Abstract
Chronic noncancer pain (CNCP) affects up to 20% of adults and can interfere with activities of daily living. Up to 4% of adults in the United States receive chronic opioid therapy and up to 57% of patients on long-term opioids for CNCP report opioid-induced constipation (OIC). OIC is essentially constipation occurring after starting opioid treatment. While laxatives are traditionally the first-line therapy for OIC, 81% of patients taking daily laxatives and opioids still reported OIC and considered that it negatively affected their quality of life. Naldemedine is a peripherally acting µ-opioid receptor antagonists (PAMORA) approved for the treatment of OIC in patients with CNCP. This article reviews the mechanism of action, efficacy, and safety of naldemedine in CNCP patients. Naldemedine improves OIC in patients with CNCP by acting as an opioid receptor antagonist in the gastrointestinal tract. It does not interfere with the analgesic properties of opioids or cause withdrawal symptoms since these effects are centrally mediated, and naldemedine does not cross the blood brain barrier. Naldemedine showed significant and sustained improvement in frequency of bowel movements, quality of life, and constipation-related symptoms. It is generally well tolerated with a higher incidence of gastrointestinal adverse events of mild or moderate severity such as diarrhea, abdominal pain, or vomiting compared to placebo. While there are no randomized, controlled trials that compare head-to-head pharmacological therapies used for treatment of OIC, network meta-analysis shows that naldemedine has an overall good benefit-risk profile compared to the other approved medications.
Introduction
Chronic pain is defined as a lasting or recurring pain that persists beyond 3 months. 1
Chronic noncancer pain (CNCP) refers to pain related to any etiology except malignancy. CNCP affects up to 20% of adults, with a higher prevalence in women and in patients older than 40 years old.2,3 CNCP interferes with activities of daily living, is associated with higher rates of work absenteeism, significantly impacts quality of life, and is commonly assessed as ‘the fifth vital sign’.2,4 Chronic pain increases healthcare utilization including more physician visits and hospitalizations. Adequate pain control has been shown to improve and sometimes normalize these parameters. 2
The World Health Organization analgesic ladder for the treatment of cancer pain entails step 1 consisting of nonopioids, step 2 mild opioids, and step 3 strong opioids. 5 However, this cancer pain analgesic ladder seems suboptimal for CNCP. 6 With the liberalization of opioid prescriptions in the late 1990s,7,8 up to 4% of adults in the United States are maintained on chronic opioid therapy for chronic pain which is mainly noncancer related, 9 up to 20% of the patients presenting to physicians with pain symptoms are prescribed opioids, 10 and up to 90% of patients with moderate or severe CNCP are on opioid therapy. 11 Opioids might provide short- or medium-term relief in CNCP; however, long-term effectiveness seems more variable. 12 Up to 22.9% of patients on oral opioids discontinue long-term therapy due to adverse events. 13 In a systematic review with 18,679 participants with CNCP, opioid usage was associated with a 42% higher risk of any adverse effect and 175% higher risk of serious adverse events compared to controls, including constipation, dizziness, drowsiness, fatigue, hot flushes, sweating, nausea, pruritis, and vomiting.14 While long-term opioid therapy is associated with development of tolerance to the therapeutic effect of opioids, leading to increased doses over time to maintain adequate pain control, there is also tolerance to the majority of side effects with the notable exception of opioid-induced constipation (OIC) which tends to persist over time, 15 and the mechanism is outlined in the following.
Pharmacology of opioid receptors
Opioid receptors located either within the central nervous system or peripherally are G protein coupled receptors which regulate pain and stress, and affect the respiratory, cardiovascular, and gastrointestinal (GI) systems. 16 Three major classes of opioid receptors have been identified, µ, δ, and κ, and their preferred endogenous ligands are β-endorphin, enkephalin, and dynorphin, respectively. 17 While µ receptors are the main mediators of the analgesia and side effects of opioids, the δ and κ receptors also mediate analgesia.18,19
In the GI system, the µ receptors are expressed chiefly in the submucosal and myenteric plexuses, and the δ receptors are present mainly in the myenteric plexus.20,21 These receptors have been identified in rodent and human GI tract, although their distributions in the GI tract differ among different species. 22 These receptors activate potassium channels causing membrane hyperpolarization and inhibition of calcium channels leading to decreased neurotransmitter release. 23 These result in decreased gastric emptying, impaired internal anal sphincter relaxation in response to rectal distention, 24 increased amplitude of nonpropulsive segmental contractions, and decreased gastric, pancreatic, and intestinal fluid secretions with increased fluid absorption in the intestines as shown predominantly in preclinical studies. However, codeine and morphine have been shown to slow gastric25,26 and colonic transit25–28 in healthy human volunteers.
Two studies have investigated the effects of other opioids on GI function and GI side effects using equi-analgesic doses of tapentadol, a combined opioid agonist and norepinephrine reuptake inhibitor, and oxycodone in healthy volunteers29,30 Marks et al. used electromagnetic capsules to measure transit, as well as tapentadol (50 mg), oxycodone (10 mg), or placebo tablets administered twice daily for 14 days. In this study, while tapentadol motility parameters and side effects were on par with those of placebo, oxycodone showed prolonged whole gut transit and rectosigmoid transit times compared to tapentadol. A separate study assessed tapentadol (75 mg), oxycodone (5 mg), or placebo each three times daily over 2 days and measured gastric emptying, small bowel (SBT measured as colonic filling at 6 h) and colonic transit over 48 h by validated scintigraphy. The latter study showed prolongation of gastric emptying by both tapentadol and oxycodone, but no difference in colonic transit with either drug relative to placebo. 30 These data suggest that longer duration of administration of exogenous opioids may be necessary to document effects on colonic transit in humans, and particularly to differentiate effects of tapentadol and oxycodone.
A plethora of GI symptoms including nausea, vomiting, anorexia, impaired digestion, abdominal spasms, and constipation may result from effects of activation of opioid receptors.31–33 The κ-opioid receptors are involved in analgesia through their action at the supraspinal, spinal, and peripheral levels. 34 Other effects also include increased diuresis and sedation. They are mainly located in the myenteric plexus and afferent neurons and can cause delayed bowel transit and visceral antinociception. κ-opioid agonists have a lower addiction potential compared to µ opioid agonists and they may be used to relieve some of the µ-opioid-induced hyperalgesia.9,35
Interestingly, while patients on opioids characteristically develop tolerance to therapeutic and adverse effects of opioids, 36 the colon appears to be resistant to tolerance. Tolerance is thought to result from β arrestin-2 related receptor internalization and desensitization but, in the colon, β arrestin-2 signaling does not seem to lead to receptor internalization and subsequent tolerance as suggested in preclinical studies.23,37,38
Opioid-induced constipation (OIC)
The Rome IV criteria for OIC are shown in Table 1 and are based on consensus recommendations. 39 Essentially, the diagnosis is constipation after starting opioid therapy.15,40 Up to 57% of patients on long-term opioids for CNCP report constipation as a side effect of opioids.41,42 OIC may occur at any time after initiation of therapy 16 at any opioid dosage 43 and despite use of laxatives which are considered the first-line therapy for OIC. 44 In 322 patients, taking daily oral opioids and laxatives, up to 81% reported OIC which negatively affected their quality of life (QOL). 45 A consensus panel has proposed the Bristol Stool Form Scale (BSFS), the Bowel Function Index (BFI), and Patient Assessment of Constipation Quality of Life (PAC-QOL) as outcome measures in OIC. 40 The BFI encompasses three variables: ease of defecation, feeling of incomplete evacuation, and personal assessment of constipation. The BFI assessment tool is useful to guide therapy in OIC: a BFI score ⩾ 30 on a scale of 0–100 appraised 1 week after starting opioids is an indication that prophylactic treatment with increased water and fiber intake and laxatives could be insufficient and that the patient might benefit from the addition of other medications for OIC.10,39,46,47
Rome IV criteria for opioid-induced constipation.

Available medications approved for OIC in CNCP differ between countries. Currently available approved treatment options for OIC in CNCP in the United States include lubiprostone, a type 2 chloride channel activator; combination of oxycodone (a semi-synthetic opioid agonist) with naloxone (a nonselective opioid antagonist with both central and peripheral actions); and naloxegol, methylnaltrexone, and naldemedine, which are all peripherally acting µ-opioid receptor antagonists (PAMORA).10,15 Naldemedine [Symproic® (Japan, USA); Rizmoic® (EU)] is a semi-synthetic opiate receptor antagonist approved in 2017 in the United States for the treatment of OIC in patients with CNCP. It is available as tablets of 0.2 mg. 48
Naldemedine
Structure and preclinical pharmacology
Naldemedine (C32-H34-N4-O6) is a PAMORA. It is an amide derivative of naltrexone, but is a larger, more polar molecule and exhibits increased binding to p-glycoprotein (P-gp). Indeed, the addition of a (2-(3-phenyl-1,2,4-oxadiazol-5-yl)propan-2-yl)acetamide to the 7-position and increased number of hydrogen bonds render the molecule more polar and thus hamper its ability to cross the blood brain barrier (BBB; Figure 1), as shown in 14C-labeled naldemedine distribution studies in animals. Another mechanism contributing to the reduced distribution of naldemedine in the brain is mediated by the P-gp efflux pump. 46

Naldemedine is an amide derivative of naltrexone, with a polar side chain (circled).
Naldemedine binds with high affinity to the three main types of opioid receptors; however, its main therapeutic effects are mediated through blocking µ receptors in the enteric nervous system. The lack of penetration of naldemedine into the central nervous system is key to preventing interference of PAMORAs with the analgesic effects of opioids. However, there would still be the potential role of naldemedine in the treatment of opioid-induced nausea and vomiting since the chemoreceptor trigger zone is outside the BBB.49–51
Pharmacokinetics in humans
After oral administration, peak concentrations (Cmax) of naldemedine were achieved within 0.5–3.0 h (Tmax) post-dose; peak concentrations occur after 0.75 h (median) in a fasted state. 52 A high-fat meal delayed Tmax to 2.5 h and decreased Cmax by approximately 35% without any appreciable change in overall absorption (as indicated by the area under the curve). 53 Area under the plasma concentration–time curve 54 and Cmax were dose dependent and were almost dose proportional with minimal accumulation following multiple daily doses. 55 Naldemedine is 93–94% bound to plasma proteins with a mean apparent volume of distribution of 155 L. 56 Naldemedine is metabolized primarily by hepatic CYP3A to nor-naldemedine and, secondarily, by UDP-glucuronosyltransferase 1A3 to naldemedine 3-G. 56 Both metabolites are less potent antagonists compared to naldemedine. Naldemedine is excreted in the urine and feces and has a terminal elimination half-life of approximately 11 h. 52
Efficacy
Clinical trials
A summary of all the clinical trials of naldemedine for CNCP is discussed in the following and in Table 2.52,55,57–60
Summary of clinical trials of naldemedine in CNCP.

∝ , proportional; ↑ , increase; ∆, change; Abd., abdominal; ADME: absorption, distribution, metabolism and excretion; AE, adverse event; BL, baseline; BM, bowel movement; CNDP, chronic noncancer pain; CSBM, complete spontaneous bowel movement; DB, double-blind; freq., frequency; GI, gastrointestinal; LSM, least square means; MAD, multiple ascending dose; MC, multicenter; PC, placebo-controlled; PG, parallel-group; PK, pharmacokinetics; QOL, quality of life; RCT, randomized controlled trial; SAD, single ascending dose; SBM, spontaneous bowel movement; SC, single-center; TEAE, treatment emergent adverse event; w/o, without.
Numbers denoting sex do not add up to total number randomized due to subject exclusion most commonly due to enrollment at two different sites.
Phase I trials. Two randomized, double-blinded, placebo-controlled phase I trials were conducted in healthy male Japanese volunteers at a single center. 55 The first trial was a single ascending dose 61 study with 56 volunteers (0.1–100 mg of naldemedine or placebo on day one) and the second was a multiple ascending dose study of 3–30 mg of naldemedine or placebo for 10 days in 36 volunteers, both randomized in a 3:1 ratio to receive naldemedine or placebo. The drug was rapidly absorbed and was well tolerated with no serious adverse events or withdrawals in both studies even at doses up to 500 times the therapeutic dose of 0.2 mg.
Subjects with any renal impairment or mild or moderate hepatic impairment had pharmacokinetics and adverse events consistent with those in healthy volunteers, and therefore, dose adjustment is unnecessary in such patients. 51
Phase II trials. Webster et al. 57 conducted a phase IIa ascending dose trial in 72 patients to evaluate the safety and efficacy of a single oral dose of naldemedine in patients with CNCP and opioid-induced bowel dysfunction (⩽5 spontaneous bowel movements (SBMs) per 2-week period). Nine patients were randomized to each naldemedine dose (0.01, 0.03, 0.1, 0.3, 1.0, and 3.0 mg) and 18 to placebo. Treatment emergent adverse events (TEAEs) were reported more frequently with naldemedine (81.5%) compared to placebo (50.0%). Abdominal pain was the most frequently reported adverse event, and prevalence increased with increased dose (placebo, 16.7%; any naldemedine dose, 46.3%: 0.01 mg, 22.2%; 0.03 mg, 11.1%; 0.3 mg, 66.7%; and 3.0 mg, 100.0%). Severe TEAEs included one drug withdrawal syndrome in a patient on 1.0 mg of naldemedine and six patients on 3.0 mg of naldemedine reporting abdominal pain, severe diarrhea in three, nausea/vomiting in two, and chills in one. Significant increase in number of SBMs from baseline to 24 h was seen with the naldemedine 0.3, 1.0, and 3.0 mg doses compared to placebo. The study showed that a single dose of naldemedine was well tolerated and the 0.3 mg dose had the best benefit-risk profile.
In a phase IIb, multicenter, 4-week trial, 244 subjects with CNCP and OIC were randomized to 0.1, 0.2, or 0.4 mg of naldemedine or placebo. Naldemedine 0.2 and 0.4 mg (but not 0.1 mg) significantly increased weekly SBM frequency compared to placebo relative to baseline in each week of the 4-week study period. 58 The weekly frequency of complete spontaneous bowel movements (CSBM; defined as a SBM with the sense of complete evacuation) was also significantly increased with 0.2 and 0.4 mg of naldemedine compared to placebo. The proportion of SBM responders, defined as patients with ⩾3 SBMs/week and an increase of ⩾1 SBM/week from baseline over the last 2 weeks of treatment, was significantly higher with 0.2 and 0.4 mg naldemedine, but not with 0.1 mg. There were no significant differences between 0.2 and 0.4 mg of naldemedine. Adverse events increased with higher naldemedine dose. The most frequently reported adverse events were GI related including abdominal pain, diarrhea, flatulence, and nausea. Based on these results, the 0.2 mg dose of naldemedine was adjudicated as the optimal dose with the best benefit and risk profile.
Phase III trials. In two identical, phase III, multicenter, 12-week trials conducted in multiple countries, 547 (COMPOSE-1) and 553 (COMPOSE-2) patients with CNCP and OIC not on laxatives were randomized to 0.2 mg naldemedine or placebo. 59
The primary efficacy endpoint was proportion of responders defined as at least three SBMs per week and an increase from baseline of ⩾1 SBM per week for at least 9 out of the 12 treatment weeks and at least 3 of the 4 last weeks of the 12-week treatment period. The proportion of responders was significantly higher in both studies with naldemedine compared to placebo: in COMPOSE-1, 130 (47.6%) of 273 patients in the naldemedine arm compared to 94 (34.6%) of 272 in the placebo arm (p = 0.002); and in COMPOSE-2, 145 (52.5%) of 276 compared to 92 (33.6%) of 274, (p < 0.0001). The proportion of responders was similar for total daily 30–100 mg and >100 mg oral morphine sulfate equivalents. Significantly greater increases were observed with naldemedine compared to placebo: mean change in frequency of SBMs per week from baseline in the first week of treatment and the last 2 weeks of the 12-week treatment period; and mean frequency per week of SBMs without straining from baseline to the last 2 weeks of the 12-week treatment period. Additional benefits of naldemedine compared to placebo were significant improvements from baseline in the mean frequency of SBMs, CSBMs, and SBMs without straining per week during each week of the 12-week treatment period, as well as shorter median times to first SBM and to first CSBM after initial dose. TEAEs were similar across both studies with higher prevalence (chiefly diarrhea and abdominal pain) with naldemedine compared to placebo.
In a double-blind, 52-week, phase III study (COMPOSE-3) conducted on patients with OIC and CNCP, 1246 patients were randomized to 0.2 mg naldemedine or placebo to evaluate the long-term safety of naldemedine. 60 The proportion of patients who reported TEAEs over the year were similar (naldemedine, 68.4%; placebo, 72.1%), but the naldemedine group had more diarrhea (11% versus 5.3% placebo), abdominal pain (8.2% versus 3.1% placebo), and vomiting (6.0% versus 3.1% placebo). Most of these events were either mild or moderate in severity, and frequency of treatment discontinuation due to GI related TEAEs was low in both groups (naldemedine, 3.7%; placebo, 1.6%). Incidences of serious adverse events, serious TEAEs, and major cardiac events were low (<2%) and comparable between groups. Proportions of opioid withdrawal were similar (naldemedine, 1.8%; placebo, 1.1%).
In addition to the safety data over the 52 weeks, frequency of bowel movements (BMs) increased significantly and remained higher throughout the 52-week treatment period in the naldemedine group (nominal p ⩽ 0.0001; Figure 2). A significant and persistent improvement was observed with naldemedine in mean overall scores compared to placebo for Patient Assessment of Constipation-Symptoms (PAC-SYM) and PAC-QOL questionnaires (p ⩽ 0.0001).

Changes from baseline (BL) in frequency of bowel movements (BM).
Naldemedine 0.2 mg improved patient-reported outcomes in the COMPOSE phase III trials59,60,62 specifically patients’ perception of meaningful change in bowel function and health-related QOL, overall PAC-SYM and PAC-QOL scores. These improvements were achieved within 2 weeks of treatment commencement and persisted for the entire study (Figures 3 and 4).
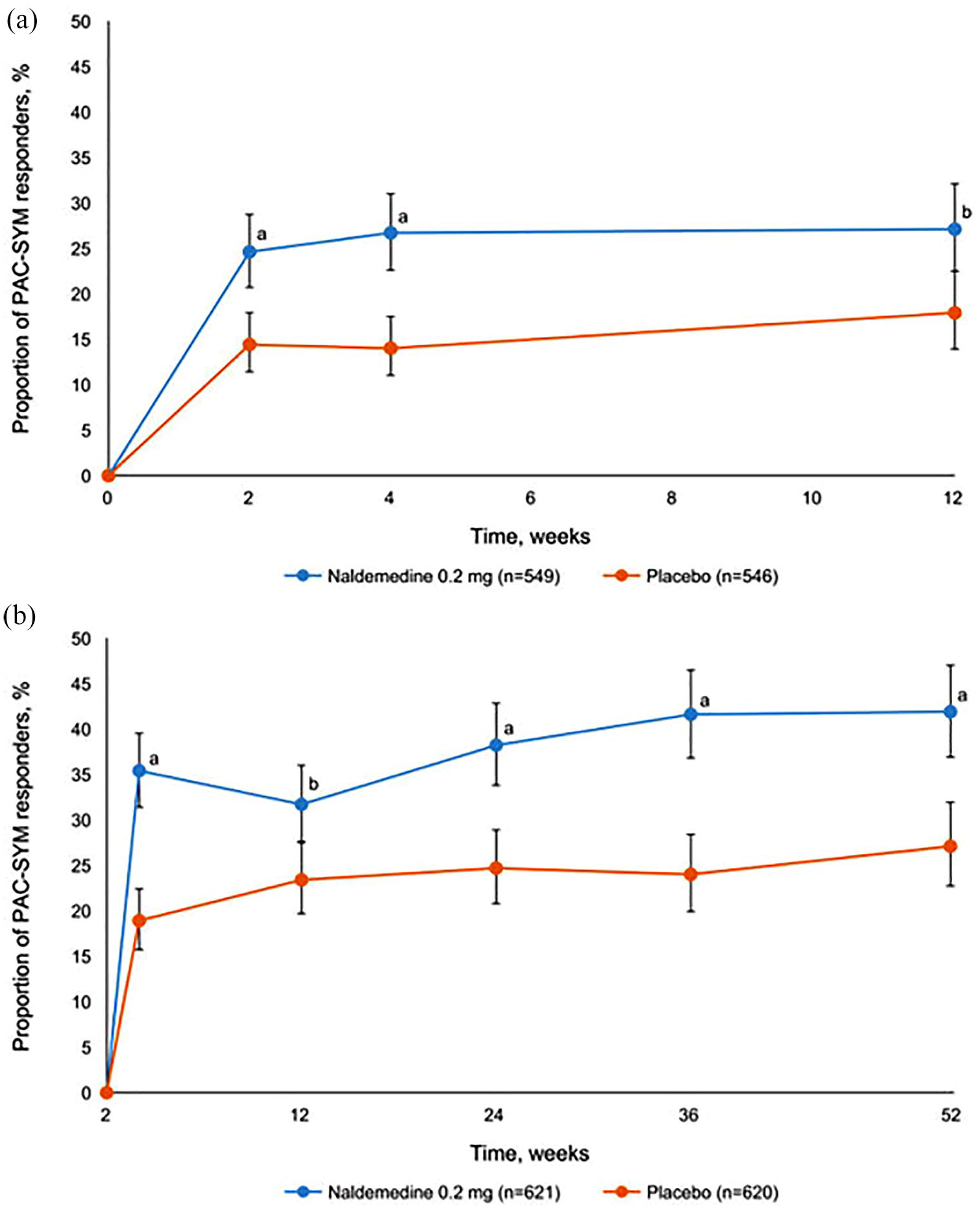
Proportion of patients achieving ⩾1.5 point decreased in patient assessment of constipation symptoms (PAC-SYM) from (a) integrated COMPOSE-1 and COMPOSE-2 data; (b) COMPOSE-3 data. Data show mean and 95% CI; (a) p < 0.001; (b) p < 0.005.

Proportion of patients achieving ⩾1.5 point decreased in patient assessment of constipation quality of life (PAC-QOL) from (a) integrated COMPOSE-1 and COMPOSE-2 data; (b) COMPOSE-3 data. Data show mean and 95% CI; (a) p < 0.001; (b) <0.005.
Wild et al. 63 analyzed the integrated data of the three COMPOSE trials in patients aged ⩾65 years old in the first 12 weeks and showed comparable overall and GI TEAEs in the overall population (47.1% and 21.8%, respectively) and in patients ⩾65 years old (45.9% and 20.2%, respectively). Opioid withdrawal was reported in 1.0% in the overall population and in 1.1% of patients ⩾65 years old on naldemedine compared to 0% and 0.6%, respectively, in the placebo group. In patients over 65 years old, there were also more responders to naldemedine (51.8%) compared to placebo (37.6%), consistent with that of the overall population.
Efficacy and safety of naldemedine has similarly been documented in patients with mild and moderate renal insufficiency, 64 consistent with effects and TEAEs and GI adverse events in the overall population.
Meta-analysis
Esmadi et al. 65 preformed a meta-analysis of naldemedine 0.2 mg in six articles, which met their inclusion criteria, comparing the efficacy of naldemedine to no naldemedine or to placebo in patients with OIC. The proportion of responders was significantly greater compared to placebo (56.4%, naldemedine versus 34.7%, placebo; p < 0.00001) and change in SBM and CSBM frequency was greater (p < 0.00001) in the naldemedine group compared to placebo, with no difference in TEAEs (mean odds ratio of 1.18 (95% CI: 0.89 to 1.55, p = 0.25)). Diarrhea was the most common of the identified adverse effects.
Adverse effects
As detailed in the discussion of all the trials, naldemedine is generally well tolerated, with overall TEAE rates similar to placebo in the phase III COMPOSE trials.59,60 However, GI-related TEAEs were more frequent in the naldemedine group, chiefly diarrhea, abdominal pain, and vomiting, with no impact on withdrawals because of TEAEs. The TEAEs reflect blocking μ-opioid receptors in the GI tract. Major adverse cardiac events are rare. Two patients developed a hypersensitivity reaction after a single dose of naldemedine, namely bronchospasm and rash. 66
Comparison of naldemedine with other pharmacological therapies
There are no randomized, controlled trials that compare head-to-head pharmacological therapies used for treatment of OIC. Relative efficacies of these pharmacological agents in the treatment of OIC can be assessed through systematic reviews and meta-analyses (SRMA) or network meta-analysis.
One SRMA identified 27 randomized, controlled trials enrolling 5390 patients receiving medical treatment and 3491 receiving placebo (total 8881). The study concluded that the μ-opioid receptor antagonists, lubiprostone, and prucalopride were superior to placebo for the treatment of OIC, with a relative risk of failure to respond to therapy of 0.70 (95% CI: 0.64–0.75) and an overall number needed to treat of 5 (95% CI: 4–7). 67
Another systematic review and network meta-analysis by Luthra et al. 68 identified 27 articles including 9149 patients comparing different pharmacological therapies to placebo. Twenty-two articles were related to µ-opioid receptor antagonists, notably naloxone, methylnaltrexone, naldemedine, alvimopan, naloxegol, and bevenopran. Three studies were related to the secretogogue and lubiprostone, and two studies were related to the prokinetic, prucalopride. The µ-opioid receptor antagonists, naloxone, naldemedine, alvimopan, and methylnaltrexone, as well as the prokinetic, prucalopride, were more efficacious than placebo for the treatment of OIC. Among these medications, only naloxone, naldemedine, and methylnaltrexone are FDA approved for the treatment of OIC in CNCP. With the primary endpoint defined as failure to achieve an average of ⩾3 BMs per week with an increase of ⩾1 BM per week above baseline or an average of ⩾3 BMs per week, naloxone ranked first [RR = 0.65 (0.52; 0.80), p = 0.84] followed by naldemedine [RR = 0.67 (0.59; 0.77), p = 0.80]. When the primary endpoint considered was failure to achieve an average of ⩾3 BMs per week with an increase of ⩾1 BM per week, naldemedine ranked first [RR = 0.66 (0.56; 0.77), p = 0.91] followed by alvimopan [RR = 0.74 (0.57; 0.94), p = 0.71]; the latter is not approved for OIC. When assessing the side-effect profile of the various medications, naloxone was considered the safest. 68
A third SRMA identified 35 trials enrolling 13,566 patients. Seven studies were related to methylnaltrexone, 7 to naldemedine, 6 to naloxone, 4 to alvimopan, 4 to naloxegol, 1 to axelopran, 1 to linaclotide, 1 to prucalopride, and 4 to lubiprostone. Endpoints considered included continuous variables such as bowel function index, number of SBMs, stool consistency based on the Bristol Stool Form Scale as well as FDA endpoints, notably ⩾3 BMs per week with an increase of ⩾1 BM per week and ⩾3 CSBMs per week with an increase of ⩾1 CSBM per week. Results for the FDA endpoints are shown in Figure 5. The study concluded that the therapeutic response in OIC was best achieved with the approved doses of the PAMORAs, methylnaltrexone, naloxegol, and naldemedine which had good benefit-risk profile. 69

Forest plot of individual medications associated with achieving the FDA endpoint for spontaneous bowel movements (i.e. at least three spontaneous bowel movements per week in 9 of 12 weeks’ treatment and increase of one spontaneous bowel movements per week over baseline) in treatment of opioid-induced constipation.
While these three SRMAs differ slightly in their results, they all concluded that naldemedine is more efficacious than placebo in the treatment of OIC. Nevertheless, it is important to recognize weaknesses or potential pitfalls in appraising the evidence from these SRMAs. In the SRMA by Nee et al., 67 an important limitation was the significant heterogeneity between the 27 included studies; the heterogeneity was attributed to the varying baseline opioid use between participants, the inclusion of multiple agents, and different subject populations (cancer and non-cancer-related pain). In the SRMA by Luthra et al., 68 only 11 out of the 27 studies were at low risk of bias and most patients were evaluated in secondary or tertiary care centers, impacting its generalizability; however, there was moderate global statistical heterogeneity in some of the analysis, and the funnel plot for the primary outcome was symmetrical excluding publication bias. In the SRMA by Vijayvargiya et al., there was the largest number of participants, all included trials had low risk of bias and most of the studies met GRADE criteria for the trial endpoints. However, a limitation of Vijayvargiya’s SRMA was that patients in different studies were on different opioid doses which could affect the homogeneity of baseline characteristics. 69
Conclusion
OIC is frequently seen in patients on chronic opioid therapy for CNCP. While patients develop tolerance to the analgesic properties and most of the side effects of opioids, OIC is a notable exception, possibly due to dysregulation at the level of β arrestin-2 signaling. Opioid discontinuation is often attributed to OIC which leads to suboptimal pain control and decreased quality of life. While the first-line treatment for OIC remains increased water and fiber intake as well as laxatives, a BFI score ⩾30 points is considered a good indicator for treatment escalation. The currently approved drugs in the United States for OIC in CNCP are lubiprostone, naloxone, and the PAMORAs naloxegol, methylnaltrexone, and naldemedine. Naldemedine is an amide derivative of naltrexone, available in 0.2 mg tablets, and exerts its effects through blocking µ-receptors in the enteric nervous system. It has been shown to significantly increase the frequency of bowel movements, has good efficacy compared to other approved OIC treatments, and has a safe side effect profile.
Footnotes
Acknowledgements
The authors thank Cindy Stanislav for excellent secretarial assistance.
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
