Abstract
Objectives:
Although effective, direct acting antiviral (DAA) therapies for genotype 1 (GT 1) hepatitis C virus (HCV) have been associated with compliance challenges. Additionally, treatment at predominantly community-based centers has been associated with low retention of patients on treatment and higher dropout rates. The OPTIMAL Phase IV interventional trial (ClinicalTrials.gov Identifier: NCT01405027) was designed to evaluate the impact of an education program for community investigator (CI) sites participating in a Chronic Liver Disease Foundation study treating chronic GT 1 HCV patients.
Methods:
This physician educational program was administered by 22 Hepatology Centers of Educational Expertise (HCEE) academic sites to 33 CI sites asked to participate from December 2011 to July 2012. The HCEE mentors from DAA-experienced academic sites educated those at CI sites on therapeutic management, practice, and patient outcomes through a series of four standardized educational sequence visits regarding the use of first generation HCV protease inhibitors and the overall treatment of HCV.
Results:
Treatment duration compliance rates for patients treated at CI sites versus those treated at HCEE academic sites were evaluable in 77 of 84 HCEE academic site patients, 102 of 113 patients treated at CI sites, and 179 of 197 overall patients. The treatment duration compliance rates for patients treated at HCEE academic sites, CI sites and overall were 85.4 ± 25.39%, 83.8 ± 27.37%, and 84.5 ± 26.48%, respectively, and did not differ statistically between the groups (p = 0.49). Almost half (47%) of the patients in the study achieved a sustained virological response for 24 weeks (SVR24) regardless of the type of site (p = 0.64). Safety profiles were similar at both HCEE and CI sites.
Conclusions:
These results demonstrated that education of CI sites unfamiliar with DAAs resulted in patient outcomes consistent with those observed at DAA-experienced academic sites.
Introduction
As recently as the beginning of the current decade, combined treatment with pegylated interferon-α (PEG) and ribavirin (RBV) were the standard of care (SOC) therapy for patients chronically infected with the hepatitis C virus (HCV). This changed in May 2011 when the first direct acting antiviral (DAA) therapies for genotype 1 (GT 1) HCV, telaprevir (TVR) and boceprevir (BOC) were approved by the US Food and Drug Administration (FDA) for use in combination with PEG/RBV for adult patients chronically infected with GT 1 HCV [Chae et al. 2013]. Both of these drugs were approved for treatment-naïve patients and patients who had been previously treated with interferon (IFN) based regimens, including those with compensated liver cirrhosis [Merck & Co, 2014; Vertex Pharmaceuticals Inc., 2013]. Additionally, both DAAs inhibit the viral NS3/4A serine protease, which has been implicated as an essential enzyme for viral replication [Merck & Co 2014; Vertex Pharmaceuticals Inc., 2013]. Although these regimens offer higher response rates [Jacobson et al. 2011; Sherman et al. 2011; Zeuzem et al. 2011; Poordad et al. 2011; Pearlman, 2012], they have been associated with several limitations [Chae et al. 2013]. For example, these drugs may provoke severe side effects including anemia [Merck & Co, 2014; Vertex Pharmaceuticals Inc., 2013]. Additionally, their complicated dosing schedules, pill burden, food effects and drug–drug interactions are cumbersome and may engender noncompliance [Asselah and Marcellin, 2012]. Therefore, efforts towards increasing compliance rates are required.
The results of the prospective, multicenter Weight-Based Dosing of PegINterferon alfa-2b and Ribavirin (WIN-R) trial, which evaluated the dose of RBV for HCV patients at both academic-based and community-based sites, revealed that treatment at predominantly community-based centers was associated with low retention of patients on treatment and higher dropout rates as a consequence [Jacobson et al. 2007]. Although the results of a more recent phase III trial showed that no new adverse events (AEs) or treatment-limiting side effects associated with BOC were observed compared with those reported in patients PEG/RBV, a higher dropout rate was found for those in the BOC treatment groups compared with the PEG/RBV group [Poordad et al. 2011]. This was also noted when TVR was added to PEG/RBV [Jacobson et al. 2011].
The high dropout and noncompliance rates associated with DAAs may be overcome in part through physician education initiatives. Such physician education activities are based on a belief that improved knowledge leads physicians to change how they practice and to better patient outcomes as a result. Continuing education is highly variable, ranging from passive, didactic, large group presentations to highly interactive learning methods such as workshops, small groups and individualized training sessions. A study evaluating the impact of continuing medical education on physician behavior and healthcare outcomes reported that a series of formal educational interventions including small groups and teleconferences demonstrated positive outcomes on physician performance and a similar impact on health care outcomes. Specifically, ‘…participating in interactive, challenging activities delivered in a longitudinal or sequenced manner, with the provision of enabling methods to facilitate implementation in the practice setting, have increased potential for positively affecting physician performance and the health of the patients they serve’ [Davis et al. 1999]. In fact, a systematic review of faculty development initiatives designed to improve teaching effectiveness in medical education revealed that the use of experiential learning, feedback, effective relationships with peers and diverse educational methods were among the most important factors in the success of such programs [Steinert et al. 2006].
Regarding DAAs and HCV in particular, the need for physician education initiatives is driven by the movement away from prior therapy and toward current all-oral therapy. Justification for such physician education initiatives in the current setting would stem from the complexity and tolerability of the treatment, which has transitioned to easier agents, but a stronger dependence on adherence and appropriate patient/treatment selection (thus retaining relevance).
The aim of the current study was to assess the effects of a physician educational program conducted by members of DAA-experienced academic sites on therapeutic management practice and patient outcomes at community investigator (CI) sites. This was achieved through the OPTIMAL Phase IV interventional trial [ClinicalTrials.gov identifier: NCT01405027].
Methods
Ethical considerations
This study was conducted in accordance with Good Clinical Practice (GCP) as outlined in the International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH) Guidelines, E6 Good Clinical Practice: Consolidated Guidance and other applicable laws and regulations. In addition, it was conducted in accordance with the USA Code of Federal Regulations. Prior to the initiation of the trial, all trial sites acquired institutional review board or independent ethics committee approval for the trial, including the protocol, informed consent and other trial documents.
Educational program
The Chronic Liver Disease Foundation (CLDF) is a nonprofit 501(c) (3) educational organization dedicated to increasing awareness of the effect of chronic liver disease in the United States. CLDF has developed a network of Hepatology Centers of Educational Expertise (HCEE) dedicated to educating health professionals about the management of chronic liver disease. These sites included experienced clinicians who had participated in clinical trials of the first HCV DAA therapies approved in 2011.
The phase IV OPTIMAL interventional trial was designed to evaluate the impact of an education program for CI sites participating in a CLDF study treating chronic GT 1 HCV patients. The educational program was administered by 22 HCEE academic sites to 33 CI sites asked to participate from December 2011 to July 2012. The program consisted of a series of four standardized educational sequence visits regarding the use of first generation HCV protease inhibitors and the overall treatment of HCV to the CI sites within the geographic region of the HCEE representative, including face-to-face meetings. Each CI site was required to have experience in treating HCV, but no previous clinical trial experience with BOC or TVR.
The first sequence visit was conducted in person, and included a review of protocol and safety/efficacy data with first generation DAAs and strategies for patient evaluation/management. The second sequence visit, which was combined with the first sequence visit, was completed prior to patient enrollment with the goal of formulating an education plan for the practice and strategies for answering questions. In the third sequence visit, records on OPTIMAL patients were reviewed and feedback was provided in person. The fourth sequence visit was conducted by teleconference within 60 days of the third sequence visit and included a review of OPTIMAL patient laboratory results and treatment responses, followed by feedback on side effect management. In addition to the sequence visits, the HCEE offered ad hoc support to CI sites via email and phone consultations.
Of the 29 CI sites who enrolled patients, the average time from sequence visits 1 and 2 to the first patient enrolled was 75 days (range: 0–257). All sites completed sequence visits 3 and 4. The average time from the first patient enrolled to sequence visit 3 was 45 days (range: 12–145). The sequence visit 4 teleconference took place 69 days on average from sequence visit 3, with 66% of the teleconferences taking place within the 60 day protocol-defined window.
Study endpoints
The primary study endpoint was the treatment duration compliance rate for patients treated at CI sites versus those treated at HCEE academic sites. Secondary endpoints included sustained virological response (SVR) and safety comparisons between CI sites and HCEE academic sites. GT 1 HCV infected patients were treated with open label BOC/PEG/RBV according to the US label [Merck & Co, 2014].
Statistical analyses
All summaries and statistical analyses were performed using SAS, version 9.2 or higher. Descriptive summaries consisted of frequencies and percentages for categorical measures and of the number of patients, mean, standard deviation, median, minimum and maximum values for continuous measures. All treatment group comparisons were tested at the α = 0.05 significance level using a two-tailed statistical test.
The primary endpoint for this study (treatment duration compliance rate) was calculated as the actual treatment duration in weeks divided by the expected duration in weeks. Mean rates were compared between groups using an analysis of variance model with corresponding 95% confidence intervals.
Results
Patient demographic and disposition data
Patient baseline demographic information is presented in Table 1. A total of 197 patients were included in the study: 84 were enrolled at HCEE academic sites and 113 at CI sites. While approximately half of all patients (51.8%) were male, the percentage of male patients seen at the HCEE academic sites (63.1%) was higher than that for patients seen at CI sites (43.4%). The majority of patients were Caucasian at HCEE academic sites, CI sites and overall (59.5%, 66.4%, and 63.5%, respectively). Additionally, the majority of patients in the study were infected with HCV GT 1a genotype (73.8%, 67.3%, and 70.1%, respectively) versus GT 1b genotype (26.2%, 31.0%, and 28.9%, respectively). The mean viral loads were 4.66 million IU/ml at the HCEE academic sites, 3.29 million IU/ml at the CI sites, and 3.88 million IU/ml for the overall patient population. A lower percentage of treatment naïve subjects were enrolled at HCEE academic sites versus CI (35.7% versus 61.1%). It should be noted that there were no differences in disease severity, such as the number of patients with cirrhosis, between the HCEE academic sites and the CI sites (Table 1). Although the overall discontinuation rates were similar at HCEE academic sites and CI sites (48.8% versus 47.8%), the reasons given for discontinuation differed (Figure 1). For example, treatment futility was a more often cited reason for patient discontinuation at academic HCEE sites (36.6%) as opposed to CI sites (24.1%), while investigator judgment, serious AEs (SAEs, and loss to follow up were more frequent in patients at CI sites compared with those at HCEE academic centers (24.1% versus 17.1%, 5.6% versus 2.4%, and 13% versus 4.9%, respectively; Figure 1).
Patient baseline demographics.
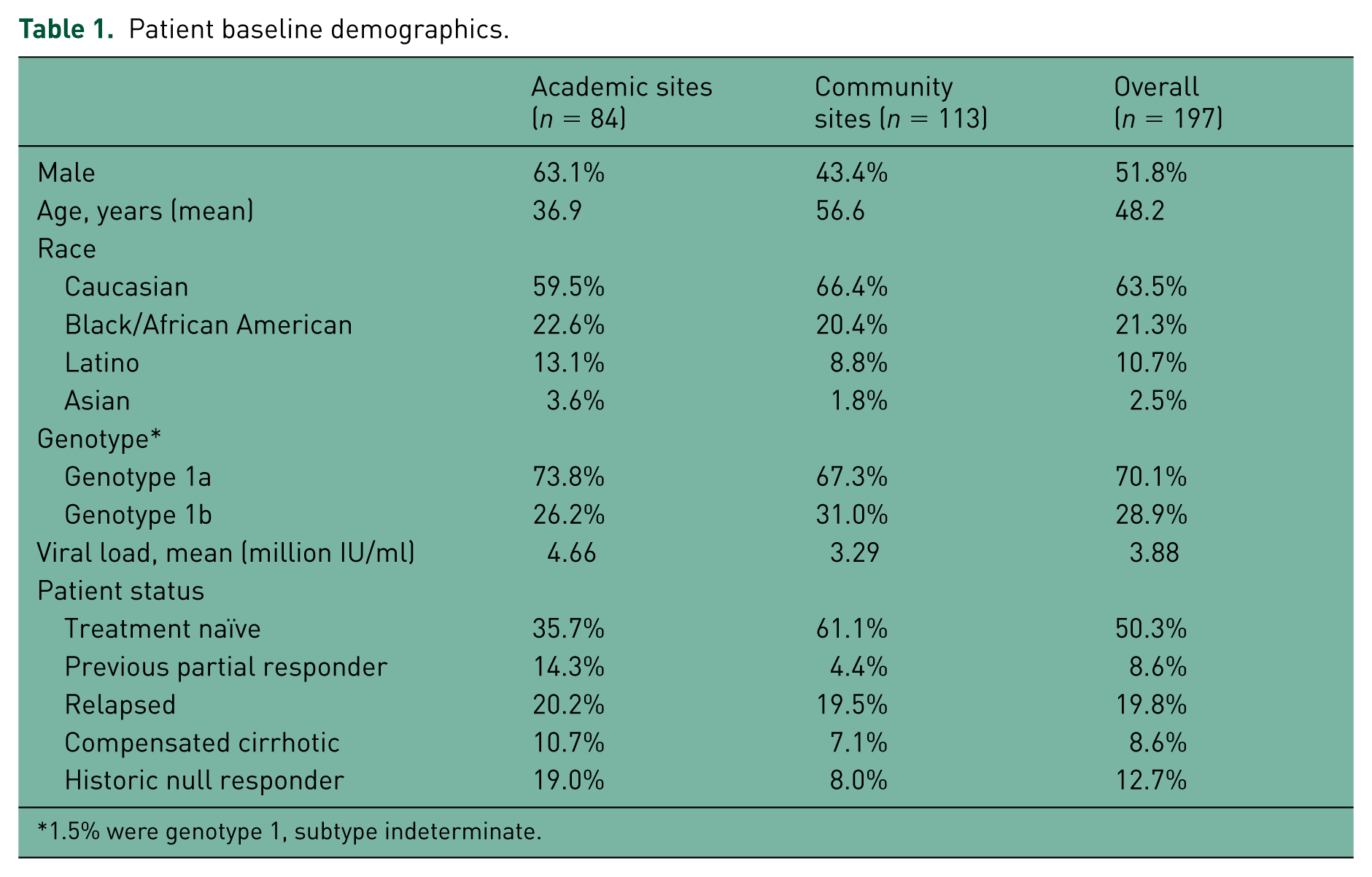
1.5% were genotype 1, subtype indeterminate.

Reasons for patient discontinuation.
Study endpoint data
The primary study endpoint, which was the treatment duration compliance rate for patients treated at CI sites versus those treated at HCEE academic sites, was evaluable in 77 of the 84 patients treated at HCEE academic sites, 102 of the 113 patients treated at CI sites, and 179 of the 197 overall patients. The mean ± standard deviation treatment duration compliance rates for patients treated at HCEE academic sites, CI sites and overall were 85.4 ± 25.39%, 83.8 ± 27.37% and 84.5 ± 26.48%, respectively, and did not differ statistically between the groups (p = 0.49).
For the secondary endpoint of SVR, 47% of the patients in the study had undetectable HCV RNA at the final follow-up visit regardless of the type of site (p = 0.64) (Figure 2). Regarding safety comparisons, the treatment-emergent AEs in descending order (>10% in either the HCEE academic group or the CI site group) were anemia (51.2% versus 40.7%), fatigue (64.3% versus 24.8%), nausea (42.9 versus 22.1%), rash (28.6% versus 23.9%) and headache (26.2% versus 15.0%).
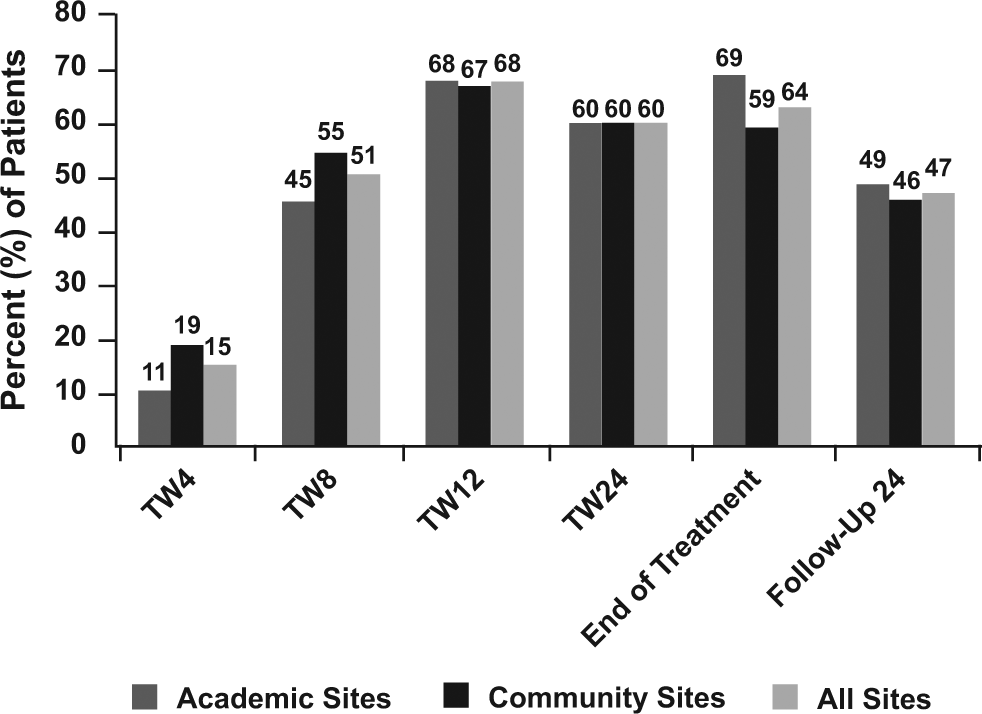
Sustained virological response rate by group.
Data pertaining to other safety measures, such as SAEs and discontinuations due to safety, revealed that anemia and depression were the only SAEs reported by more than one patient. Therefore, the SAE rate was 7.1% in each of the site-specific groups, as well as the overall study population. For the HCEE academic and CI site groups, respectively, 1.2% and 1.8% of patients had anemia reported as a SAE, and 1.2% and 0.9% had depression reported as a SAE. For the overall study population, 1.5% and 1.0% experienced anemia and depression as SAEs, respectively. No deaths were recorded, and the discontinuation rates due to safety concerns were 2.4%, 3.5% and 3.0% for the HCEE academic site group, the CI site group and the overall study population, respectively.
The nadir hemoglobin concentration was comparable between the two groups regardless of the World Health Organization (WHO) grade status. Furthermore, WHO Grade 1 hemoglobin concentrations (9.5 to <11.0 g/dl) represented the most often detected nadir level, accounting for 34.5% of patients seen at HCEE academic settings, 33.6% at CI sites and 34% in the overall study population. A similar trend was found for the nadir neutrophil absolute count, such that levels were comparable between the two groups, and those with a WHO Grade 1 nadir neutrophil absolute count (⩾1000 cells/mm3) represented the majority, accounting for 48.8% of patients seen at HCEE academic settings, 58.4% at CI sites and 54.3% in the overall study population.
It was also noteworthy that the percentages of those with hemoglobin concentrations <10.0 g/dl increased gradually from 11.3% and 7.6% in HCEE academic site patients and CI site patients, respectively, at treatment week (TW) 4, peaking for both groups at TW12 (34.7% and 40.4%, respectively).
Anemia management was at the discretion of the sites and involved RBV dose reduction and/or use of erythropoietin. Erythropoietin was more commonly used at HCEE academic sites versus CI sites (31.0% versus 13.3%). Very few patients in either group showed hemoglobin concentrations <8.0 g/dl. At the end of follow up, all patients had hemoglobin >10.0 g/dl.
Discussion
The results of the current study demonstrated that education of CI sites unfamiliar with DAAs resulted in patient outcomes consistent with those observed at DAA-experienced academic sites. The primary study endpoint, the treatment duration compliance rate for patients treated at CI sites versus those treated at HCEE academic sites, did not differ statistically between the groups. Regarding SVR, 47% of the patients in the study had undetectable HCV RNA at the final follow-up visit regardless of the type of site.
Although the educational program seemed to meet with success regarding its primary endpoint, a few differences were still noted between the two types of sites. For example, patient discontinuations due to treatment futility were higher at HCEE academic sites, which is likely attributable to a higher percentage of difficult-to-cure patients. Additionally, the lower percentage of patients who discontinued based on investigator judgment or because they were lost to follow up at HCEE academic sites may have been attributable to the larger infrastructure of site personnel familiar with patient selection and management. Safety-related differences were also noted. AEs, for example, were reported less frequently at CI sites, possibly due to less familiarity with the rigorous reporting practices used in clinical registration trials. Although more patients experienced hemoglobin concentrations ⩽10 g/dl at CI sites, likely due to a lower rate of RBV dose reductions and erythropoietin use, SAEs and discontinuations due to anemia were not increased. Perhaps most importantly, 51% of patients had undetectable HCV RNA at TW8, thereby qualifying for shorter duration of therapy.
Sustained response rates were much lower in this study compared with those reported in the phase III clinical trials as well as other literature [Merck & Co, 2014; Poordad et al. 2011; Bacon et al. 2011], likely due to the real world population treated in this trial compared with those typically included in phase III registration trials.
The treatment landscape of hepatitis C has evolved quickly over the past few years, and while this regimen will not be used to the same extent in the future, it does highlight an important concept. Even treatments involving difficult regimens with high adverse event rates can be used with success in community settings when paired with an academic site for support and education. This should be applicable to other regimens, particularly those that are all oral, and have fewer AEs. Despite the newer and safer treatment environment, current therapies will continue to challenge treaters as they consider issues pertaining to patient adherence, patient genotype, prior treatments, cirrhosis, etc. Furthermore, the ever evolving managed care landscape may present additional challenges in clinical practice. Thus, educational programs, such as the one highlighted in the present study, will continue to be of value to treaters, even in this new therapeutic environment.
Footnotes
Acknowledgements
Editorial assistance for this manuscript was provided by Lisa D. Pedicone, PhD and William R. Perlman, PhD.
Conflict of interest statement
FP discloses grants/research support from AbbVie, Achillion, Bristol-Myers Squibb, CLDF, Gilead, Idera, Intercept, Janssen, and Merck; consultant/speaker bureau for AbbVie, Gilead, Kadmon, Janssen, and Salix; and advisory board membership for AbbVie, Achillion, Bristol-Myers Squibb, Gilead, Merck, Novartis, and Tibotec/Janssen. VR discloses grants/research support from CLDF and Gilead; consultant/speaker bureau for AbbVie; and advisory board membership for AbbVie, Gilead, and Merck. VP discloses consultant/speaker bureau for Abbvie, Gilead, Salix and Bayer, and advisory board membership for Gilead. RB discloses grants/research support from AbbVie, Gilead, Janssen, and Merck; and consultant/speaker bureau for AbbVie, Gilead, Janssen, and Merck. MK discloses grants/research support, consultant/speaker bureau, and advisory board membership from/for AbbVie, Bristol-Myers Squibb, CLDF, Gilead, Janssen, Merck, Roche, and Salix. FR discloses grants/research support from Bristol-Myers Squibb, CLDF, and Merck; consultant/speaker bureau for Gilead; and advisory board membership for Gilead and Janssen. LB discloses grants/research support from AbbVie, Bayer, CLDF, GI Dynamics, Gilead, Merck, Ocera, Salix, and Takeda; consultant/speaker bureau for AbbVie, Gilead, and Janssen; and advisory board membership for AbbVie and Merck. DL discloses grants/research support from CLDF; consultant/speaker bureau for AbbVie, CLDF, and Simply Speaking; and advisory board membership for Bristol-Myers Squibb. KB discloses grants/research support from AbbVie, Bristol-Myers Squibb, CDC Foundation, CLDF, Duke, Gilead, Hyperion, Janssen, Merck, and Vertex; consultant/speaker bureau for AbbVie, CLDF, Gilead, HCV Viewpoint, Peer to Peer, and Simply Speaking; advisory board membership for AbbVie, Bristol-Myers Squibb, CLDF, Gilead, Janssen, Merck, and Vertex; editorial board involvement for the American Journal of Transplantation, CLD Journal, Images in Transplantation; and other financial material support/boards from/for ABIM, A2All, CLDF, and DSMB. SF discloses consultant/speaker bureau for Merck and Vertex. HM discloses consultant/speaker bureau and advisory board membership for Gilead, BMS, Merck, Vertex, Abbvie and Salix. SH discloses grants/research support from CLDF; consultant/speaker bureau for AbbVie, FibroGen, GI Consultants SA, Nimbus Discovery, NGM Biopharmaceuticals, CLDF, Gilead, Janssen, and Vidico; advisory board membership for CLDF; and editorial board involvement with the Hepatology Journal. MA, MB, GF, RS, MB, HA, JC, and WK disclose grants/research support from CLDF.
Funding
This work was supported by the Chronic Liver Disease Foundation through an investigator initiated study grant from Merck & Co., Kenilworth, NJ, USA. The work is a product of the authors alone with no involvement of the sponsor.
