Abstract
Pouchitis is the most common complication in patients who have undergone restorative proctocolectomy with ileal pouch-anal anastomosis (IPAA). Up to 81% of IPAA patients experience pouchitis, with 40% of patients presenting within the first year of surgery. Common risk factors include genetic mutations, extensive colitis, rheumatologic disorders, and primary sclerosing cholangitis. Currently, there are no medications with approved indications for pouchitis. As such, the conventional treatment of pouchitis is entirely off-label. This paper is intended to be a practical and up-to-date review of available therapies used for the management of pouchitis. The mainstay of treatment for acute pouchitis remains antibiotics, but newer therapeutics have also shown promise in the treatment of chronic pouchitis. Common lifestyle considerations that may play a role in pouchitis are also reviewed.
Plain language summary
The ileal pouch-anal anastomosis (“pouch”) is the most common way patients who require surgery to remove their colon are able to avoid a permanent ileostomy (“ostomy”). This pouch, created from the small intestines, serves as a reservoir to hold stool. The most common complication after pouch surgery is pouchitis. Pouchitis symptoms include more frequent bowel movements, urgency to defecate, blood in the stool, incontinence, and abdominal pain. This paper is intended to be a practical review of available therapies including medications and lifestyle changes that can be considered for the management of acute pouchitis, chronic pouchitis, and cuffitis.
Keywords
Introduction
Pouchitis is the most common complication in patients who have undergone restorative proctocolectomy with ileal pouch-anal anastomosis (IPAA). Pouchitis commonly presents with a constellation of symptoms such as increased stool frequency, watery stool, tenesmus, abdominal cramps, incontinence, and pelvic pressure. It has been reported that 93.3% of patients in all age groups maintain a functional pouch after 30 years. However, up to 81% of IPAA patients experience pouchitis.1–6 For those who develop pouchitis, up to 40% of patients will present within the first year of surgery. 5
Numerous risk factors have been associated with the development of pouchitis. Reported risk factors include mutations in NOD2/CARD15 7 and genetic polymorphisms of interleukin 1 receptor antagonists,8–10 tumor necrosis factor allele 2, and toll like receptor 1. 11 Conditions such as extensive ulcerative colitis, rheumatologic disease, primary sclerosing cholangitis, and pyoderma gangrenosum have been shown to have an increased likelihood of developing pouchitis. 12 When examining prior treatment history, a history of pre-colectomy anti-tumor necrosis factor use has been associated with an increased incidence of pouchitis. 13
The pathogenesis of pouchitis remains unclear. However, a leading hypothesis involves a developed dysbiosis of the gut microbiota. The construction of the ileal pouch creates an altered anatomy, leading to fecal stasis, an abnormal immune response, and ultimately an environment which favors inflammation. 14 Increasingly, it is being recognized that the inflammation of pouchitis appears similar to ulcerative colitis. 15
Definitions of pouchitis vary by study, 16 including a combination of criteria based on symptoms, pouchoscopy findings, or histologic features. The most commonly use scoring system for pouchitis is the Pouchitis Disease Activity Index (PDAI). 17 An overall PDAI score is calculated from three separate 6-point scales based on clinical symptoms, endoscopic findings, and histologic features. A PDAI score ⩾7 is consistent with pouchitis. A modified PDAI score (mPDAI), which excludes the histologic subscore, has also been used to diagnosis pouchitis. A mPDAI score of ⩾5 suggests a diagnosis of pouchitis. 18
When determining how to treat pouchitis, it is important to consider alternative etiologies that may mimic idiopathic pouchitis such as infection (particularly cytomegalovirus and Clostridiodes difficile), ischemia, non-steroidal anti-inflammatory drugs (NSAIDs), pre-existing versus de novo Crohn’s disease, cuffitis, postsurgical or mechanical disorders (pelvic sepsis, leak, fistulae, pouch prolapse, obstructions), and functional disorders (dyssynergic defecation, reduced pouch compliance, irritable pouch syndrome) (Figure 1). 19

Pouch conditions. (a) Normal pouch body. (b) Pouchitis. (c) Tip of J sinus tract. (d) Perianal fistula at anal verge. (e) Cuffitis. (f) Ischemic pouchitis. (g) Pre-pouch Crohn’s ileitis. (h) Pouch prolapsed.
Multiple sub-classifications exist within the diagnosis of pouchitis. Pouchitis can be acute, with symptoms lasting less than 4 weeks, or chronic, with symptoms lasting more than 4 weeks. Chronic pouchitis frequently encompasses multiple chronic inflammatory complications of the pouch including chronic antibiotic-dependent pouchitis (CADP), chronic antibiotic-refractory (CARP) pouchitis, and Crohn’s disease or Crohn’s-like disease of the pouch. It is important to note, though there are some features that suggest Crohn’s disease of the pouch, there is not a consistent definition for Crohn’s disease of the pouch. Treatments for various types of chronic pouchitis frequently overlap.
Currently, there are no medications with approved indications for pouchitis. As such, the treatment of pouchitis is entirely off-label. Just as the therapeutic armamentarium for inflammatory bowel disease continues to expand, so too does the number of possible treatments for pouchitis. This paper is intended to be a practical review of therapies used for the management of the various forms of pouchitis and cuffitis.
Primary prophylaxis
Probiotics
Intestinal dysbiosis has been implicated in the pathogenesis of pouchitis. 20 Probiotics play a role in regulating mucosal immune response through reductions in proinflammatory cytokines, thereby reducing inflammation. Probiotics have been studied for use in pouchitis as primary or secondary prophylaxis.20,21
In a double-blind, placebo-controlled trial of 40 patients who underwent IPAA for ulcerative colitis, patients were randomized immediately after ileostomy closure to either VSL#3 (Lactobacillus spp., Bifidobacterium spp., Streptococcus salivarius spp., and Thermophilus spp.) or placebo for 1 year. Patients were followed clinically, endoscopically, and histologically over the year. When comparing the VSL#3 group with the placebo group, 10% versus 40% developed pouchitis, respectively (p < 0.05), with an improved quality of life (QOL) score in the VSL#3 group. 22
In 2004, a consecutive series of 127 patients with IPAA were given daily Lactobacillus rhamnosus GG or placebo and observed over the following 2 years. Pouchitis was diagnosed based on symptoms, endoscopic appearance, and histologic features. Fewer episodes of pouchitis occurred in patients taking daily L. rhamnosus GG (cumulative risk at 3 years: 7% versus 29%; p = 0.01). 23
Most recently, a randomized placebo-controlled trial comparing nine IPAA patients who received Clostridium butyricum MIYAIRI with eight patients who received placebo found that fewer patients (1/9) in the Clostridium butyricum group developed pouchitis (based on modified PDAI score) compared with the placebo group (4/9). 24
Sulfasalazine
Sulfasalazine has been used to prevent pouchitis.25,26 Scaioli et al. performed a retrospective pilot study examining the role of sulfasalazine as primary prophylaxis for pouchitis. Sulfasalazine 2000 mg per day was given to 55 patients who underwent IPAA for ulcerative colitis. The incidence of pouchitis was significantly reduced in the sulfasalazine arm compared with the control arm (15% versus 64.5%; p < 0.001). 27
Immunomodulators and cyclosporine
Immunomodulators, such as azathioprine, along with alternative immunosuppressive therapy such as cyclosporine, have not been shown to be effective in preventing the development of pouchitis from data extrapolated from post-orthotopic liver transplant patients with IPAA.28,29
Secondary prophylaxis
Probiotics
In a randomized controlled trial by Gionchetti et al., 40 IPAA patients in clinical and endoscopic remission after 4 weeks of treatment with ciprofloxacin and rifaximin were randomized to VSL#3 6 g per day or placebo for 9 months. Three patients (15%) developed relapse in the VSL#3 group compared with 20 (100%) in the control group (p < 0.001). 22 Similar positive results were found in a follow-up study. 30 On the other hand, a prospective study of 31 patients with antibiotic-dependent pouchitis found that after induction of remission with ciprofloxacin, 74% of the patients treated with VSL#3 developed recurrence of symptoms or adverse effects within 8 months. Moreover, in the six patients who completed the 8-month course of VSL#3, there was no significant difference in mean PDAI compared with baseline PDAI at the start of the study. 31
While studies provide evidence in support of probiotics for pouchitis prophylaxis, it is important to remember that overall study sample sizes were small. Also, many commercial probiotic preparations exist, varying significantly in composition. Notably, causing considerable confusion in recent years, the De Simone formulation originally sold as VSL#3 was renamed Visbiome in 2016.
As probiotics are generally well tolerated, a trial of probiotics for pouchitis prophylaxis can be considered. Though, in our practice, the use of probiotics for prevention of pouchitis has yielded mixed results. Use of probiotics may be limited by adverse events such as abdominal cramps, vomiting, and diarrhea. 32 Fungemia, 33 bacteremia, 34 and sepsis 35 have also been reported in immunocompromised patients. 36
Acute pouchitis
Antibiotics
Antibiotics are the first-line treatment for pouchitis (Figure 2). A majority of patients with pouchitis will require treatment with antibiotics episodically. Around 39% of patients with acute pouchitis have only a single episode that responds to antibiotic therapy without recurrence. 5

Pouchitis treatment algorithm.
First-line therapy for acute pouchitis includes ciprofloxacin 500 mg BID or metronidazole 500 mg TID for 2–4 weeks (Table 1). 37 A randomized clinical trial performed by Shen et al. comparing metronidazole and ciprofloxacin for the treatment of acute pouchitis found that both were effective in reducing symptoms. However, patients treated with ciprofloxacin had significantly greater reductions in total PDAI scores (6.9 versus 3.8; p = 0.002) with fewer adverse effects (0 versus 33%) such as vomiting, dysgeusia, and transient peripheral neuropathy. 38
Pouchitis and cuffitis medication dosing.
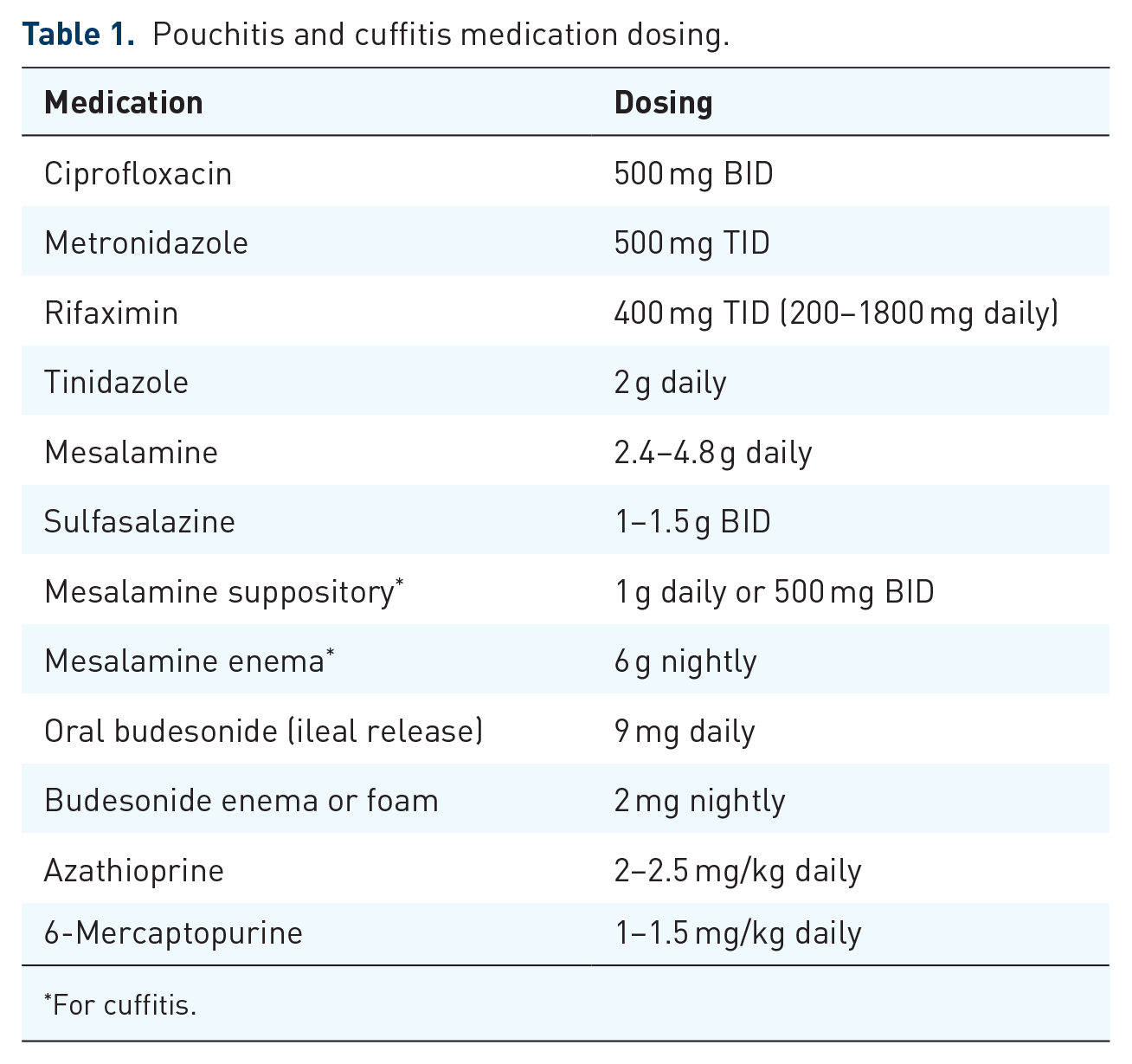
For cuffitis.
Rifaximin, administered as 400 mg TID, is not recommended as first-line therapy for treatment of pouchitis, as it has been shown to be ineffective in the treatment of acute pouchitis in a randomized, double-blind, placebo-controlled trial. 39 However, it has been shown to be effective as maintenance monotherapy in CADP at doses of 200 mg daily with the opportunity to increase up to 1800 mg per day for patients who exhibited at least partial response to the initial dose. The majority of these patients were responsive to 200 mg daily. 40 Rifaximin has also been shown to be effective in a small study using rifaximin 1 g BID in combination with ciprofloxacin 500 mg BID for use in CARP. 41
Other antibiotic agents have also been investigated in uncontrolled, small case series showing efficacy including amoxicillin-clavulanic acid, 42 topical metronidazole, 43 tetracycline, 44 and vancomycin. 45
Sulfasalazine
Data are limited on the role of sulfasalazine and mesalamine therapy for treatment of acute pouchitis. A pilot study of 22 patients evaluated sulfasalazine 3000 mg daily to treat patients with acute pouchitis. At week 8, there was a significant reduction in PDAI score (median decrease from 11 to 5; p < 0.01), and 63% were in remission (defined as PDAI <7) at the end of treatment (p < 0.01). 46 Due to lack of adequate studies, sulfasalazine and mesalamines are generally not recommended as first-line management for acute pouchitis.
Chronic inflammatory complications of the pouch
Antibiotics
After initial treatment for pouchitis, approximately 60% of patients will develop at least one recurrence and up to 20% will develop chronic pouchitis. 47 Treatment will often start with the initiation of previously used antibiotics, often requiring repeated episodic courses of antibiotics or cycling of antibiotics. The use of combination antibiotics such as ciprofloxacin with metronidazole, 48 ciprofloxacin and rifaximin, 41 or ciprofloxacin and tinidazole 49 have been beneficial in open-label trials. Long-term maintenance with antibiotics may be required. Patients should be counseled regarding symptoms of antibiotic side effects, and the antibiotic should be discontinued if there are signs of toxicity.
For IPAA patients with pouchitis refractory to antibiotics, with contraindications to antibiotics such as intolerance or toxicity, or with recurrent Clostridioides difficile, alternative treatments for pouchitis should be considered (Table 1).
Steroids
In chronic antibiotic refractory pouchitis, steroids have been used as a second-line treatment for pouchitis. Gionchetti et al. 50 demonstrated that treatment with oral budesonide at 9 mg daily for 8 weeks was effective in inducing remission in 75% of pouchitis patients refractory to antibiotics. Budesonide enemas 2 mg/100 mL at bedtime for 6 weeks have also been evaluated as an effective treatment for pouchitis, demonstrating similar improvement rates as metronidazole but with fewer side effects. 51 Oral beclomethasone dipropionate 10 mg daily achieved 80% remission rates in chronic refractory pouchitis in a small cohort of 10 patients. 52 As in ulcerative colitis, long-term use of steroids is not advised. However, a response to steroids suggests a potential role for steroid-sparing, anti-inflammatory treatments for chronic antibiotic refractory pouchitis.
Immunomodulators
Expert opinion suggests immunomodulators may play a role in the management of chronic antibiotic refractory pouchitis starting initially at doses of 50 mg/day with weight-based targets of 1.5 mg/kg/day for 6-mercaptopurine and 2.5 mg/kg/day for azathioprine. 53 In eight patients with stricturing Crohn’s-like disease or severe pouchitis, weight-based azathioprine or 6-mercaptopurine led to resolution of symptoms. 54
Anti-TNF inhibitors
The anti-tumor necrosis factor inhibitors (anti-TNFs) have shown moderate success in treating chronic refractory pouchitis and Crohn’s-like complications of the pouch. The majority of published studies on anti-TNFs in pouchitis are retrospective. In a meta-analysis by Huguet et al., a total of 24 studies were included, assessing the use of infliximab (IFX) (n = 194) and adalimumab (ADA) (n = 119) in treating pouchitis. 55 Short-term remission at 8 weeks was achieved in 56% of patients treated with IFX compared with 38% of patients treated with ADA (p = 0.20). Long-term remission at 12 months was achieved in 59% of patients treated with IFX compared with 30% of patients treated with ADA (p = 0.19). The definitions of remission varied widely across studies including endpoints such as a mPDAI score <5, absence of pouch failure, cessation of symptoms, complete closure of fistula, and endoscopic healing.
Similar meta-analyses share similar conclusions.56,57 Segal et al. 56 concluded that IFX and ADA induced remission overall in 53% of patients with chronic pouchitis (p < 0.001). Likewise, when examining three of the largest retrospective cohorts (for a total of 87 patients with pouchitis), Herfarth et al. 57 reported a partial and complete long-term response (52 weeks) of 45–58% in patients treated with IFX.
Endoscopic improvement of pouchitis has been an endpoint in several studies. Calabrese et al. 58 showed that eight of ten IPAA patients in a prospective cohort treated with IFX demonstrated complete endoscopic resolution of small bowel lesions and clinical remission at 6-month follow-up. Similarly, several case reports of pediatric IPAA patients also noted significant endoscopic improvement after IFX therapy.59–61
Prior to consideration of initiation of an anti-TNF, a thorough pre-colectomy medication history should be taken, ruling out intolerance, infusion reactions, or a history of antibodies to IFX or ADA.
Pre-colectomy lack of response to an anti-TNF, in the absence of a contraindication (immunogenicity, infusion reaction), does not necessarily preclude the use of anti-TNFs for IPAA after colectomy. 62 In one prospective study, out of 17 patients with de novo CD who had failed to respond to anti-TNFα agents before colectomy with IPAA, and who were treated with anti-TNFα therapy after surgery, 12 (71%) patients responded to treatment. On the other hand, another study suggested a lower likelihood of benefit in those with previous loss of response to anti-TNF. 63 This topic remains controversial and more studies are needed to develop a final consensus regarding anti-TNF treatment re-trials in chronic pouchitis.
Vedolizumab
Multiple studies have evaluated the role of vedolizumab (VDZ) in the treatment of chronic pouchitis.64–71 In a retrospective study of 20 patients with CADP or CARP, 55% of whom were anti-TNF experienced, the mean PDAI decreased from 10 to 3 (p < 0.01) at week 14. Clinical response to VDZ was defined as a decrease in PDAI by 3 points, which was achieved in 64% of patients. 64 There were no differences in the effectiveness of VDZ in anti-TNF naïve versus anti-TNF experienced patients, 64 as was shown in another small case series. 65 More recently, a total of 19 patients with CARP, defined as having ongoing symptoms of active pouchitis after 2 weeks of antibiotics, were given VDZ 300 mg at weeks 0, 2, 6 and 14. Mean mPDAI score improved in 32% of patients (p = 0.03). Some 74% had improvement in mPDAI and endoscopic findings with median improvement of 2 points (p = 0.031). 71 The largest study to date was a retrospective multicenter cohort study at five academic centers in the United States including 83 IPAA patients with endoscopically confirmed inflammation of the pouch. Some 87% of patients were previously treated with antibiotics for pouchitis, and 51% previously were treated with anti-TNFs for pouchitis. After at least 3 months follow-up, 71% of patients exhibited a clinical response, and 19.3% achieved clinical remission; 54% had endoscopic improvement while 17.6% achieved endoscopic healing. Patients who developed pouchitis symptoms within 1 year post-operatively were found to be less likely to respond to VDZ. 70 A randomized, double-blind, placebo-controlled, phase IV study evaluating VDZ for treatment of chronic pouchitis in 102 patients has been completed; reporting of results is anticipated shortly (EARNEST trial; NCT02790138).
Ustekinumab
Few case reports and case series have been published describing the use of ustekinumab (UST) for chronic pouchitis. In the largest series, Weaver et al. reported a retrospective, multicenter cohort’s experience with UST for treatment of Crohn’s-like disease of the pouch and chronic pouchitis. Clinical response and remission were assessed after 6 months of treatment. This was a highly biologic-experienced cohort. Of 56 patients, 73% were previously treated with anti-TNF, VDZ, or both after IPAA. Of 42 patients who completed 6 months of therapy, 83% of Crohn’s-like and chronic pouchitis patients demonstrated clinical response. Only 11% of those with Crohn’s-like disease of the pouch and none of those with chronic pouchitis were in clinical remission at 6 months follow-up. Of the responders with chronic pouchitis, 60% were able to stop all antibiotic therapy at 6 months follow-up. There was no difference in response comparing those who were biologic naïve and biologic experienced. Body mass index (BMI) at the time of induction was higher in UST non-responders compared with responders (mean BMI 26.3 versus 23.7; p = 0.033). Male sex was associated with nonresponse to UST (p = 0.014). 72
Similar smaller studies have shown clinical and endoscopic improvement with use of UST in chronic pouchitis. One study reported 50% of patients exhibited a clinical response at 13 months. Among these 13 patients, nine had an ulcerated surface area greater than 10% before UST treatment; after treatment with UST, only four patients still had an ulcerated surface area of greater than 10%. 73
In a retrospective study of 46 patients with chronic pouchitis, cuffitis, or Crohn’s disease of the pouch treated with UST, physicians reported clinical response of 80% at 2–4 months after start of treatment, 59% with improvement in PDAI, 53% improvement in inflammation on pouchoscopy, and 31% with antibiotic cessation within 12 months. 74 Smaller case reports have also shown clinical75,76 and endoscopic response.73,75 The SOCRATES study is an open-label, multicenter pilot study in Belgium studying UST for treatment of relapsing and chronic antibiotic refractory pouchitis after an initial 4 weeks of ciprofloxacin or metronidazole (NCT04089345).
Tacrolimus
Tacrolimus, a calcineurin inhibitor, acts by suppressing T-cell proliferation and activation. Data are limited to small case series describing the treatment of chronic inflammatory complications of the pouch with tacrolimus.77–80 One study reported use of oral tacrolimus (0.1 mg/kg daily) in two patients with chronic pouchitis. Both patients achieved clinical remission, with normalization of inflammatory laboratory parameters, and absent acute inflammatory changes on imaging studies for a period of at least 8 weeks. 81 A single study described the use of tacrolimus enemas in patients with chronic pouchitis. Ten patients with chronic pouchitis, defined as ⩾3 episodes per year or persistent symptoms requiring long-term continuous antibiotics, were given daily tacrolimus enemas (0.08 mg/kg per 100 mL aqueous enema solution). After 8 weeks, the mean PDAI score decreased significantly from 15.9 points to 7.8 points (p < 0.01) with 70% in clinical remission with a clinical PDAI subscore of 0. None of the patients achieved endoscopic remission but did have a significant improvement in endoscopic appearance (p < 0.01). There were no reported adverse outcomes. 77 The reported improvement in pouchitis with tacrolimus oral and topical formulations is intriguing, but more studies are warranted to explore the efficacy of tacrolimus for treatment of chronic inflammatory conditions of the pouch.
Tofacitinib
Tofacitinib inhibits the JAK-STAT signaling pathway and reduces the production of inflammatory cytokines. Though tofacitinib has been approved for the treatment of ulcerative colitis, only small cases series have evaluated its use in chronic inflammatory disorders of the pouch. One case reported improved clinical and endoscopic healing with the use of tofacitinib in a patient with refractory chronic pouchitis who had previously failed conventional therapy including antibiotics, anti-TNF therapy, and VDZ. 82 Another case series of two patients treated with tofacitinib 10 mg twice daily for treatment of Crohn’s-like disease of the pouch reported endoscopic improvement in one patient and complete healing in the other patient. When decreasing the dose to 5 mg twice daily, symptoms recurred in both patients but again resolved when increasing back to 10 mg twice daily. 83 On the other hand, in a case series of seven patients with chronic pouchitis, treatment with tofacitinib failed to demonstrate improvement in PDAI symptom sub-scores. 84 At this time, more data are needed before making a recommendation for the use of tofacitinib in the treatment chronic inflammatory disorders of the pouch.
Alicaforsen enema
Alicaforsen, a human ICAM-1 anti-sense oligonucleotide, is a therapeutic target in inflammatory bowel disease and pouchitis. ICAM-1, a transmembrane glycoprotein expressed on the surface of intestinal epithelial cells and vascular endothelial cells, 85 contributes to leukocyte adhesion, migration, stimulation, and intestinal T-lymphocyte trafficking. 86
In 2004, Miner et al. conducted an open-label, uncontrolled study in 12 patients with chronic refractory pouchitis (defined as failing alternative therapy with ongoing symptoms after 4 weeks). Patients were treated with 240 mg of alicaforsen enemas nightly for 6 weeks. At week 6, 58% of patients were in remission with an average decrease in PDAI score by six points. 87 A second study evaluating alicaforsen enemas studied 13 patients with chronic antibiotic refractory pouchitis. Patients were evaluated at baseline and after a 6-week course. Both clinical and endoscopic disease activity were significantly reduced. However, after a median of 16 weeks, 82% of patients experienced recurrent pouchitis.88,89 In 2019, a phase III, randomized, double-blind, placebo-controlled trial evaluating alicaforsen enemas for treatment of active, chronic antibiotic refractory pouchitis did not reach the primary efficacy endpoints of reduction in bowel frequency at week 10 and endoscopic remission. Alicaforsen is currently not approved for use.
Fecal microbiota transplant
Microbial dysbiosis is thought to be an important factor in the development of pouchitis. Several small studies have studied the role of fecal microbiota transplant (FMT) in the treatment of chronic pouchitis. One prospective, placebo-controlled, double-blind trial of patients with CADP received a single endoscopic FMT followed by daily FMT for 2 weeks. The study was ended prematurely due to failure of response for the first six patients enrolled. 90 Several other studies did not show a significant improvement in PDAI, endoscopic, or histologic scores after FMT.91–93 However, Selvig et al. 91 did report an improvement in bowel frequency (from 9 to 7 bowel movements per day; p = 0.03). Two small case series reported improved clinical PDAI, endoscopic appearance, and fecal calprotectin levels with FMT in chronic pouchitis.94,95 In a systematic review of nine studies (44 patients) of FMT used to treat chronic pouchitis, clinical response after FMT was reported in 14 (32%) patients and clinical remission in 10 (23%) patients. 96 Most studies included were case series or case reports. No serious adverse outcomes were reported, but minor self-limited symptoms such as nausea, bloating, abdominal pain, fever, dizziness, and fatigue were reported.
At present, FMT cannot be recommended for medical management of chronic pouchitis, but the promise of correcting the dysbiosis associated with the ileal pouch warrants further study. Certainly, further research regarding FMT in pouchitis, as well as inflammatory bowel disease (IBD) in general, may lead to novel therapeutic applications. Multiple studies are ongoing (NCT02049502, NCT0242836, NCT03545386, NCT04100291).
Cuffitis
Cuffitis is inflammation of the remnant rectum known as the “rectal cuff.” Cuffitis, in its classic form, is a form of ulcerative colitis at the cuff; however, non-classically could be caused by Crohn’s disease of the pouch, anastomotic separation, ischemia, or prolapse. 97 First-line therapy for the management of the classic cuffitis is topical mesalamine suppositories. 98 One small study showed mesalamine suppositories at 500 mg given twice daily decreased the number of bloody bowel movements in an open-label study of 14 patients with cuffitis. 98 In a case series of 120 patients with cuffitis, 40 patients (33.3%) had mesalamine/steroid-responsive cuffitis; 22 (18.3%) had mesalamine/steroid-dependent cuffitis, and 58 (48.4%) developed mesalamine/steroid-refractory cuffitis after a median follow-up of 6 years.99
Hydrocortisone or budesonide topical therapy can be used as second-line therapy for cuffitis if no improvement on topical mesalamines. Treatment with topical steroids may be prescribed for 2–4 week courses but should not be used chronically given long-term risks of steroid use.
Lifestyle modifications
In addition to medical treatments for pouchitis, patients frequently inquire about lifestyle modifications they may pursue to prevent pouchitis, either in lieu of or to complement medical therapy. Here we discuss a few common lifestyle modifications studied in pouchitis.
Smoking
Smoking in ulcerative colitis has been shown to be protective against development of ulcerative colitis and has even been studied as an intervention in refractory ulcerative colitis. 100 The role of smoking in pouchitis, however, is unclear. Previous studies have identified smoking cessation as a risk factor for pouchitis.101,102 In a study comparing nonsmokers, former smokers, and current smokers, smoking was surmised to be potentially protective against development of pouchitis. Pouchitis occurred in 18 of 72 (25%) nonsmokers, 4 of 12 (33%) former smokers, and 1 of 17 (6%) smokers. 103 On the other hand, a prospective study demonstrated that former smoking or active smoking can increase the risk of pouchitis. 104 A meta-analysis of 15 studies evaluating current, former, or never smokers found no association of smoking status with risk of development of pouchitis in patients with an IPAA. 16 Patients should be counseled that smoking is not recommended to prevent or treat pouchitis.
NSAID use
NSAIDs can cause bowel damage and exacerbate disease activity in IBD.105–107 NSAIDs have been identified as a risk factor for pouchitis.12,108 In a cohort of 17 IPAA patients with pouchitis with chronic daily use of NSAIDs (more than 6 months), withdrawing NSAIDs led to a significant reduction in mean PDAI scores (3.6 points; p < 0.02) and an improvement in global QOL scores (p < 0.05.) 109 However, in an IBD Partners study of IPAA patients, NSAID use within 6 months was not found to be associated with poorer patient-reported outcomes. 110 In a small case series of 27 patients with ulcerative colitis, Crohn’s disease, or pouchitis who used selective COX 2 inhibitors (celecoxib or rofecoxib) for a median duration of 9 months, 14 (52%) reported clinical benefit with only two (7%) experiencing disease worsening. 111
In general, we advise routine avoidance of oral NSAIDs in patients with a history of pouchitis. However, limited topical or oral NSAIDs are considered acceptable for intermittent use as needed if other analgesics are ineffective. Chronic NSAID use is not recommended.
Diet
Diet plays a role in the gut microbiome, 112 innate immunity, and adaptive immunity. 113 Data are limited regarding the role of diet and the development of pouchitis. In patients without pouchitis, a diet low in FODMAPs has been shown to decrease stool frequency. 114 However, in patients with pouchitis, a low FODMAP diet did not change stool frequency.
A more recent prospective study evaluated nutrition and fecal microbiota in 172 patients with ulcerative colitis who underwent IPAA. Patients in the lowest tertile of fruit consumption had higher rates of pouchitis (30.8% versus 3.8% p = 0.03). Fruit consumption correlated with microbial diversity (p = 0.002). 115 Previous studies have also shown an increase of fruit consumption potentially decreasing the risk of pouchitis. 116
Adherence with the Mediterranean diet has been associated with decreased fecal calprotectin in IPAA patients. The Mediterranean diet was associated with higher dietary fiber and antioxidant intake. Subgroup analysis suggested that adherence to the Mediterranean diet was inversely associated with onset of pouchitis. 117
In general, based on expert opinion, we recommend small, frequent meals focusing on lean protein, moderate carbohydrates, and soluble fiber for IPAA patients. Limiting high FODMAP foods is recommended for symptomatic improvement. Raw fruits and vegetables are recommended, if tolerated. IPAA patients are encouraged to avoid or limit high-fat foods such as fried foods and fatty meats. Soluble fiber supplements can be used to bulk stools, except in patients with demonstrated poor pouch compliance and small capacity on ano-pouch manometry. More studies are needed to determine the optimal diet for IPAA patients.
Conclusions
The mainstay of treatment for acute pouchitis remains antibiotics. However, for patients with chronic pouchitis who cannot take or are refractory to antibiotics, more traditional IBD treatments such as corticosteroids, biologics, small molecule drugs, and others can be considered. Lifestyle factors may play a role in the development or management of pouchitis. Physicians should counsel IPAA patients with pouchitis on both medical treatments and lifestyle adaptations.
