Abstract
Background:
Linaclotide is approved for the treatment of moderate-to-severe irritable bowel syndrome (IBS) with constipation (IBS-C) in adults. This study aimed to assess factors predictive of a clinical response and improvements in non-IBS symptoms with linaclotide treatment in a Spanish patient population.
Methods:
In this open-label phase IIIb study, patients with moderate-to-severe IBS-C received linaclotide 290 μg once daily for 12 weeks. The primary endpoint was clinical response at week 12, defined as >30% reduction in IBS symptom severity score (IBS-SSS) or IBS-SSS <75 plus self-reported response of feeling ‘better’ or ‘much better’ versus the baseline. Digestive nonintestinal and extra-digestive symptom scores were assessed. Baseline characteristics and week 4 clinical response were assessed as predictors of week 12 clinical response.
Results:
A total of 96 patients were eligible; 91 were female and the mean age was 47.4 years. Mean (SD) baseline IBS-SSS was 371 (72.5). In the intention-to-treat and per-protocol populations, 22.9% and 31.7% were clinical responders at week 4, respectively, and 25.0% and 36.7% were clinical responders at week 12. Digestive nonintestinal and extra-digestive symptom scores were significantly improved at weeks 4 and 12. Baseline characteristic was not associated with week 12 clinical response; however, clinical response at week 4 was predictive of response at week 12 (OR: 6.5; 95%IC: 2.1–19.8). The most common adverse event was diarrhea inclusive of loose or watery stools (35.4%).
Conclusions:
Linaclotide improves IBS-C symptoms, including digestive nonintestinal and extra-digestive symptoms. A clinical response at week 4 may predict response at week 12.
Introduction
Irritable bowel syndrome (IBS) is a common and chronic gastrointestinal (GI) disorder characterized by recurrent abdominal pain and altered bowel movements. 1 IBS is a disorder of gut–brain interaction 2 and is subtyped by its predominant stool patterns including IBS with constipation (IBS-C), IBS with diarrhea, IBS with a mixed stool pattern, and unclassified IBS.1,3
IBS affects approximately 11% of the global population and 11.5% in Europe, including an estimated 10% of people in Spain.4,5 IBS-C is estimated to account for approximately one-third of all IBS cases, and is one of the most common reasons for patient visits to both primary care physicians and gastroenterologists. 6 Mean overall costs for IBS-C in a United States (US) community center analysis were US$11,406 over a 2-year period, and in Europe, the mean annual overall costs to the healthcare system for moderate-to-severe IBS-C were €4639.7,8
While some patients with IBS-C may experience mild or intermittent symptoms, for some, these symptoms are severe and debilitating. IBS is also associated with a significant GI and non-GI comorbidity burden, 9 which contributes to the significant impact of IBS-C on patients’ health-related quality of life. As a result, IBS-C is associated with a high economic and humanistic burden due to greater use of healthcare resources and work productivity loss, including presenteeism and activity impairment.10,11
IBS-C management options include nonpharmacological measures such as dietary modifications, increasing physical activity, and psychological therapy. 12 Pharmacological therapy of IBS-C includes antispasmodics, over-the-counter medications (e.g. laxatives), as well as approved prescription medications such as lubiprostone and linaclotide.12–14 In two phase III trials, linaclotide significantly improved pain, constipation, and abdominal bloating in patients with IBS-C and is therefore approved by the European Medicines Agency for moderate-to-severe IBS-C in adults.15,16
IBS-C is a chronic condition requiring long-term management, and patient dissatisfaction with available treatment options or lack of response to treatment can result in negative outcomes such as repeated physician consultations, use of multiple treatments (over-the-counter or prescription medications), medication switching, use of unproven medications, or unnecessary surgery.13,17 Therefore, it is important to understand the effectiveness of any treatment over time. The ability to identify patients who will or will not respond to a treatment, both in the short and long term, would be of substantial clinical value and will be important in establishing reasonable patient, prescriber, and payer expectations of the effectiveness of a treatment. Current treatment recommendations for linaclotide suggest a first follow-up visit 4 weeks after initiation, with a second visit at 12 weeks and periodically thereafter, depending on the individual patient’s needs.18,19
The aim of this study was to identify factors that may help predict a short- and long-term clinical response to linaclotide and to assess the impact of linaclotide on IBS-C symptoms, including GI, digestive nonintestinal, and extra-digestive symptoms after 12 weeks.
Methods
Study design
This was an open-label, uncontrolled, single-arm phase IIIb clinical trial conducted at 12 hospital centers across Spain from November 2014 to February 2016. Physicians at the hospital centers invited patients who met all inclusion and no exclusion criteria to participate. Written informed consent was obtained from all patients or their legal representatives before any procedure or participation in the trial commenced. Following consent, patient information was entered into an electronic case report system. Linaclotide 290 µg capsules were administered once daily for 12 weeks, in accordance with its approved indication, and taken at least 30 minutes before meals. 18
Patients completed a 4-week screening period (weeks −4 to 0) and a baseline visit at week 0, followed by study visits for assessment of efficacy and safety at weeks 4, 8, and 12 (Table S1). After completion of the 12-week treatment period, an additional safety visit was scheduled at week 16. Treatment adherence was evaluated through capsule counting. All laxatives, enemas, and suppositories were stopped during the screening period and allowed as rescue medication for a maximum of 12 days during the treatment period. Medications that were not permitted included opiates, spasmolytic drugs, and herbal therapies, which included probiotics. Physicians also stopped medications such as antihistamines, proton pump inhibitors, antacids, selective serotonin reuptake inhibitors, and calcium channel blockers during screening or immediately before study entry. However, these medications could be restarted, if clinically indicated.
The study protocol was conducted in accordance with the Helsinki Declaration, approved by the World Medical Association (version revised at the 59th World Medical Association General Assembly, Seoul, Republic of Korea, October 2008), Oviedo Convention (1977), Good Clinical Practices (GCP of ICH, 1996), and current regulatory requirements. The study protocol was approved by the ethics committees of all study centers and by the Spanish Agency of Medicines and Health Products. The trial protocol was registered with EudraCT (EudraCT Number: 2013-001248-67).
Study population
Eligible patients were aged ⩾18 years and had a diagnosis of moderate-to-severe IBS-C based on Rome III criteria 20 and a baseline IBS symptom severity score (IBS-SSS) of >175. Additionally, patients must have shown blood analysis without anemia in the previous 6 months and, if aged >50 years, must have had a normal colonoscopy in the previous 5 years.
Exclusion criteria included diagnosis of any chronic digestive disease other than IBS, any abdominal surgery (except hysterectomy, cholecystectomy, and appendectomy), a diagnosis of cancer or chronic inflammatory disease, or a diagnosis of other disorders that could affect IBS (cardiologic, neurologic, pneumologic, endocrinologic, or renal disorders). Patients taking opioids, anticholinergics, or calcium antagonists were also excluded.
Study assessments
IBS severity was measured using the IBS-SSS, a validated measure consisting of five domains [abdominal pain frequency, abdominal pain severity, abdominal distention (bloating) severity, satisfaction with bowel habit, and impact of IBS on daily activities], scored from 0 to 100 on a visual analog scale (VAS), producing an overall reported score on a scale of 0–500, with higher scores representing more severe symptoms. Disease severity was stratified by IBS-SSS score as follows: normal: <75; mild: 75 to <175; moderate: 175 to <300; severe: ⩾300. 21 A reduction in IBS-SSS score of ⩾50 points is considered to represent a clinically meaningful improvement. 21 A validated Spanish version of the IBS-SSS is available which, in addition to the VAS, features a numerical scoring system for each domain on a scale of 0–12, with higher scores indicating more severe symptoms. 22 The IBS-SSS VAS (0–100) was used to evaluate individual symptom scores for abdominal pain severity, abdominal bloating severity, and satisfaction with bowel habit, where higher scores indicate greater pain/bloating severity and lower satisfaction with bowel habit.
Global improvement was evaluated through the question: ‘Overall, how do you feel about your IBS symptoms compared with how you felt before beginning treatment?’, using a five-point Likert scale with five response options ranging from ‘much worse’ to ‘much better’.
Digestive nonintestinal symptom score was measured using a validated Spanish version of the Rome III questionnaire.20,23 It consists of six questions regarding symptom frequency (heartburn and acid reflux, postprandial fullness, early satiety, epigastric pain, nausea, and vomiting) in the last 4 weeks, scored from 0 (never) to 3 (most of the time, ⩾3 days weekly). The total score for digestive nonintestinal symptoms was the sum of these responses.
Extra-digestive symptom score was measured using the patient health questionnaire 12 somatic symptom scale, 24 which consists of questions on extra-digestive symptoms such as back pain, headache, chest pain, dizziness, fainting spells, shortness of breath, joint pain, feeling tired, pain or problems during sex, menstrual cramps, and trouble sleeping. Bothersomeness of each symptom is scored on a three-point scale, where 0 is ‘not bothered at all’ and 2 is ‘bothered a lot’, and summed to provide a total score on a scale of 0–24. 24
Constipation-related quality of life was measured using the validated quality of life in constipation-20 questionnaire. 25 It consists of four domains (emotional, general physical, rectal physical, and social) and includes 20 items scored on a five-point Likert scale with responses ranging from ‘always’ to ‘never’.
Pain/constipation index was obtained by calculating the ratio of the pain severity score and bowel habit satisfaction score components of the IBS-SSS. An index score >1 was defined as pain predominance, while an index score <1 was defined as constipation predominance. Anxiety and depression were measured using the hospital anxiety and depression scale. 26
The safety of linaclotide was assessed via the observation and recording of adverse events (AEs) throughout the trial duration. AEs were collected by the investigators according to their clinical judgment. A definition of diarrhea was not prespecified in the protocol; those AEs described as ‘loose/watery stools’, ‘increased number of bowel movements’, or ‘diarrhea’ were coded using the Medical Dictionary for Regulatory Activities version 17.1.
Efficacy endpoints
The primary endpoint was clinical response at week 12, assessed using the IBS-SSS and global improvement scores. A clinical response was defined as meeting both of the following criteria: firstly, either a decrease in IBS-SSS score >30% versus baseline or an IBS-SSS score <75; and secondly, a self-reported response on the five-point Likert scale of feeling ‘better’ or ‘much better’ versus baseline. Patients had to meet both criteria at week 4 and week 12 to be considered a responder.
Statistical analyses
The intention-to-treat (ITT) population was defined as all patients who completed the screening phase and received at least one dose of study medication. The population that completed the protocol, or protocol completers, 27 was defined as all patients who completed the treatment period of 12 weeks. Statistical analyses were performed for both the ITT and CP (protocol completer) populations. Sample size was determined by logistic regression analyses where the dependent variable was clinical response and potentially associated factors were included. A 5% significance level was used for all tests.
For descriptive analyses, quantitative variables were reported as mean and standard deviation (SD) with 95% confidence interval (CI). For univariate analyses, the study-dependent variable was clinical response at week 12. Association between qualitative independent variables and clinical response was evaluated through Pearson’s Chi-squared test or Fisher’s exact test. Relative risks were reported with 95% CI. For quantitative independent variables, a comparison of means through a Student’s t test was performed and a nonparametric test of the median was performed for quantitative variables that were not adjusted to a normal distribution. Mean differences were reported with 95% CI.
To identify predictive factors independently related to clinical response at week 12, a binary regression model was generated. Variables with significance p < 0.05 in the univariate analysis and the following baseline variables were included in the model as potential predictive factors in a backward stepwise selection: age, time since IBS diagnosis, IBS-SSS score, abdominal pain severity score, digestive nonintestinal symptom score, extra-digestive symptom score, anxiety/depression score, fear/concern score, drug confidence score, and pain/constipation index. Relative risks were reported with 95% CI and model classification power was assessed with area under the curve analysis at 95% CI. In order to assess whether a response at week 4 is predictive of a response at week 12, a logistic regression model was generated using predictive factors identified in the binary model above, with the additional variable of clinical response at week 4. The area under the curve of both models were compared to assess improvement in predictive capacity from the addition of week 4 response.
Exploratory Pearson’s correlation analyses were conducted to assess the relationship between anxiety/depression score and digestive nonintestinal and extra-digestive symptom scores at screening (week −4) and during the treatment phase (ITT population), and to evaluate the association between change in IBS-SSS score and change in digestive nonintestinal and extra-digestive symptom scores during the treatment phase (ITT population).
Results
Patient disposition, demographics, and baseline characteristics
A total of 121 patients were screened; 109 patients completed the screening phase and 96 entered the treatment phase (ITT population). During the study, 36 patients discontinued linaclotide treatment. The reasons for discontinuation were AEs (n = 12, 12.5%), withdrawal of consent (n = 3, 3.1%), protocol deviation (n = 9, 9.4%), and lack of efficacy (n = 12, 12.5%). A total of 60 patients completed the treatment phase (CP population; Figure 1).

Study design and patient disposition.
In the ITT population, the mean (SD) age was 47.4 (14.7) years (range 18–80) and 94.8% of patients were female. Patients had been previously diagnosed with IBS-C for a mean (SD) duration of 7.5 (10) years. At the screening visit, the IBS-SSS total mean (SD) score was 371.0 (72.5), and the IBS-SSS domain mean (SD) scores were 68.4 (19.5) for abdominal pain severity, 76.3 (20.2) for abdominal bloating severity, and 65.9 (21.7) for satisfaction with bowel habit (Table 1).
Patient demographics and characteristics at screening.

Scale of 0–500.
Assessed with IBS-SSS VAS of 0–100, where higher scores indicate more severe symptoms/lower satisfaction with bowel habits.
Based on Rome III questionnaire.
Assessed with PHQ-12, scale of 0–24, where higher scores indicate greater bothersomeness of symptoms.
BMI, body mass index; CP, protocol completer; IBS-C, irritable bowel syndrome with constipation; IBS-SSS, irritable bowel syndrome symptom severity score; ITT, intention-to-treat; PHQ-12, patient health questionnaire 12 somatic symptom scale; SD, standard deviation; VAS, visual analog scale.
Efficacy
Primary endpoint: clinical response at week 12
A clinical response in the ITT and CP populations, based on meeting both the IBS-SSS and global improvement from the responder definition, was achieved in 25.0% and 36.7% of patients, respectively, at week 12 [Figure 2(a)]. In terms of the IBS-SSS component alone, 32.3% and 46.7% of patients showed a response at week 12 in the ITT and CP populations, respectively [Figure 2(b)]. From global improvement alone, 55.2% and 70.0% were responders at week 12 in the ITT and CP populations, respectively [Figure 2(c)]. At week 12, 61.5% and 80.0% of patients met either of the individual criteria in the ITT and CP populations, respectively (Figure 2).

Proportions of patients displaying a clinical response (a); response based on the IBS-SSS component only (b); and response based on global improvement only (c): ITT and CP populations.
Change in IBS-SSS total score and symptom scores
Between baseline (week 0) and week 12, approximately half (47.9%) of patients in the ITT population had a decrease in IBS-SSS of ⩾50 points and one-third (33.3%) had a decrease of ⩾100 points (Figure 3). Similarly, in the CP population, 56.7% of patients had a decrease in IBS-SSS of ⩾50 points and 45.0% had a decrease of ⩾100 points (Figure 3).
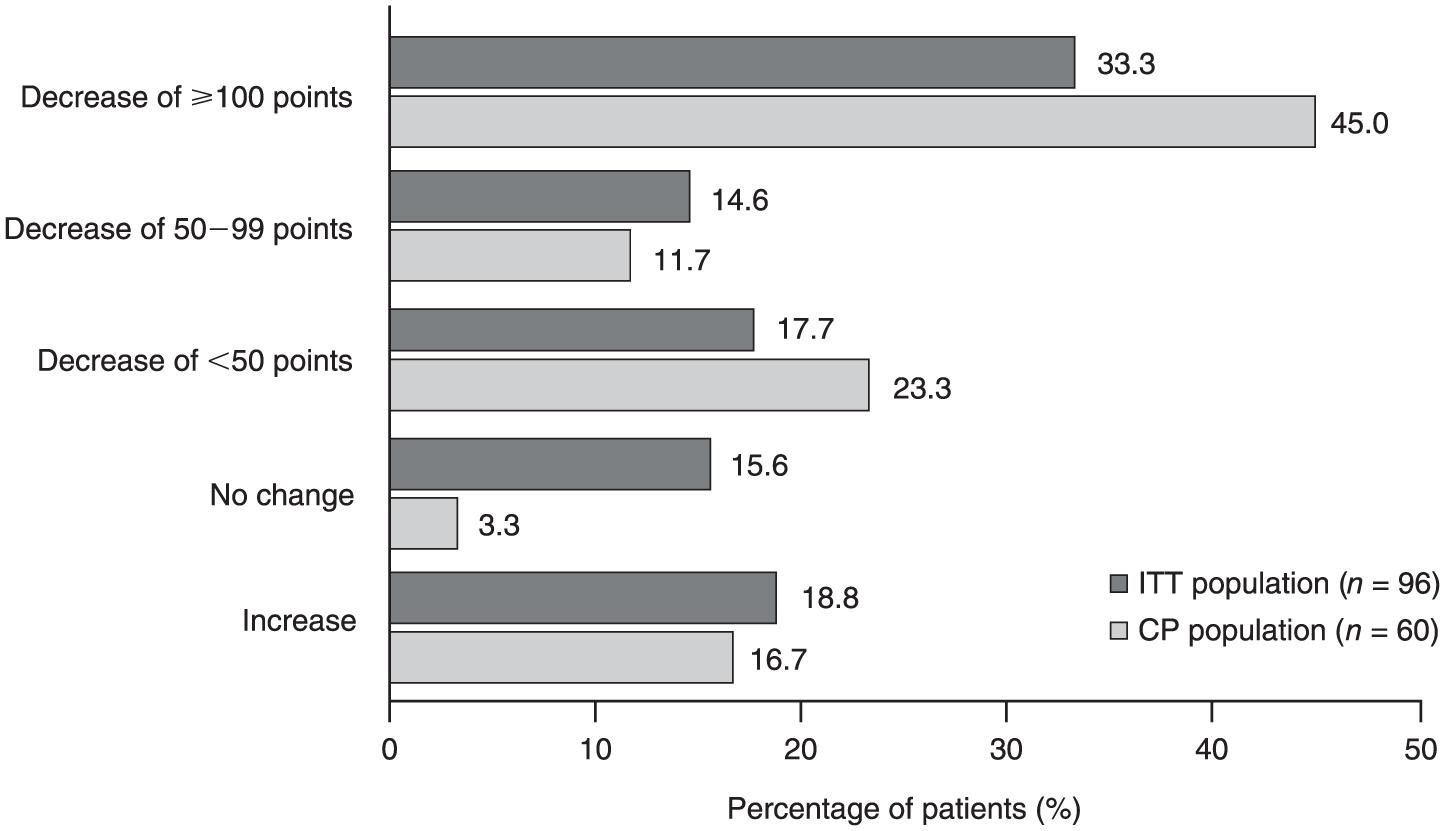
Change from baseline in IBS-SSS overall score at week 12: ITT and CP populations.
At weeks 4, 8, and 12, individual symptom scores were all significantly lower compared with baseline in both the ITT and CP populations (Figure 4). In the ITT population at week 12, the mean abdominal pain severity score decreased from 66.6 to 49.2 (p < 0.001) [Figure 4(a)], the mean abdominal bloating severity score decreased from 79.1 to 72.0 (p < 0.001) [Figure 4(b)], and the mean bowel habit satisfaction score improved from 64.1 to 45.5 (p < 0.001) [Figure 4(c)].

IBS-SSS scores from baseline (week 0) through week 12: abdominal pain severity (a); abdominal bloating severity (b); and bowel habit satisfaction (c): ITT and CP populations.
Digestive nonintestinal and extra-digestive symptoms
Digestive nonintestinal symptom scores were significantly lower at all subsequent visits compared with the baseline (Figure 5). In the ITT population, the mean digestive nonintestinal symptom score remained stable during the screening phase from week −4 (8.89) to baseline (8.65) (p > 0.05) and decreased from baseline to 6.81 at week 12 [21.2% improvement; p < 0.001; Figure 5(a)]. Extra-digestive symptom scores slightly worsened from week −4 (11.71) to baseline (12.65) (p < 0.05), and significantly decreased at weeks 4, 8, and 12. In the ITT population, the mean extra-digestive symptom score decreased from baseline to 7.39 at week 12 [41.5% improvement; p < 0.001; Figure 5(b)].

Digestive nonintestinal (a) and extra-digestive (b) symptoms from screening start (week −4) to end of treatment (week 12): ITT and CP populations.
An exploratory analysis revealed that at week −4 (screening), anxiety/depression score correlated with digestive nonintestinal symptom score (r = 0.39; p < 0.001) and extra-digestive symptom score (r = 0.49; p < 0.001). However, in the ITT population, change in anxiety score during the treatment phase did not show a correlation with change in digestive nonintestinal symptom score (r = 0.11; p = 0.365) or extra-digestive symptom score (r = −0.06; p = 0.713). Change in IBS-SSS score during the treatment phase in the ITT population was moderately correlated with change in digestive nonintestinal symptom score (r = 0.44; p ⩽ 0.001) but not with change in extra-digestive symptom score (r = −0.14; p = 0.309).
Use of concomitant medications for constipation
At baseline, 42.7% of patients in the ITT population reported use of laxatives, enemas, or suppositories; use of rescue medication was reported in 12.5% of ITT patients between baseline and week 4, and there was a further decrease to 11.5% between weeks 4 and 12 (Table S2).
Quality of life
Linaclotide treatment was associated with an improvement in the mean (SD) global quality of life in constipation-20 questionnaire score, from 32.3 (13.8) at baseline to 37.2 (14.2) at week 12 (p = 0.005) in the ITT population [Figure S1(a)], and from 33.1 (13.6) at baseline to 38.0 (13.9) at week 12 (p = 0.001) in the CP population [Figure S1(b)]. Significant improvements at week 12 were observed in the physical (p < 0.001, ITT and CP populations), emotional (p < 0.005, ITT population; p = 0.007, CP population), and rectal physical role (p ⩽ 0.001, ITT and CP populations) subscales; however, there was no significant change in the social role subscale (Figure S1).
Prediction of clinical response at week 12
None of the evaluated baseline demographics or clinical characteristics were independently associated with a clinical response at week 12 (Table S3). However, clinical response at week 4 was independently associated with a clinical response at week 12, with an odds ratio of 6.5 [95% CI: 2.1, 19.8; Figure S2 and Table S3].
Safety
In the ITT population, 67.7% of patients experienced at least one AE, with diarrhea being the most common, occurring in 34 (35.4%) patients (Table 2) and leading to treatment discontinuation in 12 (12.5%) patients. In total, 42 incidences of diarrhea were reported, 41 of which were deemed treatment-related; of these cases, 43.9% were rated mild, 51.2% were moderate, and 4.9% were severe. Nearly half (48.8%) of the AEs of diarrhea related to study medication occurred in week 1. The median duration of diarrhea related to study drug was 9 days, with 36.6% of episodes lasting ⩽7 days. No serious AEs or deaths occurred.
Summary of safety and AEs occurring during the treatment period, regardless of relatedness to study drug.

AEs recorded using the Medical Dictionary for Regulatory Activities version 17.1.
AE, adverse event; ITT, intention-to-treat.
Discussion
This single-country, multicenter, open-label study provides real-world evidence of the efficacy and safety of linaclotide in a Spanish population of adults with moderate-to-severe IBS-C, supporting the findings of the phase III linaclotide clinical trial program. Improvements in both digestive and extra-digestive symptoms were observed with linaclotide treatment, and linear regression analysis demonstrated that clinical response at week 4, based on simultaneous improvement in symptom severity and global improvement, was predictive of sustained clinical response at week 12.
In this study, approximately one-third of patients achieved a clinical response at week 12, based on the restrictive definition of both an improvement in IBS-SSS and a patient-reported global improvement. However, this finding does not necessarily indicate that only one-third of patients obtained a benefit from linaclotide. A clinically meaningful reduction (⩾50-point decrease) in IBS-SSS score was achieved at week 12 in almost 50% and 60% of patients in the ITT and CP populations, respectively. In addition, approximately two-thirds of patients reported that they felt ‘better’ or ‘much better’ at week 12 versus baseline. Treatment with linaclotide also led to improvements in individual symptom scores for abdominal pain, abdominal bloating, and bowel habit satisfaction. Use of rescue medication was minimal during the treatment phase, reflecting the efficacy of linaclotide. Further, bowel habit satisfaction score improved with sustained linaclotide treatment.
The proportion of week 12 clinical responders in the present study (ITT population: 25.0%; CP population: 36.7%) supports the previously reported findings of two large phase III trials of linaclotide.16,28 These trials utilized the US Food and Drug Administration-mandated responder criteria of ⩾30% reduction in average daily worst abdominal pain score and an increase by ⩾1 complete spontaneous bowel movement from baseline in the same week for at least 50% of weeks, and reported responder proportions of 33.7% and 33.6%, respectively, after 12 weeks of linaclotide treatment.16,28
The overall safety profile of linaclotide in this trial was consistent with that previously reported, with diarrhea being the most frequent AE. Diarrhea was reported in 35.4% of patients and led to discontinuation in 12.5%, regardless of relatedness to study medication; only 96 patients were included in this analysis, which should be taken into consideration when interpreting these data. The majority (95%) of AEs of reported diarrhea were mild or moderate and typically not prolonged, with a median duration of 9 days. In the previous two phase III studies, treatment-emergent AEs of diarrhea with linaclotide treatment were reported in 19.7% and 19.5% of patients, respectively, and AEs of diarrhea with linaclotide treatment led to discontinuation in 4.5% and 5.7% of patients, respectively.16,28 No new safety signals were observed in the present study.
Interestingly, linaclotide treatment was also associated with an improvement in digestive nonintestinal and extra-digestive symptoms. These symptoms are known to contribute to the overall impact of IBS on quality of life,10,11 and this finding may be important in the clinical setting, since IBS is often comorbid with GI and non-GI disorders (e.g. functional heartburn, dyspepsia, fibromyalgia). The results presented in this study support the hypothesis that an improvement in digestive nonintestinal and extra-digestive symptoms leads to an improvement in other symptoms related to non-GI disorders.
Improvement of digestive nonintestinal symptoms with a pharmacological treatment has not been reported previously, and no medication for IBS has previously been investigated in this regard. In an exploratory analysis, improvement in IBS symptoms was correlated with improvement in digestive nonintestinal symptoms (possibly from the actions of improved bowel function on enteroenteric reflexes), but not with improvement in extra-digestive symptoms. Neither improvement in digestive nonintestinal symptoms nor extra-digestive symptoms were correlated with a decrease in anxiety. As linaclotide is known to be locally active with minimal bioavailability,29,30 these effects of linaclotide treatment are unlikely to be attributable to central effects. Further research on how linaclotide may mediate the observed improvements in digestive nonintestinal and extra-digestive symptoms is warranted. Changes in the gut microbiota have been shown to modulate brain activity in healthy individuals, 31 so exploring the potential impact of linaclotide on the gut microbiota in IBS-C may be of interest.
Patients with IBS experience increased mucosal immune activation, predominantly characterized by the infiltration of mast cells and T lymphocytes that produce serine proteases, histamine, and prostaglandins.32,33 This immune activation in low-grade inflammation may be the consequence of a perturbation in gut microbiota, and may promote changes in intestinal sensory motor function, sensitizing both the intrinsic primary efferent and extrinsic primary afferent neurons. IBS-C could therefore lead to neural and muscle dysfunction (e.g. altered intestinal motility and visceral sensitivity), resulting in symptom development. 34 The role of linaclotide in modulating these mechanisms and its influence on the intestinal mucosal barrier remains to be elucidated in the clinical setting.
This study aimed to identify whether easily recognized clinical features may predict a response to linaclotide; however, none of the baseline patient characteristics evaluated in a logistic regression model could predict a clinical response to linaclotide at week 12. In line with linaclotide’s mechanism of action, a higher pain/constipation index, indicating pain predominance, was hypothesized to be associated with treatment response; however, the response to linaclotide was independent of pain or constipation predominance. In addition, these data indicated that a response to linaclotide is independent of factors such as the presence of psychological comorbidities (anxiety and depression) and symptom severity. These results support the findings of earlier phase III studies in which the efficacy of linaclotide was similar, irrespective of baseline disease severity. 35 However, the key finding of this study (i.e. that a clinical response to linaclotide treatment at week 4 was predictive of a response at week 12) may facilitate the identification of patients in clinical practice who will have a long-term response. This finding also supports the current recommendation that a follow-up visit be conducted 4 weeks after initiation of linaclotide treatment. 36 However, not all patients responding at week 12 were predicted to respond based on their results at week 4, suggesting that in some cases, the maximum efficacy of linaclotide may only be observed after >1 month of treatment. This is supported by the phase III clinical trial data, which showed that the time to maximum treatment effect varied by symptom, with mean change from baseline in worst abdominal pain scores reaching a plateau after 8–12 weeks of linaclotide treatment.16,28
With the majority of patients in the ITT and CP populations (>90% in both) being female, this study is limited by a potential selection bias. However, given a higher prevalence of IBS in women, 4 the study was likely to enroll more female patients with IBS. Other study limitations include the relatively small number of patients entering the treatment phase and the lack of a placebo comparator. In addition, this was an open-label study primarily conducted in hospital centers in a single country and therefore may not be representative of the wider IBS patient population in a real-world setting. However, the prospective, multicenter nature of the study lends validity to its findings, which represent a broad geographic distribution of IBS patients in Spain. The results should therefore be viewed in light of these limitations.
Conclusion
This trial demonstrated that linaclotide is an effective treatment for IBS-C that improves GI, digestive nonintestinal, and extra-digestive symptoms related to the disorder. In line with previous findings, linaclotide is effective in patients with IBS-C, regardless of factors such as baseline disease severity and the presence of anxiety or depression. Furthermore, a clinical response to linaclotide after 4 weeks of treatment, based on simultaneous improvement in symptom severity and global improvement, may predict a response with continued use; results from this study suggest that improvements may be observed by week 12. Taken together, these findings may allow for better clinical decision-making, as they validate the comparable efficacy of linaclotide in patients with severe IBS symptoms and provide a basis for evaluation of response to linaclotide over time in clinical practice.
Supplemental Material
Supplementary_Material – Supplemental material for Clinical response to linaclotide at week 4 predicts sustained response in irritable bowel syndrome with constipation and improvements in digestive and extra-digestive symptoms
Supplemental material, Supplementary_Material for Clinical response to linaclotide at week 4 predicts sustained response in irritable bowel syndrome with constipation and improvements in digestive and extra-digestive symptoms by Blanca Serrano-Falcón, Sílvia Delgado-Aros, Fermín Mearin, Constanza Ciriza de los Ríos, Jordi Serra, Miguel Mínguez, Miguel Montoro Huguet, Antonia Perelló, Cecilio Santander, Ángeles Pérez Aisa, Inmaculada Bañón-Rodríguez and Enrique Rey in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
This study was sponsored by Allergan plc, Dublin, Ireland. Writing and editorial assistance was provided to the authors by Beatrice Vetter-Ceriotti, Helen Woodroof, and Madeeha Aqil, PhD of Complete HealthVizion, Inc., Chicago, IL, USA and funded by Allergan plc, Dublin, Ireland. All authors met the ICMJE authorship criteria. Neither honoraria nor payments were made for authorship.
Some of the data presented in this work have previously been submitted as an abstract and presented as a poster (Asociación Española de Gastroenterología 2017, Madrid, Spain, 8–10 March 2017) and oral presentation (Digestive Disease Week® 2017, Chicago, Illinois, USA, 6–9 May 2017).
Author contributions
BSF, SDA, FM, CCR, JS, MM, MMH, AP, CS, and APA served as investigators, performed the research, and collected and analyzed the data. IBR designed the research study and analyzed the data. ER served as the coordinating investigator, designed the research study, performed the research, and collected and analyzed the data. All authors met ICMJE criteria and approved the final version of the manuscript.
Author note
Some of these data have previously been submitted as an abstract and presented as a poster (Asociación Española de Gastroenterología 2017, Madrid, Spain, March 8–10, 2017) and oral presentation (Digestive Disease Week® 2017, Chicago, Illinois, USA, May 6–9, 2017).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Allergan plc and Almirall S.A. It received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
S. Delgado-Aros has acted as a consultant for Allergan plc, Almirall, and Shire Pharmaceuticals Iberia and has received compensation from Abbot Laboratories. F. Mearin has received grant support from Alfa Wassermann, Allergan, and Menarini. C. Ciriza de los Ríos has acted as a consultant for Allergan and Bayer. J. Serra has acted as a consultant and has participated in advisory boards or in invited conferences for Allergan, Almirall, Norgine, Reckitt Benckiser, Bayer, and Zespri. M. Mínguez has acted as a consultant for Allergan plc, Farmasierra SL, and Bayer. M. Montoro Huguet has served as advisor for MSD, Almirall, and Vifor Pharma. A. Pérez Aisa has acted as a consultant for Allergan and Shionogi and has received compensation from Mylan, Norgine, and Cassen for formative actions. I. Bañón-Rodríguez is a former employee of Allergan plc. E. Rey has acted as a consultant for Allergan plc, Norgine Iberia, Farmasierra SL, and Bayer, and has received research grants from Allergan plc and Norgine Iberia. B. Serrano-Falcón, A. Perelló, and C. Santander have no conflicts of interest to declare.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
