Abstract
Introduction:
Anti-tumor necrosis factor (TNF) dose intensification represents an effective method of overcoming secondary loss of response (LOR); however, a subset of patients may not respond (tertiary non-response), or fail to demonstrate durable response (tertiary LOR) to intensified dosing. This systematic review and meta-analysis aimed to evaluate these outcomes to determine the clinical effectiveness of empiric dose intensification in Crohn’s disease.
Methods:
Multiple databases including MEDLINE and EMBASE were interrogated to identify studies that reported outcomes following anti-TNF dose intensification to address secondary LOR in Crohn’s disease. Studies that used anti-TNF levels as the primary basis for dose intensification were excluded. Studies that reported (1) tertiary response and tertiary non-response within 6 months or (2) tertiary response and tertiary LOR beyond 6 months, were pooled using a random effects model with risk ratio (RR) derived, quantifying the effect of each comparison.
Results:
Twenty-six studies reported outcomes following anti-TNF dose intensification to address secondary LOR. Short-term response within 12 weeks of any dose-intensification strategy was 33–90%, while sustained response (⩾48 weeks) was achieved in 25–85%. Tertiary non-response occurred in up to 45% of intensified patients within 6 months of anti-TNF dose intensification, while tertiary LOR beyond 6 months occurred in up to 64% of patients. Tertiary response was more likely than tertiary non-response within 6 months (RR 2.58, 95% CI (1.76, 3.79), I2 = 82%, 12 studies), while sustained response beyond 6 months compared to tertiary LOR (RR 1.10 (0.75, 1.61) I2 = 85%, 7 studies) was less convincing.
Conclusion:
Although anti-TNF dose intensification is clinically effective in patients with Crohn’s disease, particularly within the first 6 months, a proportion of patients will fail to demonstrate short-term and/or sustained clinical response. Hence, clinical reassessment following anti-TNF dose intensification, particularly beyond 6 months, remains important to differentiate between effective and ineffective dose-intensification strategies.
Introduction
Despite the efficacy of anti-tumor necrosis factor (TNF) agents in Crohn’s disease, up to 30% of patients exhibit primary non-response, with a further 46% of anti-TNF responders demonstrating features of secondary loss of response (LOR) within 12 months of anti-TNF initiation.1 –3 Several studies have demonstrated the clinical effectiveness of anti-TNF dose intensification to overcome LOR using dosing strategies including one or more of: shortening the interval between anti-TNF doses; increasing the baseline anti-TNF dose while maintaining the dosing interval, and; anti-TNF re-induction.4 –7
Two recent systematic reviews, one of which also undertook a meta-analysis, evaluated the clinical effectiveness of anti-TNF dose intensification to address LOR across Crohn’s disease and ulcerative colitis.8,9 While comprehensive in their approach, neither review distinguished between anti-TNF dose intensification undertaken to address secondary LOR and anti-TNF dose intensification undertaken to address pooled primary non-response and secondary LOR. It is, however, important to differentiate between both of these entities, given the disparate clinical effectiveness of anti-TNF dose intensification between primary responders and primary non-responders. Thus, targeted evaluation of the clinical effectiveness of anti-TNF dose intensification to address secondary LOR is needed.
Moreover, amid the growing use of intensified anti-TNF dosing to address LOR in Crohn’s disease, it remains important that clinicians recognise that a subset of patients may not respond, or fail to demonstrate durable response to intensified anti-TNF dosing. These outcomes, notionally termed, ‘tertiary non-response’ and ‘tertiary LOR’, have not previously been well described, nor compared with tertiary response (Figure 1). Hence, in addition to evaluating the clinical effectiveness of anti-TNF dose intensification to address secondary LOR, this review will evaluate outcomes such as ‘tertiary non-response’ and ‘tertiary LOR’ reflective of ineffective dose intensification. Factors associated with these outcomes will also be described.

Defining response, non-response, and loss of response to anti-TNF therapy in inflammatory bowel disease.
Methods
This systematic review and meta-analysis was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines; however, it was not prospectively registered on PROSPERO. 10
Selection criteria
Studies eligible for inclusion in this systematic review included clinical trials (randomised/non-randomised) and cohort studies (retrospective/prospective) that investigated: (a) Population: adult patients with Crohn’s disease who demonstrated primary response to adalimumab, certolizumab, or infliximab; (b) Intervention: empiric adalimumab, certolizumab, or infliximab dose intensification to address secondary LOR following standard induction and maintenance dosing; (c) Outcome: proportion demonstrating clinical response/remission, non-response and LOR following anti-TNF dose intensification. Studies that did not clearly define criteria for secondary LOR prior to dose intensification and/or clinical response/remission following anti-TNF dose intensification, were excluded. Similarly, studies that reported outcomes following anti-TNF dose intensification to address combined primary non-response and secondary LOR were reported separately, unless outcomes specific to secondary LOR were reported or could be imputed.
Definitions
Anti-TNF dose intensification was defined as empiric dose intensification based on clinical symptoms as judged by the treating clinician, with or without objective disease assessment. Studies that used serum anti-TNF trough levels in the absence of clinical symptoms as the primary basis for directing therapeutic intervention following secondary LOR, were excluded. This decision was made to minimise heterogeneity in baseline disease activity and its impact on subsequent assessments of tertiary response, non-response and LOR following anti-TNF dose intensification.
Anti-TNF dose intensification was defined as one or more of: shortening the interval between anti-TNF doses; increasing the baseline anti-TNF dose while maintaining the dosing interval, and; anti-TNF re-induction. Tertiary response was defined as the number of patients demonstrating clinical response following anti-TNF dose intensification undertaken to address secondary LOR relative to all patients who underwent anti-TNF dose intensification. Tertiary non-response was defined as lack of response occurring within 6 months of anti-TNF dose intensification. Tertiary LOR was defined as LOR occurring more than 6 months following anti-TNF dose intensification. Anti-TNF dose intensification following secondary LOR was defined as empiric dose intensification following partial or complete response to standard induction dosing. Studies that included ‘non-response’ in their criteria for anti-TNF dose intensification were deemed to reflect dose intensification undertaken to address primary non-response and were thus reported separately.
Search strategy
A search of the medical literature published in English was conducted, using Ovid MEDLINE, EMBASE, Cochrane Central Register of Controlled Trials and Cochrane Database of Systematic Reviews through to 30 July 2021. Search algorithms included a combination of terms reflecting the disease of interest (Crohn(s) disease) in combination with treatment (anti-TNF, anti-TNF, TNF-alpha, adalimumab, certolizumab pegol, or infliximab), anti-TNF dosing regimen (intensif*/escalat*) and outcome (response/ LOR) of interest without restriction. Two authors (AS/RG) independently reviewed titles and abstracts of studies identified by the search and excluded those that were clearly unrelated on the basis of pre-specified inclusion and exclusion criteria. This process was undertaken with the assistance of the Covidence software programme. 11 Full-text of selected articles was appraised to determine suitability for inclusion, with conflicts in study selection resolved by consensus and referring back to the original article, in consultation with a senior investigator (PDC). The reference lists of relevant studies were manually searched to identify additional publications of relevance.
Data synthesis and statistical analysis
Systematic review
The primary outcome of interest was tertiary response, that is, the proportion of patients demonstrating clinical response following anti-TNF dose intensification to address secondary LOR. Tertiary non-response, tertiary LOR and clinical remission following anti-TNF dose intensification were also assessed. Factors associated with these outcomes were also evaluated. Where studies reported outcomes following several dose-intensification strategies, outcomes were described collectively, based on anti-TNF, and per dose-intensification strategy, where possible. Studies in which anti-TNF dose intensification was undertaken to address both primary non-response and secondary LOR were reported separately.
Meta-analysis
The purpose of the meta-analysis was to evaluate the clinical effectiveness (tertiary response) of anti-TNF dose intensification against measures of clinical ineffectiveness (tertiary non-response /tertiary LOR). Hence studies that reported (1) tertiary response and tertiary non-response within 6 months or (2) tertiary response and tertiary LOR beyond 6 months, were included in the meta-analysis.
Data were combined to provide risk ratio (RR) with 95% confidence intervals (CIs) to summarise the effect of each comparison using a statistical significance threshold of p value < 0.05. Study heterogeneity was analysed using the I2 statistic: with heterogeneity thresholds as follows: not important (I2 < 40%), moderate (40–75%), and considerable (>75%). A Begg’s funnel plot was used to estimate the possibility of publication bias. 12 Sensitivity analyses were performed for each meta-analysis subgroup by excluding studies that were identified as potentially introducing a critical risk of bias that could likely modify the outcome. Data were analysed using Review Manager (version 5.4).
Data extraction
The following characteristics were extracted from each eligible study: first author name, year of publication, country where study was undertaken, study design, number of patient who were dose intensified, anti-TNF agent, dose-intensification strategy, duration of anti-TNF therapy prior to dose intensification, proportion on immunomodulator co-therapy at dose intensification, proportion with prior biologic failure at baseline, and proportion with perianal disease at baseline. Key study definitions including the basis for secondary LOR prior to anti-TNF dose intensification, basis for clinical response/remission following anti-TNF dose intensification, and the basis for clinical non-response/LOR following dose intensification were also documented. Finally, outcome measures pertaining to the proportion of patients demonstrating clinical response/remission and clinical non-response/LOR following dose intensification along with corresponding timepoints were recorded.
Risk of bias assessment
The methodological quality of all studies was assessed by two authors (AS and RG) with agreement reached by consensus if discrepancies arose. Each randomised controlled trial (RCT) was assessed using the Cochrane Collaboration tool, evaluating bias across the several domains including selection bias, reporting bias, performance bias, detection bias, attrition bias, and other bias. 13 Similarly, observational studies were evaluated using the Newcastle–Ottawa Scale (NOS), with the quality of each study evaluated independently. Representativeness of the exposed cohorts, ascertainment of exposure, demonstration that the outcome of interest was not present at start of study, assessment of the outcome, and adequacy of the follow-up were assessed for each included study. Two criteria of the NOS, 14 namely selection of the non-exposed cohort and comparability of the cohorts, were not used because cohorts not exposed to intensified anti-TNF therapy were not included.
Results
Search results
Following removal of duplicate records, 1024 articles were identified for assessment (Figure 2). Records deemed irrelevant or not suitable based on predetermined inclusion criteria were excluded, leaving 148 articles for detailed evaluation. We identified 34 eligible full-text articles of adult patients with Crohn’s disease that underwent anti-TNF dose intensification to address secondary LOR, comprising 3 RCTs, 3 post hoc analyses of RCTs, 7 prospective observational studies, and 21 retrospective studies (Table 1).4–7,15–45 Thirteen studies reported outcomes following infliximab dose intensification, 15 studies reported outcomes following adalimumab dose intensification, and six following both adalimumab and infliximab dose intensification. No studies evaluating certolizumab dose intensification met eligibility criteria.
Characteristics of all studies included in the systematic review.

NR, not reported; immunomodulator: thiopurine or methotrexate.
Across entire study, not just intensified subgroup.

Study selection flowchart as at 30 July 2021.
Assessment of bias
The risk of bias assessments for RCTs and observational studies are presented in Supplementary Tables 1 and 2, respectively. Of three included RCTs, the study by Watanabe et al. 37 was identified as potentially subject to selection bias owing to insufficient description of the randomisation and allocation concealment processes, while the studies by Steenholdt et al.15,29 were subject to detection bias as outcome assessment was not blinded, thus potentiating a higher risk of bias. Most observational studies exhibited a low to moderate risk of bias when applying the modified NOS scale (out of 6). Notably, observational studies were largely retrospective in design, with study outcomes generally present at study outset, reflecting an obvious source of bias.
Systematic review
Defining LOR prior to anti-TNF dose intensification
Clinical disease assessment
The definition of LOR prior to anti-TNF dose intensification was heterogeneous (Table 2). Several studies incorporated definitions based on validated clinical indices including the Crohn’s Disease Activity Index (CDAI) and Harvey–Bradshaw Index (HBI); however, disease activity thresholds associated with clinical LOR prior to dose intensification were variable.7,15–17,20,22,23,25,27,29,35–39 Eleven studies incorporated assessments of CDAI into the definition of LOR, with four studies documenting absolute CDAI thresholds.15–17,20,22,25,27,29,35,37,38 Four studies incorporated assessments of HBI into the definition of LOR, each documenting one or both of an absolute HBI threshold and/or change from baseline HBI, reflective of clinical LOR.7,23,36,39 The remaining studies ostensibly relied on physician-determined clinical deterioration, or definitions of symptomatic disease that did not incorporate validated clinical indices, to define LOR prior to anti-TNF dose intensification.
Tertiary response and remission following anti-TNF dose intensification.
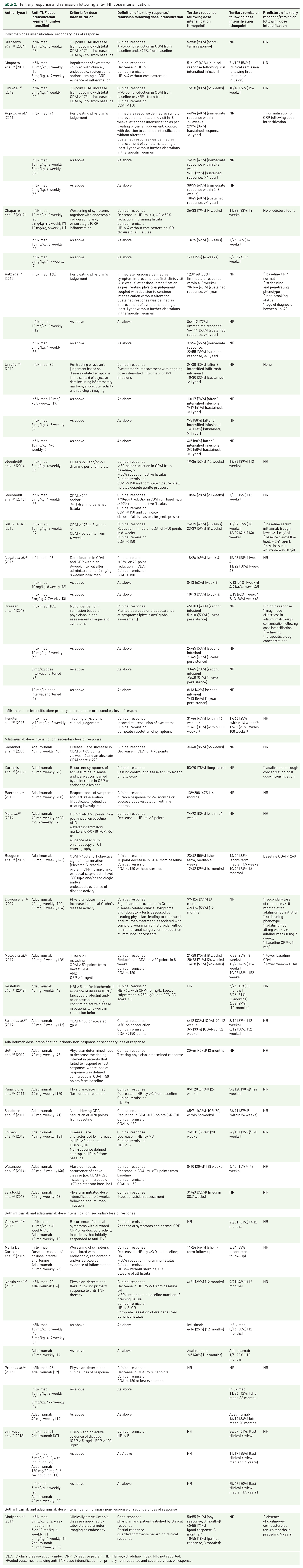
CDAI, Crohn’s disease activity index; CRP, C-reactive protein; HBI, Harvey–Bradshaw Index; NR, not reported.
Pooled outcomes following anti-TNF dose intensification for primary non-response and secondary loss of response.
Objective disease assessment
Six studies required both clinically active disease based on clinical indices (CDAI or HBI) and objective disease activity defined as one or more of elevated C-reactive protein (CRP), faecal calprotectin, evidence of radiologic, and/or endoscopic inflammation.7,17,20,23,38,39 An additional eight studies required physician-determined symptomatic disease plus objectively assessed disease activity prior to anti-TNF dose intensification.6,21,26,28,32,41,42,45
Indication for anti-TNF dose intensification
Outcomes across eight studies did not differentiate between anti-TNF dose intensification undertaken to address primary non-response and secondary LOR.30,33–37,40,41 The remaining 26 studies reported clinical outcomes following anti-TNF dose intensification specifically undertaken to address secondary LOR.
Anti-TNF dose-intensification strategies
Of 34 eligible studies, clinical outcomes following anti-TNF dose intensification were reported across 45 patient cohorts, comprising 23 infliximab cohorts, 17 adalimumab cohorts, and 5 pooled adalimumab/infliximab cohorts. Thirty-seven cohorts reported outcomes following anti-TNF dose intensification to address secondary LOR, while eight cohorts reported pooled outcomes following anti-TNF intensification for primary non-response and/or secondary LOR, and were thus reported separately.
Infliximab dose intensification
Infliximab dose intensification was undertaken exclusively for secondary LOR across 17 of 19 eligible studies, with all but one study including patients who were anti-TNF experienced. Several studies evaluated more than one dose-intensification regimen, including infliximab 5 mg/kg 4–7 weekly (n = 10), infliximab 10 mg/kg 8 weekly or 5 mg/kg 4 weekly (n = 17), high-dose infliximab (⩾10 mg/kg, 4–7 weekly) (n = 5) and infliximab re-induction (n = 2).7,15,17,19,21–30 These regimens were evaluated across 19 studies, 13 of which evaluated outcomes following infliximab dose intensification alone, with the remaining six studies evaluating outcomes across mixed adalimumab and infliximab cohorts.
Clinical outcomes following infliximab dose intensification
Clinical response and remission across all infliximab dose-intensification studies, strategies and timepoints ranged from 25% to 90% and 19% to 58%, respectively. Short- to medium-term clinical response following infliximab dose intensification undertaken to address secondary LOR using any dose-intensification strategy ranged from 40% to 90% at 4–8 weeks to 28–80% at 12–24 weeks; with longer-term response after at least 1 year sustained in 25–83% of patients.4,5,15,16,19–21,25–29,43 The proportion of patients who achieved short-term clinical remission using any infliximab dose-intensification strategy to address secondary LOR at 4–12 weeks was 19–58%, while long-term clinical remission was reported in 41–56%.15,16,20,26–29,43,44 Only one study adopted infliximab dose intensification to address both non-response and secondary LOR, and despite the use of high-dose infliximab intensification (⩾ 10 mg/kg 4–7 weekly), reported short-term and sustained clinical response in 47% and 34% of patients, respectively. 30
Infliximab 10 mg/kg 8 weekly
Eleven studies included patients who underwent infliximab 10 mg/kg 8 weekly dose intensification, of which seven studies reported clinical response and three studies reported clinical remission outcomes specific to this dosing strategy. Seven studies reported short-term clinical response in 52–90% of patients within 12 weeks.5,16,19–21,25,28 Three studies reported sustained clinical response in 41–50% of patients following at least 12 months of intensification.5,19,21 Three studies reported clinical remission in 28–54% of patients within 4–8 weeks of intensification.16,20,28 Two studies reported sustained clinical remission after 40–48 weeks of intensification in 41–44% of patients.16,20
Infliximab 5 mg/kg 4 weekly
Six studies included patients who underwent infliximab 5 mg/kg 4 weekly dose intensification, of which four studies reported clinical response and three studies reported clinical remission outcomes specific to this dose-intensification strategy. Three studies reported clinical response in 28–66% of patients within 20 weeks of intensification.5,15,29 Two studies reported sustained clinical response in 39–83% of patients following at least 12 months of intensification.5,27 Two studies reported clinical remission in 19–39% of patients after 12-weeks of intensification.15,29 Only one study reported sustained clinical remission following 54-weeks of intensification in 56% of patients. 27
Infliximab 5 mg/kg 4–7 weekly
Six studies included patients who underwent infliximab 5 mg/kg 4 weekly dose intensification, of which five studies reported clinical response and two studies reported clinical remission outcomes specific to these dose-intensification strategies. Five studies reported clinical response in 15–88% of patients within 18 weeks of intensification.4,19–21,28 Three studies reported sustained clinical response in 13–51% of patients following at least 12 months of intensification.4,19,21 Two studies reported clinical remission in 57–62% of patients 4 weeks following intensification.20,28 Only one study reported sustained clinical remission following 48 weeks of intensification in 54% of patients. 20
High dose infliximab ⩾10 mg/kg 4–7 weekly
Five studies included patients who underwent high-dose infliximab dose intensification, of which three studies reported clinical response and one study reported clinical remission outcomes specific to this dose-intensification strategy. Three studies reported short-term clinical response in 47–80% of patients after 2–3 intensified infusions, and sustained clinical response in 34–54% of patients following at least 12-months of intensification.19,21,30 One study reported short-term clinical remission within 16-weeks in 25% of patients and sustained clinical remission within 100 weeks in 34% of patients. 30
Factors associated with response and/or remission following infliximab dose-intensification
Several factors have been associated with favourable clinical outcomes following infliximab dose intensification. Katz et al. 5 found that immediate clinical response was associated with a stricturing (OR 4.1, 95% CI (1.8–9.1)) or penetrating (OR 4.1 (1.8–9.1)) phenotype and normal baseline CRP (OR 3.2, (1.2–9.4)) via multivariate analysis. Similarly, Kopylov et al. 4 reported that normalisation of CRP following infliximab dose intensification was associated with immediate clinical response (OR 4.2 (1.2–15.2)), relative to persistent CRP elevation. Katz et al. 5 also documented that sustained response at 1 year was associated with younger age (16–40 years) at disease diagnosis (OR 2.7 (1.1–7.7)), and normal CRP (OR 4.0 (1.5–10.3)) at LOR. The proportion of patients achieving clinical remission at week 40 was found to be higher in patients with serum infliximab trough levels ⩾1 mg/mL, plasma interleukin (IL)-6 levels ⩽2.41 pg/mL and/or serum albumin level ⩾3.8 g/dL at baseline by Suzuki et al. 16 Dreesen et al. also noted that patients who achieved biologic response (defined by reduction in CRP by 50% or CRP ⩽ 5 mg/L from baseline CRP > 5 mg/L) and remission (CRP ⩽ 5 mg/L from baseline CRP > 5mg/L) following infliximab dose intensification were more likely to have higher infliximab trough levels. 19 Moreover, a therapeutic (>3.0 ug/mL) infliximab trough concentration was also positively associated with biologic response. 19
Adalimumab dose intensification
Adalimumab dose intensification was undertaken exclusively for secondary LOR in 14 of 21 studies. Unlike infliximab, the majority (n = 17) of adalimumab studies included patients who were anti-TNF naïve. Adalimumab dose-intensification strategies evaluated included adalimumab 40 mg weekly (n = 17), adalimumab 80 mg 2 weekly (n = 6), and adalimumab re-induction (n = 1).6,7,17,22–24,31–45 These regimens were evaluated in 21 studies, 15 of which evaluated outcomes following adalimumab dose intensification alone, with the remaining six studies evaluating outcomes across mixed adalimumab and infliximab cohorts.
Clinical outcomes following adalimumab dose intensification
Clinical response and remission across all adalimumab dose-intensification studies, strategies, and timepoints ranged from 20% to 85% and 15% to 84%, respectively. Short-term clinical response within 12 weeks of adalimumab dose intensification undertaken to address secondary LOR across all dose-intensification strategies ranged from 33% to 79%, while clinical response within 24 weeks ranged from 29% to 80%, and sustained clinical response following at least 1 year of intensified adalimumab therapy ranged from 33% to 85%.6,17,22–24,31,32,38,43 Short-term clinical remission following adalimumab dose intensification to address secondary LOR was achieved at 8–12 weeks in 16–67% of patients, while medium-term (12–24 weeks) and sustained (⩾52 weeks) clinical remission was reported in 24–43% and 20–84% of patients, respectively.17,22,38,39,43,44
Comparatively, six studies used adalimumab dose intensification to address both non-response and secondary LOR, with overall adalimumab response ranging from 20% to 72%.31,33–37,40 Of these six studies, three reported short-term (⩽24 weeks) response in 58–72% of patients, while three studies reported sustained response (⩾52 weeks) in 20–72% of patients.33–37,40
Adalimumab 40 mg weekly
Seventeen studies included patients who underwent adalimumab 40 mg weekly dose intensification, of which nine studies reported clinical response and six studies reported clinical remission outcomes specific to this dose-intensification strategy. Four studies reported clinical response in 35–71% of patients following 3–6 months of intensification.6,33,34,36 Five studies reported sustained clinical response in 40–85% of patients following at least 12 months of intensification.31,32,35,40,43 Three studies reported clinical remission in 16–35% of patients following 3–6 months of intensification.34,36,39 Three studies reported sustained clinical remission following at least 12 months of intensification in 20–84% of patients.35,43,44
Adalimumab 80 mg 2 weekly
Six studies included patients who underwent adalimumab 80 mg 2 weekly dose intensification, of which four studies reported clinical response and four studies reported clinical remission outcomes specific to this dose-intensification strategy. Three studies reported short- to medium-term clinical response in 39–75% of patients within 6 months of intensification.17,22,38 Three studies also reported sustained clinical response in 20–57% of patients following at least 12 months of intensification.16,17,37 Similarly, three studies reported clinical remission in 24–67% of patients within 6-months of intensification and sustained clinical remission following at least 48-weeks of intensification in 15–50% of patients.17,22,37
Adalimumab 40 mg weekly or adalimumab 80 mg 2 weekly
Two studies reported pooled outcomes following adalimumab 40 mg weekly and 80 mg 2 weekly. Short-term clinical response was achieved in 79–80% of intensified patients between 3 and 6 months, while sustained clinical response at 12 months was achieved in 60%.23,24
Factors associated with response and/or remission following adalimumab dose intensification
Duveau et al. 24 found that adalimumab 40 mg weekly rather than 80 mg 2 weekly (OR 3.6, 95% CI (1.3–10.4)) and CRP ⩽ 5 mg/L at dose intensification (OR 6.6 (1.4–27.5)) were associated with 12 month clinical response to adalimumab dose intensification. Secondary LOR that developed 10 months or more (OR 2.6 (1.0–6.5)) after adalimumab initiation and stricturing disease (OR 4.4 (1.4–14.0)) were also found to be associated with clinical response at 3 months. 24 Motoya et al. 17 also noted that CDAI at baseline and at 4 weeks were each lower in patients that achieved 24-week clinical remission.
Pooled infliximab and adalimumab dose intensification
Clinical outcomes following pooled infliximab and adalimumab dose intensification
Six studies reported outcomes across cohorts that underwent both infliximab and adalimumab dose intensification, including two studies that reported outcomes across infliximab and adalimumab subgroups.7,41–45 The remaining four studies only reported pooled outcomes following infliximab and adalimumab dose intensification, including one study by Ghaly et al. 41 which included patients that underwent dose intensification for primary non-response.7,42,45 Pooled outcomes across these four studies using any infliximab or adalimumab dose-intensification strategy, reported clinical response in 46–91% (⩽3 months) and sustained clinical remission in 61–81% of patients.7,41,42,45
Factors associated with response and/or remission following pooled infliximab and adalimumab dose intensification
Ghaly et al. 41 reported that the absence of continuous corticosteroids for ⩾6 months in the 5 years preceding anti-TNF dose intensification was the sole predictor of durable steroid-free remission over the 12 months following dose intensification (OR 3.5, 95% CI 1.05–12.05).
Non-response and LOR following anti-TNF dose intensification
Nineteen studies reported or allowed imputation of clinical outcomes pertaining to tertiary non-response and tertiary LOR following infliximab and/or adalimumab dose intensification (Table 3).4–7,16,17,21,23–25,28–30,33,35,38,41,44,45
Tertiary non-response and loss of response following anti-TNF dose intensification.

NR: not reported; CRP C-reactive protein; CDAI: Crohn’s disease activity index; HBI: Harvey–Bradshaw Index
Pooled outcomes following anti-TNF dose intensification for primary non-response and secondary loss of response.
Tertiary non-response
Tertiary non-response within 6 months of anti-TNF dose intensification undertaken to address secondary LOR was reported in 10–45% of patients across 10 studies, including two studies that reported outcomes following both infliximab and adalimumab dose intensification.6,16,17,21,24,28,29,38,44,45 When outcomes were analysed by anti-TNF agent, tertiary non-response occurred in 10-58% and 14-45% of patients following infliximab and adalimumab dose intensification, respectively. Comparatively, three studies included patients who underwent anti-TNF dose intensification for both primary non-response and secondary LOR, reporting tertiary non-response in 7–37% of patients.30,33,41
Tertiary LOR
Tertiary LOR following at least 6 months of anti-TNF dose intensification to address secondary LOR was reported in 7–64% of patients across 10 studies; including 16–64% and 7–57% of patients following infliximab and adalimumab dose intensification, respectively.4,5,7,16,17,21,23–25,28 Comparatively, two studies included patients who underwent anti-TNF dose intensification for both primary non-response and secondary LOR, reporting tertiary LOR in 30–38% of patients.30,35
Predictors of tertiary non-response and tertiary LOR following anti-TNF dose intensification
Baert et al. 6 reported that failure of dose intensification was associated with elevated CRP at baseline, while Ma et al. 23 ascribed the need for concurrent corticosteroid therapy at initial adalimumab induction to a reduced likelihood of response to adalimumab dose intensification following secondary LOR. Duveau et al.24 reported that tertiary LOR occurred less frequently over time with adalimumab 40 mg weekly than 80 mg 2-weekly and with a CRP ⩽ 5 mg/L compared with a CRP > 5 mg/L prior to dose intensification. Ma et al. also reported that factors including CRP > 10 mg/L and prior anti-TNF exposure were associated with an increased risk of tertiary LOR on univariate analysis, while elevated CRP was predictive of shorter time to tertiary LOR on Kaplan–Meier analysis23. In a pooled cohort who underwent anti-TNF re-induction and/or dose interval shortening with infliximab and adalimumab, longer time to treatment failure was associated with higher baseline serum albumin, male sex, and thiopurine co-therapy on multiple regression analyses. 7
Meta-analysis
Differentiating between effective and ineffective dose intensification
Studies evaluated as part of each meta-analysis below, that is, (1) tertiary response versus tertiary non-response and (2) tertiary response versus tertiary LOR, were assessed across comparable timepoints.
Evaluating tertiary response and tertiary non-response
Twelve studies reported both tertiary response and tertiary non-response within 6 months of infliximab (n = 5), adalimumab (n = 5) or co-reported infliximab/adalimumab (n = 2) dose intensification, including nine studies that used dose intensification to address secondary LOR alone.6,16,17,21,24,28–30,33,38,41,45 Clinical assessment of tertiary response and tertiary non-response was undertaken after a median of 14 weeks (range 4–24 weeks). Anti-TNF dose intensification using any strategy was more likely to result in tertiary response than tertiary non-response within the first 6 months (RR 2.58 [95% CI 1.76, 3.79, I2 = 82%], Figure 3(a)).

Forest plot comparison of: (a) tertiary response versus tertiary non-response within 6 months of anti-TNF dose intensification (b) tertiary response versus tertiary loss of response beyond 6 months of anti-TNF dose intensification.
Evaluating tertiary response and tertiary LOR
Seven studies reported both tertiary response and tertiary LOR beyond 6 months of infliximab (n = 4) or adalimumab (n = 3) dose intensification, including five studies that used dose intensification to address secondary LOR alone.4,5,17,21,24,30,35 Sustained tertiary response to anti-TNF dose intensification beyond 6 months (RR 1.10 (95% CI 0.75, 1.61, I2 = 85%), Figure 3(b)) was comparable to tertiary LOR after a median of 12 months (range 9–23 months).
Potential sources of heterogeneity were evaluated by subgroup analysis, including by anti-TNF (infliximab. adalimumab or pooled infliximab/adalimumab) or indication for dose intensification (secondary LOR versus pooled primary non-response/secondary LOR). The results remained similar in all cases with no variations of significance, although heterogeneity remained high.
Discussion
This systematic review affirms that empiric anti-TNF dose intensification represents a clinically effective strategy to address secondary LOR to adalimumab and infliximab therapy in patients with Crohn’s disease. Short-term clinical response following any anti-TNF dose-intensification strategy to address secondary LOR within 12 weeks ranged from 33% to 90%, while sustained clinical response lasting at least 48 weeks ranged from 25% to 85%.4–6,15–17,19–29,31,32,38,43,45 Similarly, short-term clinical remission following any anti-TNF dose-intensification strategy to address secondary LOR was 16–67% within 12 weeks, while longer-term clinical remission was reported in 20–84%.7,15–17,20,22,26–29,38,39,42–45
Nevertheless, empiric anti-TNF dose intensification is not always effective, and even when effective, may not provide a durable clinical response. Hence, the current systematic review also evaluated tertiary non-response and tertiary LOR following anti-TNF dose intensification. Tertiary non-response within 6 months of any anti-TNF dose-intensification strategy occurred in 10–45% of patients, while tertiary LOR following at least 6 months of intensified therapy occurred in 7–64% of patients. Hence, despite the clinical effectiveness of anti-TNF dose intensification in addressing LOR, clinicians must remain cognisant that a proportion of patients may not respond, or fail to demonstrate durable response to anti-TNF dose intensification. This highlights the need to differentiate between effective and ineffective dose intensification. Our meta-analysis demonstrated that patients are more than twice as likely to achieve tertiary response then experience tertiary non-response within the first 6 months following anti-TNF dose intensification; however, on the basis of currently available data, the clinical effectiveness of longer-term anti-TNF dose intensification remains to be clarified. These findings reflect real-world data wherein the likelihood of response is highest immediately following anti-TNF initiation, with diminishing response over time.
Given that some patients will respond to empiric anti-TNF dose intensification, while others may not, it remains important to identify factors associated with response, non-response and LOR following anti-TNF dose intensification. Baseline characteristics associated with favourable clinical response following either adalimumab or infliximab dose intensification included younger age at diagnosis (16–40 years), 5 stricturing/penetrating phenotype,5,24 absence of continuous (⩾6 months) corticosteroid use within 5 years of dose intensification, 41 LOR after ⩾10 months of adalimumab, 24 non-smoking status, 5 lower baseline CDAI, 17 lower baseline CRP,5,24 baseline albumin ⩾3.8 g/dL, 16 baseline serum infliximab trough levels ⩾1 mg/mL, 16 and plasma IL-6 levels ⩽2.41 pg/mL. 16 Other factors associated with clinical response included dose intensification using adalimumab 40 mg weekly rather than 80 mg 2-weekly, 24 lower CDAI 4 weeks following dose intensification, 17 and normalisation of CRP following dose intensification. 4 Similarly, characteristics associated with tertiary non-response and tertiary LOR-included corticosteroid use at adalimumab induction, 23 higher baseline CRP,6,23,24 and prior anti-TNF exposure, 23 while adalimumab 40 mg weekly (versus adalimumab 80 mg 2 weekly), 24 male sex, 7 higher baseline albumin, 7 and thiopurine co-therapy 7 were all associated with more favourable outcomes.
This systematic review and meta-analysis is, to the best of our knowledge, the first to evaluate tertiary response, tertiary non-response, and tertiary LOR following empiric anti-TNF dose intensification undertaken specifically to address secondary LOR in patients with Crohn’s disease. Strengths of our study include the rigorous and extensive literature search, evaluation of both favourable and unfavourable post-intensification clinical outcomes, patient, disease and treatment characteristics associated with these outcomes, and evaluating post-intensification outcomes specific to secondary LOR; however, we also acknowledge several limitations. Methodological heterogeneity impacts the interpretation of the studied outcomes, including differences between studies relating to study design, study population, and analytical approaches. Moreover, while current treatment algorithms advocate that secondary LOR be defined by the presence of both clinical and objective disease activity prior to anti-TNF dose intensification, only 15 of 34 eligible studies included validated clinical indices in their definition of LOR, of which six studies also required objective disease activity prior to anti-TNF dose intensification. Hence, a lack of standardisation between definitions of secondary LOR and clinical outcomes following dose intensification, likely reduces the reliability of direct comparison between studies. This also highlights the current unmet need for a more standardised approach to clinical assessment of secondary LOR and/or clinical response following anti-TNF dose intensification.
There is accumulating evidence to suggest that the first biologic agent may offer the most favourable response; reflected by studies documenting that a greater proportion of patients require adalimumab and infliximab dose escalation with second and third-line therapy. 46 This review noted that studies that reported outcomes following infliximab dose intensification were more likely to have included patients who were biologic experienced relative to studies that reported outcomes following adalimumab dose intensification across largely biologic naïve cohorts. However, the impact of prior biologic exposure on clinical outcomes, particularly tertiary response and tertiary non-response, following anti-TNF dose intensification was difficult to ascertain owing to incomplete reporting of prior biologic exposure across patient subsets who were dose intensified. For similar reasons, it was difficult to ascertain the comparative effectiveness of empiric anti-TNF dose intensification undertaken for primary non-response and secondary LOR across studies that reported pooled clinical outcomes.
Despite the clinical effectiveness of anti-TNF dose intensification based on clinical and/or objective disease assessment alone, strategies that incorporate anti-TNF drug levels at the time of secondary LOR have been purported to increase the likelihood of therapeutic success.2,47,48 This implies that the optimal clinical approach to secondary LOR involves assessment of clinical and objective disease activity in conjunction with anti-TNF trough levels; highlighting the need to update existing model of care to facilitate this approach. Our group has demonstrated the utility of a virtual biologic clinic-led approach in executing such a strategy; showcasing that a virtual clinic led model-of-care is supported by processes that promote more appropriate dose intensification and more frequent treatment success than standard outpatient care alone. 18
In conclusion, although empiric anti-TNF dose intensification is clinically effective in patients with Crohn’s disease, particularly within the first 6 months, a proportion of patients will fail to demonstrate short-term and/or sustained clinical response. This highlights the need for clinical reassessment following anti-TNF dose intensification, particularly beyond 6 months, to differentiate between effective and ineffective dose-intensification strategies. The ideal paradigm for disease reassessment following dose intensification is yet to be fully elucidated in Crohn’s disease, but should accommodate early recognition of response and non-response to facilitate discontinuation of ineffective therapy in cases of tertiary non-response, and consideration of further therapeutic optimisation or switching in cases of partial or incomplete tertiary response, respectively. Such an approach embodies many of the principles central to the modern-day treat-to-target paradigm.
Supplemental Material
sj-docx-1-tag-10.1177_17562848211070940 – Supplemental material for Systematic review and meta-analysis: evaluating response to empiric anti-TNF dose intensification for secondary loss of response in Crohn’s disease
Supplemental material, sj-docx-1-tag-10.1177_17562848211070940 for Systematic review and meta-analysis: evaluating response to empiric anti-TNF dose intensification for secondary loss of response in Crohn’s disease by Ashish Srinivasan, Robert Gilmore, Daniel van Langenberg and Peter De Cruz in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
AS is supported by an Australian Government Research Training Programme (RTP) scholarship, Monash University Graduate Excellence Scholarship and Monash University Department of Medicine Postgraduate Faculty Award. PDC is supported by a National Health & Medical Research Council (NH&MRC) Early Career Fellowship
Author contributions
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AS has served as a speaker for Sandoz, and received research support from Pfizer. PDC has served as a consultant, an advisory board member, or a speaker for AbbVie, Baxter, Ferring, Janssen, Celltrion, Emerge Health, Shire, and Takeda, and received research support from Ferring, Shire, Janssen, AbbVie, and Takeda. DvL has received educational grants or research support from Pfizer, Takeda, Ferring, and Shire and has received consultancy and/or speaker’s fees from Pfizer, Janssen, AbbVie, Ferring, Vifor, and Emerge Health. RG has no disclosures.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Guarantor of the article
A/Prof Peter De Cruz.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
