cancer (CRC) is one of the leading causes of cancer-related mortality worldwide.1–3 Although endoscopic mucosal resection (EMR) is the most common treatment approach for superficial colorectal lesions, when dealing with larger lesions, en-bloc EMR might be unfeasible and unsafe and a piecemeal resection may hinder histologic assessment and lead to an increased risk of local recurrence.4,5 Endoscopic submucosal dissection (ESD) may overcome these limitations, allowing dissection of larger lesions in one piece. Indeed, the European Society of Gastrointestinal Endoscopy suggests to consider ESD for the removal of colorectal lesions with a high risk of superficial submucosal invasion, in case they cannot be removed en-bloc by standard polypectomy or EMR. 6 It is also true that ESD is technically demanding and time-consuming, and an increased risk of adverse events has been reported.7,8
The main difficulties are related to the instability of the operating field, due to the physiologic peristalsis, and to the loss of traction, due to the single operating channel. Recently, several new techniques and devices have been developed to facilitate ESD and to overcome the difficulties related to challenging situations.9–11
Tissue retraction system (TRS; ORISE, Boston scientific Co., Marlborough, MA, USA) is a new endoscopic platform designed to stabilize the intraluminal space, to help visualizing the dissection plane, and to provide tissue retraction and counter traction. It consists of an expandable and dynamically controlled intraluminal chamber, mounted on a flexible overtube, and two associated specifically designed retractor graspers. The system is front-loaded over the endoscope and introduced into the colon, theoretically designed to reach at least the splenic flexure. When the target area is reached, the cage is deployed creating an expanded, optimally reconfigured and stable operating field around the target lesion. Then endoscopic removal of the lesion is performed using standard endoscopic instruments (injection needles, knives, snares, etc.) through the operating channel of the scope with assistance of one or two retractor graspers handled by a second operator. Each accessory within the TRS can be moved forward and backward, left or right, rotated 360°, and can be advanced out and pulled in, regardless of the TRS, allowing for the maximal tensile force needed to visualize the dissection plane.12–14
The platform is not yet commercially available, and only two case reports of TRS-assisted ESDs have been published so far.14,15 No studies have been performed yet in humans to evaluate its feasibility and safety in speeding up colorectal ESDs.
We performed a pilot study to evaluate the feasibility, the efficacy, and the safety in patients undergoing ESD of colorectal lesions with the assistance of the TRS.
Methods
Patients and study design
We prospectively enrolled all consecutive patients ⩾18 years who underwent a sigmoid colon or rectal ESD in our institution, Humanitas Research Hospital. A detailed consultation and written informed consent were obtained from all patients prior to the procedure.
Out of Good Clinical Practice, we enrolled only patients whose lesions could not have been optimally and/or radically removed with standard polypectomy or EMR. 6 All patients whose lesions expressed any risk for deep submucosal invasion such as distorted pit (Kudo’s type V) and/or capillary (Sano’s type III) patterns 16 were excluded from this trial. We did not enroll patients in poor general condition (American Society of Anesthesiologists score ⩾ 3), known for coagulation disorders, those who were pregnant and/or breastfeeding, and those who were not able to sign the informed consent.
The study was conducted in accordance with the declaration of Helsinki and was registered on the ClinicalTrial.gov (NCT:03553199). The study protocol was approved by the local Institutional Review Board. The methods of our study were based on the STROBE recommendations. 17
Endoscopic procedure
All procedures were performed by expert operators (AR and RM), defined as endoscopists who had already performed at least 80 ESDs, 6 and were carried out under deep sedation administered by a dedicated anesthesiologist.
A standard gastroscope (ELUXEO, EG-760R; FUJIFILM Co., Tokyo, Japan) with CO2 insufflation was used for all the procedures.
After submucosal injection with saline tinged with methylene blue, we delimited the resection area by performing a circumferential mucosal incision keeping a 4–5 mm margin from the lesion using a T-type Hybridknife (ERBE, Marietta, GA, USA). Once the incision was performed, we pulled out the endoscope defining the end of the circumferential incision time (CIT) and starting time of TRS assemblage time (AT). The TRS was thus mounted and delivered: once the endoscope with the overtube was reinserted, we defined the ending AT (ORISE tissue retractor system components are illustrated in Figure 1).

ORISE tissue retractor system. (a) Tissue retractor system cage architecture details. (b) Tissue retractor system devices. (B1) OIGs which are flexible conduits, guiding graspers. The OIG is available in three (3) tip configurations; with 45°, 60°, and 90° tip bend angles. (B2) OTR overtube consisting of a handle and a flexible shaft with expandable distal end (Cage); (B3 and B4) graspers; and (B5) cage. (c) Tissue retractor system assembled over a gastroscope with two inserted retractor graspers.
The ORISE TRS was then placed over the lesion. The lesion edge was then grasped with the dedicated grasper to provide traction, and submucosal dissection was performed (Figure 2).

TRS-aided ESD. (a) Rectal lesion. (b) TRS luminal distension and stabilization properties. (c) Lesion retracted cranially by the two graspers allowing easy injection/incision with the ESD knife.

TRS-aided ESD.
After resection, the cage was closed and the specimen was retrieved. The specimen was finally placed on a polystyrene rigid support and fixed with pins, then saved in formalin solution, and sent to the pathologist. All procedural adverse events were recorded.
Endoscopists’ perception and feelings through the different procedural steps (assemblage, resection assistance, specimen retrieval) were graded according to a Visual Analogue Scale.
Follow-up
Inpatients clinical status was assessed the day after the procedure and daily until discharge. Outpatients received a phone call on day 1, day 7, and day 14 after the endoscopic procedure to investigate the possible postprocedural delayed AEs related to the procedure.
Patients with complete endoscopic excision and no indication for surgery based on final histology were advised to undergo the first endoscopic surveillance at 6 months, 18 then at intervals of 12, 36, and 60 months. 6 If any recurrence was detected, this was resected and sent for histology; in the case of suspicious invasive neoplasia, biopsies were taken.
Outcomes and definitions
Complete resection was defined according to current guidelines. 6 The resection was considered complete and defined as R0 when the neoplastic/dysplastic tissue was removed en bloc with free lateral and vertical margins. The endoscopic resection was considered incomplete in two cases: (1) when the lateral or vertical margins were positive for neoplastic/dysplastic invasion (R1) and (2) when the margins were not evaluable because of artificial burn effects (RX). Adverse events were defined according to ASGE lexicon as the occurrence of either intraprocedural or immediately postprocedural unintentional perforation, peritonitis, pneumoperitoneum, bleeding requiring hemostasis or transfusion, and procedure-related death. 19 The endoscopist’s (first operator) feelings about assemblage, resection, and specimen retrieval was graded from 1 to 10. According to the study by Suzuki et al., we defined submucosal dissection time (SDT) as the time (min) from the creation of the first traction with the TRS to the end of ESD. The area of the specimen was calculated by multiplying the halves of both height and length of the resected piece, then multiplied by 3.14. Submucosal dissection speed (SDS) was defined by dividing the specimen area by the SDT (min). 20
Finally, considering also both the CIT and the AT as previously defined, total ESD procedure time was as follows:
Data collection
Data were collected on a dedicated CRF spreadsheet using Windows Microsoft Excel (version 16, Microsoft Corp, Redmond; WA. USA). It included patient demographics, characteristics of the lesion (size, location, macroscopic appearance, pit, and vascular patterns), piecemeal or en-bloc removal, resection status (R0/R1), histological report, number of graspers used, CIT, TRS AT, SDT, SDS, and rate and type of adverse events related to the procedure and/or to the device.
Statistical analysis
Quantitative variables were expressed as means [standard deviation or median (range)]. Qualitative variables were described by frequencies with percentages. STATA (version 15) was used for the statistical analysis.
Results
Baseline
We enrolled 10 patients (M/F 4/6, age: 70.4 ± 11.0 years old). Most of the lesions (n = 7) were laterally spreading tumor (LST) granular mixed, two were non-granular LSTs, and the last one was an LST granular type. Eight out of 10 lesions were located in the rectum. Two lesions were located in the sigmoid colon. Average lesion size was 31.2 ± 2.7 mm, and mean lesion area was 1628.88 ± 205.3 mm2. Baseline characteristics are summarized in Table 1 and lesions characteristics are extensively reported in Supplemental Table 1.
Baseline characteristics.

LST, laterally spreading tumors; SD, standard deviation.
Outcomes
Procedure
The two sigmoid lesions were removed through standard ESD, because the platform assemblage failed after several attempts due to difficulties in positioning and narrower lumen. On the other hand, an en-bloc resection was successfully completed in all the rectal cases (8/10, 80%), using the TRS assistance (Table 2).
Outcomes.

AT, assembly time; SD, standard deviation; SDS, submucosal dissection speed; SDT, submucosal dissection time.
The average total procedure time was 60.5 ± 23.3 min. The average time spent for assembling TRS was 6.5 ± 4.8 min. SDT resulted to be 45.1 ± 19.7 min, while SDS was 30.6 ± 23.9 min/mm2. In all, but two procedures, only one of the two graspers were used. Procedural characteristics and endoscopist’s feelings are extensively reported in Supplemental Table 2.
A complete resection (R0) was achieved in 7 (87.5%) out of 8 patients. Only one lesion (pt. 2) resulted in a rectal cancer (adenocarcinoma), with deep submucosal involvement and positive deep margins. The patient was indeed referred for surgery. Other lesions were adenomatous lesions (six high-grade and one low-grade adenomas).
None of the eight patients developed neither intraprocedural nor postprocedural adverse events.
Follow-up
After 6 and 12 months from the procedures, all the seven patients underwent a follow-up endoscopy: the scar was regular in all the cases, with no signs of residual adenomatous tissue.
Discussion
ESD is a well-established method for endoscopic removal of colorectal lesions in the East. Recently, it has also been gaining recognition in the Western world, where CRC prevalence is even more relevant. As a matter of fact, by allowing en-bloc excision regardless of the lesion size, ESD aims to increase the possibility of curative resection even for superficially invasive CRC. Nevertheless, ESD is a technically challenging, time-consuming procedure, which implies a significantly higher risk of adverse events if compared to EMR, necessitating ongoing innovations to overcome its complexity. Conventional ESD has a limited armamentarium of devices and maneuvers helping the endoscopist in improving the access to the submucosal space and the identification of the proper dissection plane (i.e. distal attachment, fluid injections, patient repositioning).
According to our pilot study, TRS-assisted ESD is a feasible option when used in the rectum, with promising results in helping the operator in achieving adequate efficacy and safety outcomes as shown by the 100% of en-bloc resection coupled with the almost 90% of R0 resection on one side and the absence of procedural-related adverse events on the other.
The theoretical advantage of the TRS platform over the number of accessories and techniques previously proposed for ESD assistance is the ability to provide both dynamic tissue retraction and instrument triangulation, which are essential for ideal visualization and access to dissection plane. In this regard, in our opinion, the TRS system was not taken the full advantage, as the endoscopist only used the two graspers twice in our series. However, it could be argued that as ‘pure endoscopists’, the authors are not used to the ‘surgical concept’ and this device could have been used better. Furthermore, the platform provided a stable operating space during the procedure and works as a convenient specimen-retrieval device after resection. These technical advantages aim at first to improve the safety and efficacy outcomes of colorectal endoscopic resections; however, they might also consistently affect resection speed, overcoming one of the key barriers preventing the definitive recognition of ESD potential over EMR. As a matter of fact, considering the number of colorectal lesions that Western endoscopists need to manage, the systematical adoption of ESD for large colorectal lesions would add an unaffordable burden on most of the western endoscopy services. Significantly reducing the procedural times would be the real game changer for ESD affirmation. In this regard, if comparing the SDS with TRS in our series with the SDS of western expert endoscopists in previously published series, we showed a faster dissection speed (30.6 ± 23.9 versus 16.7 mm2/min, respectively). 21 At the same time, this allowed to reach a dissection speed comparable to the one reported by eastern expert endoscopists (30.6 ± 23.9 versus 29.1 ± 3.2 mm2/min, respectively).20,22
In our opinion, despite the several advantages of this new technology, few drawbacks may still prevent its large-scale use, and should be overcome. First, despite improving over the procedures due to the endoscopist learning curve, the AT should be further reduced by re-designing the deployment strategy of the working chamber. The inconvenient assemblage strategy prevented the use of the TRS platform for endoscopic resection proximally to the rectum, where the deployment complexity is increased. It could be argued that the rectum is where patients would benefit the most of an en-bloc resection given the higher risk of submucosal invasion. Nevertheless, overcoming the technical limitations of standard ESD even in other colonic tracts would have been even more relevant for a new resection-assisting platform.
Moreover, even assuming the use of the TRS platform only for rectal resections, a second limitation is due to the design of the overtube, developed for reaching the splenic flexure.
This forces the endoscopist to work far away from the patient when performing rectal ESD, losing part of the technical gain in terms of scope control. Considering that the TRS platform had been developed aiming to improve the procedural outcomes through an endoscopist-friendly assistance, this limitation may paradoxically may have a role in decreasing the willingness to use the device in daily practice for rectal lesions.
The main drawbacks of the study are the lack of comparison with conventional ESD and the limited sample size, preventing conclusive statement on TRS. However, reassuring on feasibility and safety, this pilot experience permits to design future comparative studies. Moreover, its strict protocol, coupled with the prospective setting, permitted to lucidly analyze its pros and cons as discussed.
Conclusion
In conclusion, the TRS platform provides a stable working space, dynamic tissue retraction, and instrument triangulation. In spite of few technical limitations which would need to be overcome, the time for technology to permit a decisive step toward a mini-invasive intraluminal surgery has finally come.
Supplemental Material
sj-docx-1-tag-10.1177_17562848221104953 – Supplemental material for Pilot study on a new endoscopic platform for colorectal endoscopic submucosal dissection
Supplemental material, sj-docx-1-tag-10.1177_17562848221104953 for Pilot study on a new endoscopic platform for colorectal endoscopic submucosal dissection by Roberta Maselli, Marco Spadaccini, Piera Alessia Galtieri, Matteo Badalamenti, Elisa Chiara Ferrara, Gaia Pellegatta, Antonio Capogreco, Silvia Carrara, Andrea Anderloni, Alessandro Fugazza, Cesare Hassan and Alessandro Repici in Therapeutic Advances in Gastroenterology
Footnotes
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
Drs Roberta Maselli, Andrea Anderloni, Alessandro Fugazza, and Alessandro Repici received consultancy fees from Boston Scientific. Dr Silvia Carrara received in the last 36 months a research Grant from Boston Scientific on FNB Needles. No further conflict of interest to be disclosed.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.