Abstract
Irritable bowel syndrome with diarrhea (IBS-D) is a common, chronic functional gastrointestinal disorder with symptoms that can be distressing for patients and often result in substantially impaired quality of life. This review focuses on providing clinicians with information on practical, evidence-based treatment for IBS-D. Current therapies commonly used for the treatment of IBS-D, including pharmacologic and nonpharmacologic interventions, are briefly reviewed, followed by discussion of the emergent pharmacologic treatments (rifaximin and eluxadoline) and medical foods (IBgard® and EnteraGam®). Given the lack of a standard treatment algorithm for IBS-D and the emergence of new pharmacologic therapies, treatment needs to be tailored to the individual patient and take into account the severity of disease. In this context, the latter part of this manuscript examines how treatments for IBS-D can be used in clinical practice by presenting three patient case scenarios with varying degrees of IBS-D severity. For each case, the patient’s medical history and clinical presentation are related to the Rome Foundation multidimensional clinical profile (MDCP) and potential treatment options with current and emergent therapies are reviewed. The interplay of gastrointestinal symptoms and their psychosocial impact, as well as the importance of a patient-centered approach to therapy, are discussed. Consideration is given to the potential need for combination therapies and how emergent treatments could fit into the treatment pathway for mild, moderate, and severe cases of IBS-D in clinical practice.
Keywords
Introduction
Irritable bowel syndrome (IBS) is a common, chronic functional gastrointestinal (GI) disorder characterized by recurring abdominal pain, bloating, loose or frequent stools and/or constipation in the absence of structural, major inflammatory, or biochemical abnormalities [Chey et al. 2015b]. IBS symptoms can be distressing for patients and often result in substantially impaired quality of life, reduced work productivity, and increased utilization of healthcare resources [Hulisz, 2004; Paré et al. 2006].
Prevalence of IBS varies by geographic region, with differences apparent in diagnostic criteria, study populations, and methodology [Canavan et al. 2014; Lovell and Ford, 2012]. Globally, the pooled prevalence of IBS is 11% [Lovell and Ford, 2012], and the prevalence in adults in the US is ~10–15% [Saito et al. 2002]. IBS is further categorized into three subtypes depending on the predominant bowel habit: IBS with constipation (IBS-C), IBS with diarrhea (IBS-D), and IBS with a mixed bowel pattern (IBS-M) [Longstreth et al. 2006], which have a prevalence in the US of 28%, 26%, and 44%, respectively [Su et al. 2014].
Diagnosis of IBS is based on clinical symptom criteria and the exclusion of certain organic diseases only in patients presenting with concerning features [Chey et al. 2015b]. The recently released Rome IV diagnostic criteria for IBS are: recurrent abdominal pain, on average, at least 1 day per week in the past 3 months, associated with two or more of the following criteria: (1) related to defecation, (2) associated with a change in frequency of stool, (3) associated with a change in form (appearance); with these criteria fulfilled for the past 3 months, with symptom onset at least 6 months before diagnosis [Mearin et al. 2016]. Although the Rome diagnostic criteria for IBS are widely used in clinical trials, there is recognition that they are not often used in clinical practice [Ford et al. 2014a; Olafsdottir et al. 2012].
Additionally, IBS can coexist with non-GI somatic pain disorders, including fibromyalgia, as well as with other GI disorders, such as gastroesophageal reflux disease and functional dyspepsia, with several studies suggesting shared pathophysiologic mechanisms for these conditions [Kim and Chang, 2012]. The overlap of these disorders increases in prevalence in tertiary referral patient populations.
The focus of this review is on IBS-D and the aim is to provide information on practical, evidence-based pharmacologic treatment of IBS-D, predominantly of relevance to clinicians in North America. A brief discussion of the pathophysiology of IBS will be followed by a review of the current and emergent pharmacologic therapies for IBS-D along with other commonly used treatments, such as lifestyle modifications, medical foods, and psychological therapies. The goal will be to examine these treatments in the context of clinical practice by describing three patient case scenarios with varying degrees of IBS-D severity and the potential treatment options for each situation.
Pathophysiology of irritable bowel syndrome
The pathophysiology of IBS is complex and multifactorial, including genetic predisposition, visceral hypersensitivity, autonomic nervous system dysregulation, abnormalities in GI motility, secretory function, permeability, immune activation, and the composition of the gut microbiome [Cheng et al. 2013; El-Salhy, 2015; Hasler, 2011; Simrén et al. 2013]. There is also evidence that brain–gut axis dysregulation [Fichna and Storr, 2012; Koloski et al. 2012], sex- and gender-related effects [Mulak et al. 2014], and psychosocial factors, such as a history of abuse [Drossman, 2011; Fichna and Storr, 2012; Halland et al. 2014], may be important in the development of IBS.
Dysregulations in the brain–gut axis can cause changes in gut motility and visceral perception, which is considered an important mechanistic factor for the development of abdominal pain in IBS [Fichna and Storr, 2012]. Patients with IBS often experience general psychological comorbidities, such as depression and anxiety, as well as symptom-specific anxiety, which can negatively impact quality of life, severity of symptoms, and treatment response [Fadgyas-Stanculete et al. 2014; Fichna and Storr, 2012; Fond et al. 2014; Lackner et al. 2013, 2014], and increase healthcare utilization [Johansson et al. 2010].
Current treatments for irritable bowel syndrome with diarrhea
At present, there is no standard treatment algorithm for IBS-D and therapeutic options focus on alleviating symptoms, often encompassing lifestyle and dietary modifications [Buchanan et al. 2014; Johannesson et al. 2011; Moayyedi et al. 2014; Shepherd and Gibson, 2013], and medical foods for initial and adjunctive treatment, over-the-counter medications, prescription medications, and psychological therapies [Chey et al. 2015b; Ford et al. 2014a].
Lifestyle and dietary modifications
Modulating diet and, more recently, exercise and sleep, have been some of the therapeutic tools used for treating IBS. A low-fat diet is often recommended, as fatty foods may cause painful contractions in patients with IBS [Simrén et al. 2007a, 2007b]. The recommended amount of fiber in the diet is 20–28 g/day for women and 31–38 g/day for men [Food and Nutrition Board Institute of Medicine, 2005]. While soluble fiber, such as psyllium (isphagula), may help relieve abdominal pain in some patients with IBS [Nagarajan et al. 2015], there is no evidence for the use of insoluble fiber, which may actually cause bloating and abdominal discomfort [Ford et al. 2014a]. Several studies have demonstrated that gluten in patients without celiac disease (non-celiac gluten sensitivity), and other carbohydrates may contribute to symptoms in IBS [Biesiekierski et al. 2011, 2013; Ong et al. 2010; Wilder-Smith et al. 2013, 2014]. A number of studies have also shown that a gluten-free diet can improve IBS symptoms in some patients [Rodrigo et al. 2014; Vazquez-Roque et al. 2013], suggesting that at least a subset of IBS patients might benefit from a diet with no, or reduced, gluten. However, the effects attributed to gluten may be triggered by nonabsorbable carbohydrates such as fructans and galactans present in wheat [Gibson and Muir, 2013]. In the past few years, a diet low in fermentable oligosaccharides, disaccharides, monosaccharides, and polyols (FODMAPs) has been embraced by patients with IBS and may be worth trying in patients with IBS-D or patients who have symptoms of bloating [Böhn et al. 2015; de Roest et al. 2013; Halmos et al. 2014; Lacy, 2015; Pedersen et al. 2014; Staudacher et al. 2011, 2012]. A recent study showed that gluten exclusion was not the primary cause of the beneficial effect of a low FODMAP diet [Biesiekierski et al. 2013]. In a controlled, randomized, crossover study of patients with IBS, a low FODMAP diet significantly reduced functional GI symptoms, including overall GI symptoms, bloating, abdominal pain, and passage of flatus, with stool frequency and consistency also improved in the subgroup of patients with IBS-D [Halmos et al. 2014]. In another recent randomized study, a low FODMAP diet significantly reduced IBS symptoms but it did not differ from traditional IBS dietary advice, which included a regular meal pattern, avoidance of large meals, and reduced intake of fat, insoluble fibers, caffeine, and gas-producing foods such as beans, cabbage, and onions, and had greater emphasis on when, rather than what, to eat [Böhn et al. 2015]. However, this traditional diet recommended the avoidance of some FODMAP foods. Practically speaking, a low FODMAP diet is quite restrictive, not balanced, and often difficult for patients to follow on a long-term basis; hence, patients undertaking this diet should do so under the instruction of an experienced dietitian. Furthermore, it may adversely influence the gut microbiome [Halmos et al. 2015]. In general, patients should try to reintroduce some of these foods slowly with the guidance of their physician or a dietitian, if available. Regular exercise has been shown to decrease the symptoms of IBS [Johannesson et al. 2011], while disturbed sleep increases next-day symptoms of abdominal pain and, therefore, good sleep hygiene should be encouraged [Buchanan et al. 2014].
Pharmacologic treatments
Until recently, alosetron (Lotronex®; Prometheus Laboratories, Roswell, GA) was the only pharmacologic treatment approved by the US Food and Drug Administration (FDA) specifically for IBS-D [US Food and Drug Administration, 2010]. Alosetron is a selective 5-HT3 antagonist that reduces abdominal pain and bowel movement (BM) frequency and urgency, but is only indicated for women with more severe IBS-D who have not responded to conventional treatments (Table 1) [US Food and Drug Administration, 2010]. Due to concerns over serious cases of ischemic colitis and complications of constipation associated with alosetron, it is subject to a risk evaluation and mitigation strategy (REMS) [Ford et al. 2014a; US Food and Drug Administration, 2010]; however, the incidence of such events is low [Tong et al. 2013]. Overall, the strength of evidence for alosetron use in IBS-D is moderate [Chang et al. 2014; Ford et al. 2014a], with a number needed to treat (NNT) of 8 [Ford et al. 2014a]. Recently, a generic version of alosetron became available in the US; alosetron is not approved by the European Medicines Agency (EMA) for use in Europe.
Current Food and Drug Administration-approved pharmacologic and commonly used pharmacologic and medical food treatments for irritable bowel syndrome with diarrhea.

AE, adverse event; BID, twice daily; BM, bowel movement; FDA, US Food and Drug Administration; GI, gastrointestinal; IBD, inflammatory bowel disease; IBS, irritable bowel syndrome; IBS-D; irritable bowel syndrome with diarrhea; MOA, mechanism of action; NNH, number needed to harm; NNT, number needed to treat; PO, oral administration; QD, once daily; QID, four times daily; QOL, quality of life; SAE, serious adverse event; SNRIs, serotonin–norepinephrine reuptake inhibitors; SSRIs, selective serotonin reuptake inhibitors; TCAs, tricyclic antidepressants; TID, three times daily.
Other pharmacologic treatments for IBS-D that are not FDA or EMA approved but are often used in clinical practice include the antidiarrheal agent loperamide, antidepressants, and antispasmodics (Table 1). In the US, antidepressants are more commonly used for IBS compared with Europe and other parts of the world, where antispasmodics are more commonly used as initial treatment.
Although the quality of evidence for loperamide in IBS-D is low, it is an effective antidiarrheal agent and is a commonly used over-the-counter medication (Table 1) [Chang et al. 2014; Ford et al. 2014a]. However, the efficacy of loperamide in reducing abdominal pain has not been convincingly proven.
Antidepressants have an overall NNT of 4 (Table 1). Tricyclic antidepressants (TCAs) may provide global IBS symptom relief, including reducing abdominal pain in IBS [Chang et al. 2014; Ford et al. 2014a, 2014b; Trinkley and Nahata, 2014]. In a per-protocol analysis excluding participants with nondetectable blood levels, desipramine improved symptoms compared with placebo in women with moderate-to-severe diarrhea-predominant functional GI disorders [Drossman et al. 2003], presumably partly due to its anticholinergic effects on gut motility [Clouse et al. 1994; Saha, 2014]. There are contradictory reports of the efficacy of selective serotonin reuptake inhibitors (SSRIs) in improving global symptoms and abdominal pain in IBS and the quality of evidence supporting their use is low [Chang et al. 2014; Ford et al. 2014a, 2014b; Trinkley and Nahata, 2014]. However, SSRIs may enhance overall wellbeing by improving the perception of symptoms by reducing coexistent psychological symptoms that may contribute to IBS symptoms. Data relating to serotonin–norepinephrine reuptake inhibitor efficacy in IBS are extremely limited [Brennan et al. 2009; Grover and Drossman, 2011; Kaplan et al. 2014], but their beneficial effect on chronic somatic and neuropathic pain could conceivably be applicable to visceral pain. In clinical practice, centrally acting agents are used to potentiate the effects on the GI tract and their side effects may be beneficial for the symptoms of IBS. For example, TCAs have a constipating side effect [US Food and Drug Administration, 2014b] that makes them useful for IBS-D.
Antispasmodics, such as peppermint oil, have been shown to be effective in improving IBS symptoms and abdominal pain, albeit with low or moderate quality of evidence and variable efficacy for individual agents (Table 1) [Chang et al. 2014; Ford et al. 2014a; Trinkley and Nahata, 2014]. In addition to peppermint oil, hyoscyamine and dicyclomine are commonly used in the US, while in Europe and other parts of the world, agents such as mebeverine, phloroglucinol, and trimebutine are also often tried [Annaházi et al. 2014].
There is some evidence suggesting certain probiotics may help relieve overall symptom burden in some patients with IBS [Hungin et al. 2013]; however, specific recommendations regarding their use in IBS have not been made (Table 1) [Cash, 2014; Ford et al. 2014a]. Probiotics are used in both the US and Europe, though the use of medical foods in the US likely means that probiotics are currently used more widely outside of the US.
Additionally, patients with IBS-D symptoms may show evidence of bile acid malabsorption [Wedlake et al. 2009], and some physicians use an empirical trial of a bile acid sequestrant in such patients. Bile acid sequestrants have shown some initial evidence of efficacy in recent pilot studies in patients with IBS-D [Bajor et al. 2015; Camilleri et al. 2015]. A fecal bile acid assay is currently available and may help to identify patients with IBS-D symptoms who have bile acid diarrhea and who respond to a bile acid sequestrant [Camilleri et al. 2014; Shin et al. 2013].
Nonpharmacologic psychological therapies
A recent meta-analysis showed that nonpharmacologic psychological therapies, such as cognitive behavioral therapy (CBT), hypnotherapy, multicomponent psychological therapy, and dynamic psychotherapy, were significantly more effective than usual management, supportive therapy, or placebo in patients with IBS, with an overall NNT of 4 (Table 2) [Ford et al. 2014b]. It is important for physicians to understand how psychological therapies, used either alone or in conjunction with pharmacologic treatment, may provide clinical benefits for patients [Ford et al. 2014b]. Currently, a lack of skilled practitioners and poor patient and clinician acceptance of these therapies, amongst other factors, limits their adoption in clinical practice [Chey et al. 2015b]. However, there is increasing evidence that patients with mild-to-moderate IBS symptoms may achieve a benefit from internet-based or self-administered (minimal contact) CBT programs [Lackner, 2007; Ljótsson et al. 2011a, 2011b]. Employing these modalities may help ease the problems of availability and time constraints.
Psychological therapies for irritable bowel syndrome [Ford et al. 2014b].

CBT, cognitive behavioral therapy; CI, confidence interval; IBS, irritable bowel syndrome; NS, not significant.
Emergent treatments for irritable bowel syndrome with diarrhea
Two new pharmacologic treatments recently received approval from the FDA for the treatment of IBS-D in adults: eluxadoline (Viberzi®; Allergan, Parsippany, NJ), a mixed µ-opioid receptor agonist, κ-opioid receptor agonist, and δ-opioid receptor antagonist that is locally active in the GI tract [US Food and Drug Administration, 2015a; Wade et al. 2012], and rifaximin (Xifaxan®; Salix Pharmaceuticals, Bridgewater, NJ), a minimally absorbed broad-spectrum antibiotic that acts locally in the GI tract and inhibits bacterial transcription and ribonucleic acid synthesis [Iorio et al. 2015; US Food and Drug Administration, 2015b].
Emergent treatments for IBS-D that are non-FDA approved but likely to be used in clinical practice are the medical food products enteric-coated sustained-release microspheres of peppermint oil (IBgard®; IM HealthScience, Boca Raton, FL) and serum-derived bovine immunoglobulin/protein isolate (SBI) [EnteraGam®; Entera Health, Ankeny, IA]. Of note, medical foods are not simply foods for special dietary use; they are foods that are specially formulated or processed and are intended to meet distinctive nutritional requirements of a disease or condition, used under medical supervision and intended for the specific dietary management of a disease or condition [US Food and Drug Administration, 2013b]. As medical foods are not drugs, they do not have to undergo premarket review or approval by the FDA. However, any facility involved in manufacturing, processing, packing, or holding medical foods must be registered with the FDA.
Eluxadoline
Eluxadoline [75 or 100 mg twice daily (BID)] has demonstrated efficacy in simultaneously relieving the symptoms of abdominal pain and diarrhea associated with IBS-D through 12 weeks (FDA-required primary endpoint) and through 26 weeks (EMA-required primary endpoint) in two large, double-blind, randomized, placebo-controlled phase III trials [IBS-3001 (n = 1282) and IBS-3002 (n = 1146)] (Table 3) [Lembo et al. 2016]. The treatment effect of eluxadoline over placebo was observed within the first week of treatment, with 22.8% and 24.5% of patients treated with eluxadoline 75 mg and 100 mg, respectively, having a composite response in weeks 1–4 compared with 12.5% of patients receiving placebo (p < 0.001) [Lembo et al. 2016]. Eluxadoline also significantly improved multiple other abdominal and bowel symptoms associated with IBS-D compared with placebo, such as abdominal discomfort and bloating, and BM urgency, frequency, and incontinence through 26 weeks of treatment [Harris et al. 2015]. Use of eluxadoline on an as-needed basis has not been evaluated in clinical trials.
Emergent Food and Drug Administration-approved pharmacologic agents and medical food treatments for irritable bowel syndrome with diarrhea.

AE, adverse event; ALT, alanine transaminase; BID, twice daily; FDA, US Food and Drug Administration; GERD, gastroesophageal reflux disease; GI, gastrointestinal; HIV, human immunodeficiency virus; IBS, irritable bowel syndrome; IBS-D, irritable bowel syndrome with diarrhea; IBS-M, irritable bowel syndrome with a mixed bowel pattern; MCH, mean corpuscular hemoglobin; NNH, number needed to harm; NNT, number needed to treat; OATP1B1, organic anion transporter family member 1 B1; QD, once daily; QID, four times daily; SAE, serious adverse event; TID, three times daily.
Eluxadoline was generally well tolerated in clinical studies, with the most common adverse events (AEs) of constipation, nausea, and abdominal pain occurring in ~7–9% of patients receiving eluxadoline 100 mg BID [Lembo et al. 2016]. The majority of AEs related to eluxadoline occurred soon after treatment initiation (within the first 2 weeks) [Cash et al. 2015b]. As eluxadoline is a µ-opioid receptor agonist, there is a potential for increased risk of sphincter of Oddi (SO) spasm. Cases of SO spasm manifesting as pancreatitis or abdominal pain with elevated hepatic enzyme levels occurred in 0.54% of patients treated with eluxadoline 75 or 100 mg BID in phase III trials [Lembo et al. 2016]. All cases occurred in patients without a gallbladder and subsequently resolved on discontinuation of eluxadoline. Therefore, it is recommended to use the lower dose of 75 mg BID in patients who are postcholecystectomy [US Food and Drug Administration, 2015a]. There is also a potential increased risk of pancreatitis not associated with SO spasm in patients receiving eluxadoline. Overall, cases of pancreatitis occurred in 0.3% of patients who received eluxadoline 75 or 100 mg BID in phase III clinical trials, with the majority of these events associated with excessive alcohol intake [Lembo et al. 2016]. Consequently, eluxadoline is contraindicated in patients with a history of alcohol abuse or addiction, or who drink more than three alcoholic drinks per day, and patients should avoid chronic or excessive alcohol intake in combination with eluxadoline [US Food and Drug Administration, 2015a].
Rifaximin
Rifaximin treatment [550 mg three times daily (TID) for 14 days] is a short-term regimen but it can only be repeated up to two times if symptoms recur [US Food and Drug Administration, 2015b]. In two large, double-blind, randomized, placebo-controlled phase III trials in IBS patients without constipation [TARGET 1 (n = 623) and TARGET 2 (n = 637)], a significantly greater proportion of patients who received rifaximin achieved adequate relief of IBS symptoms for at least 2 of the 4 weeks after treatment than patients who received placebo (Table 3) [Pimentel et al. 2011]. Rifaximin also significantly improved IBS-related bloating, abdominal pain or discomfort, and stool consistency for at least 2 of the 4 weeks after treatment [Pimentel et al. 2011]. Significantly more patients in the rifaximin group than in the placebo group had a composite response of simultaneously relieving symptoms of abdominal pain and stool consistency (46.6% versus 38.5%, p = 0.04, in TARGET 1; 46.7% versus 36.3%, p = 0.008, in TARGET 2). In a subsequent study [TARGET 3 (n = 2579)], of 1074 patients with IBS-D who initially responded to open-label rifaximin based on the composite response, ~60% experienced symptom recurrence at a median time of 10 weeks after first treatment [US Food and Drug Administration, 2015b]. Patients who relapsed were randomized to receive rifaximin 550 mg TID or placebo for up to two 14-day repeat treatment periods separated by 10 weeks. Rifaximin significantly improved IBS symptoms based on composite response compared with placebo in the month after both retreatment periods (period 1: 33% versus 25%, p = 0.02; period 2: 35% versus 27%, p = 0.03) [Chey et al. 2015a].
The safety profile of rifaximin is comparable with placebo during both the treatment and post-treatment periods (overall duration 12–16 weeks) after a single course of treatment [Schoenfeld et al. 2014]. There does not appear to be an increased risk of Clostridium difficile infection [Schoenfeld et al. 2014], although cases of C. difficile-associated colitis have been reported in postmarketing surveillance of rifaximin in other indications [US Food and Drug Administration, 2015b]. The potential risk for development of antimicrobial resistance with repeated courses of rifaximin appears to be small, but additional long-term safety data are needed [Schoenfeld et al. 2014].
Enteric-coated peppermint oil
Enteric-coated peppermint oil (IBgard®) uses a novel triple-coated microsphere formulation to facilitate sustained release in the small intestine, and to reduce AEs associated with peppermint oil such as heartburn, abdominal pain, or anal burning [Cash et al. 2015a; Khanna et al. 2014]. In a small, randomized, placebo-controlled study (n = 72), treatment with enteric-coated peppermint oil (IBgard®) [180 mg TID] for 4 weeks significantly improved total IBS symptom score and individual symptoms of abdominal pain, bloating, pain at evacuation, and urgency compared with placebo in patients with IBS-D or IBS-M (Table 3) [Cash et al. 2015a]. Enteric-coated peppermint oil was well tolerated, with only two treatment-emergent AEs in enteric-coated peppermint oil-treated patients of dyspepsia and upper respiratory tract infection.
Serum-derived bovine immunoglobulin/protein isolate
SBI is believed to act by providing nutritional support for epithelial barrier function and maintaining immune balance in the GI tract [Wilson et al. 2013]. In a pilot double-blind, randomized, placebo-controlled, 6-week study in patients with IBS-D (n = 66), SBI 10 g/day and 5 g/day significantly reduced the number of days with any symptom between weeks 2 and 6 compared with baseline (it was assumed that 3 weeks’ treatment would be needed before benefits would be observed) (Table 3) [Wilson et al. 2013]. The benefits tended to be greater for the 10 g/day group. SBI exhibited a similar safety profile to placebo. Additionally, SBI appeared to be effective in improving abdominal and bowel symptoms in a case series report in patients with IBS-D (n = 7) who received SBI therapy for 17–32 weeks [Good et al. 2015]. Given the limited data at the present time, the value of SBI for IBS-D is yet to be established and larger and longer-term clinical studies are required to assess the efficacy and safety of SBI in IBS-D.
Incorporating pharmacologic treatments for irritable bowel syndrome with diarrhea into clinical practice
Given the lack of a standard treatment algorithm and the emergence of new pharmacologic treatments for IBS-D, treatment needs to be tailored to the individual patient and take into account the severity of disease. Unfortunately, IBS-D severity is not precisely defined and in practice is usually based empirically on frequency of symptoms and the impact of symptoms on patients’ daily lives. In 2011, clinical profiles were proposed for patient-rated severity in IBS based on both physiologic and psychological features, and estimated the prevalence of mild, moderate, and severe IBS as 40%, 35%, and 25%, respectively [Drossman et al. 2011]. Severity in IBS was defined as a biopsychosocial composite of patient-reported GI and extra-intestinal symptoms, degree of disability, and illness-related perceptions and behaviors. It was also noted that both visceral and central nervous system (CNS) physiologic factors affect severity and that as severity increases, the CNS provides a greater contribution. More recently, the Rome Foundation advanced the multidimensional clinical profile (MDCP) to capture various dimensions of patients’ experiences of functional disorders and integrate the information into a meaningful plan of clinical care [Drossman, 2015]. For example, the MDCP helps characterize the IBS illness state, which can then be applied to a treatment plan. The MDCP includes five dimensions: categorical diagnosis, clinical modifiers, impact on daily activities, psychosocial modifiers, and physiological modifiers of function and biomarkers (Table 4).
Rome Foundation multidimensional clinical profile for functional gastrointestinal disorders: category definitions related to irritable bowel syndrome [Drossman, 2015] and irritable bowel syndrome with diarrhea case scenario diagnostic classifications.

GI, gastrointestinal; IBD, inflammatory bowel disease; IBS, irritable bowel syndrome; IBS-C, irritable bowel syndrome with constipation; IBS-D, irritable bowel syndrome with diarrhea; MDCP, multidimensional clinical profile; SIBO, small intestinal bacterial overgrowth.
To show how current and emergent treatments could be applied in clinical practice for the treatment of IBS-D, we present three case scenarios of patients with differing levels of IBS-D severity (mild, moderate, and severe), relate each patient’s medical history and clinical presentation to the MDCP categories, and discuss the potential treatment options for each situation.
Patient scenario 1: Mild irritable bowel syndrome with diarrhea
A 22-year-old woman presents with a 2-year history of abdominal pain and diarrhea. She experiences intermittent episodes of lower abdominal cramping and bloating associated with 4–6 loose BMs occurring three times a month. The abdominal cramping seems to occur shortly after meals, or at times of increased stress, and subsides after BM. Her symptoms are not associated with intake of milk products. She drinks coffee daily and likes to chew gum. Recently, she started to reduce some activities such as hiking or jogging. She has not dated for the past 6 months over concerns that fecal urgency may interfere with intimacy. She cannot predict what will trigger an episode and is worried about the availability of bathrooms. The patient would like to find out what is causing her symptoms and how to get rid of them. A summary of the patient’s symptoms, medical and psychosocial history, physical and laboratory examination results, including celiac serologies, and previous interventions is given in Figure 1.
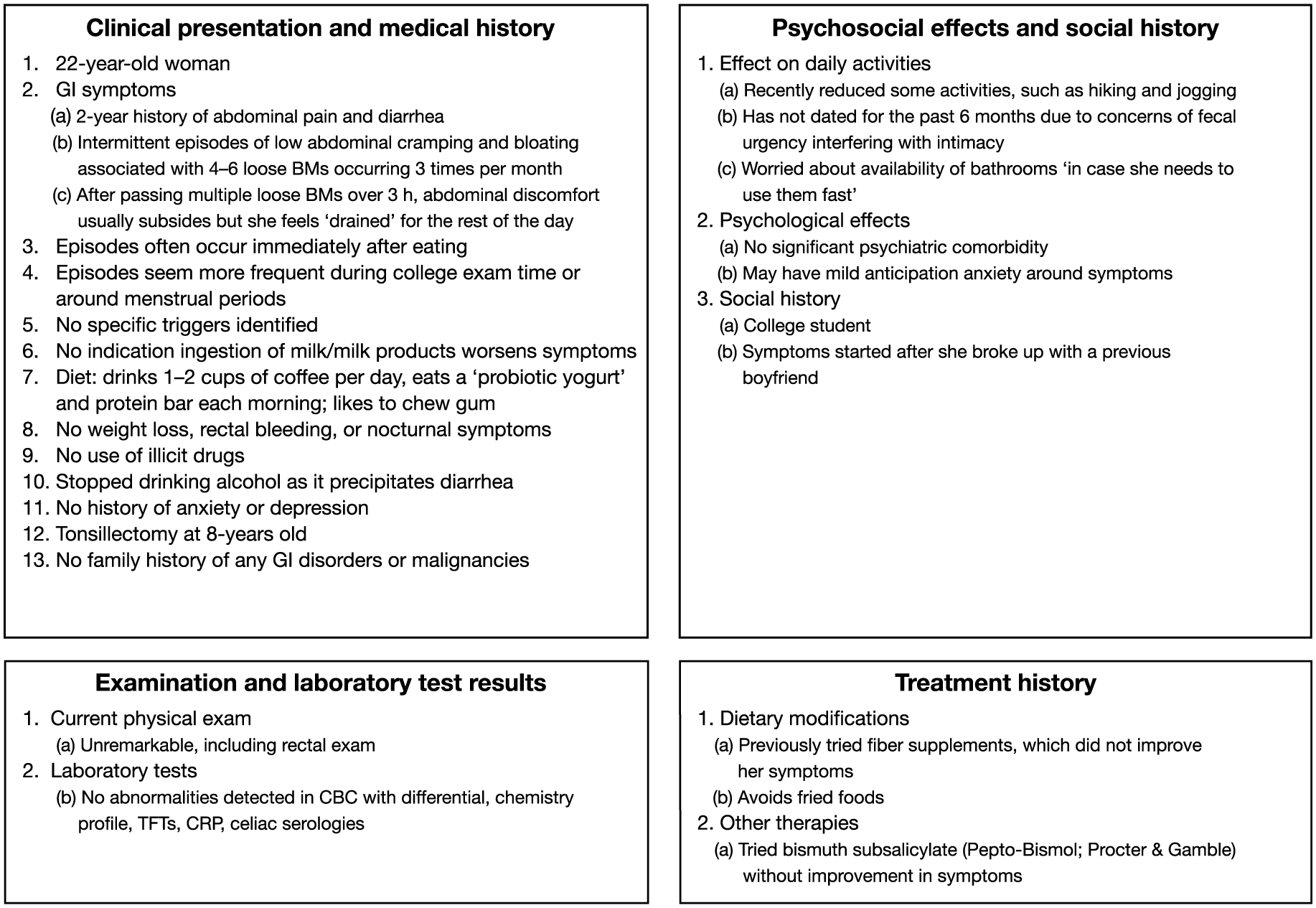
Summary of mild irritable bowel syndrome with diarrhea case scenario.
Comment, treatment options, and management
The patient fulfills the MDCP diagnostic classification [Drossman, 2015] for mild IBS-D (Table 4). She has no red-flag symptoms or signs necessitating structural evaluation of the GI tract. She has no evidence of celiac disease and it is unlikely she has lactose intolerance. Her IBS symptoms are relatively infrequent but interfere with some of her activities and her social life, and negatively impact her health-related quality of life, with fecal urgency being the predominant concern. She may have mild anticipation anxiety around the symptoms but overall, there is no significant psychiatric comorbidity.
As the patient is unable to predict episodes, keeping a symptom diary would allow the patient to take ownership of her care and help her identify potential symptom triggers and prevent additional triggers. Furthermore, education about IBS and obtaining more information, including from reputable patient-oriented websites, for example, the International Foundation for Functional Gastrointestinal Disorders website, may be empowering for her. It should be emphasized that IBS does not lead to cancer or colitis and does not decrease life expectancy. Importantly, the physician should also convey that they will work with the patient to better manage her IBS symptoms.
For dietary modifications, it may be helpful to reduce her caffeine intake, as it stimulates gut motility, and avoid chewing gum or sugarless gum, which may worsen fructose intolerance or contain sorbitol and worsen diarrhea, respectively. As the patient’s symptoms often start after meals, a low FODMAP diet may be suggested, although her IBS symptoms are mild and infrequent and this diet is quite restrictive and difficult to maintain. With regard to treatment, gut-acting agents, such as antispasmodics like dicyclomine, which blunt the gastrocolonic reflex responsible for the postprandial urge to defecate, are appropriate as first-line pharmacologic treatment in this case [Chey et al. 2015b]. These agents would be preferably used on an as-needed basis and are a good choice for this patient. Generally, 10–20 mg taken 30 min before meals may be helpful in reducing, or even preventing, her postprandial symptoms. Over-the-counter enteric-coated peppermint oil may also have a beneficial effect on the patient’s overall IBS-D symptoms, as well as on specific symptoms such as abdominal pain, bloating, and fecal urgency.
In terms of emerging treatments, a trial of rifaximin 550 mg TID for 2 weeks may be considered in preference to eluxadoline, as the patient’s IBS symptoms are mild and intermittent and do not warrant chronic treatment. Rifaximin has also demonstrated efficacy in relieving bloating [Pimentel et al. 2011], which is a bothersome symptom in this patient.
The patient was seen 4 weeks later and reported that avoiding caffeine has reduced her stool urgency to some extent. She has been taking dicyclomine 10 mg as needed for abdominal cramping. She has also tried taking dicyclomine before meals to prevent symptoms on several occasions with good results. Following these measures, she reports a marked improvement, of ~70%, in severity of abdominal pain, bloating, and diarrhea. She has only had one ‘big episode’ since her last visit. The fecal urgency is greatly improved and the patient is considering dating again. She is very pleased with her clinical improvement.
Patient scenario 2: Moderate irritable bowel syndrome with diarrhea
The patient is a 35-year-old man, an attorney, with a 5-year history of abdominal pain and diarrhea, presenting for a second opinion after previously being treated by a different gastroenterologist. His GI symptoms have recently worsened from two semi-formed BMs per day to 3–4 BMs per day that have begun to interfere with his work. He experiences abdominal cramping daily, which is relieved by defecation. On days he is in court, he does not eat until later in the day, as he is worried about fecal urgency. On the weekends, he participates in activities with his children, aged 4 and 7 years, but ‘likes to be near a bathroom’. He goes camping, but drinks bottled water, and his GI symptoms do not worsen after his camping trips. He has never had fecal incontinence but is anxious about this possibility and he has also not been sleeping well. A summary of the patient’s symptoms, medical and psychosocial history, current physical and previous laboratory examination results, including celiac serologies and endoscopic results, and treatment history is presented in Figure 2.

Summary of moderate irritable bowel syndrome with diarrhea case scenario.
Comment, treatment options, and management
Using the MDCP diagnostic classification [Drossman, 2015], this patient has moderate IBS-D (Table 4). Given his camping experiences, a parasitic infestation such as Giardia lamblia was considered. However, stool analysis was negative for any bacterial or viral infections, or a parasitic infestation. It is unlikely he has celiac disease, given negative blood testing and negative duodenal biopsies a year ago. Microscopic colitis and inflammatory bowel disease were also diagnostic considerations, but his colonoscopic biopsies from the left and right colon were within normal limits.
As dietary modifications had provided limited benefit for the patient, he tried the medical food bovine serum immunoglobulin isolate 10 mg BID for 6 weeks and observed a mild decrease in his symptoms. His stool consistency improved to being soft to formed and he experienced less urgency. The patient had previously tried enteric-coated peppermint oil [Cash et al. 2015a] but it did not improve his IBS-D symptoms. Given the patient’s lack of response to dicyclomine and hyoscyamine, an antispasmodic was not suggested.
The patient’s sleep disturbance could potentially be treated with specific medications for insomnia or medications that have sedating effects, such as TCAs. In a post hoc analysis [Drossman et al. 2003], TCAs were efficacious in improving abdominal pain and diarrhea, presumably due to their anticholinergic side effects. Therefore, the patient was given a trial of a TCA, amitriptyline, with an initial dose of 10 mg nightly, increasing to 50 mg over several weeks. Amitriptyline reduced his IBS-D symptoms considerably and improved his sleep quality. Having better control of his IBS-D symptoms also helped with the patient’s symptom-related anxiety. In conjunction with pharmacotherapy, the patient received CBT to reduce maladaptive coping behaviors, as patients with mild-to-moderate IBS symptoms may achieve a benefit [Lackner, 2007; Ljótsson et al. 2011a, 2011b].
At a 6-week follow-up appointment, the patient reported improvement in his IBS-D symptoms but mentioned he was experiencing dry mouth, a known side effect of TCAs [US Food and Drug Administration, 2014b]. Consequently, the amitriptyline dose was not increased. One could also consider using desipramine as an alternative TCA, as its anticholinergic effects are generally less than for amitriptyline. The use of additional agents was considered at this point. The patient received rifaximin 550 mg TID for 2 weeks, but it was not effective. Eluxadoline would be an option and, given its efficacy and safety profile, could have been used earlier in the treatment pathway. Whether to choose eluxadoline as a first- or second-line therapy may be determined by a patient’s most bothersome IBS-D symptoms; for example, a patient who has more severe abdominal pain may benefit from eluxadoline as a first-line treatment. As for any treatment, potential side effects, cost, and availability may also help govern this choice. In general, both rifaximin and eluxadoline could be used after a trial of loperamide or as first-line treatments.
Overall, with a combination of antidepressant treatment and CBT, the patient’s IBS-D symptoms improved to the point he was having 1–2 nonurgent, soft-to-formed BMs per day. In combination with a TCA, CBT helped improve his sleep quality and anxiety.
Patient scenario 3: Severe irritable bowel syndrome with diarrhea
The patient is a 45-year-old woman who has a 25-year history of recurrent abdominal pain, fecal urgency, and loose stools. Over the past 1–2 years her GI symptoms have worsened from 1–2 ‘bad days’ per week, during which she experiences severe abdominal pain and fecal urgency associated with 6–8 loose-to-watery BMs per day, to 4–5 ‘bad days’ per week. She has experienced a few episodes of fecal incontinence when she could not reach a bathroom in time. She has also recently been diagnosed with fibromyalgia. The patient is currently experiencing a period of high stress both at work and in her personal life. Her job requires a lot of travel and she worries about prolonged travel because she needs to be near a bathroom. She now misses work at least several days per month. A summary of the patient’s symptoms, medical and psychosocial history, physical and laboratory examination and endoscopic results, and treatment history is shown in Figure 3.

Summary of irritable bowel syndrome with diarrhea case scenario.
Comment, treatment options, and management
Using the MDCP diagnostic classification [Drossman, 2015], this patient has severe IBS-D (Table 4). She has longstanding symptoms, but they have recently worsened during a more stressful period in her life. As in the previous moderate IBS-D case, microscopic colitis is a consideration, which has a higher prevalence in middle-aged women [Chey et al. 2010]; however, her colonoscopy with biopsies was negative. The patient’s symptoms are severely impacting her daily activities, she has developed symptom-specific anxiety due to the severity and unpredictability of her symptoms, and she also has coexistent fibromyalgia.
In patients with severe symptoms, it is important to implement a multidisciplinary, patient-centered approach to therapy. It is helpful to identify the most bothersome GI symptoms, psychosocial factors that impact symptoms, the patient’s expectations and beliefs about their IBS-D, and response to prior treatments to determine the current treatment options. Establishing a therapeutic patient–provider relationship is a cornerstone of IBS therapy regardless of severity, but it is essential in severe disease. Education, reassurance, and addressing realistic expectations and psychosocial factors can improve a patient’s health status and treatment response. Generally, for a patient with severe IBS-D symptoms, learning to better manage their symptoms and improve daily functioning is a more realistic expectation than anticipating complete resolution of symptoms.
Treatment for severe IBS-D includes pharmacologic and nonpharmacologic approaches. In this case, most IBS-D treatments did not significantly improve the patient’s symptoms, or were associated with bothersome side effects (Figure 3). However, she experienced some symptom improvement with a low FODMAP diet.
Further pharmacologic options for this patient include a 5HT3 antagonist. Although several 5HT3 receptor antagonists (alosetron, ondansetron, and ramosetron) have shown efficacy in IBS-D, ramosetron is not available in the US, and ondansetron appears to be less effective in patients with severe diarrhea, and it does not significantly decrease abdominal pain [Chiba et al. 2013; Cremonini et al. 2012; Fukudo et al. 2014, 2015; Garsed et al. 2014; Krause et al. 2007]. Given the severity of the patient’s symptoms, alosetron may be the more effective option, as it has demonstrated efficacy in improving abdominal pain, fecal urgency, diarrhea, and health-related quality of life in women with severe IBS-D at a dose of 0.5 mg BID [Cremonini et al. 2012; Krause et al. 2007]. Eluxadoline 100 mg BID with food could be suggested, as this medication has shown efficacy in improving fecal urgency [Lembo et al. 2016].
Psychological therapies are also options for this patient and can be used in conjunction with other treatments. CBT may help reduce her symptom anxiety and enable her to develop effective coping strategies to deal with her GI and non-GI symptoms. Hypnotherapy directed toward relaxation and control of intestinal motility followed by ego strengthening may also be helpful. Mindfulness meditation may be useful, as it has been shown to reduce IBS symptom severity [Gaylord et al. 2011]. The choice of psychological therapy is dependent on the patient’s interest and motivation, time, cost, and availability of a trained therapist with experience in IBS.
This patient worked with her physician to institute a combination of treatments that progressively improved her IBS-D symptoms. She continued her low FODMAP diet but consulted with a dietitian to help gradually reintroduce certain foods into her diet. After discussing the benefits and risks associated with eluxadoline and alosetron, the patient agreed to try eluxadoline and she was prescribed a dose of 100 mg BID. Subsequently, her diarrhea, urgency, and incontinence substantially decreased and the number of ‘bad days’ decreased to 1 day per week. Concurrently, the patient began CBT and within 2 months, noted that she felt less anticipatory anxiety, and perceived better control of her symptoms. She also learned how to self-monitor her symptoms and improve her stress management. Although she has missed fewer days of work, she is seriously considering changing jobs to one with less travel, particularly as she needs time to care for her parents.
Summary and conclusion
Currently, there is no standard treatment algorithm for IBS-D, and therapeutic options focus on alleviating symptoms, such as diarrhea and abdominal pain. Establishing a therapeutic relationship with the patient is an essential part of treating IBS. In practice, treatment for IBS-D often encompasses combinations of both pharmacologic and nonpharmacologic therapies. With the emergence of new pharmacologic treatments, medical foods, and diets for IBS-D, treatment needs to be individualized to each patient’s situation. The choice of treatment depends on the predominant symptom, severity of the disorder, presence of comorbidities, prior treatment experiences, and patient and clinician preferences. Combining gut and centrally acting agents is an effective strategy, particularly for severe IBS.
To provide clinicians with information on how current and emergent treatments for IBS-D could be applied in clinical practice, we briefly reviewed the available therapies and presented mild, moderate, and severe IBS-D case scenarios for which potential treatment options were discussed. The importance of the interplay of GI symptoms and their psychosocial impact, as well a patient-centered approach to therapy, were discussed. Additionally, for each case, the need for combinations of treatments to enable patients to achieve the most effective relief of their IBS-D symptoms and better management of their disorder was considered.
Over the past 2 decades, clinical trials of investigational agents for IBS have focused on the treatment of specific symptoms. In the future, more specific therapies should become available, as our understanding of the complex, multidimensional pathophysiologic mechanisms involved in IBS advances, such as the role of altered epithelial barrier, secretion, bile acid metabolism, neurohormonal regulation, intestinal immune function, gut microbiota, and brain–gut regulation, among others [Wadhwa et al. 2015]. Furthermore, while extremely challenging, investigating combinations of treatments in clinical trials of IBS-D would shed more light on this complex disorder, particularly as in ‘real-world’ clinical practice, combination treatments, such as those described in the clinical scenarios in this manuscript, are commonly utilized for patients with IBS-D. Thus, evidence-based data for combination treatments would be very useful for clinicians.
Footnotes
Acknowledgements
All authors met the International Committee of Medical Journal Editors authorship criteria. The authors take full responsibility for the scope, direction, and content of the manuscript, participated in drafting and critically reviewing the manuscript, and approved the submitted manuscript. Neither honoraria nor payments were made for authorship. They would like to thank Gail Rickard, PhD, of Complete HealthVizion, Inc., Chicago, IL, for editorial assistance in the writing and revision of the draft manuscript on the basis of detailed discussion and feedback from all the authors.
Funding
The editorial assistance provided to the authors was funded by Allergan plc, Parsippany, NJ.
Conflict of interest statement
Financial arrangements of the authors with companies whose products may be related to the present report are listed below, as declared by the authors. Susan Lucak has served as an advisor or consultant for Actavis, Inc., a subsidiary of Allergan plc, Takeda, Salix Pharmaceuticals, Ironwood Pharmaceuticals, and Prometheus, and has served as a speaker or received lecture fees from Actavis, Inc., a subsidiary of Allergan plc, Takeda, Salix Pharmaceuticals, and Ironwood Pharmaceuticals. Lin Chang has participated in advisory boards for Actavis, Inc., a subsidiary of Allergan plc, Commonwealth Laboratories, QOL Medical, Takeda, AstraZeneca, Synergy, BioAmerica, and Ardelyx Pharmaceuticals. Albena Halpert has served as an advisor or consultant for Allergan plc, Ironwood Pharmaceuticals, Prometheus, and Takeda, and has previously received unrestricted educational grants from Forest Laboratories, a subsidiary of Allergan plc, and Takeda. Lucinda A. Harris has participated in advisory boards for Actavis, Inc., a subsidiary of Allergan plc, Commonwealth Laboratories, QOL Medical, Ironwood Pharmaceuticals, Salix, and Synergy, has received grant support from Alvine Pharmaceuticals, Inc. and Rhythm Pharmaceuticals, has received fees for CME lectures from Forest Laboratories, a subsidiary of Allergan plc, and Ironwood Pharmaceuticals, and has served as a consultant for Synergy.
