Abstract
Introduction:
Diarrhea is common in persons living with HIV (PLWH)/AIDS. With the increasing utilization of multiplex gastrointestinal PCR panel (GI panel) testing, we aimed to characterize the roles of CD4 count and hospitalization in GI panel assessments of PLWH with acute diarrhea.
Methods:
We performed a cross-sectional study of adult PLWH with acute diarrhea who underwent GI panel testing at two urban academic centers. Demographic, HIV disease, GI panel result, and hospitalization data were collected, and patients were cohorted by CD4 count (CD4 < 200, CD4 200–499, CD4 > = 500). The primary outcome was enteric infection as detected by GI panel, and hospitalization.
Results:
Of 298 PLWH, 119 (39.9%) had a CD4 count below 200, 195 (65.4%) were hospitalized, and 137 (46.0%) had enteric infection. Bacterial infection correlated with higher CD4 count (41.9% (CD4 > = 500) vs 31.2% (CD4 200–499) vs 25.2% (CD4 < 200), p = 0.041). Hospitalization correlated with poorly controlled HIV and fewer enteric infections (34.4% vs 68.0%, p < 0.001). After adjusting for HIV disease severity, a negative GI panel remained independently associated with hospitalization (adjusted odds ratio (aOR) 5.32, 95% confidence interval (CI) 2.72–10.9), even in patients tested within 72 hours of hospitalization. Despite better HIV control, men who have sex with men (MSM) had more frequent infectious diarrhea, including from E. coli, giardiasis, and multiple pathogens. MSM status independently predicted enteric infection (aOR 1.93, 95% CI: 1.02–3.67).
Conclusions:
GI panel results vary by HIV disease severity and hospitalization in PLWH. Clinicians – especially in the inpatient setting – should carefully consider these factors when interpreting GI panel results. Further characterization of diarrheal etiology in PLWH with a negative GI panel is needed.
Plain Language Summary
Diarrhea is common in people living with HIV (PLWH) and has a variety of causes, including infections, medications, and HIV itself. Multiplex polymerase chain reaction (PCR) stool testing simultaneously evaluates for a variety of common viral, bacterial, and parasitic infections of the gastrointestinal tract, and is increasingly being used in patients with diarrhea. However, patients with HIV and diarrheal illness may have uncommon infections not typically present in those with normal immune function – and thus not routinely evaluated for in stool testing. It is not known what factors, if any, might affect the results of PCR testing in HIV-related diarrhea.
In this study, we examined all PLWH who underwent stool PCR testing for diarrhea over a 4-year period. We separated the patients into groups based on HIV disease severity as measured by CD4 T-cell count, or the count of the immune cells affected by HIV. We examined whether there were differences among groups in infection rates as detected by PCR stool testing. Separately, we studied the role of hospitalization in stool PCR test results.
Of 298 PLWH who underwent stool PCR testing for diarrhea, 119 had a CD4 count less than 200 (low CD4 count), 195 were hospitalized at time of testing, and 137 had a positive stool PCR test. Compared to those with a low CD4 count, subjects with less severe HIV disease were more likely to have a bacterial infection on stool PCR testing and less likely to be hospitalized. Hospitalized patients were more likely to have a negative PCR stool test, regardless of CD4 count. Many patients with a low CD4 count had diarrheal etiologies not evaluated by multiplex stool PCR. In PLWH who experience diarrhea, stool PCR testing results vary by CD4 count and hospitalization. Providers should be mindful of these factors when interpreting stool PCR test results.
Introduction
Despite advances in antiretroviral therapy (ART), diarrhea is common in people living with HIV (PLWH) and negatively affects quality of life.1–3 Although diarrheal illness from opportunistic infections has declined in the ART era, the overall prevalence of diarrhea remains unchanged, affecting an estimated 28%–60% of PLWH.1,4,5 This is attributed to a concurrent rise in non-infectious diarrhea in patients with AIDS, with postulated etiologies including HIV-associated enteropathy, ART side effects, HIV-related enteral malignancy, and irritable bowel syndrome.1,2,6
A similar trend is apparent in hospitalization rates in PLWH. Though all-cause hospitalization in PLWH declined between 1996 and 2016, PLWH remain twice as likely to be hospitalized than the general population.7,8 Among PLWH, those with a CD4 T-cell count below 200 are three times more likely to be hospitalized.7,9
Guidelines recommend evaluating PLWH with diarrheal symptoms for Cryptosporidium, Cyclospora, Cystoisospora, microsporidia, Mycobacterium avium complex (MAC), and cytomegalovirus (CMV) in addition to conventional stool culture, microscopy, and stool ova and parasite examinations.2,10 Recently, multiplex gastrointestinal pathogen polymerase chain reaction panels (GI panels) are being added to conventional stool testing, which typically include bacterial culture with ova and parasites exam, in the assessment of diarrhea in all patients. However, the role of the GI panel in PLWH with acute diarrheal illness remains largely unexplored. In addition, although AIDS is a risk factor for non-infectious diarrhea and hospitalization, it is not known whether CD4 count or hospitalization status affect GI panel results. In this study, we aimed to characterize the role of CD4 count and hospitalization in the evaluation of enteric infection by GI panel in PLWH with acute diarrhea.
Methods
Patient selection
We performed a cross-sectional study of the electronic medical records at two urban quaternary care institutions serving New York City (NYU Langone Health and NewYork-Presbyterian-Columbia University Medical Center). Adult patients with HIV who underwent stool testing between 2015 and 2019 with the FilmArray GI pathogen panel (BioFire Diagnostics, Salt Lake City, UT) for acute diarrhea were included in the study. HIV diagnosis was evaluated using International Classification of Disease-10 coding 11 and confirmed by manual chart review. Acute diarrhea was ascertained by description in provider chart documentation and defined as ⩾ 3 unformed stools in 24 hours for less than 14 days. Patients who did not have a CD4 T-cell count evaluated within three months of diarrheal symptoms were excluded from the analyses. Consent for treatment was obtained from all patients at the time of medical evaluation, and all patient data was de-identified at the time of data collection.
Variables and definitions
The following demographic data were collected: age, sex, race, ethnicity, and Charlson comorbidity index. 12 Self-identification as a man who has sex with men (MSM) was also collected based on provider chart documentation. Data on testing context, testing setting, and symptomatology were collected. HIV disease severity was evaluated using CD4 T-cell count and HIV RNA viral load collected within 3 months of GI panel. Patients with a CD4 count below 200 cells/mm3 at time of GI panel testing were classified as having a low CD4 count rather than AIDS, as the presence of opportunistic infection was not evaluated for. An unsuppressed HIV RNA viral load was defined as ⩾ 20 copies HIV RNA/mL. Antibiotic management, including initiation of empiric antibiotic therapy (prior to result of GI PCR testing) and initiation of directed antibiotic therapy (after result of GI PCR testing) was recorded. Hospitalization at time of testing was noted. For hospitalized patients, length of stay (LOS) and the time interval between admission date and test date were calculated. Time interval was recorded as a negative time value for specimens collected in the emergency department prior to admission.
Only the first GI panel tested for diarrheal symptoms was included for each patient. Enteric infection was defined as a positive result on the GI panel. The GI panel is capable of identifying the nucleic acids of 23 pathogens (14 bacteria, 5 viruses, and 4 parasites) within one hour, and is performed on stool samples stored in Cary Blair transport medium. The test has a clinical sensitivity and specificity of 94.5% to 100%.13,14 The pathogens are: Campylobacter jejuni, Clostridioides difficile (toxin A/B), Plesiomonas shigelloides, Salmonella species, Yersinia enterocolitica, Vibrio parahaemolyticus, Vibrio vulnificus, Vibrio cholerae, enteroaggregative Escherichia coli (EAEC), enteropathogenic E. coli (EPEC), enterotoxigenic E. coli (ETEC) lt/st, Shiga-like toxin-producing E. coli (STEC) stx1/stx2, E. coli O157, Shigella/enteroinvasive E. coli (EIEC), adenovirus F40/41, astrovirus, norovirus GI//GII, rotavirus A, sapovirus (I, II, IV, and V), Cryptosporidium species, Cyclospora cayatenesis, Entamoeba histolytica, and Giardia lamblia. At both institutions, a separate PCR test was utilized to evaluate for Clostridioides difficile infection (Xpert C. difficile; Cepheid, Sunnyvale, CA).
Outcome and statistical analyses
The primary outcome was presence of enteric infection by stool GI panel. Occurrences of ED visit, surgery, death, endoscopy, and hospitalization within 30 days of stool testing were examined as secondary outcomes. If patients were hospitalized at time of testing, only re-hospitalization was considered a 30-day hospitalization event.
The primary outcome was first assessed by CD4 count (CD4 < 200, CD4 200–499, CD4 ⩾ 500) and then by comparing hospitalization status. Categorical variables were analyzed using chi-square testing. Continuous variables were analyzed using either the Mann–Whitney test or the ANOVA test. To further investigate the relationship between hospitalization, CD4 count, and GI panel result, two multivariable analyses were performed using logistic regression modeling with GI panel result and hospitalization each being the dependent variable, and adjusted for covariates of age, sex, race, MSM status, Charlson’s comorbidity index, viral load, and CD4 category. To minimize confounding from hospital-acquired diarrhea, a sub-analysis was performed by limiting hospitalized patients to only those who underwent GI panel testing within 72 hours of admission. A second sub-analysis performed on male patients evaluated the effect of MSM status on study outcomes. All statistical analyses were conducted using R version 3.3.3. 15 A p value < 0.05 was considered statistically significant. The study was approved by the New York University Institutional Review Board (i18-01121; 8/16/2018) and is reported in accordance with the STROBE statement. 16
Results
Characteristics and outcomes
Of 342 adult PLWH evaluated with the GI panel from 2015 to 2019 for acute diarrhea, 298 had a CD4 count evaluated within 3 months of GI panel testing (Table 1). The median age at testing was 51 years (interquartile range (IQR) 40.3–58 years). Of all patients, 79 (26.5%) were female, and 126 (42.3%) identified as MSM. There were 119 patients (39.9%) with low CD4 T-cell count, and 148 (49.7%) with unsuppressed viral loads. Of 92 patients at one center (NYU) in the study, 5 were diagnosed with HIV within 1 year prior to testing; the median duration of HIV disease was 18.4 years (IQR 6.4–27.2 years).
Baseline characteristics.

IQR, interquartile range.
Enteric infection revealed by the GI panel was present in 137 (46.0%) patients: 41 with viral pathogens, 95 with bacterial pathogens, 34 with parasitic pathogens, and 43 with multiple pathogen types (Table 2). The most common pathogens included EPEC, norovirus, Campylobacter species, Shigella/EIEC, Cryptosporidium, EAEC, and Giardia lamblia (Supplementary Figure 1). At the time of evaluation, 195 patients (65.4%) required hospitalization. Antibiotics were prescribed to 193 (64.8%) of patients; 125 (41.9%) received empiric antibiotic therapy, and 84 (28.2%) received directed antibiotic therapy (Table 2). Within 30 days of stool testing, 23 (7.7%) patients died, 24 (8.1%) underwent endoscopy, 20 (6.7%) underwent surgery, and 39 (13.1%) were either hospitalized or re-hospitalized. There was no correlation between GI panel positivity and mortality (p = 0.262). Of 17 patients with low CD4 count and a negative GI panel at one center (NYU Langone Health), 7 (41%) had infectious diarrhea from opportunistic pathogens not included on the GI panel such as MAC, histoplasmosis, and Pneumocystis carinii. These alternate infectious etiologies were identified through serum, urine, or endoscopic evaluation. Of 20 patients with a CD4 count > 200 and a negative GI panel, there were no opportunistic infections identified as the cause of diarrhea (Supplementary Table 1).
Results of gastrointestinal (GI) panel.

EAEC, enteroaggregative Escherichia coli; EIEC, enteroinvasive E. coli; EPEC, enteropathogenic E. coli; ETEC, enterotoxigenic E. coli; GI, gastrointestinal; STEC, Shiga-like toxin-producing E. coli.
Comparison among CD4 groups
In correlation analyses comparing three CD4 groups (CD4 < 200, CD4 200–499, CD4 > = 500), there were no differences in overall enteric infection rates among groups (p = 0.159). Bacterial infection was more common in higher CD4 count groups (41.9% (CD4 > = 500, n = 36) vs 31.2% (CD4 200–499, n = 29) vs 25.2% (CD4 < 200, n = 30), p = 0.041; Figure 1). There were trends toward higher Cryptosporidium and viral infections and lower Campylobacter infections in patients with a low CD4 count, although this was not significant (Table 2). Patients with a CD4 count of 500 or higher were less likely to be hospitalized (40.7% (CD4 > = 500, n = 35) vs 61.3% (CD4 200–499, n = 57) vs 86.6% (CD4 < 200, n = 103), p < 0.001) and more likely to identify as MSM. Patients with a low CD4 count were more likely to receive empiric antibiotic therapy, while patients with normal CD4 counts were more likely to receive directed antibiotic therapy. There were no differences among CD4 groups in 30-day secondary outcomes.

Distribution of pathogen classes within positive GI panels of each CD4 count group. Those with CD4 count < 200 were less likely to have a bacterial enteric infection (p = 0.041).
Comparison between hospitalized and non-hospitalized patients
Hospitalized patients were less likely to have a positive GI panel result than non-hospitalized patients (34.4% (n = 67) vs 68.0% (n = 70), p < 0.001; Figure 2, Table 2). Specifically, there were fewer bacterial (23.6% (n = 46) vs 47.6% (n = 47.6), p < 0.001) and parasitic (8.2% (n = 16) vs 17.5% (n = 18), p = 0.017) infections in hospitalized patients compared to non-hospitalized patients. Those not hospitalized were more likely to have enteric infections from Campylobacter, EAEC, and EPEC. Unsuppressed viral load and the presence of low CD4 count were associated with hospitalization. Hospitalized patients were more likely to receive empiric antibiotic therapy while non-hospitalized patients were more likely to receive directed antibiotic therapy. Hospitalized patients had higher 30-day mortality than non-hospitalized patients.
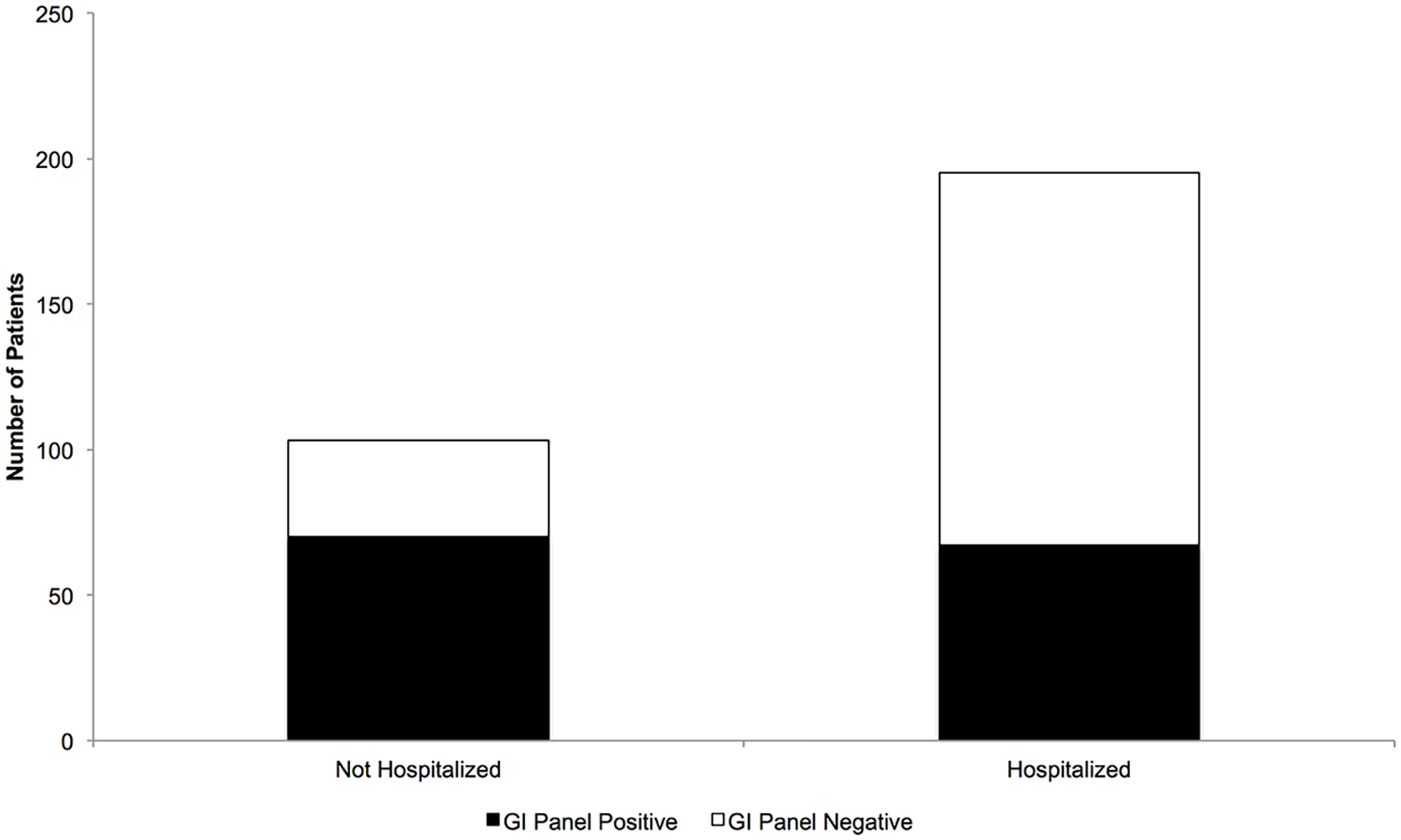
Hospitalized patients are less likely to have a positive GI panel.
Multivariable analyses
In a multivariable analysis adjusted for age, sex, race, MSM status, and Charlson’s comorbidity index, an unsuppressed viral load was associated with a positive GI panel (adjusted odds ratio (aOR) 2.65, 95% Confidence Interval (CI) 1.38–5.25; Table 3). Hospitalized patients were less likely to have a positive GI panel (aOR 0.209, 95% CI:: 0.196–0.400). Separately, a negative GI panel was associated with hospitalization after controlling for age, sex, race, MSM status, medical comorbidities, and HIV disease severity (aOR 5.32, 95% CI: 2.72–10.9). A low CD4 count and unsuppressed viremia were also associated with hospitalization (Table 3).
Adjusted multivariable regressions of the study.

GI, gastrointestinal. Bold values are statistically significant.
Sub-analysis A: limiting inpatients to those tested within 72 hours
Among hospitalized patients, earlier testing was associated with a positive GI panel (median time to test 1.00 days (IQR 0.489–1.97) for positive GI panel vs 2.00 days (IQR 0.899–4.63) for negative GI panel, p < 0.001). To evaluate for confounding from hospital-acquired non-infectious diarrhea, a sub-analysis was conducted limiting hospitalized patients to those who underwent GI panel testing within 72 hours of admission. In the sub-analysis, 59.3% of hospitalized patients tested within 72 hours of admission had a negative GI panel, while 32.0% not hospitalized had a negative GI panel (p < 0.001; Supplementary Figure 2). In an additional multivariable analysis, hospitalization was associated with negative GI panel (aOR 4.08, 95% CI 2.01–8.62; Supplementary Table 2) in the limited cohort.
Sub-analysis B: the MSM population
A separate sub-analysis was conducted in male patients to further characterize the effect of MSM status. In correlation analyses, patients identifying as MSM had higher rates of a positive GI panel compared to non-MSM patients (62.7% (n = 79) vs 41.9% (n = 39), p = 0.002; Supplementary Table 3). MSM patients were more likely to have a parasitic infection (19.0% (n = 24) vs 8.6% (n = 8), p = 0.031), specifically giardiasis (11.9% (n = 15) vs 2.2% (n = 2), p = 0.008), an E. coli infection (31.7% (n = 40) vs 19.4% (n = 18), p = 0.040), and multiple pathogens (25.4% (n = 32) vs 10.8% (n = 10), p = 0.007) compared to non-MSM patients. MSM patients had better HIV control by viral load and CD4 count compared to non-MSM patients (Supplementary Table 3). Among male patients, MSM status remained associated with positive GI panel result (aOR 1.93, 95% CI 1.02–3.67; Supplementary Table 4) in the multivariable analysis controlling for HIV disease control.
Discussion
In this dual-center study of PLWH with acute diarrhea, the rates of enteric infection detection by multiplex GI panel were comparable among CD4 count groups, although patients with a low CD4 count were less likely to have bacterial enteric infections. In addition, hospitalized patients were more likely to have worse HIV disease control and more likely to have a negative GI panel. These associations remained true even in inpatients tested within 72 hours of hospitalization. In hospitalized PLWH with unsuppressed HIV, diarrhea may be from infections not evaluated by the GI panel or from non-infectious etiologies.
The GI panel has advantages over traditional stool immunoassay testing in PLWH, including faster test turnaround time and improvement in antibiotic stewardship via detection of viral enteric infections not identified on conventional stool testing. 17 As the GI panel is increasingly used over conventional stool testing in this population, it is critical to understand factors that may affect the interpretability of its results.
In this study, the overall rate of enteric infection by GI panel was 46%, similar to a previous study in PLWH with diarrhea (53%), and higher than previous studies of diarrhea in the general population (35–37%).17,18 Despite higher rates of enteric infection in PLWH, we found no differences in overall enteric infection rates by CD4 count within the PLWH population. In patients with a low CD4 count, however, there were fewer bacterial enteric infections. While this may be related to the more frequent prescription of prophylactic antimicrobials and consequent inhibition of potential bacterial enteric pathogens in these patients in our study, it would be unusual for patients non-adherent to ART to be adherent to antimicrobial prophylaxis. Alternatively, this may be explained by the concurrent trend toward higher viral and Cryptosporidium infections in patients with a low CD4 count. Low CD4 count status nearly doubles the risk of parasitic enteric infections, especially from Cryptosporidium.19–22 Patients with a low CD4 count may be more susceptible to parasitic and viral pathogens compared to those with higher CD4 counts. Thus, they experience relatively fewer bacterial enteric infections and similar overall enteric infections compared to other CD4 groups.
E. coli, Cryptosporidium, Microsporidia, Cystoisospora belli, CMV, and MAC are the most common microorganisms implicated in diarrhea in PLWH.10,23 In this study, E. coli and Cryptosporidium were among the most prevalent pathogens identified on the GI panel. The most common pathogen overall was EPEC, which is known to be common in PLWH with diarrheal illness and was the most commonly detected organism in a multinational study of community acquired gastroenteritis.17,24 Norovirus, a common viral pathogen in PLWH with diarrhea, was the most common viral pathogen identified by GI panel in our study.25,26
Hospitalization and negative GI panel
Interestingly, there were fewer enteric infections by GI panel in hospitalized compared to non-hospitalized PLWH, even after adjustment for HIV disease severity. Since GI panel testing after 72 hours of hospitalization has low diagnostic yield, 27 we confirmed this association in a sub-analysis limiting the hospitalized patient population to those tested within 72 hours of hospitalization – suggesting against confounding from hospital-acquired diarrhea. One possible explanation for this relationship is that hospitalized patients had worse HIV control in our study and thus may have been more likely to experience infectious diarrhea caused by an organism not evaluated by the GI panel. Lower CD4 count is associated with the successful identification of endoscopically diagnosed opportunistic infections (such as CMV and MAC).28,29 Indeed, in one center in our study, 41% of patients with a low CD4 count who tested negative on the GI panel had infectious diarrhea from opportunistic pathogens such as MAC, histoplasmosis, and Pneumocystis carinii, whereas none with a CD4 count 200 or above had opportunistic infections identified as the cause of diarrhea.
Another possible mechanism of CD4 count influence on the relationship between hospitalization and negative GI panel could be that a low CD4 count is a risk factor for non-infectious diarrhea as well.1,2 Non-infectious etiologies common in those with a low CD4 count include HIV-related enteropathy and HIV-associated malignancies, such as Kaposi sarcoma and Non-Hodgkin lymphoma,2,30–32 although these etiologies were not specifically evaluated for in our study. Protease inhibitors have been implicated in ART-related diarrhea through their mediation of apoptosis of intestinal epithelium, ultimately promoting water secretion into the gut lumen. 33 Though non-infectious diarrhea could be caused by ART medications, this is less likely in our study as fewer hospitalized patients were on ART at time of GI panel testing than non-hospitalized patients. Irritable bowel syndrome, another non-infectious etiology, is more prevalent in PLWH compared to patients who are HIV-negative. 6
MSM sub-analysis
Despite improved HIV control, MSM patients had higher rates of infectious diarrhea overall, including from E. coli, Giardia, and multiple pathogens. MSM patients were also less likely to be hospitalized in our study than non-MSM patients; this is consistent with prior studies in which MSM PLWH are less likely to experience all-cause hospitalization.7,8 Bacterial enteric pathogens that appear to specifically affect MSM patients regardless of HIV status include E. coli, Shigella, and Campylobacter species. 34 In our study of PLWH, E. coli and Shigella infections were more common in MSM patients. Giardiasis, the most commonly identified parasite in a prior study of all MSM patients with diarrhea, 34 was associated with MSM status in our study. Multipathogen enteric infection was associated with MSM status and was present in 25% of MSM patients in our study; multiple pathogens have been present in up to 43% of all MSM patients. 34 It is likely that sexual practices among MSM patients – regardless of degree of immunosuppression – place them at elevated risk of unintentional fecal-oral transmission of these organisms and subsequent enteric infection. 35 These findings illustrate the need for new strategies to prevent sexual transmission of enteric pathogens in MSM PLWH.
Study limitations
This study was limited by its retrospective nature; diarrhea is commonly underreported as a symptom in PLWH 2 and may have been missed if not specifically asked about. Further, although diarrheal symptoms at time of testing were manually confirmed by chart review, diarrhea may not have been the predominant symptom at time of GI panel testing; this may affect the generalizability of our results. Symptom chronicity may have been a potential confounder; although we manually restricted our cohort to patients with acute diarrhea, the retrospective nature of our study makes it possible that patients with chronic – and thus more likely non-infectious – diarrhea were included. These patients could be disproportionately more likely to seek evaluation and be hospitalized. In addition, the study was limited to only those patients evaluated with the GI panel. Although this was done to evaluate factors potentially affecting the results of this increasingly utilized test, it is possible that our results missed outcomes from PLWH with diarrhea who were not evaluated with a GI panel.
Our exclusion of individuals without a CD4 count within 3 months of testing could potentially bias our study population toward those with recent HIV diagnosis or those not on ART. However, this is less likely given that the median duration of HIV at one study center was 18.4 years. In addition, it is unlikely that patients with diarrheal disease severe enough to necessitate medical care and diagnostic evaluation with stool testing would not also undergo CD4 count testing to evaluate for medication non-adherence and to guide clinical decision-making. Removal of this criterion could lead to the false labeling of subjects non-adherent to ART as having well-controlled HIV disease, precluding accurate investigation of the relationship between HIV severity and the GI panel result.
Two potential confounders that were not examined in this study include intravenous drug use and admission to the intensive care unit. Intravenous drug use is a known risk factor for hospitalization in PLWH, and intensive care unit admission is associated with higher rates of non-infectious diarrhea in patients evaluated with GI panel.8,36
The GI panel may not reliably distinguish between infection and colonization by specific pathogens, as the isolation of genetic material by PCR testing is not necessarily equivalent to viable organisms present in the gastrointestinal tract. The clinical significance of this discrepancy is not well established. While we were able to quantify the prevalence of opportunistic infectious diarrhea in patients with a low CD4 count and negative GI panels at one site, we did not specifically evaluate for pathogens not tested for by the GI panel at both sites; given our findings of higher rates of negative GI panels in hospitalized patients with worse HIV control, this needs to be further explored. Finally, we did not evaluate for a history of opportunistic infections, precluding our ability to precisely define the population of patients with AIDS in our study. Instead, we used a low CD4 T-cell count to approximate AIDS status, as has been done previously.1,21,22
Conclusion
Diarrheal disease in PLWH continues to impact quality of life and morbidity despite advancements in ART. Results of the GI panel–a contemporary method for testing for enteric infection – vary based on HIV disease severity and hospitalization. Clinicians caring for PLWH should be cognizant of these factors when interpreting GI panel results. While these data suggest that opportunistic infection may underlie diarrhea in patients with severe HIV and a negative GI panel, further studies are needed to verify whether this relationship is driven by infectious causes not identified by the GI panel, as this would inform the necessity of additional testing.
Supplemental Material
sj-docx-3-tag-10.1177_17562848221092593 – Supplemental material for The influence of hospitalization and HIV severity on gastrointestinal PCR panel evaluation of HIV-related acute diarrhea in New York City: a retrospective, cross-sectional study
Supplemental material, sj-docx-3-tag-10.1177_17562848221092593 for The influence of hospitalization and HIV severity on gastrointestinal PCR panel evaluation of HIV-related acute diarrhea in New York City: a retrospective, cross-sectional study by Abhishek Verma, Ashley M. Hine, Andrew Joelson, Rena Mei, Robert A. Pitts, Benjamin Lebwohl and Jordan E. Axelrad in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-4-tag-10.1177_17562848221092593 – Supplemental material for The influence of hospitalization and HIV severity on gastrointestinal PCR panel evaluation of HIV-related acute diarrhea in New York City: a retrospective, cross-sectional study
Supplemental material, sj-docx-4-tag-10.1177_17562848221092593 for The influence of hospitalization and HIV severity on gastrointestinal PCR panel evaluation of HIV-related acute diarrhea in New York City: a retrospective, cross-sectional study by Abhishek Verma, Ashley M. Hine, Andrew Joelson, Rena Mei, Robert A. Pitts, Benjamin Lebwohl and Jordan E. Axelrad in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tiff-1-tag-10.1177_17562848221092593 – Supplemental material for The influence of hospitalization and HIV severity on gastrointestinal PCR panel evaluation of HIV-related acute diarrhea in New York City: a retrospective, cross-sectional study
Supplemental material, sj-tiff-1-tag-10.1177_17562848221092593 for The influence of hospitalization and HIV severity on gastrointestinal PCR panel evaluation of HIV-related acute diarrhea in New York City: a retrospective, cross-sectional study by Abhishek Verma, Ashley M. Hine, Andrew Joelson, Rena Mei, Robert A. Pitts, Benjamin Lebwohl and Jordan E. Axelrad in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tiff-2-tag-10.1177_17562848221092593 – Supplemental material for The influence of hospitalization and HIV severity on gastrointestinal PCR panel evaluation of HIV-related acute diarrhea in New York City: a retrospective, cross-sectional study
Supplemental material, sj-tiff-2-tag-10.1177_17562848221092593 for The influence of hospitalization and HIV severity on gastrointestinal PCR panel evaluation of HIV-related acute diarrhea in New York City: a retrospective, cross-sectional study by Abhishek Verma, Ashley M. Hine, Andrew Joelson, Rena Mei, Robert A. Pitts, Benjamin Lebwohl and Jordan E. Axelrad in Therapeutic Advances in Gastroenterology
Footnotes
Author contribution(s)
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JEA receives research support from BioFire Diagnostics and has received consultancy fees from Janssen, Abbvie, and BioFire Diagnostics. JEA receives research support from the Crohn’s and Colitis Foundation and The National Institute of Diabetes and Digestive and Kidney Diseases [Grant Number: K23DK124570]. AV: No disclosures. AMH: No disclosures. AJ: No disclosures. RM: No disclosures. BL: No disclosures.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
