Background:
Thiopurine methyltransferase (TPMT) plays a significant role in the metabolism of thiopurines, and, for patients with inflammatory bowel disease (IBD), it is useful to perform TPMT genotyping prior to azathioprine (AZA) treatment. In this study, we determined TPMT gene polymorphisms in a cohort of IBD patients in Latvia.
Methods:
DNA samples were obtained from 244 IBD patients, and qPCR was performed for detection of rs1800462, rs1800460, and rs1142345 single-nucleotide polymorphisms (SNPs). Three common, non-functional TPMT alleles (TPMT*2, *3B, and *3C) were identified (women, 51%; men, 49%). TPMT*2, *3A, *3B, and *3C allelic variants detected using qPCR were consistent with restriction fragment length polymorphism (RFLP) data.
Results:
Among patients, 78% had ulcerative colitis and 22% had Crohn’s disease, with 93.9% of the former carrying a wild-type homozygous TPMT*1/*1 genotype and 6.1% carrying heterozygous genotypes. The most frequent polymorphisms were TPMT*1/*3A (5.3%: two variants: TPMT*3B and TPMT*3C), TPMT*1/*3C (0.4%), and TPMT*1/*2 (0.4%). None of the patients carried a TPMT*3B polymorphism and no patients were homozygous for any mutation.
Conclusion:
This is the first study to identify TPMT gene polymorphisms in adult IBD patients in Latvia. The results indicate that the frequency of common TPMT alleles is similar to that of other European populations.
Introduction
The number of patients with inflammatory bowel disease (IBD) is increasing worldwide, and, in this regard, Latvia is no exception. Azathioprine (AZA), the prodrug of mercaptopurine (MP), is used widely for the treatment of IBD.1−3 AZA is characterized by a glucocorticoid-sparing effect, which is beneficial to patients who are unable to maintain IBD remission using glucocorticoids.
4
However, adverse effects have been recorded in approximately 10% of patients using AZA for the treatment of IBD. Among these patients, approximately 10−20% need to discontinue treatment due to these side effects.2,5,6 Most of the adverse events occur within the first 3 months of treatment.
7
It has been observed that, at 1 and 3 months, 26% and 93%, respectively, of patients on a full dose of AZA develop complications,
8
with the most commonly reported side-effects being gastrointestinal intolerance, hepatotoxicity, infections, and bone marrow toxicity.1,7,8 Additional complications include pancreatitis, malignancies, and allergic skin reactions.
AZA is initially metabolized in the liver via a non-enzymatic pathway to 6-MP. Subsequently, 6-MP is converted to its metabolites via an intracellular multi-enzymatic process involving three enzymes: hypoxanthine phosphoribosyl transferase (HPRT), thiopurine S-methyltransferase (TPMT), and xanthine oxidase (XO). The process whereby thiopurine S-methyltransferase is metabolized in the human body has yet to be fully established, and severe life-threatening bone marrow toxicity can result from the excess production of a drug-derived thioguanine nucleotide (TGN).3,9,10 In terms of the therapeutic effect of AZA, it appears that 6- thioguanine (6-TG) mediates the majority of this drug’s effects,6,10,11 and accumulation of 6-TG can lead to AZA-associated adverse effects; for example, the incorporation of 6-TG into DNA can initiate delayed cytotoxicity and induce apoptotic cell death by inhibiting intracellular signalling pathways.
10
TPMT is required for the detoxification of 6-TG through S-methylation.
12
Thus, if the thiopurine dosage is not administered based on an individual’s TPMT activity level, toxicity due to TPMT deficiency can result in treatment interruption.13−15 In contrast, a higher than the normal TPMT activity may render patients refractory to conventional dosages of AZA.
16
Thus, TPMT plays a significant role in the metabolism of thiopurines, with low TPMT activity being associated with altered thiopurine metabolism, overproduction of cytotoxic metabolites, and myelosuppression. In patients with IBD, TPMT genotyping can be performed prior to treatment to evaluate the treatment risks; however, this does not exclude that patients may or may not tolerate thiopurines. Current guidelines for thiopurine treatment mandate regular hematologic monitoring to detect leukopenia. In this study, we sought to identify TPMT gene polymorphisms in a cohort of IBD patients in Latvia.
Patients and methods
A total of 244 IBD patients identified from the Genome Database of the Latvian Population were included in the study after obtaining informed consent and the completion of health and heredity questionnaires, as described previously.
17
The study was performed in accordance with the Declaration of Helsinki and was approved by the Central Medical Ethics committee of Latvia (protocol no. 22.03.07/A7, no. 3/18-02-21).
From each IBD patient, we collected 20 ml of blood in an ethylenediaminetetraacetic acid (EDTA)-containing tube and 7 ml of blood in a clot-activator tube. Serum, plasma, and white blood cells were separated within 2 days of blood collection. DNA was extracted using the phenol–chloroform extraction method. Aliquots of plasma, serum, white blood cells, and DNA were stored at −80°C, avoiding cycles of re-freezing and thawing cycles.
TPMT genotypes were determined by real-time polymerase chain reaction (qPCR) using TaqMan Fluorescent Probes (TaqMan Drug Metabolism Genotyping Assays) for detection of the rs1800462, rs1800460, and rs1142345 single-nucleotide polymorphisms (SNPs). The three common non-functional TPMT alleles (TPMT*2, *3B, and *3C) were determined. The PCR reactions amplified probes binding to DNA copies at sites of the TPMT gene that might contain polymorphisms and emitted fluorescent signals. All polymorphisms were analyzed using a StepOneTM Software version 2.3 Real-Time PCR System. TPMT*2, *3A, *3B, and *3C allelic variants detected by qPCR were confirmed by performing PCR-restriction fragment length polymorphism (RFLP) analysis and allele-specific PCR. The results of qPCR and the alternative PCR assays were found to be entirely consistent. The DNA fragments were separated and analysed in 2.5% agarose gels and visualized by staining with ethidium bromide.
The following data were collected for each patient on the basis of responses to the health and heredity questionnaires: demographics, gender, age, nationality, and the region of Latvia where the patient was born (Vidzeme, Kurzeme, Latgale, or Zemgale). The questionnaires assessed the patient’s medical history, lifestyle, and other important factors, such as smoking status, physical activity, possible risk factors of anamnesis, allergic reactions, medication intolerances, regular medications, and comorbidities.
Study participants
Our study population comprised 244 adult IBD patients, with an almost equal gender ratio (51% women and 49% men). The mean age of the participants was 43 ± 16 years. Among these patients, 78%, with a median age of 41 years (Q1−Q3 = 29.8−54.3), had ulcerative colitis, and 22%, with a median age of 43 years (Q1−Q3 = 30.8−55.0), had Crohn’s disease (p = 0.57). Women comprised 47% and 63% of the ulcerative colitis and Crohn’s disease groups, respectively (Table 1).
Statistical analysis
Continuous variables are presented as the median and interquartile range (Q1−Q3) and were compared using the Mann–Whitney test. The categorical variables are expressed as the frequency and percentage and were compared using Pearson’s chi-squared test with Fisher’s exact test or Cramer’s V effect size as appropriate. Odds ratios (ORs) are presented with 95% confidence intervals (CI). All statistical analyses were performed using SPSS Statistics version 23.0. A p-value of < 0.05 was considered statistically significant.
Results
TPMT genotyping
TPMT alleles were identified in all 244 patients, among whom we found that 93.9% were carrying a wild-type homozygous TPMT*1/*1 genotype and 6.1% were heterozygous and harbored polymorphisms (4.9% of whom had ulcerative colitis) (Table 2). However, we found that TPMT polymorphisms were not consistently associated with IBD (OR: 1.15, 95% CI: 0.31−4.28, p = 0.99). The most frequent polymorphisms (5.3%) were TPMT*1/*3A genotype with TPMT*3B and TPMT*3C alleles. Only two patients had TPMT*1/*3C and TPMT*1/*2 genotypes independently. We were unable to detect any patients carrying the TPMT*3B polymorphism and no patient was found to be homozygous for any mutation.
Association of TPMT polymorphisms with different clinical factors
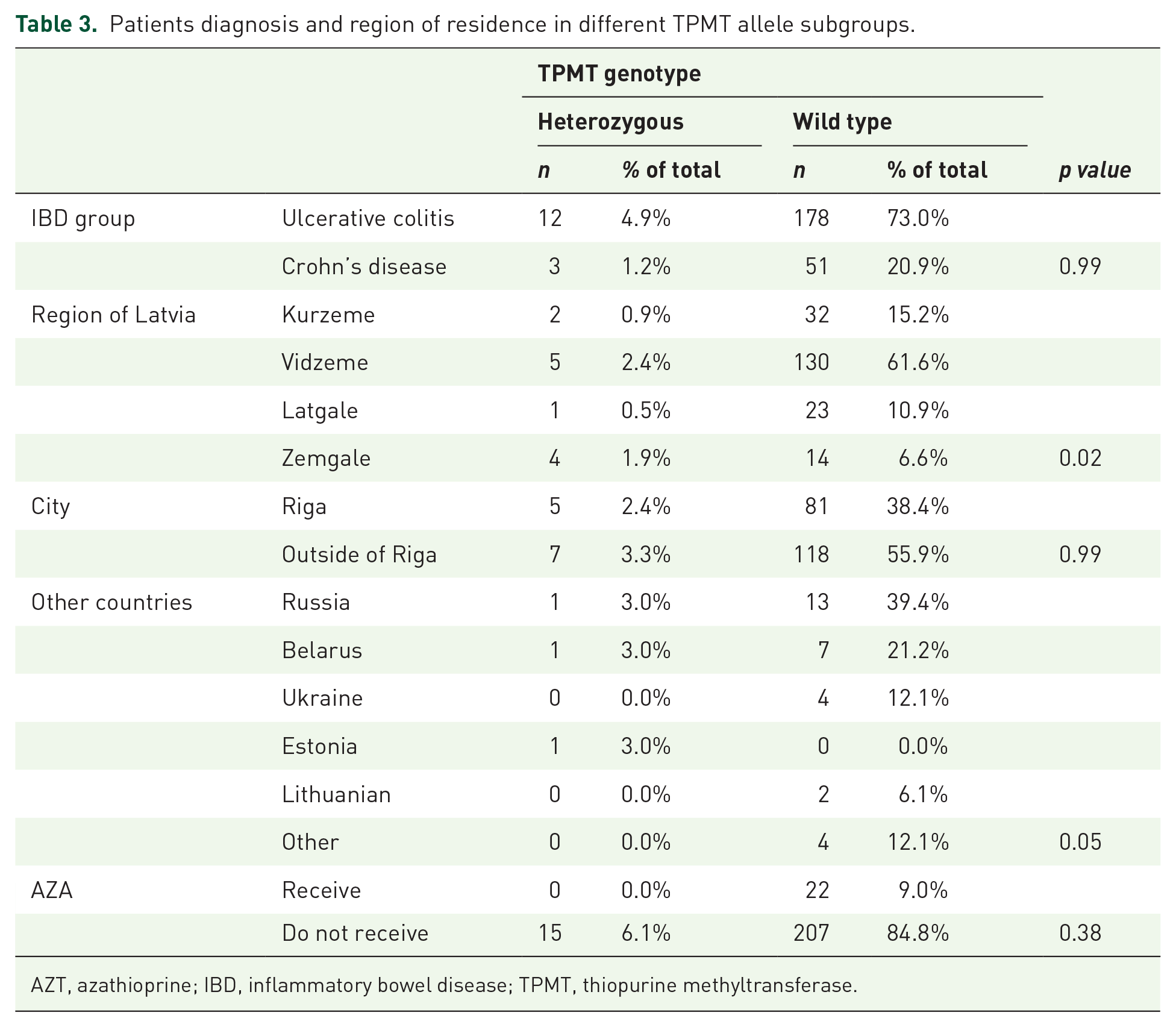
We examined whether different clinical factors were associated with TPMT polymorphisms (Table 3). We accordingly observed no significant association between gender and TPMT polymorphisms (p = 0.21). The majority of the patients with TPMT polymorphisms were Caucasians and were born in Latvia outside the capital Riga. A moderate association was found for patients born in the Vidzeme region (Cramer’s V = 0.2). Fisher’s exact test showed a nominal statistical association between TPMT polymorphisms and the possible risk factors of anamnesis (working with chemicals, dust, aerosols, and lacquers, and working in chemical factories) (p = 0.04).
Among all the included patients, 80% regularly took IBD medications and 18% had allergic reactions to antibiotics, analgesics, and other drugs. Although no statistical association was found between TPMT polymorphisms and drug allergy (p = 0.78), 0.8% of all patients with positive TPMT polymorphisms had previously used AZA and had experienced adverse drug reactions (ADRs), such as myelosuppression and gastrointestinal intolerance. These patients had a TPMT*1/*3A genotype. Myelosuppression was objectively characterized as decreased white blood cells (neutropenia) and gastrointestinal intolerance was characterized as vomiting, nausea and stomach cramps. Furthermore, we found that 15% of all the patients were smokers, with a median smoking duration of 14 years (Q1−Q3 = 10.0−30.0), whereas 28% of the patients were previous smokers, with a median smoking duration of 10 years (Q1−Q3 = 4.0−20.0). However, Fisher’s exact test did not reveal any statistical associations between TPMT polymorphisms and smoking status (OR: 1.19, 95% CI: 1.12−1.26, p = 0.14).
Discussion
Different studies recommend that the TPMT status of patients should be determined prior to the commencement of thiopurine therapy.1,2,10,14,18 This can be achieved by one of two methods, namely, by determining the TPMT phenotype by estimating TPMT enzyme activity in the circulating red blood cells (RBC), or through genotyping of known TPMT variants associated with enzyme deficiency using PCR.14,15,18,19 The activity of the TPMT enzyme is related mainly to rs1800462, rs1800460, and rs1142345 SNPs, which are inherited co-dominantly.10,20 In terms of AZA toxicity, large inter-individual differences are observed due to the genetic heterogeneity of the TPMT gene, which has more than 40 reported allelic variants.18,21 Nevertheless, three main patterns of TPMT enzyme activity can generally be distinguished: (a) homozygous patients with two mutant non-functional TPMT gene alleles and low TPMT activity, (b) heterozygous individuals with one functional and one non-functional allele and intermediate TPMT activity, and (c) homozygous wild-type (normal) individuals with two functional alleles and normal or high TPMT activity.10,20–22
Although known TPMT alleles tend to vary among different ethnic groups, four specific non-functional alleles have been identified as being more prevalent, namely, TPMT*2, TPMT*3A, TPMT*3B, and TPMT*3C. It is believed that these alleles account for between 80% and 95% of observed decreases in TPMT enzyme activity. Accordingly, these four alleles tend to be routinely targeted in most of the genotyping assays.3,20,21 In general, the most frequently encountered allele in all populations is TPMT*3A, followed by TPMT*3C,4,6,10,23 which is consistent with the findings of the present study.
The most common TPMT genotype in Caucasian populations is homozygous for the wild-type TPMT gene. Several studies have shown that 85–95% of patients have a wild-type genotype (93.9% in this study). In most populations, approximately 10% of individuals are heterozygotes and a further 0.3% carry homozygous variants of the non-functional TPMT alleles.14,20,21,24,25 In the present study, none of the participating patients were identified as being homozygous for any mutation. TPMT genotyping has been demonstrated to show high sensitivity,19,23,26 with reported sensitivities and specificities of 88.9% (81.6–97.5%) and 99.2% (98.4–99.9%), respectively. Comparatively, the approximate sensitivity and specificity of TPMT phenotyping are 91.3% (86.4–95.5%) and 92.6% (86.5–96.6%), respectively.
19
Our study data indicated that 9% of IBD patients were undergoing thiopurine therapy at the time they were included in the study. This percentage does not include patients who have used thiopurines before and stopped, or those who did not use thiopurines at the start of the study. Indeed, most of the patients included in the study had not yet started thiopurine treatment.
With regard to the cost-effectiveness of genotype-led treatment, very recent data published by Sluiter et al. showed that genotype-guided thiopurine treatment in IBD patients reduces the risk of ADR among patients carrying a TPMT variant, without increasing overall healthcare costs and resulting in a quality of life comparable with that of standard treatment.
27
The Clinical Pharmacogenetics Implementation Consortium (CPIC) has published recommendations for AZA dosing based on TPMT genotype. In patients with two functionally active TPMT alleles, the activity of the TPMT enzyme is in most cases normal or high. For these patients, the CPIC recommends the initiation of AZA therapy at normal dosage and thereafter adjusting the dose based on disease-specific guidelines. In heterozygous patients, TPMT enzyme activity is intermediate, and these patients are at an increased risk of AZA-induced myelosuppression depending on the dose. The CPIC accordingly recommends that, for heterozygous patients, AZA treatment should be initiated at 30–70% of the target dose and to titrate the dose based on tolerance. For homozygous patients with two non-functional TPMT alleles and consequently low TPMT enzyme activity, an alternative treatment is suggested.1,8,21 Interestingly, in this regard, a previous study that sought to determine a safe dosage of AZA in patients with intermediate or low TPMT enzyme activity, found that TPMT genotyping could facilitate a reduction in adverse haematological effects by as much as 89%.
28
Given that TMPT can vary quite extensively, depending on age, sex, ethnicity, red blood cell lifespan, red blood cell transfusion, leukemia, and treatment-related factors, these various factors should ideally be taken into consideration when interpreting the results of TPMT activity analyses.20,22 In addition, assessments of TPMT activity may require repeated estimations, as treatment with AZA itself can induce an increase in TPMT activity. Similarly, other medications, such as 5-aminosalicylates, can reversibly inhibit TPMT activity.29,30 Therefore, TPMT genotyping is considered to be a more precise and reliable method.
8
Typically, the overall concordance between TPMT genotype and phenotype is between 90% and 95%, although some studies have reported genotype–phenotype concordance values of approximately 60–70% in patients with low TPMT activity, whereas those for heterozygous patients are approximately 70–86%.10,20
However, even though the identification of TPMT genotype and/or phenotype can make a valuable contribution towards identifying patients with a higher risk of developing bone marrow toxicity, additional therapeutic drug monitoring is advised for those patients on AZA therapy.
11
Routine laboratory tests consist of complete blood counts (CBCs), liver chemistries, platelet counts (PC), and creatinine clearance. Some authors recommend monitoring CBC and PC at weekly intervals during the first month of AZA treatment, followed by twice monthly monitoring during the second and third months, and monthly checks thereafter. Furthermore, liver function tests should be performed at 3-monthly intervals.
19
If, however, signs of myelosuppression develop, AZA therapy should be immediately discontinued.
A recent publication by Ribaldone et al. describes a meta-analysis investigating the associations between TPMT polymorphisms and AZA-induced adverse events in patients with autoimmune diseases.
31
The results showed that TPMT polymorphisms were significantly associated with AZA-induced overall adverse effects, bone marrow toxicity, and gastric intolerance. However, the subgroup analysis according to ethnicity showed a significant association between TPMT polymorphisms and AZA-induced bone marrow toxicity in Asian populations, but not in Caucasian populations. The authors concluded that TPMT polymorphisms can explain a variable proportion but not all episodes of AZA-related adverse events, and, furthermore, a normal TPMT genotype cannot exclude the development of side effects.
31
Thus, TPMT genotyping before starting AZA therapy cannot replace the current practice of periodic monitoring of white blood cell count. However, this is a challenge that requires additional future research, particularly as some severe toxicities leading to life-threatening conditions remain unexplained.
It is important to note that the randomized controlled trial, TOPIC, conducted by Coenen et al. showed that pretreatment TPMT genotyping is also relevant for patients who are heterozygous for a variant in TPMT, not only for homozygous carriers of a genetic variant in TPMT.
32
The results of the TOPIC trial showed no overall effect of pretreatment TPMT screening followed by personalized dosing on hematologic ADRs. However, in combination with other literature, the TOPIC study shows that pretreatment TPMT screening followed by personalized dosing reduces the risk of leukopenia in patients carrying a genetic variant in TPMT and indicates that pharmacogenetic TPMT testing should be used as standard care to individualize thiopurine treatment of IBD patients.
32
Thiopurines still remain very effective in inducing and maintaining long-term remission in up to 70% of patients with IBD, and it is important to remember that patients with allelic variants should not be denied the therapeutic option of AZA, as they may tolerate this drug.
31
Conclusion
In conclusion, our results indicate that the frequency of common TPMT alleles is similar to those of other European populations. In this study we verified the homozygous wild-type TPMT*1/*1 genotype as the most frequently encountered genotype in ulcerative colitis and Crohn’s disease patients’ groups, and that TPMT*3A is the most prevalent polymorphism in the study population. Further, we noted the absence of both the TPMT*3B polymorphism and homozygous variant TPMT genotypes in this population. To our knowledge, this is the first study to identify TPMT gene polymorphisms in adult IBD patients in Latvia. We recommend that TPMT genotyping should be prioritized in specialized IBD centres and risk group patients for the prediction of thiopurine-induced adverse drug reactions among IBD patients, and that this genotyping should be applied with respect to personalized therapy. In the future, additional genotyping of patients experiencing adverse effects due to thiopurine treatment will be required to identify potential gene/allele–dose effects.