Abstract
Diabetes is the leading cause of chronic kidney disease and end stage renal disease (ESRD) globally. Diabetic kidney disease, previously known as diabetic nephropathy, is a general term used for deterioration of kidney function in patients with diabetes. It is important to diagnose and manage diabetic kidney disease early, so as to reduce the incidence of ESRD and the cardiovascular complications of diabetes. This article looks at the epidemiology, presentation and management of diabetic kidney disease.
Clinical case scenario
A 56-year-old patient attended his annual diabetic review with urine and blood tests performed prior to the appointment. He was diagnosed with type 2 diabetes 5 years ago. His HbA1c last year was 52 mmol/mol; no oral hypoglycaemic treatment was prescribed. His updated HbA1c has risen to 66 mmol/mol. His albumin-to-creatinine ratio (ACR) is 6.2 mg/mmol, blood pressure (BP) 122/80 mmHg and body mass index (BMI) 31 kg/m2. He is a non-smoker with an alcohol intake within recommended limits. He works as a tree surgeon and is physically active. His regular medications are ramipril 10 mg once daily and atorvastatin 20 mg at night. What do you think of his HbA1c, ACR? What management options would you consider at this point?
Introduction
Albuminuria is the hallmark of the beginning of diabetic kidney disease. Normally albumin, a type of protein needed for muscle and tissue repair and immune response against infections, is filtered in a healthy kidney and not evident in the urine in large quantities. In chronic kidney disease (of any aetiology) albumin leaks into urine in increasing quantities (see Fig. 1). In the past, terms such as microalbuminuria (ACR >2.5 mg/mmol for men and >3.5 mg/mmol for women) and macroalbuminuria (urine ACR >30 mg/mmol) were used. There is a move away from using the terms micro and macro-albuminuria to albuminuria with subsequent quantification of the measured excreted amount, which helps to classify diabetic kidney disease and estimate its prognosis (see Table 1). Albuminuria can be quantified by a variety of methods including an early morning/first-void spot sample or a random spot urine test.
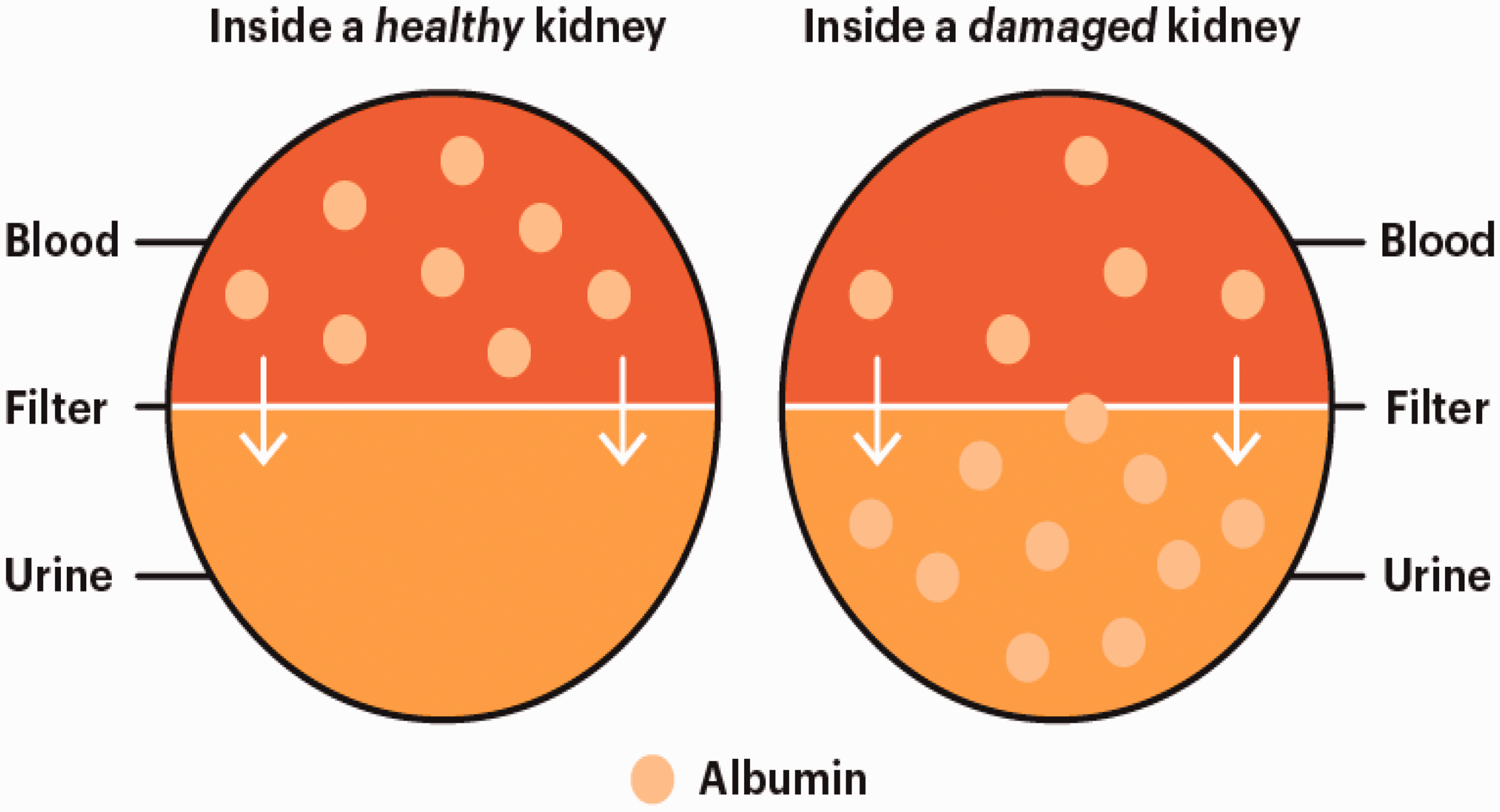
Albuminuria in kidney disease.
Prognosis of chronic kidney disease.
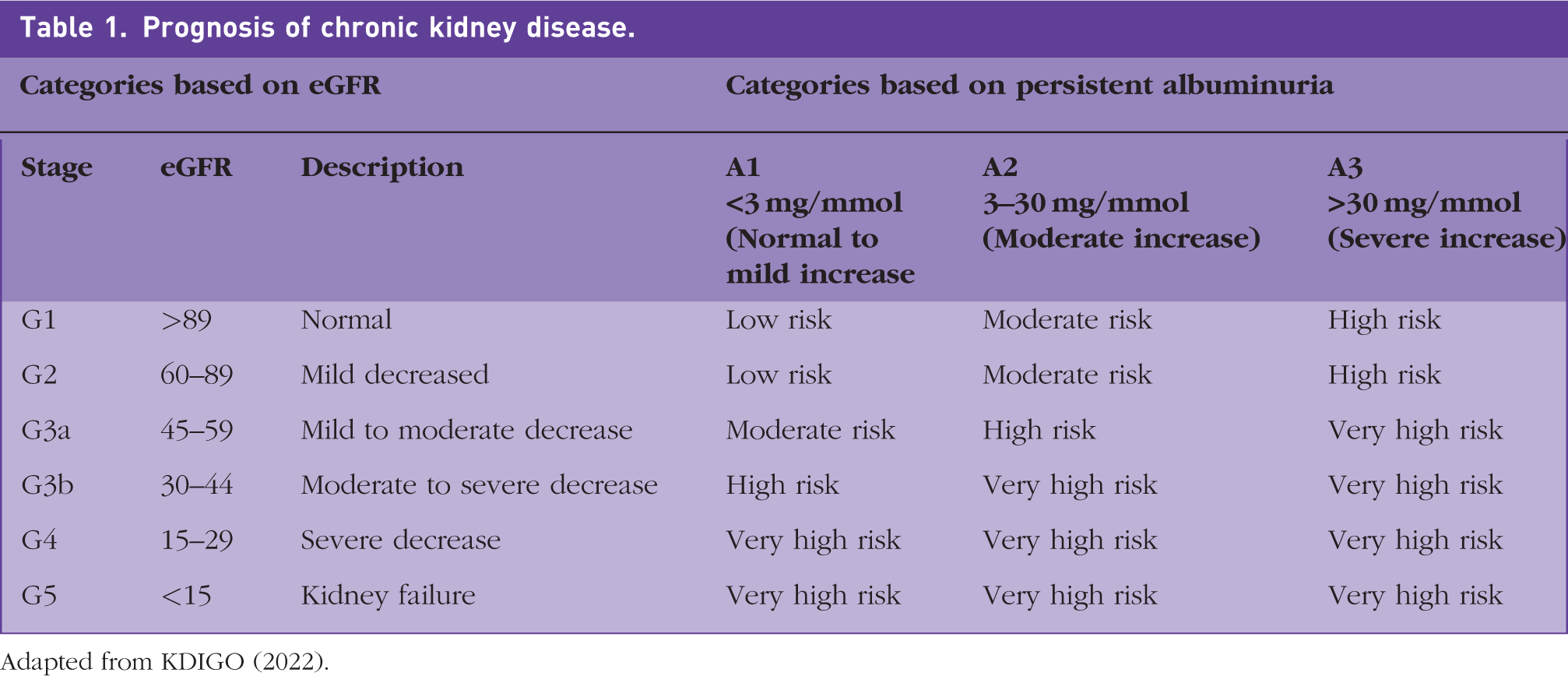
Adapted from KDIGO (2022).
Albuminuria is an independent marker of chronic kidney disease (CKD) even in the presence of normal blood tests for kidney function (i.e., estimated glomerular filtration rate (eGFR) >59 ml/min/1.73 m2). The presence of moderate levels of albuminuria is predictive of ESRD, cardiovascular disease and increased morbidity and mortality. The onset of mild albuminuria is associated with doubling the risk of cardiovascular disease in people with diabetes (SIGN, 2010). The national kidney foundation has an easy to remember traffic light system to understand the severity of albuminuria.
Epidemiology
The rising incidence of diabetic kidney disease in the UK is caused by an increase in the prevalence of diabetes, a younger age of onset of type 2 diabetes, and longer life expectancy among people with diabetes. The prevalence of the CKD (stage G3a, see Table 1) is approximately 18% in the diabetic population. There are also racial differences, the prevalence of proteinuria and severe CKD (stages 4 and 5) are higher in black and Asian communities compared with the white population (Dreyer et al., 2009). CKD is known to be more prevalent in the elderly, women, racial minorities, and in people with diabetes and hypertension.
Pathogenesis
The pathophysiology behind development of microalbuminuria includes glomerular enlargement, glomerular basement membrane (GBM) thickness, and effacement of podocyte foot process, which leads to leakage of albumin from the glomeruli. GBM effectively works as a complex biological sieve and the above changes can compromise its function (see Fig. 1). Dysfunction of GBM clinically presents as an intermittent albuminuria, which with time, progresses to persistent albuminuria, and thus, eventually increasing amounts of albuminuria.
Vascular endothelial dysfunction is known to be associated with microalbuminuria and cardiovascular disease. Risk factors for diabetic kidney disease - advancing age, smoking, reduced physical activities, obesity, dyslipidaemia and insulin resistance - are known to cause endothelial damage. Both hyperglycaemia and dyslipidaemia can contribute to diabetic renal disease by causing renal arteriosclerosis.
Clinical presentation
Initially, diabetic kidney disease is asymptomatic. Signs and symptoms in the later stages could be lower leg oedema, breathlessness (due to fluid overload), dark urine (due to haematuria) and tiredness (due to anaemia). As CKD does not present with symptoms initially, it is important to screen for microalbuminuria annually as part of the annual diabetic review. An early diagnosis and intensive treatment can slow the progression of CKD.
Urine albumin excretion can be temporarily increased by physical exertion, intercurrent infections and illnesses including diabetic ketoacidosis. Interestingly, orthostatic albuminuria is common in tall, thin, young individuals aged <30 years. Therefore, SIGN guideline suggests having two positive results out of three, before albuminuria is confirmed (SIGN, 2010). Common causes of temporary and persistent albuminuria have been highlighted in Table 2.
Common causes of albuminuria.

Investigations
National Institute for Health and Care Excellence (NICE) guidelines (NICE, 2023a) suggest an annual review for a person with diabetes to include blood pressure and blood tests (for HbA1c, full blood count (FBC) and renal function) along with urine ACR (see Box 1). In cases of a new or worsening proteinuria, assessment should include urine analysis, culture for urine infection and consideration of a renal ultrasound. Annual diabetic review in primary care.
BMI
Blood test (for FBC, HbA1c, renal function and lipid profile)
Urine test (for ACR and albuminuria)
Discuss urine/ blood result, any new or worsening symptoms, medicine compliance and side effects
Blood pressure (home or ambulatory BP target <145/85, in 80 years or above, and <135/85 in <80 years; If concomitant CKD, then target is <130/80)
Foot screening
The presence of diabetic retinopathy has strong association with development of diabetic kidney disease. The onset of diabetic kidney disease can lead to onset or progression of diabetic retinopathy (SIGN, 2010). It is possible to have non-diabetic aetiology of renal disease in people with diabetes. For example, consider a non-diabetic cause of renal disease should there be an absence of progressive diabetic retinopathy or other factors present in Box 2. A referral to secondary care would be suitable in these cases. Indications for referral to secondary care.
If non-diabetic cause of renal disease suspected, as in following instances –
sudden development of proteinuria or heavy proteinuria (>100 mg/mmol)
absence of progressive retinopathy or
presence of systemic illness
Risk of renal replacement therapy over 5 years (determined by using four-variable kidney failure risk equation) is > 5%
ACR > 30 mg/mmol plus, unexplained haematuria
Co-existing treatment resistant hypertension (poorly controlled BP despite being on therapeutic doses of four different anti-hypertensives.
A sustained reduction in eGFR of 15 ml/min/1.73 m2 or 25% or more in a year
Suspected rare causes of CKD, including renal artery stenosis
Management
Management of diabetic kidney disease incudes management of the risk factors presented in Box 3. Managing the four modifiable risk factors for diabetic kidney disease- smoking, hyperglycaemia, hypertension and dyslipidaemia – can delay the onset and slow the progression of CKD in diabetes. The management can be divided into non-pharmacological (lifestyle intervention) and pharmacological treatment. Risk factors for development and progression of diabetic nephropathy. Non-modifiable:
Family history
Male gender
Advancing age
Long duration of diabetes Modifiable:
Smoking
Hyperglycaemia
Hypertension
Hypercholesterolemia
The non-pharmacological treatments include weight loss, increase in physical activity and smoking cessation. These have overall beneficial effects for glycaemic control and also reduce the risk of macrovascular (cardiovascular disease) and microvascular complications of diabetes (nephropathy, retinopathy, and neuropathy). The dietary recommendation in diabetic kidney disease is maintaining a healthy, balanced diet with an increase in vegetables, whole grains, fruits, plant-based proteins and unsaturated fats with less processed meats and simple carbohydrates (KDIGO, 2022). In the later stages of CKD, a reduction in total protein intake may be recommended, as protein is associated with high levels of phosphate, which is detrimental to kidney function. Specialist dietetic input for these patients is essential.
Hyperglycaemia
The onset of diabetic kidney disease can be prevented or delayed by good control of blood glucose and blood pressure. For example, a study showed that reduction in HbA1c from 63 to 53 mmol/mol, was associated with a reduction in albuminuria by 24% (UKPDS, 1998). Similarly, the Diabetes Control and Complications Trial showed that intensive glycaemic control in people with type 1 diabetes delays the onset and slows the progression of microvascular complications of diabetes (nephropathy, retinopathy and neuropathy).
Hypertension
Studies have shown that irrespective of the baseline blood pressure, treatment with anti-hypertensives reduces the risk of diabetic renal disease. The target BP for people with CKD (for those both with and without type 1 or type 2 diabetes) is more stringent than people without CKD. In those with CKD and in ACR < 70, target clinic BP is <140/90 (aim for systolic BP 120–139), whereas in those with an ACR ≥70 the target BP is <130/80 (aim for systolic BP 120–129).
For individuals with diabetes, CKD and hypertension, use an angiotensin converting enzyme inhibitor (ACE-I) or an angiotensin receptor blocker (ARB) first line. Both the ACE-I and ARBs reduce proteinuria by preferentially dilating efferent renal arteriole, which leads to reduction in intraglomerular blood pressure. By doing so, they both confer reno-protective effects. Both of these medicines can reduce the level of albuminuria and slow the progression of diabetic kidney disease.
The NICE guidelines committee for management of CKD in diabetes did not find any difference between the results of ACE-I or ARBs for albuminuria, ESRD, cardiovascular or all-cause mortality (NICE, 2021). However, SIGN guidelines suggest that irrespective of the BP, a type 1 diabetes patient with albuminuria should be treated with an ACE-I, and a type 2 diabetes patient with albuminuria with either an ACE-I or an ARB (SIGN, 2010). It is important to note that although most clinical trials have no race-specific outcomes, treatment with an ACE-I and ARBs tend to be more effective in Caucasian populations than black populations, due to higher renin-angiotensin activity.
Albuminuria
Albuminuria is an independent risk factor for the progression of diabetic kidney disease. Latest NICE guidelines (NICE, 2023a) for patients with type 2 diabetes and CKD suggest titrating an ACE-I or ARB to the maximum tolerated dose and:
Offering a sodium-glucose cotransporter-2 (SGLT-2) inhibitor to patients with ACR above 30 mg/mmol Considering SGLT-2 inhibitor if the ACR is between 3 and 30 mg/mmol
Prescribe the SGLT-2 inhibitors, which are licensed for this use (currently only dapagliflozin) and meet the relevant eGFR threshold (for dapagliflozin do not initiate if eGFR < 15). It is important to be aware of the reduced hypoglycaemic efficacy of SGLT-2 inhibitors with reducing eGFR (e.g. the British National Formulary advises consideration of additional anti-hypoglycaemic agent if eGFR goes below 45 in a patient on dapagliflozin).
Dyslipidaemia
Lipid-lowering drugs can reduce the progression of renal arteriosclerosis. Many randomised controlled trials, cohort studies and meta-analysis have shown that statins can reduce proteinuria, as well as slow down the onset and progression of diabetic renal disease. In all patients with CKD offer 20 mg atorvastatin. There is no need to assess cardiovascular risk.
Complications
The complications of diabetic kidney disease include anaemia, renal bone disease, ESRD and an increase in mortality and morbidity. The onset and progression of cardiovascular disease in diabetes is exacerbated by the onset of diabetic kidney disease (Mishra, 2022). NICE states that 21% of mortality in people with type 1 diabetes is attributable to the diabetic kidney disease. In people with type 2 diabetes, nephropathy can result in a quadrupling of cardiovascular mortality (NICE, 2023a). Anaemia develops at a younger age in patients with diabetic kidney disease compared with CKD secondary to other causes. Other complications e.g. renal bone disease and metabolic acidosis is common in CKD of any aetiology. Patients with diabetes who have the complications described above should be referred to secondary care for further management.
Key points
Diabetic kidney disease is the leading cause of ESRD in the UK; the annual diabetic review should include screening for albuminuria, which is the earliest sign of diabetic kidney disease
There are four modifiable risk factors of diabetic kidney disease: smoking, hyperglycaemia, hypertension and hyperlipidaemia
Onset of diabetic kidney disease can lead to progression of diabetic retinopathy
Patients with diabetes and albuminuria (ACR ≥ 3) should be offered an ACE-I or ARB, titrated to the maximum tolerated dose
SGLT2-inhibitors should be considered in type 2 diabetes with an ACR >3 mg/mmol and offered with an ACR >30 mg/mmol, while on highest tolerated dose of ACE-I or ARBs
For women of child-bearing age consider contraception or pregnancy preparation including medication review
