Abstract
Hepatorenal syndrome (HRS) is a serious form of renal dysfunction in patients with cirrhosis and ascites. It is an important component of the acute-on-chronic liver failure (ACLF) syndrome. Significant recent changes in the understanding of the pathophysiology of renal dysfunction in cirrhosis include the role of inflammation in addition to hemodynamic changes. The term acute kidney injury (AKI) is now adopted to include all functional and structural forms of acute renal dysfunction in cirrhosis, with various stages describing the severity of the condition. Type 1 hepatorenal syndrome (HRS1) is renamed HRS-AKI, which is stage 2 AKI [doubling of baseline serum creatinine (sCr)] while fulfilling all other criteria of HRS1. Albumin is used for its volume expanding and anti-inflammatory properties to confirm the diagnosis of HRS-AKI. Vasoconstrictors are added to albumin as pharmacotherapy to improve the hemodynamics. Terlipressin, although not yet available in North America, is the most common vasoconstrictor used worldwide. Patients with high grade of ACLF treated with terlipressin are at risk for respiratory failure if there is pretreatment respiratory compromise. Norepinephrine is equally effective as terlipressin in reversing HRS1. Recent data show that norepinephrine may be administered outside the intensive care setting, but close monitoring is still required. There has been no improvement in overall or transplant-free survival shown with vasoconstrictor use, but response to vasoconstrictors with reduction in sCr is associated with improvement in survival. Non-responders to vasoconstrictor plus albumin will need liver transplantation as definite treatment with renal replacement therapy as a bridge therapy. Combined liver and kidney transplantation is recommended for patients with prolonged history of AKI, underlying chronic kidney disease or with hereditary renal conditions. Future developments, such as the use of biomarkers and metabolomics, may help to identify at risk patients with earlier diagnosis to allow for earlier treatment with improved outcomes.
Introduction
Renal dysfunction is the most common complication in patients with liver cirrhosis and ascites, occurring in 20–49% of patients.1–3 For a long time, renal dysfunction in cirrhosis was synonymous with type 1 hepatorenal syndrome (HRS1), a condition associated with a fatal outcome in days to weeks if left untreated. After defining the diagnostic criteria for hepatorenal syndrome (HRS) in 1996, 4 there followed treatment guidelines for the condition. Further understanding of the pathophysiology of HRS led to refinement of diagnostic criteria for HRS in 2007. However, it soon became clear that the threshold of serum creatinine (sCr) set for the diagnosis of HRS represented very advanced renal failure; therefore, treatment beginning at such a late stage of renal impairment was compromising the chance for renal recovery. Therefore, there was a need to change the diagnostic criteria for HRS. The term acute kidney injury (AKI) was borrowed from the nephrology and intensive care communities of specialists, who describe renal dysfunction by a dynamic change rather than by a fixed threshold in the sCr (Table 1), 5 as these small dynamic changes in sCr have been associated with negative outcomes in other patient populations. 6 AKI in cirrhosis was finally defined in 2015, and HRS1 became a special sub-category of AKI, and renamed as HRS-AKI. 7 These new diagnostic criteria will allow for prompt diagnosis and early initiation of treatment.
Diagnostic criteria of AKI in cirrhosis.
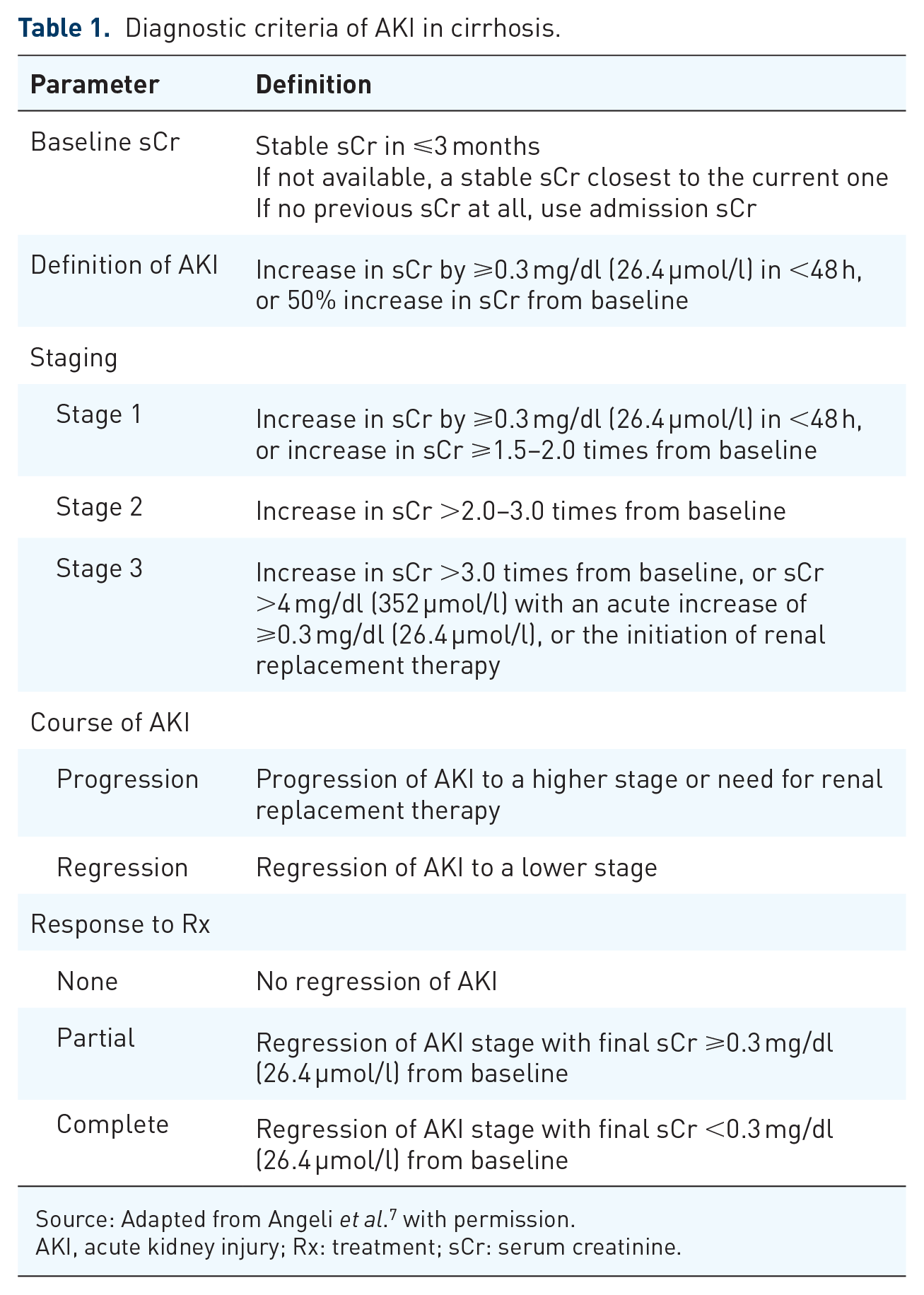
Source: Adapted from Angeli et al. 7 with permission.
AKI, acute kidney injury; Rx: treatment; sCr: serum creatinine.
There are several phenotypes of AKI and may include functional and structural etiologies. Functional AKI is related to functional changes in the kidneys or renal circulation; and in cirrhosis, is usually related to hemodynamic abnormalities affecting the kidneys as a result of advanced cirrhosis. Functional AKI can include volume responsive prerenal azotemia or HRS-AKI. In contrast, structural causes of AKI involve structural injury to the kidneys such as glomerulonephritis, interstitial nephritis or tubular damage. A common prototype would be acute tubular necrosis (ATN). Overall incidence of ATN (15–60%) and prerenal azotemia (15–45%) in cirrhosis are more common than HRS, which affects 10–40% of patients. 8 HRS is the subset of AKI that does not respond to volume resuscitation alone and is considered one of the dreaded complications of cirrhosis with median survival without treatment in days to weeks. 9 Recent advances in the understanding and management of the HRS, combined with diagnosis of renal dysfunction at an earlier stage of its natural history, have made treatment more effective. In this review, we discuss the recent advances in the management of HRS.
Definition of AKI in cirrhosis
Diagnostic criteria of HRS1 were laid down in 1996 as a rapid rise in sCr, doubling from the baseline value within 2 weeks to at least 2.5 mg/dl without any evidence of structural renal disease. 4 After the introduction of the AKI nomenclature for the cirrhotic population, AKI is now defined as rise in sCr of ⩾0.3 mg/dl in <48 h, or a 50% increase in sCr from the lowest sCr level within the previous 3 months (Table 1). 5 HRS-AKI is then defined as at least stage 2 AKI while fulfilling all the other diagnostic criteria of HRS (Table 2). 7 Inclusion of urine output in the diagnostic criteria of AKI as in other patient populations has not been generally accepted by the hepatology community for patients with cirrhosis, despite the fact that oliguria in critically ill patients with cirrhosis has been shown to have a negative impact on hospital mortality. 10 This is because patients with cirrhosis and ascites usually have reduced urine output related to their avid renal sodium and water retention, and therefore making it difficult to assess the extent of renal injury using urine output criteria alone. The entity previously known as chronic, or type 2 HRS (HRS2) is characterized by slow progression of renal dysfunction over time occurring in patients with ascites. According to the new nomenclature, HRS2 is a form of chronic kidney disease (CKD), which in some literature is a subclass of HRS-non-AKI. 11
Diagnostic criteria of HRS-AKI.

Source: Adapted from Angeli et al. 7 with permission.
AKI, acute kidney injury; HRS, hepatorenal syndrome.
Pathophysiology of AKI in cirrhosis
Splanchnic and systemic arterial vasodilatation are the hallmarks of advanced cirrhosis. Overproduction of various vasodilatory substances like nitric oxide, carbon monoxide and endocannabinoids are responsible for splanchnic and systemic arterial vasodilatation in cirrhosis (Figure 1). 12 Bacterial translocation due to increased intestinal permeability related to portal hypertension transfers increased amounts of bacterial and bacterial products from the gut lumen to the splanchnic circulation. 13 Some of these pathogen-associated molecular patterns (PAMPs) have vasodilatory properties and can contribute to the splanchnic and systemic arterial vasodilatation. This clinically manifests as systemic hypotension. The physiological response is an increased cardiac output and activation of various vasoconstrictor systems such as the renin–angiotensin–aldosterone system (RAAS), sympathetic nervous system, and non-osmotically related increase in vasopressin release in order to maintain hemodynamic stability. The renal circulation is very sensitive to the vasoconstrictive effects of these vasoconstrictors, and the end result is renal vasoconstriction. 14 There is also renal sodium and water retention in order to compensate for the relative reduction in intravascular volume. Excess renal vasoconstriction predisposes the patient to the development of AKI, whereas excess water retention over excess sodium retention leads to hyponatremia.

Pathophysiology of hepatorenal syndrome.
More recently, it is becoming increasing evident that another contributor to the pathogenesis of AKI in cirrhosis is systemic inflammation. 15 It has been noted that at least one-third of patients with HRS-AKI have evidence of inflammation without any documented infection. 16 Inflammation can be due to sterile inflammatory process like alcoholic hepatitis and drug-induced liver injury 17 or due to infective process like bacterial infection.18,19 These inciting inflammatory events with their excess production of PAMPs from infection or damage-associated molecular patterns (DAMPs) from sterile hepatocyte injury can increase chemokine and cytokine production through activation of the immune system.15,20 Many of these molecules can directly damage renal tubules by forming microthrombi in the renal microcirculation through immunologic mechanisms and leukocyte/platelet activation, 19 thereby contributing to the renal dysfunction.
AKI often occurs as a part of multi-organ failure scenario in the syndrome known as acute-on-chronic liver failure (ACLF), 21 which is characterized by one or more extrahepatic organ failures and associated with high short-term mortality. 21 Renal failure is considered one of the important extrahepatic organ failures that defines mortality. 22 The presence of other organ failures may also enhance the inflammatory milieu, which can significantly worsen renal function, analogous to inflammation worsening the severity of liver dysfunction. 15
Diagnosis of HRS-AKI
HRS-AKI in cirrhosis is a diagnosis of exclusion, after prerenal azotemia and structural causes of AKI such as ATN have been ruled out. Therefore, it is important to take a careful history, enquiring about excess diuretic use, copious diarrhea from high lactulose doses, or the presence of hematemesis and/or melena. Examination of patients should also aim at assessing the fluid status of the patient. The urine should be examined for the presence of proteinuria, hematuria, or various casts. Once prerenal azotemia and structural renal diseases have been excluded, then we can confidently diagnose HRS-AKI (Figure 2). The majority of the HRS-AKI episodes are precipitated by an acute event. It is important to identify all the precipitating events and treat them appropriately. Bacterial infection can precipitate AKI due to production of excessive inflammatory cytokines. A complete septic workup and initiation of empiric antibiotics is recommended in all patients with HRS-AKI

Suggested management algorithm for acute kidney injury including hepatorenal syndrome.
Role of kidney biomarkers in diagnosing HRS-AKI
Despite the various limitations of sCr, it is the only parameter currently used to define and stage AKI in cirrhosis. Recently, the role of various kidney biomarkers is being recognized as being able to differentiate the structural from functional causes of AKI. Serum cystatin C, which is released by all nucleated cells and eliminated by glomerular filtration, has been studied extensively for accurate estimation of glomerular filtration rate (GFR).23–27 The combination of cystatin C and sCr was able to calculate GFR more accurately compared to sCr alone, especially when GFR is below 60 ml/min. 28 This should allow for a more accurate representation of a reduction of GFR. However, this has not been incorporated into the diagnostic criteria for HRS-AKI. Multiple other biomarkers like neutrophil gelatinase-associated lipocalin (N-GAL), interleukin 18 (IL-18), and kidney injury molecule-1 (KIM-1) have been investigated in different studies for the differentiation of HRS-AKI from other causes of AKI.29,30 The most widely studied biomarker for this purpose is N-GAL, which was able to diagnose ATN in 86% of cases with the cut-off value of 220–244 µg/g of creatinine.31,32 However, clinical usefulness of these biomarkers remains unclear due to limited availability for clinical use and lack of widely accepted cut off value.
Treatment of HRS
Recognition of AKI is the most important first step in its management in cirrhosis (Figure 2). It requires frequent monitoring of the sCr levels, especially in patients with potential precipitating factors. These include events that can either disturb the already precarious hemodynamics further or worsen the extent of inflammation. Examples include infection, be it bacterial or fungal, intravascular volume loss such as excessive diuresis or diarrhea, or an inflammatory condition such as alcoholic hepatitis. Once the diagnosis of AKI is confirmed, the initial step in the management is to remove and treat the precipitating factors to prevent further worsening of the kidney injury. 33 It is also important to decide the stage of kidney injury to initiate appropriate therapy without delay. A urinalysis is always recommended to rule out structural causes of AKI by identifying hematuria or proteinuria, or various casts. A full septic workup including culturing samples from all possible sites as infection was found to be the most common precipitating factor of AKI in patients with cirrhosis. 34 Other general measures in the management of AKI includes withdrawal of diuretics, volume expansion or resuscitation, and adjustment of lactulose dose if there is lactulose-induced diarrhea leading to dehydration. Appropriate volume resuscitation with colloid solutions, such as albumin, should be initiated in patients with intravascular volume loss due to excessive diuretics use and large-volume paracentesis (LVP). Patients with gastrointestinal blood loss should be resuscitated with packed red blood cells. Use of nephrotoxic drugs or radiographic dye should be avoided. Non-selective beta blockers (NSBBs) are commonly used in patients with decompensated cirrhosis as a prophylaxis against variceal bleeding. In patients with AKI who are on an NSBB, withholding NSBB should be considered, especially if there is systemic hypotension. 35 These general measures may lead to improvement in renal function, especially in patients who have prerenal azotemia. The diagnosis of HRS-AKI is therefore one of diagnosis of exclusion. Patients with HRS-AKI are not volume responsive and therefore their sCr either stays unchanged or increases further despite these measures. Patients with HRS-AKI by definition have stage 2 AKI. As urine output in these patients is invariably low, it is tempting to insert a urinary catheter to monitor the urine output. However, that also increases the chance for urinary tract infection, and therefore should be avoided if possible.
Albumin
Albumin constitutes 60% of plasma protein in healthy individuals and its main physiologic function is to maintain colloid osmotic pressure, thereby maintaining intravascular volume. 36 Hypoalbuminemia in cirrhosis is due to both decreased synthesis and increase catabolism. 36 Albumin is used in patients with cirrhosis not only for its volume expanding effect but also for its antioxidant, immune modulating, and endothelial stabilizing properties. 37 The circulating albumin in the cirrhotic patients is structurally modified and functionally impaired, thereby unable to perform in full functional capacity. 38 The normal antioxidant and scavenging properties of albumin due to the presence of free cysteine moiety at position 34 (cys-34) helps in clearing reactive oxygen species, cytokines, and various bacterial products.37,39 These antioxidant and anti-inflammatory properties of albumin also have beneficial effects on cardiac output in patients with advanced cirrhosis by improving cardiac contractility. 40 However, increased oxidative stress in patients with cirrhosis transforms the albumin to an oxidized state from its normal reduced state.41,42 As such, albumin is unable to perform the scavenger and detoxification function as it is unable to bind to the free radicals.
Although a recent study suggests that routine use of albumin in decompensated cirrhosis admitted into hospital was unable to prevent renal dysfunction, 43 the inflammation suppressing effect of albumin is seen in higher doses. Therefore, a meta-analysis found that a total of 600 g of albumin given over the course of HRS treatment provided patients a significant survival benefit compared to patients who received a total dose of 200 g. 44 The International Club of Ascites (ICA) recommends using albumin at a dose of 1 g/kg body weight, with a maximum dose of 100 g/day to differentiate prerenal causes of AKI from HRS-AKI; thereafter, albumin is recommended to be given with vasoconstrictors at the dose of 20–40 g/day, 45 although there has been no dose responsive study related to albumin to derive at this dosing regimen. The use of albumin in the management of HRS-AKI has also been recommended by various academic liver societies.46,47
Vasoconstrictors
Vasoconstrictors are the mainstay of treatment for HRS-AKI. This is because their mode of action can help to improve the hemodynamic abnormalities that have led to the development of HRS-AKI. They are recommended to be used in combination with albumin46,47 and should be started as soon as the diagnosis of HRS-AKI is made. Vasoconstrictors used in the treatment for HRS1 includes terlipressin, norepinephrine, a combination of midodrine and octreotide, or dopamine with furosemide, 48 with terlipressin being the most widely used vasoconstrictor worldwide. Splanchnic vasoconstrictors like terlipressin and octreotide act by reducing portal inflow, thereby transferring some of the intravascular volume to the systemic circulation, improving filling of the central compartment, while reducing the portal pressure at the same time. This also improves the renal circulation by decreasing the compensatory activation of various systemic vasoconstrictors. 49 Systemic vasoconstrictors, such as midodrine and norepinephrine, improve renal circulation by increasing mean arterial pressure, and hence the renal perfusion pressure. 49 All the studies published so far have used the traditional definition of HRS1 for patient inclusion. As the new diagnostic criteria will allow us to diagnose HRS earlier at a lower sCr level, 7 future patients would have received pharmacotherapy approximately 4 days earlier with lower sCr at the time of initiation of treatment. 50 Hopefully, this will lead to better outcomes with earlier treatment start as lower pretreatment sCr is more likely to be associated with a transient course of AKI, 51 although we will have to wait for the results of future studies to confirm this hypothesis.
Terlipressin
Terlipressin is a vasopressin analog and induces vasoconstriction via V1 receptor of the vascular smooth muscle cells. Terlipressin decreases portal pressure by reducing portal inflow and also intrahepatic resistance. Subsequent redistribution of blood volume to the systemic circulation will lead to an increase in mean arterial blood pressure and improvement in renal circulation. 52 Terlipressin has also been shown to ameliorate systemic inflammation by reducing bacterial translocation in decompensated cirrhosis and hence helps to reduce the extent of vasodilatation in the presence of infection. 53
The most recently published randomized control trial on the use of terlipressin versus placebo, both with albumin, in the treatment of HRS1 from North America is the largest trial to date and it included 300 patients 54 (Table 3). The study results clearly showed that terlipressin is more effective than placebo in improving renal function. Terlipressin was given as intermittent boluses at an initial dose of 1.0 mg every 4–6 h, increasing to 2 mg every 4–6 h to a maximum daily dose of 12 mg if there was less than 30% reduction in the sCr by day 4 of treatment after at least 10 doses. 54 Treatment should be continued for up to 14 days or stopped earlier if the patient had a complete response. Treatment could also be stopped on day 4 for non-response defined as the sCr continuing to rise despite treatment. 54 The median duration of treatment was 6 days. Verified HRS reversal, defined as lowering of sCr with treatment twice to <1.5 mg/dl 2 h apart while remaining free of renal replacement therapy (RRT) and alive for 10 days, was reported in 32% in the terlipressin group, compared to 17% in placebo group (p = 0.006). HRS reversal was durable at day 30 without the need for renal replacement in 32% of patients in the terlipressin arm compared to 16% in the placebo arm (p = 0.003). 54 However, there was no difference in the overall survival or transplant-free survival up to 90 days after completion of treatment.
Published studies related to vasoactive drugs in the treatment of type 1 hepatorenal syndrome.
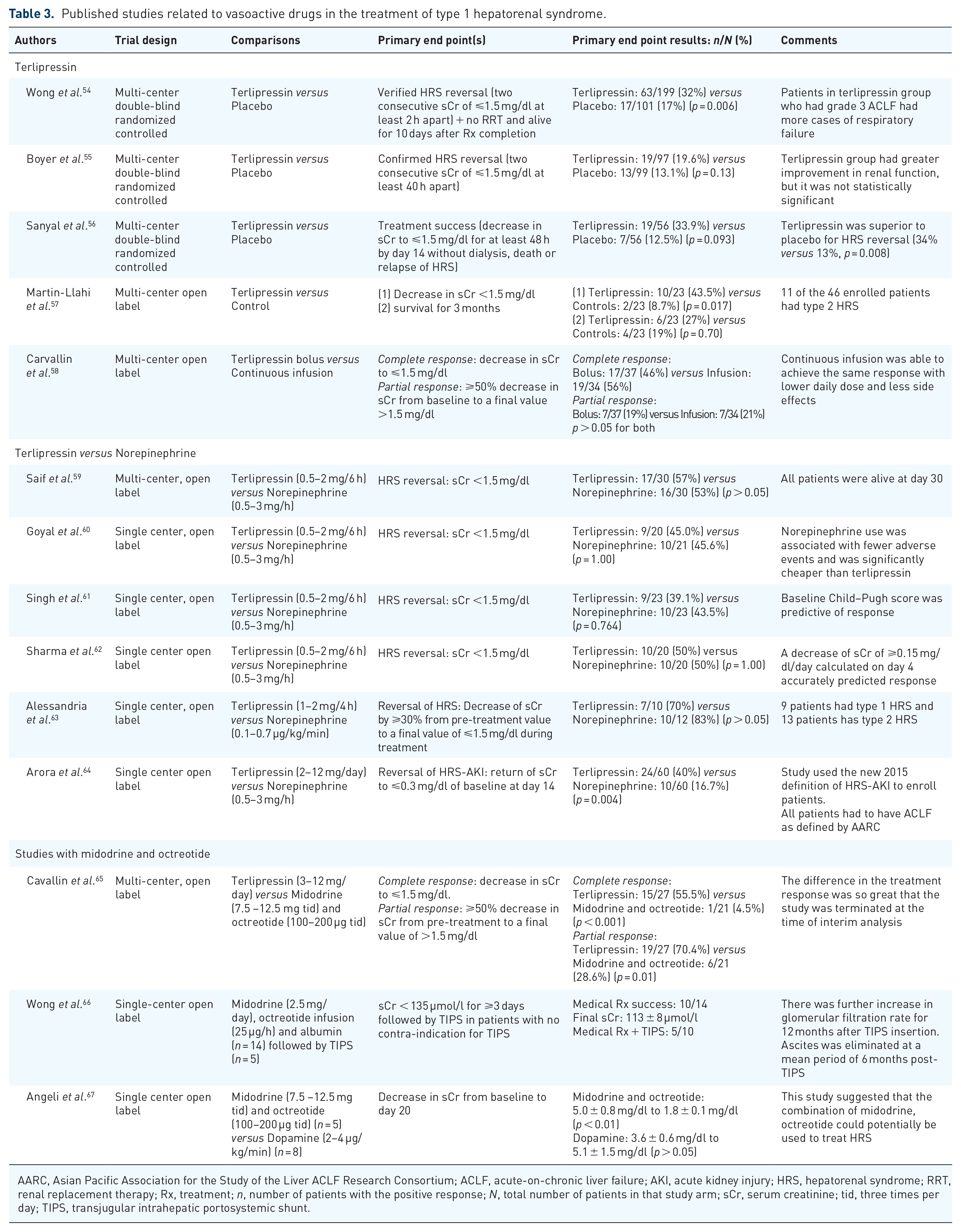
AARC, Asian Pacific Association for the Study of the Liver ACLF Research Consortium; ACLF, acute-on-chronic liver failure; AKI, acute kidney injury; HRS, hepatorenal syndrome; RRT, renal replacement therapy; Rx, treatment; n, number of patients with the positive response; N, total number of patients in that study arm; sCr, serum creatinine; tid, three times per day; TIPS, transjugular intrahepatic portosystemic shunt.
An important complication emerged from this latest randomized controlled trial (RCT) but not reported in previous trials were higher rates of respiratory failure noted in terlipressin group (16 of 200 patients or 8%), compared to placebo (3 of 100 patients or 3%) (p = 0.14). 68 Most of the cases in the terlipressin group (12 out of 16 patients) were observed in patients with grade 3 ACLF as defined by the European Association for the Study of Liver Disease-Chronic Liver Failure (EASL-CLIF) Consortium, whereas no case of respiratory failure was observed amongst patients with grade 3 ACLF in the placebo group (p = 0.01). 68
Other published randomized trials comparing the same dose regimen of terlipressin versus placebo, all with concomitant albumin, have come from Spain (n = 1) and North America (n = 2) (Table 3).55–57 Two of these three studies showed a significant improvement in renal function in those receiving terlipressin plus albumin compared to the albumin alone group with reversal of HRS1 occurring in 36–44% of patients.55–57 Although none of the studies showed an improvement in overall survival with terlipressin use compared to placebo; however, for patients who showed a response to treatment, there was a significant improvement in survival at 90 days when compared to non-responders. Even a partial response with a 20% decrease in sCr at the end of treatment was associated with an improvement in survival. 69 In fact, a meta-analysis has shown that with any vasoconstrictor treatment, for every 1 mg/dl drop in sCr, there was a 27% reduction in the relative risk for mortality. 70
Baseline bilirubin of <10 mg/dl, a baseline sCr of <5 mg/dl, and lower stage of ACLF were found to be the predictors of response to terlipressin.71–73 Other factors determining the renal recovery following terlipressin use include lower MELD score, the presence of systemic inflammation, alcoholic hepatitis, and sepsis.53,54,74,75 A sustained increase in the mean arterial pressure by 5–10 mmHg with treatment has been identified as the predictor of response in one study. 69
A recent study has demonstrated beneficial effect of continuous infusion of terlipressin by using less total daily dose (2–4 mg/day) with lower side effects compared to bolus dosing. 58 Commonly noted cardiovascular side effects include arrhythmia, angina, and digital ischemia. Abdominal ischemia leads to abdominal pain and diarrhea. Therefore, terlipressin should not be given in patients who have a history of ischemic heart disease or peripheral vascular disease. The emergence of an increased incidence of respiratory failure with terlipressin use in patients with grade 3 ACLF suggests patients with baseline respiratory compromise should also not be given terlipressin. 68 Other significant predictors of respiratory failure with albumin use include a high baseline international normalized ratio and a high baseline mean arterial blood pressure. 68 At the time of writing of this review, terlipressin is still not available for commercial use in North America.
Norepinephrine
Norepinephrine is an alpha agonist and therefore a systemic vasoconstrictor. It activates the alpha-1 adrenergic receptor in the vascular smooth muscle cells. The resultant increased peripheral vascular resistance in advanced cirrhosis can improve the mean arterial pressure and hence the renal perfusion pressure. Norepinephrine is less expensive than terlipressin, but the requirements of central venous catheter and cardiac monitoring in an intensive care unit (ICU) setting may offset the cost benefit. The dose varies from 0.5 to 3 mg/h, adjusted to maintain the mean arterial blood pressure to ensure renal perfusion.
To date, there have not been any studies comparing norepinephrine with placebo. Most published trials have evaluated the efficacy of norepinephrine versus terlipressin in the treatment of HRS1. All are small randomized studies totaling 96 patients in the norepinephrine arm and 99 patients in the terlipressin arm.59–63 Efficacy of norepinephrine to reverse HRS1 has been reported to be between 39% and 70%59–63 and is equivalent to terlipressin in this regard.76–78 However, all the reported studies have only included a small number of patients and therefore are at high risk for bias. In the setting of ACLF as defined by the Asian Pacific Association for the Study of the Liver ACLF Research Consortium, terlipressin was superior to norepinephrine in reversing HRS-AKI in patients with decompensated cirrhosis. 64 Another comparative study showed higher rates of full response with HRS reversal with norepinephrine compared to combination of midodrine and octreotide. 79 Further assessment of the use of norepinephrine was reported in a small feasibility study from North America where norepinephrine was used in the non-ICU setting in the treatment of HRS in patients who were non-responders to midodrine and octreotide. 80 Forty-five percent of patients showed a complete or partial response with the median duration of treatment of 2 days. Improved transplant-free survival at 90 days was also noted in responders. 80
Midodrine and octreotide
Midodrine is an alpha-adrenergic agonist, commonly used in North America to treat HRS1 due to non-availability of terlipressin. It increases the mean arterial pressure by systemic vasoconstriction, thereby improving the renal perfusion pressure and hence the renal circulation. Midodrine is used in combination with octreotide and albumin in the treatment of HRS1. Octreotide is a nonspecific antagonist to various splanchnic vasodilatory substances, especially glucagon. Octreotide alone has been shown to be ineffective in the treatment of HRS1. All the published studies using the combination of midodrine, octreotide, and albumin only included very small number of patients,66,67,81 but the response is very slow, and can take up to weeks to show improvement in renal function, although reversal of HRS is possible,66,82 The effects of the combination on HRS reversal has been shown to be significantly inferior to that of terlipressin. 65
Renal replacement therapy
The use of RRT in patients with cirrhosis and HRS1 has been controversial and is typically dependent on the status of the patient with respect to liver transplantation. The procedure is associated with risks and complications in the critically ill patient. These include systemic hypotension and hemodynamic instability during dialysis, risk for cardiac events, venous access issues, and potential for bleeding and infection. 83 RRT does not offer survival benefit; therefore, decision to initiate RRT should be individualized based on the goal of care, otherwise it just lengthens hospital stay without improving survival. 8 RRT is indicated in patients with HRS-AKI unresponsive to pharmacologic therapy who have either volume overload, electrolyte derangements, or uremia. 84 Continuous RRT (CRRT) is preferred over intermittent RRT in these subsets of patients as maintenance of hemodynamic stability and adequate volume management is always challenging. Rapid fluid and solute shift associated with intermittent RRT is complicated by hypotension during dialysis and cerebral edema. 83 Use of anticoagulation during RRT in critically ill patients with increased bleeding risk, such as patients with decompensated cirrhosis, has remained controversial. Although unfractionated heparin is the most commonly used anticoagulation during RRT, but its safety is not established in patients with liver failure. Regional citrate anticoagulation (RCA) is associated with fewer adverse events and has a prolonged circuit life span compared to systemic heparin anticoagulation in critically ill patients. 85 A systematic review and meta-analysis concluded that RCA can be safely used during RRT in liver failure patients without experiencing side effects like hypocalcemia and acid–base imbalance related to citrate accumulation. 86 Careful monitoring of electrolytes and acid–base status has been recommended for patients undergoing RRT with the use of RCA. 87 RRT is regarded as a bridge to liver transplantation in those who are liver transplant candidates. In selected patients, RRT may prolong life expectancy for several weeks to months. 88 However, prolonged RRT pre-transplant has a negative impact on post-transplant patient and renal outcomes. 89 Recent data suggest that in patients with HRS-AKI as part of the ACLF syndrome, improving renal function with RRT may decrease 28-day mortality by decreasing the number of organ failure.88,90 Therefore, RRT may be beneficial in this very selected group of patients with HRS-AKI irrespective of their transplant candidacy.
Therapeutic plasma exchange
Therapeutic plasma exchange has been shown to be beneficial for acute liver failure in an open randomized study by improving survival in patient who were not transplanted. 91 Therapeutic plasma exchange would allow recovery of native liver function by facilitating liver regeneration through reducing systemic inflammatory mediators and removing toxic substances. Studies related to therapeutic plasma exchange have shown improvement in systemic hemodynamics and encephalopathy in patients with ACLF, 92 but none of these studies have assessed renal function improvement specifically in patents with HRS-AKI.
Liver transplantation
Liver transplantation is the only curative treatment of HRS-AKI as this corrects the underlying portal hypertension and liver dysfunction. Therefore, patients with HRS-AKI should be referred for liver transplant assessment as soon as possible. Once accepted onto the liver transplant waiting list, patients with HRS-AKI are prioritized due to an increase in sCr, and the associated rise in MELD score. There has been some controversy as to whether patients with HRS-AKI should be treated with vasoconstrictor therapy, as a positive response would lower the MELD score and adversely affect the priority for liver transplant. However, response to vasoconstrictor therapy before liver transplant is associated with improved survival post-transplant. 93 Furthermore, treatment with vasoconstrictor therapy reduces the likelihood for pre- and post-transplant RRT requirement. 94 In patients who require RRT pre-transplant, liver transplantation should not be delayed, as the most important predictor of post-transplant renal function recovery is the duration of pre-transplant RRT. The cut-off duration for predicting renal function recovery has been reported to be 14 days. 34 For patients who are not transplant candidates, palliative care should be offered.
Simultaneous liver–kidney transplantation
There is a lot of controversy regarding simultaneous liver–kidney transplantation (SLKT) for patients with HRS-AKI, as this takes away an organ from patients on the kidney transplant waiting list. It is also a contentious topic among transplant fraternity due to questionable survival benefit following SLKT, particularly among those who are not on dialysis during transplant. 95 In general, SLKT is considered for those patients with HRS-AKI who are unlikely to have renal recovery with liver transplant alone, or in patients with underlying CKD and hereditary renal conditions (Table 4). With the demographic change in the indication of liver transplantation with increase in non-alcoholic steatohepatitis as the primary indication, many of the patients may have underlying chronic kidney dysfunction. 96 According to current Organ Procurement and Transplantation Network policy in the United States, the use of SLKT requires patients to be on dialysis or have an estimated GFR of ⩽25 ml/min for a minimum of consecutive 6 weeks. 97 With the creation of a ‘safety net’ in the recent allocation policy, priorities are given to those who are placed on the kidney waiting list within 1 year of liver transplant. 97 However, factors that may impact SLKT decision, such as age, gender, race, and co-morbid conditions, are not included in the recent policy to decide about the allocation. In general, patients with structural renal disease, probably secondary to diabetes mellitus, hypertension are likely to have shorter survival compared to those with HRS-AKI or ATN post-liver transplant, 98 as the associated CKD with these conditions has a negative impact on survival, 99 and therefore should be offered SLKT. A recent publication, which assessed the impact of the 2017 policy change on renal outcomes, found that there was an increased access to deceased donor kidney transplantation for liver transplant alone recipients with kidney disease without negatively affecting outcomes. 100
OPTN selection criteria for SLKT.

Source: Adapted and modified from Maiwall and Sarin. 92
AKI: acute kidney injury; CKD, chronic kidney disease; Cr Cl, creatinine clearance; eGFR, estimated glomerular filtration rate; ESRD, end-stage renal disease; HUS, hemolytic uremic syndrome; OPTN, Organ Procurement and Transplantation Network; SLK, simultaneous liver–kidney; SLKT, simultaneous liver–kidney transplant.
Prognosis
The prognosis of HRS1 is very poor with a median survival measured in days to weeks if left untreated. 101 Treatment of HRS1 with terlipressin does not improve the overall or transplant-free survival.54,69 However, for patients who respond to terlipressin, the 90-day transplant-free survival is consistently better than that of non-responders.69,58,93 For patients with other organ failures as part of the ACLF syndrome, the prognosis is even worse. 72 High lactate levels, a sign of tissue ischemia, also has been identified as a sign of poor prognosis. 102
For patients who have developed HRS-AKI superimposed on CKD, the episode of HRS tends to have a much more aggressive course. 51 The presence of CKD also predisposes the patient to the development of other organ failure. 103 In fact, for every 10 ml/min drop in estimated GFR, there is a 10.5% increased risk for circulatory failure, 7.0% for brain failure, and 5.8% for respiratory failure. 103 The presence of CKD, as indicated by proteinuria, provides 82.4% sensitivity and 80% specificity for the development of AKI when the urinary protein/creatinine ratio is >30. 104 Therefore, it is imperative for patients who have chronic conditions, such as diabetes or systemic hypertension, to have these potential etiologies of CKD meticulously managed, so to improve the overall prognosis of these patients.
Prevention of HRS
HRS-AKI is most often precipitated by a precipitating event. 105 Identifying and preventing the precipitating event is the key to the prevention of HRS-AKI. Several well-established strategies are recommended by various academic associations.46,47 These include regular monitoring of renal function when patients are started on diuretics, avoidance of excessive diuretics use, start regular LVP when patients have developed refractory ascites, use of albumin with LVP of >5 l to prevent paracentesis-induced circulatory disturbances, prophylaxis against bacterial infection such as during an episode of gastrointestinal bleed or after an episode of spontaneous bacterial peritonitis (SBP), as well as avoidance of nephrotoxic drugs or the use of non-steroidal anti-inflammatory agents.
As bacterial infections are the most common cause of HRS-AKI, 34 primary prophylactic against SBP in patients with advanced liver disease defined as Child–Pugh score of ⩾9, a serum bilirubin level ⩾5 mg/dl, a sCr level of ⩾1.2 mg/dl, blood urea nitrogen level of ⩾42.6 mg/dl, and serum sodium of ⩽130 mmol/l has been shown to be associated with reduced incidence of HRS and overall mortality. 106 However, a recent prospective study found that primary prophylaxis against SBP was associated with a higher incidence of AKI (48% versus 30%; p = 0.04), and higher 90-day mortality (35% versus 22%; p = 0.02) compared to patients who were on secondary SBP prophylaxis, 107 possibly related to changed microbiota with continued antibiotic use. It has been suggested that a personalized approach to antibiotic prophylaxis, weighing out risks and benefits, is the most appropriate strategy. 108 The results of a recent network meta-analysis have thrown considerable uncertainty about whether antibiotic prophylaxis against SBP is beneficial, and if beneficial, which antibiotic prophylaxis is most beneficial in people with cirrhosis and ascites with low protein or history of SBP. 109 Therefore, further studies are needed to better define this evolving issue.
Finally, long-term weekly albumin infusion to patients with decompensated cirrhosis and non-refractory ascites compared to standard of care has been shown to reduce the incidence of HRS and improved overall survival in a large RCT. 110 However, this does not appear to be the definitive answer to the chronic use of albumin in these patients, 111 as two further RCTs among liver transplant listed outpatients using albumin and midodrine versus placebo, or among admitted patients with decompensated cirrhosis did not prevent the development of complications including HRS.43,112
Future directions
Early diagnosis of HRS-AKI to initiate vasoconstrictor therapy is crucial in the management of HRS-AKI. Existing tools, such as urinalysis and fractional excretion of sodium, have significant limitation in differentiating the different phenotypes like prerenal, HRS-AKI, and structural kidney injury. Metabolomic profiling may be the new tool to identify biomarkers of renal dysfunction in patients with cirrhosis, allowing early diagnosis of HRS, prediction of response to HRS treatment, and native kidney recovery after liver transplantation, all important in the decision-making regarding the need for SLKT.113,114 The future should also see further developments in the prediction models of morbidity and mortality in patients with cirrhosis and HRS-AKI, so to improve our management of these patients.
Conclusion
HRS-AKI is a form of AKI in cirrhosis. It is a diagnosis of exclusion. Recent changes in diagnostic criteria means that HRS-AKI can now be diagnosed at a lower level of sCr, thereby allowing earlier institution of treatment. Vasoconstrictors are the mainstay of treatment with terlipressin being the most commonly used worldwide, but terlipressin is not yet available in most parts of North America. Vasoconstrictors are usually given in conjunction with albumin, but care needs to be taken not to overdose patients with albumin. This is because albumin increases the cardiac preload by improving the volume status of the patients, whereas vasoconstrictors by increasing the afterload may cause cardiac failure with pulmonary edema in these patients. Timely assessment for liver transplant is necessary, as these patients still have a high mortality rate despite response to pharmacotherapy because of their advanced liver dysfunction. RRT may be used as a bridging therapy, but patients should not be maintained on long-term RRT while waiting for a liver transplant, as this has a negative impact on post-transplant patient outcomes including non-reversal of HRS. Combined liver kidney transplant should be considered for patients with underlying CKD or with prolonged pre-transplant RRT. The future will see the availability of new techniques and biomarkers to identify the susceptibility for HRS development, progression of HRS, and response to treatment. This will allow for personalized targeted treatment to improve patient outcomes.
Footnotes
Author contribution(s)
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
Mallinckrodt Pharmaceutical: Consultant and research grant provided to the institution.
