Abstract
Background:
5-fluorouracil (5-FU) and mitomycin-C (MMC) with radiotherapy (RT) remain an established treatment for patients with anal cancer (AC). Genetic mutations in two major metabolizing enzymes for 5-FU; dihydropyrimidine dehydrogenase (DPYD and thymidylate synthetase (TYMS), have been associated with clinical response and toxicity. However, their place in the treatment of AC remains undetermined.
Methods:
We retrospectively reviewed 21 patients with AC, including T2-4, N0-1, M0 or T1-4, N2-3, and M0 treated between 2012 and 2018. All patients were treated with 5-FU 1,000 mg/m2/day via continuous intravenous (IV) infusion 1–4 and 29–32, MMC 10 mg/m2 IV bolus days 1 and 29 plus RT. Patients who developed ⩾3 grade toxicities were tested for the DPYD and TYMS genes. Treatment was either modified with reduced doses or changed to MMC 10 mg/m2 day 1 and 29 with cisplatin 25 mg/m2/week plus RT. Toxicities and responses were collected.
Results:
Six out of 21 patients who developed ⩾3 grade toxicities including pancytopenia, neutropenia, thrombocytopenia, mucositis, nausea, rash, and nephritis were found to have genetic mutations: TYMS 2RG/3RC (n = 2), 3RG/3RC (n = 1), 2R/2R (n = 2), TYMS 3′UTR del/Ins (n = 2), and DPYD c.2864A > T heterozygous (n = 1). Two patients received 5-FU at a 50% reduced dose on days 29–32; one patient refused to receive 5-FU (continued with MMC and RT); one patient received only radiation therapy due to persistent pancytopenia despite the use of growth factors; two patients received an alternative regimen consisting of MMC 10 mg/m2 on day 29 with cisplatin (CDDP) 25 mg/m2/week plus RT; and two patients received cisplatin/MMC with RT from the beginning as they were prospectively detected to have TYMS abnormalities prior to dosing the chemotherapy. These patients tolerated treatment very well with only grade 2 toxicities. All the patients (4/4) on cisplatin/MMC achieved clinical complete response (cCR), while four patients (4/15) on 5-FU/MMC reached cCR at the first assessment. Radiological response showed complete response at the end of 24 weeks assessment.
Conclusions:
Molecular testing for DPYD and TYMS genes can allow us to identify patients who are most likely to respond or face severe toxicity to 5-FU in a potentially curable cancer. Combining radiation with CDDP with MMC in patients with AC is feasible. A prospective study based on pharmacogenetic testing comparing MMC/cisplatin with MMC/5-FU is indicated in patients with AC.
Keywords
Introduction
The combination of 5-fluorouracil (5-FU) and mitomycin-C (MMC) with concurrent radiotherapy (RT) remains the gold standard treatment for patients with locally-advanced anal cancer (AC).1–5 5-FU is considered among the most widely-prescribed anticancer agents worldwide to treat numerous solid tumors, notably gastrointestinal (GI) and breast. Following the administration of 5-FU, approximately 80% is catabolized by dihydropyrimidine dehydrogenase (DPYD), the rate-limiting enzyme for 5-FU metabolism.6–10 The remaining part of 5-FU is further anabolized into an active form that inhibits the synthesis of both DNA by competitive inhibition of the thymidylate synthase (TYMS) enzyme or RNA by direct incorporation of cytotoxic metabolites into nucleic acids. The TYMS enzyme provides the only de novo source of thymidylate for DNA synthesis.11,12 Although other markers have been implicated in 5-FU-associated toxicity and efficacy, there is emerging data that suggests TYMS is over-expressed in cancer cells, making it a potential predictive marker to assess response to chemotherapy, such as 5-FU.11–14
The most common side effects (>30%) in patients following 5-FU may include diarrhea, nausea/vomiting, mucositis, photophobia, and bone marrow suppression, notably neutropenia. 15 On the other hand, MMC is mainly associated with myelosuppression (>30%), which is usually delayed in onset. GI toxicities are less common compared with 5-FU. Bladder inflammation (urinary frequency, burning, cramping, and pain) is seen following intravesical administration of MMC therapy. 16
DPYD deficiency syndrome is a well-recognized pharmacogenetic syndrome which typically manifests as severe or fatal diarrhea, mucositis/stomatitis, myelosuppression, and even rare toxicities, such as hepatitis, encephalopathy, and acute cardiac ischemia following a first or second dose of 5-FU.6–10,17 On the other hand, data related to the TYMS gene is scarce.11–14,18,19
The human TYMS gene is polymorphic, with either double or triple tandem repeats of a 28 base-pair sequence downstream of the cap-site in the 59-terminal regulatory region.11,12 Previous studies have suggested that the TYMS genotype predicts TYMS mRNA expression in metastasized colon tumors and serves as a surrogate predicted of response, as well as toxicity, to 5-FU.18,19
There are currently few guidelines available to select upfront those patients who are at risk for either drug resistance or toxicity to 5-FU or CAP. 20 This topic is of clinical significance, as per recent publications, especially a report by Jones et al.21–23 We here present a study that analyzed TYMS genotyping and its role with outcome in patients treated with 5-FU and MMC for AC.
Patients and methods
Approval from the Institutional Review Board (IRB) at the Tufts University Medical Center was obtained before the initiation of this retrospective chart review to analyze patients treated for AC between July 2012 and September 2018. The methods adopted in this retrospective study were in accordance with the ethical standards of the institutional IRB, as well as the Declaration of Helsinki and approved by the IRB. All patients were consented both for pharmacogenetic testing as well as for chemotherapy agents used in the treatment.
We retrospectively reviewed 21 patients AC. The patient demographic and clinical data was collected and entered into a customized case report form. The primary endpoint of the study was to assess toxicity in patients with DPYD and TYMS mutations treated for AC and their outcome when treated with either modified or alternative regimens.
Eligibility criteria
The inclusion criteria included the patients who had histologically confirmed squamous cell AC, with the following TNM staging: primary tumor T2–T4 tumors (T2 = diameter of the primary cancer >2 cm but <5 cm; T3 = 5 cm; and T4 = invading adjacent organs) with any N category (pelvic or inguinal defined by clinical examination, biopsy, or diagnostic radiology), M0 (absence of metastases). Patients included had Eastern Cooperative Oncology Group (ECOG) performance status ⩽2, hemoglobin ⩾9.0 g/dl, neutrophils (ANC) ⩾1500 k/ul, platelets >100,000/cm2, and adequate renal (creatine <1.5 mg/dl) and hepatic organ function treated (total bilirubin <1.5 mg/dl, AST/ALT <2.5 mg/dl). Patients were excluded if they had severe comorbid conditions including acquired immune deficiency syndrome (AIDS), or major malignancy (unless successfully treated and disease-free for at least 5 years, or concurrent uncontrolled medical illness. Before treatment, written informed consent was obtained from all patients. The study was approved by institutional scientific and research ethics committees.
Pre-treatment assessment
Prior to any therapy, patients underwent direct rectal examination (DRE), imaging with computed tomography (CT) of abdomen, pelvis and chest or magnetic resonance imaging (MRI) of the abdomen/pelvis to establish the stage of disease. In addition, blood and serum chemistry evaluations were performed to determine the adequacy of hepatic, renal, and bone marrow functions according to institutional guidelines.
Treatment plan
All patients were planned to receive MMC at a dose of 10 mg/m2 IV bolus on days 1 and 29 (not to exceed 20 mg per course), with 5-FU at a dose of 1000 mg/m2/d by continuous infusion on days 1–4 and 29–32, as previously published. Patients received appropriate pre-medications and hydration per National Comprehensive Cancer Network (NCCN) guidelines. Radiation therapy consisted of a minimum dose of 45 Gy in 25 fractions of 1.8 Gy over 5 weeks to the primary cancer and for patients with T3, T4, node-positive disease, or patients with T2 residual disease, they received an additional boost of 10–14 Gy (total dose of 55–59 Gy) as previously published.2,3 Initial radiation fields also included the pelvis, anus, perineum, and inguinal nodes, with the superior field border at L5-S1 and the inferior border to include the anus with a minimum margin of 2.5 cm around the anus and tumor as previously described.2,3
Patients were monitored for toxicities and laboratory abnormalities on a weekly schedule in the medical oncology clinic. Acute toxicity and chronic toxicities were confirmed during weekly multidisciplinary tumor boards. The worst grade was scored from start of treatment until 30 days after the last fraction of radiotherapy. Toxicities were collected and graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events (NCI-CTCAE), v4.03. 24
If a patient developed severe toxicities concerning for pharmacogenetic syndrome, DPYD and TYMS were tested while treatment was held for toxicities to resolve. DPYD and TYMS were analyzed by polymerase chain reaction (PCR)/single nucleotide extensions (SBE)/fragment analysis (FA) as determined by the Associated Regional and University Pathologists (ARUP) Laboratories. 25
Dose modifications for each chemotherapeutic agent were performed according to the providers instructions, as approved by the Federal Drug Agency (FDA) and institutional practices. If no pharmacogenetic abnormality was detected and toxicities recovered to <grade 1, the doses of both MMC and 5-FU were modified according to pre-specified criteria as previously described. In case of grade 4 neutropenia or febrile neutropenia, the doses of both MMC and 5-FU were reduced by 50%. Temporary suspension of chemoradiation was allowed for grade 3 or 4 toxicities. In patients who developed ⩾3 grade toxicities, chemoradiation was either suspended temporarily until recovery to grade 2 or lower grade.
In some patients, the chemotherapy agent was modified with reduced doses of chemotherapy or omitted while radiation was continued following multidisciplinary team (MDT) discussion at the tumor board.
However, if DPYD and/or TYMS abnormality was found, then the chemotherapy was changed to an alternative regimen, preferably a, non-FU containing regimen after informed consent was gained following discussion in the multidisciplinary clinics. We adopted a mitomycin-cisplatin regimen consisting of MMC 10 mg/m2 on day 1 and 29, with cisplatin 25 mg/m2/week plus RT based on European Organization for Research and Treatment of Cancer phase II study 22011-40014.26,27 It is important to note here that affected by the data on earlier patients included in this study, five patients included in this data underwent pre-treatment pharmacogenetic testing for TYMS and DPYD. Two of these five patients were found to have pharmacogenetic abnormalities; as a result, they were initiated on the alternative chemotherapy regimen consisting of cisplatin with MMC instead of MMC and 5-FU from day 1 along with concomitant radiation.
Eight weeks after therapy, patients underwent reevaluation similar to baseline, including tumor response by DRE, palpation of inguinal notes during treatment, and radiological imaging. A full-thickness biopsy was optional. Based on previous data as responses can be delayed in many cases, we also selected 26 weeks = 182 days as the time point to assess a complete clinical response (cCR) after chemoradiation therapy. Patients were followed up every 3 months for 1 year and every 6 months for the 2nd year, then yearly as per NCCN guidelines.
Results
Demographics
A total of 21 patients (Caucasians 80%; 7 females, 14 males; age range: 42–68 years.) were included in the study who were treated for LAC including T2-4, N0-1, M0 or T1-4, N2-3, M0 treated between 2012 and 2018 (Figure 1). In addition, two patients received cisplatin/MMC with RT from the start as they were detected to have TYMS abnormalities during pre-treatment screening as previously described.

Study schema/patient disposition.
Six of the 21 patients who developed severe toxicities consisting of grade ⩾3 pancytopenia, neutropenia, thrombocytopenia, mucositis, nausea, rash, and nephritis tested positive for abnormities in DPYD and/or TYMS, while four showed pharmacogenetic abnormalities. Attribution of toxicities to each chemotherapeutic agent was determined by the MDT.
MDT, multidisciplinary team.
Pharmacogenetics
Six of the 21 patients were found to have a TYMS (28%) mutation, while one had both TYMS and DPYD mutations (1/21: 5%) as summarized in Table 1.
Pharmacogenetics in the study related to 5-FU.

5-FU, 5-fluorouracil; DPYD/DPD, dihydropyrimidine dehydrogenase; SNP, single nucleotide polymorphisms; TSER, thymidylate synthase enhancer region; TYMS, thymidylate synthase; UTR, untranslated region; VNTR, variable number tandem repeat.
Toxicities
The time to onset of severe toxicities was at a median of week 3 (range: 2–4 weeks) with MMC/5-FU with RT. Chemotherapy was suspended in all patients until they recovered to grade 1 toxicity, while radiation continued as per the schedule. Table 2 summarizes grade 3 and 4 toxicities encountered in this study.
Grade 3 and 4 toxicities.
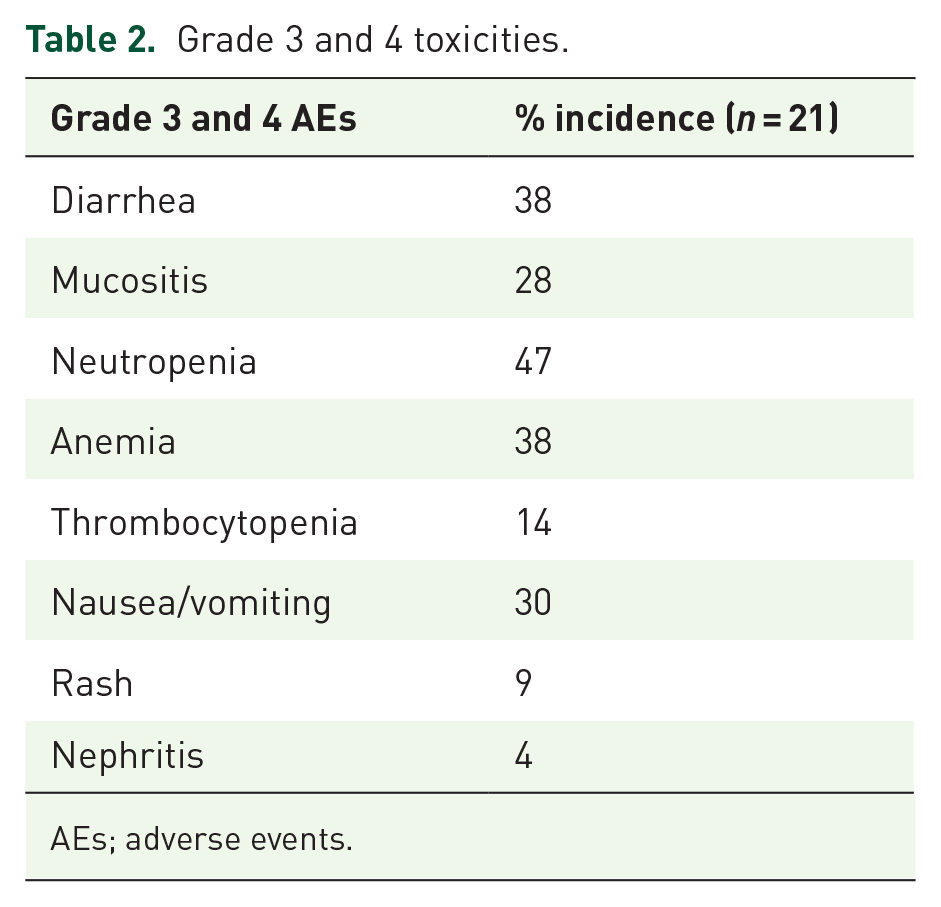
AEs; adverse events.
Two patients received 5-FU at 50% reduced dose (500 mg/m2/d by continuous infusion on days 29–32); one patient refused to receive 5-FU (continued with MMC and RT); and one patient received only radiation therapy due to persistent pancytopenia despite the use of growth factors.
Two patients, including one with both TYMS and DPYD abnormalities, and one who developed grade 4 toxicity following their first week of MMC/5-FU with radiation, were changed to an alternative regimen consisting of MMC 10 mg/m2 on day 29 with cisplatin 25 mg/m2/week plus RT adapted from the European Organization for Research and Treatment of Cancer (EORTC) studies: 22953 and 22011-40014 (Figure 2).26,27
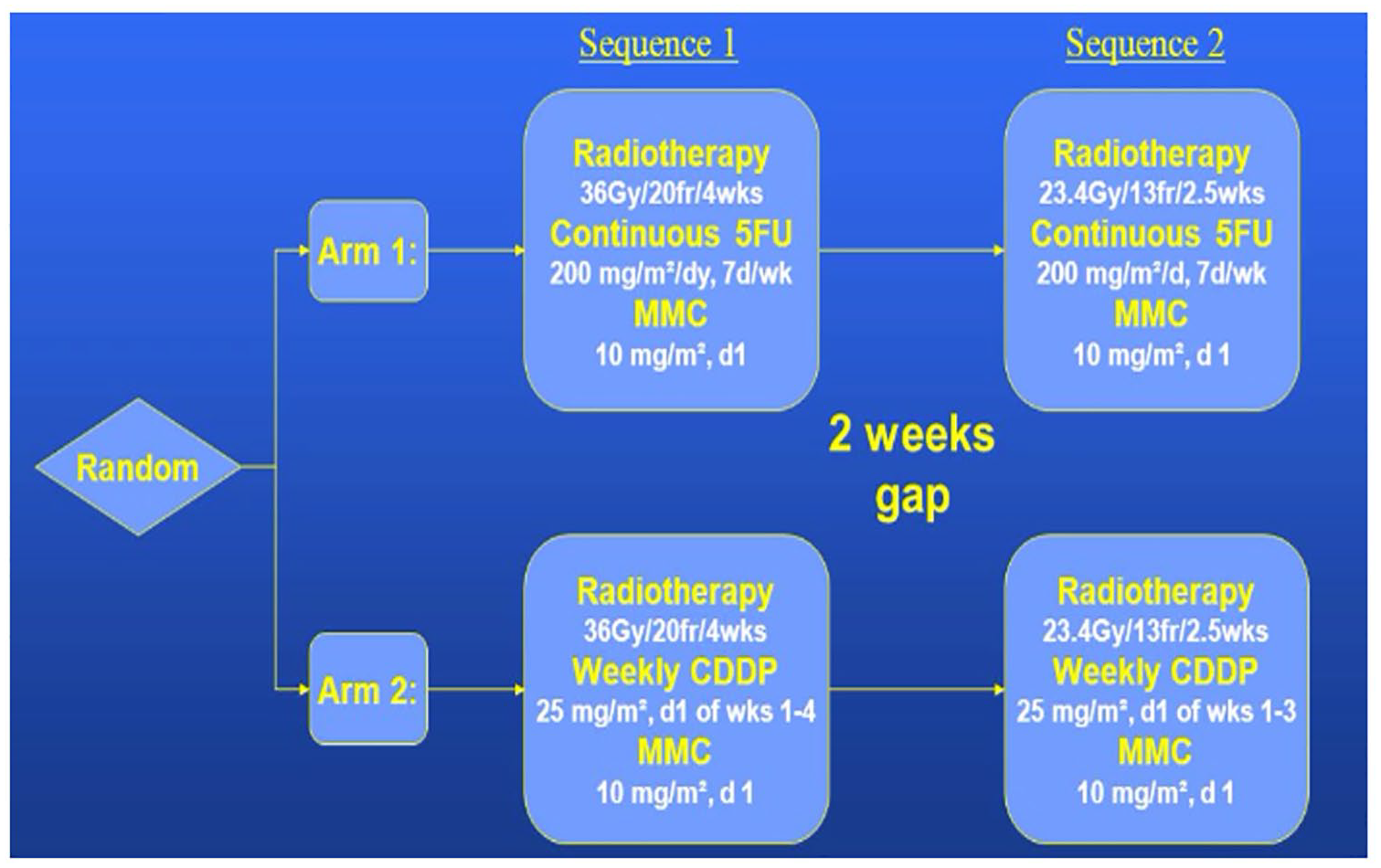
Alternative treatment regimen: EORTC 22011-40014.
In addition, two patients received cisplatin/MMC with RT from the beginning as they were prospectively detected to have TYMS abnormalities prior to dosing the chemotherapy. These patients tolerated treatment very well with only grade 2 toxicities.
Efficacy
Overall, all the patients showed response, indicating the patients who received even a lower dose of 5-FU with 3′-untranslated region (UTR) 6 bp deletion (TTAAAG) (rs16430) or 5′-thymidylate synthase enhancer region (TSER) G>C single nucleotide polymorphism (SNPs) in the second repeat of 3R allele (3RC) (rs34743033) as shown in Table 2. The four patients who received the alternative regimen of MMC and cisplatin achieved cCR when evaluated at the end of therapy. Radiological response showed CR at the end of 24 weeks assessment. All six patients remained cancer free at the conclusion of the second year since completing the therapy.
Discussion
Our study, although small, provides evidence of personalizing treatment of AC based on pharmacogenetic testing for TYMS and DPYD. Moreover, this study further augments other recent reports on documenting on pharmacogenetics related to 5-FU or capecitabine in treating AC.21–23 Currently, there is no biomarker which can predict response to treatment and, despite the development of novel chemotherapeutic agents, 5-FU and MMC remain the standard regimen delivered to patients with AC with concurrent radiation therapy.
Previous studies have indicated that the TYMS genotype predicts not only for toxicity, but also for efficacy to 5-FU. Studies in colorectal cancer have revealed that patients who are homozygous for the 3R allele exhibit lower objective responses to TYMS-targeted therapies.12,18 It is evident from the studies cited above that FU and CAP remain persistently active and play an integral part of treatment for AC. We found only one study in which the investigators studied CAP-MMC combination with RT and assessed the role of TYMS polymorphism as a predictor to toxicity 23 but found no relation to efficacy. Forty percent of patients with a low expression genotype experienced grade 3-4 toxicity versus 18% of patients with a high expression genotype. Moreover, TYMS is also now know to form ribonucleoprotein complexes with several other mRNAs, including p53 and c-myc, which further supports the need to investigate the role of TYMS in cancer therapy. The association between TYMS and 5-FU related outcomes remains a topic of constant debate. 12 Some critiques argue that the differences in outcomes could be related to differences in study design or a lack of statistical power. However, we would like to mention here that our study and others do indicate that, in selected patients with TYMS abnormalities such as 6 bp deletion (TTAAAG; rs16430) or 3RC/3RC or 2R/3RC, they may see improved response to 5-FU even if administered a lower dose of 5-FU. This will need to be validated in prospective trials.
AC, though a rare malignancy, is potentially curable following chemoradiotherapy. The initial studies that determined that AC carcinoma is highly-sensitive to concurrent chemoradiation have led to the develop of numerous variants in chemotherapy as well as radiotherapy regimens, with the overall aim being to improve the outcome in patients with AC.1–5,28 The primary objective is to preserve the sphincter and avoid colostomy; as a result, surgery has become a salvage or secondary therapy.
The optimal dose of radiation with concurrent chemotherapy to optimize local control and minimize sphincter toxic effects was determined in the RTOG-9208 study, which showed it to be in the 45 Gy to 60 Gy range. 29 The RTOG-9811 study showed that CDDP/5-FU was not superior to MMC/5-FU for DFS (the primary endpoint).28,30 In addition, the cumulative colostomy rate was significantly worse in the CDDP/5-FU arm compared with the MMC/5-FU arm. However, the overall survival (OS) in both arms was similar. This reinforced the idea that MMC/5-FU should persistently be the standard of care of patients with AC. More recently, EORTC studies showed that RR was 91.9% with MMC/CDDP versus 79.5% with MMC/5-FU.26,27 event free survival (EFS) at 12 months was 94.5% versus 76.9% between the two arms, respectively. As a result, favoring EFS with RT + CCDP/MMC. CCDP/MMC also achieved RR of 75% at the end of 8 weeks. Both studies have shown the feasibility, tolerability, and responses. Therefore, we adopted this regimen in our selected patients after an open discussion in a multidisciplinary meeting. As far as we are aware, EORTC has abandoned the study of CDDP with MMC at present. The three-year EFS rates with RT + MMC/5-FU were anticipated to be 55% during the design of the phase II study; however, due to a lack of long-term follow-up and the small number of patients treated, conclusions cannot be drawn at the moment.
Capecitabine, an oral pro-drug of 5-FU, has been tested in clinical trials to replace infusional 5-FU, offering an alternative to 5-FU as well as being more convenient for patients.23,31,32 Akin to chemotherapy, improvement and modifications in radiotherapy also remain of research interest to investigators. Radiotherapy is generally administered using a two- or three-field technique to a total dose of 45–50.4 Gy over 4–5 weeks, sometimes followed by a boost of up to 59.4 Gy and, more recently, in the form of intensity-modulated radiation therapy (IMRT).33,34
Compliance with the overall treatment time is crucial in AC, as evidenced by post hoc analyses of data from the ACT II trial. 35 Our study also suggests that, if you continue the RT and switch chemotherapy drugs, then these patients will have superior outcomes to those who were rested form radiation. Even with IMRT, this poor compliance is an issue. A pooled analysis of patients treated with IMRT showed failure to complete treatment or interruptions (any extension >2 days over the planned overall treatment) occurred in 5.2% of patients. This was associated with a hazard ratio (HR) of 5.80 (1.96–17.29) for persistent disease (p = 0.001) compared with treatment delivered per-protocol. These data underline the importance of the compliance issue and continuity of therapy in a potentially curable disease.
In summary, TYMS and polymorphism, in addition to DPYD, least to 5-FU-associated toxicities and probably outcome. 36 Investigators have tried to pharmacokinetically-based 5-FU in some studies, but such studies have not been collaborated with pharmacogenetic abnormalities.37,38 Taken together, our current study supports a rational to consider TYMS and DPYD testing both before initiating therapy with patients with AC. A prospective randomized phase II study is warranted to further validate the clinical implications of 5-FU pharmacogenetics in a curable cancer such as AC.
Footnotes
Authors’ note
Presented as a Poster at GI Symposium 2019 (Abstract # 605).
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
