Abstract
Autonomic nervous system (ANS) activity is a fundamental component of emotional responding. It is not clear, however, whether positive emotional states are associated with differential ANS reactivity. To address this issue, we conducted a meta-analytic review of 120 articles (686 effect sizes, total N = 6,546), measuring ANS activity during 11 elicited positive emotions, namely amusement, attachment love, awe, contentment, craving, excitement, gratitude, joy, nurturant love, pride, and sexual desire. We identified a widely dispersed collection of studies. Univariate results indicated that positive emotions produce no or weak and highly variable increases in ANS reactivity. However, the limitations of work to date – which we discuss – mean that our conclusions should be treated as empirically grounded hypotheses that future research should validate.
People feel many different positive emotions, including amusement while watching stand-up comedy (Gross & Levenson, 1995), awe while viewing beautiful landscapes (Gordon et al., 2017), pride while winning a competition (Tracy & Matsumoto, 2008), craving while looking at delicious food (Nederkoorn et al., 2000), and sexual desire while seeing attractive others (Bos et al., 2013). These positive emotions differ in many ways (e.g., Tong, 2015), but it is not yet clear whether and to what extent they produce different physiological responses (Kreibig, 2010). Initial work suggests physiological differences (e.g., Shiota et al., 2011), but more work is needed to test whether differences in physiological responses between different positive emotions are robust and replicable in larger samples and across different elicitation conditions. Moreover, with increasing numbers of studies on similar emotions and physiological responses, it becomes possible to test whether the magnitude of physiological responses depends on participant characteristics, such as age or sex, or methodological factors, such as the type of stimulus used (e.g., contact with real food vs. observing others eating).
The current state-of-the-art contains hundreds of individual studies, one qualitative review (Kreibig, 2010), and three meta-analyses (Cacioppo et al., 2000; Lench et al., 2011; Siegel et al., 2018) that have examined the relation between positive emotions and autonomic nervous system (ANS) activity. However, it is not known whether positive emotions differ in their ANS responses. This is because previous meta-analyses have employed a broad perspective on positive and negative emotions in general. Consequently, these works focused on happiness rather than contrasting a broader range of positive emotions against each other (Cacioppo et al., 2000; Lench et al., 2011; Siegel et al., 2018). Some previous meta-analyses concluded that positive emotions produce weak ANS reactivity, though this reactivity is not different from neutral conditions (Lench et al., 2011; Siegel et al., 2018). Other integrative work indicated that some positive emotions–contentment, relief, and love–cause decreases in ANS activity, whereas other positive emotions–joy and happiness–cause increases in ANS activity (Kreibig, 2010). This suggests that substantial variability may be expected and revealed by synthesizing primary research in a rigorous meta-analysis.
The main goal of the present investigation is to synthesize findings from past research that measured a physiological component of positive emotions. In this article, we draw upon prior theory and empirical work to identify 11 positive emotions best represented in the literature. We provide a short overview of ANS activity as the key feature of emotional responding. We also evaluate models of ANS reactivity specific to positive emotions and present possible moderators of ANS reactivity to positive emotions. Finally, we employ a meta-analytic design to test whether discrete positive emotions cause specific physiological reactivity. We summarize and discuss how our new findings impact emotion research. The novelty of this contribution lies in its quantitative test of ANS reactivity resulting from the elicitation of discrete positive emotions.
Positive Emotions
Our focus is on emotions characterized by positive valence or pleasure felt in response to a stimulus (e.g., object or event). Valence is the most fundamental and well-studied aspect of the emotional response (Mauss et al., 2005), and positive valence is elicited by favorable situations, e.g., smiling people or amusing events (Lang et al., 1997; Marchewka et al., 2014). As a class of emotions, positive emotions evolved around the pursuit of opportunities to obtain material, social, and/or informational resources that are key to promoting adaptive fitness (Fredrickson, 2013; Shiota et al., 2017). Positive emotions have received considerable attention with an emphasis on the common function of all positive emotions, e.g., broadening the thought-action repertoire and the urge to build psychosocial resources (Fredrickson, 2001, 2013), but less emphasis has been put on differences between positive emotions (Ekman & Friesen, 1986).
Over the years, theorists have moved beyond the assumption of a single, common positive emotion and suggested a number of discrete positive emotions such as joy and interest (Tomkins, 1962; Izard, 1977); amusement, contentment, excitement, pride in achievement, relief, satisfaction, sensory pleasure (Ekman, 1999); joy, interest, contentment, pride, and love (Fredrickson, 2001); seeking, lust, care, and play (Panksepp & Watt, 2011); amusement, ecstasy, excitement, fiero, happiness, interest, rejoicing, relief, naches, schadenfreude, sensory pleasures, and wonder (Ekman, & Cordaro, 2011); enjoyment (playing), interest (exploration), love (attachment), and relief/contentment (soothing) (Levenson, 2011); amusement, awe, gratitude, hope, inspiration, interest, joy, love, pride and serenity (contentment) (Fredrickson, 2013); anticipatory and consummatory love, amusement, compassion, flow, gratitude, interest, joy, pride, and surprise (Kreibig, 2014); amusement, attachment love, awe, contentment, enthusiasm, gratitude, liking/pleasure, nurturant love, pride, and sexual desire (Shiota et al., 2017).
These distinctions have inspired empirical work that has identified a number of distinctive positive emotions, such as awe, calmness, excitement, happiness, joy, love, and pride (Fehr & Russell, 1984); admiration, confidence, love, pride, secure, respect, and self-worthiness, (De Rivera & Grinkis, 1986); contentment, happiness, liking, love, and pride (Storm & Storm, 1987); love and joy (Shaver et al., 1987); amusement, awe, contentment, interest, joy, love, and pride (Campos et al., 2013); amusement, awe, challenge, compassion, contentment, gratitude, hope, interest, joy, pride, relief, romantic love, and serenity (Tong, 2015); admiration, adoration, aesthetic appreciation, amusement, awe, calmness, craving, entrancement, excitement, interest, joy, nostalgia, relief, romance, satisfaction, and sexual desire (Cowen & Keltner, 2017); admiration, amusement, attachment love, awe, contentment, empathy, enthusiasm, gratitude, hope, interest, nurturant love, romantic love, schadenfreude, sympathy, and tenderness (Weidman & Tracy, 2020).
We chose to focus on 11 discrete positive emotions: amusement, attachment love, awe, contentment, craving, excitement, gratitude, joy, nurturant love, pride, and sexual desire (for the full description, see Table 1). As emphasized in the previous paragraphs, this is not an exhaustive list of all positive emotions. We chose to study these 11 specific positive emotions because they are not only widely represented in the positive emotion models but also are targets of considerable and increasing psychophysiological research. Some positive emotions such as hope or schadenfreude have not been investigated in the psychophysiological literature yet. Moreover, the overview of existing empirical and theoretical models indicates not only a variety of discrete positive emotions but also a variety of labels used to describe them. For instance, while presenting humorous or funny situations, researchers aimed to elicit amusement (Kreibig et al., 2013), mirth (Foster et al., 2003), and happiness (Kring & Gordon, 1998). To clarify the 11 discrete positive emotions that were selected for inclusion in the present meta-analytic review, we provide a definition for each of the 11 emotions as well as information on both the emotion elicitation methods and the constructs and names that are similar to the emotion studied here (see Table 1). In Table 1, we also address how other discrete positive emotions may overlap with those included in the present review.
Discrete positive emotions.

We excluded from our list of emotions three potentially positive states, namely challenge, compassion, and interest. Challenge is a motivational state elicited in goal-oriented, important situations, where individuals evaluate their resources as meeting or exceeding the situational demand required for success (Blascovich, 2008). Compassion is the emotional response to another’s suffering, accompanied by the motivation to help (Stellar et al., 2017). Compassion is part of a group of states, such as empathic concern, pity, and sympathy (Goetz et al., 2010). Interest is a cognitive state of focused attention (Ekman & Cordaro, 2011; Lazarus, 1991). We excluded challenge because it is a motivational rather than an emotional state (Blascovich, 2008); compassion (and similar emotions) because it is not clearly a positive emotion, but rather a mixed emotion (Condon & Barrett, 2013; Myrick & Oliver, 2015); and interest because it is a cognitive rather than an emotional state (Ekman & Cordaro, 2011).
Positive Emotions and Autonomic Nervous System Reactivity
The autonomic nervous system (ANS) is a neural network that coordinates the work of organs distributed throughout the human body (Levenson, 2014). There are three basic regulatory functions of ANS, namely maintaining optimal, steady body conditions (i.e., homeostasis); activating body systems to provide necessary resources to respond to challenges and opportunities; and deactivating body systems when action is no longer performed (Levenson, 2003). Two ANS branches have been the main focus of psychophysiology, namely the sympathetic and parasympathetic nervous systems (SNS and PNS). The role of the SNS is to provide oxygenated blood from the heart to the other body organs such as limbs, brain, and lungs, and it is often related to the fight-or-flight response. The greatest SNS activation occurs with metabolically demanding situations like exercising. The PNS plays a complementary role to the SNS and is responsible for stimulating "rest-and-digest" or "feed-and-breed" activities, often related to resting states. However, the SNS and PNS do not operate in isolation, and bodily changes may also be influenced by the mutual combination of SNS and PNS stimulation and withdrawal, known as coactivation (Berntson, 2019; Berntson et al., 1991; Levenson, 2014). Thus, autonomic measures are often influenced by the combination of SNS and PNS. Furthermore, within SNS and PNS branches, there are further mechanisms of influence that often diverge (e.g., within SNS, there are different types of receptors such as alpha-adrenergic, beta-adrenergic, and cholinergic) (Levenson, 2014).
If we accept that emotions evolved to deal with fundamental life tasks (e.g., avoiding harm or restoring calm), then emotions might be thought to involve ANS reactivity that provides optimal support to respond to those tasks (Ekman & Cordaro, 2011; Tooby & Cosmides, 1990). This assumption leads to identifying physiological reactivity as a key element of emotional response along with subjective experience and a behavioral/expressive response (Gross, 2015; Mauss et al., 2005). In affective science, the most commonly used ANS measures are derived from cardiovascular, electrodermal, and respiratory reactivity (see Table 2 for the commonly used ANS measures) (Berntson et al., 1991; Cacioppo et al., 2000; Larsen et al., 2008).
ANS measures commonly used in psychophysiological studies.
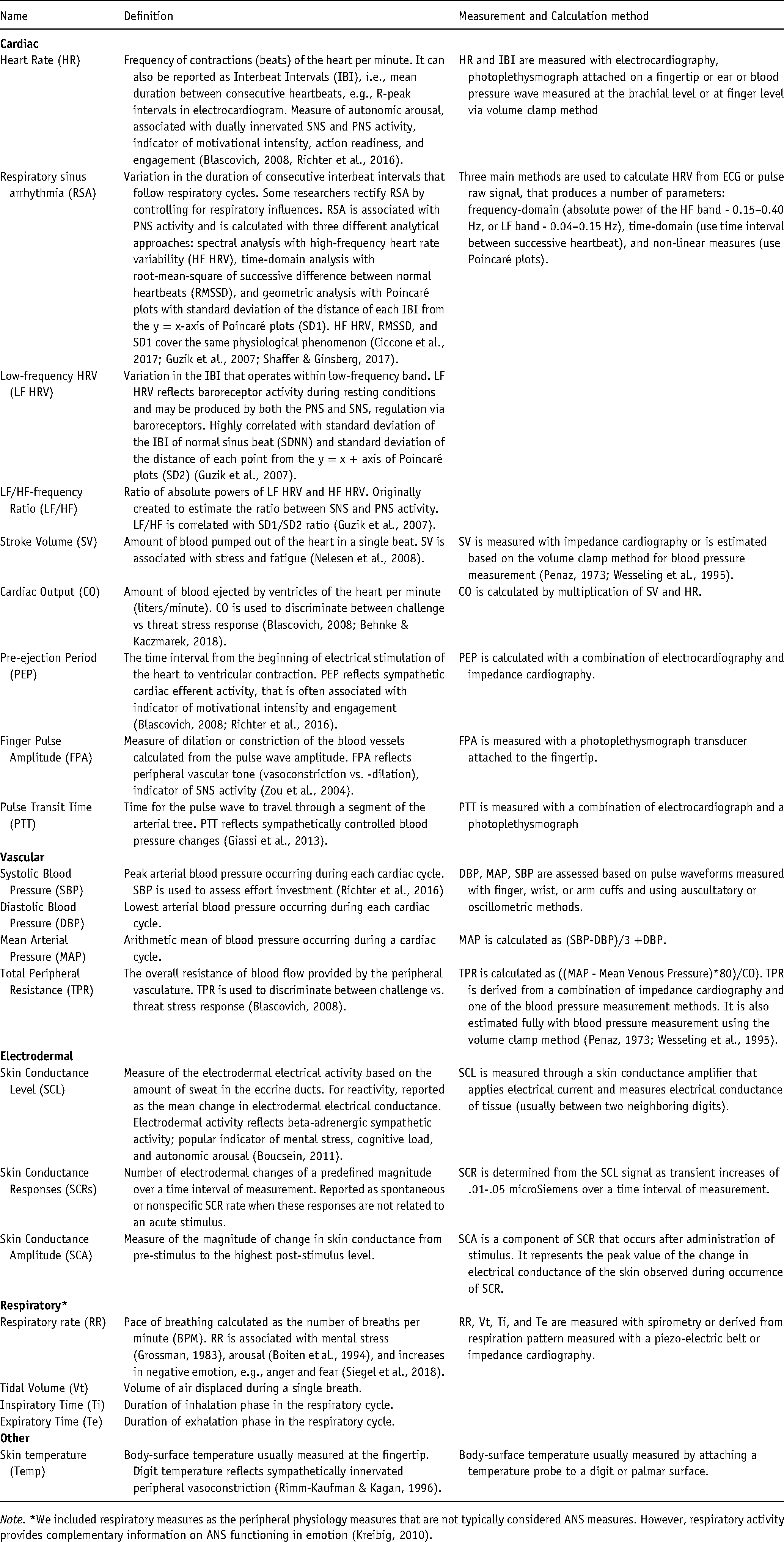
Note.
Within positive emotions, several theories have focused on the functions of psychophysiological emotional responding, namely undoing (or soothing) of sympathetic reactivity to stress (Fredrickson & Levenson, 1998; Fredrickson et al., 2000; Fredrickson, 2013; Levenson, 1988, 1999; Porges, 2011), and approach-avoidance models (Gable & Harmon-Jones, 2010; Harmon-Jones et al., 2013).
The undoing hypothesis was the first theoretical proposition that explicitly addressed the physiological effects of positive emotions (Fredrickson & Levenson, 1998). The undoing hypothesis stems from an initial observation that, in contrast to negative emotions, “positive emotions such as happiness, amusement, and contentment, did not seem to move autonomic levels away from their baseline states” (Levenson, 1999, p. 494). If this is true, certain positive emotions might play a different physiological role. For instance, they might be involved in the process of undoing (or down-regulating) physiological arousal caused by stress and negative emotions (Levenson, 1988). This might be primarily because positive emotions reflect (Tomkins, 1962) and facilitate (Levenson, 1988) tension reduction. This hypothesis received support in some studies (Fredrickson & Levenson, 1998; Fredrickson et al., 2000; Yuan et al., 2010). Furthermore, the polyvagal theory clarifies why positive emotions do not elicit sympathetic activation, suggesting that positive emotions cause an increase in vagal parasympathetic activation that inhibits stress-related influences on the heart (e.g., Cosley et al., 2010; Pressman & Cohen, 2005; Steptoe et al., 2005; Porges, 2011). However, the polyvagal theory focuses on the social process. Thus, it most closely relates to social positive emotions, like attachment (Porges, 2003) or nurturant love (Porges, 1998). It may not apply to more self-oriented positive emotions, like excitement or pride.
In summary, although these models do not strictly apply to ANS reactivity of all positive emotions, they present important contexts that shaped the way researchers think about the psychophysiology of positive emotion. Furthermore, these models were built upon the premise that all positive emotions do not produce SNS activity but share a similar physiological function, that is, producing PNS activity.
Models that focus on motivational tendencies propose differences between high-approach and low-approach positive emotions in terms of ANS reactivity (Gable & Harmon-Jones, 2010; Harmon-Jones et al., 2013). Although positive valence and approach tendency are usually highly correlated - individuals are likely to approach situations that elicit pleasant feelings and avoid situations that elicit unpleasant feelings (Cacioppo et al., 1999; Marchewka et al., 2014) - recent work has demonstrated that high-approach positive emotions differ from low-approach positive emotions in several ways, including their neural correlates (Harmon-Jones et al., 2008), their effects on cognition (Gable & Harmon-Jones, 2008; Li et al., 2018), and their effects on behavior (Fawver et al., 2014; Mouras et al., 2015). Moreover, studies showed at least some differentiation between high-approach and low-approach positive emotions in terms of their associated physiological responses, for instance, joy or happiness versus contentment or awe (for the review, see Kreibig, 2010). Some positive emotions, like excitement or enthusiasm, are associated with narrowing attentional focus and characterized by strong goal-attainment, approach tendencies, and increases in sympathetically mediated arousal across cardiac, vascular, and electrodermal systems (Kreibig et al., 2010; Shiota et al., 2017). In contrast, other positive emotions, like awe or contentment, are associated with broadening attentional focus and characterized by preparation for stillness and withdrawal of sympathetic influence on the heart (Kreibig, 2010). These initial findings indicate differences between discrete positive emotions and that they might have a different impact on ANS reactivity.
The differences between the theoretical models contribute to the longstanding debate over whether different emotions elicit unique ANS reactivity and, if so, in what form (for reviews, see Kreibig, 2010, 2014; Mendes, 2016, Siegel et al., 2018). Some theorists have argued for the specificity of ANS reactivity in discrete emotions (Ekman & Cordado, 2011; Stemmler, 2004), and some have argued for more generality (Barrett, 2006, 2013, 2017; Cacioppo et al., 2000; Quigley & Barrett, 2014; Siegel et al., 2018) (for a full discussion, see Mendes, 2016). In light of this issue, we examined in the present meta-analytic review whether research findings within positive emotions support the generalizability or differentiation hypothesis. Specifically, we aimed to examine whether all positive emotions do not show significant SNS reactivity and some PNS reactivity (in line with the group of undoing or soothing models) or whether at least some discrete positive emotions have the potential to elicit robust SNS reactivity (in line with motivation-oriented models).
To determine whether discrete emotions activate distinctive ANS responses or largely a uniform ANS response, previous studies focused on the specificity and consistency of ANS reactivity (Barrett, 2006; Quigley & Barrett, 2014; Siegel et al., 2018). Meta-analyses that tested the specificity or uniqueness of ANS responses between emotion categories showed that there are at least some categories of emotions that display different autonomic responses (Cacioppo et al., 2000; Lench et al., 2011; Siegel et al., 2018). Meta-analyses that tested the consistency of responses within the given emotion category showed moderate to high heterogeneity of effect sizes within emotion categories (low consistency), which suggests that the association between emotions and ANS activity is affected by different moderators (Cacioppo et al., 2000; Lench et al., 2011, Siegel et al., 2018). However, tests of expected moderators failed to show significance (Siegel et al., 2018), leaving neither side supported. Given the considerable increase in primary research on positive emotions in recent years, meta-analytic research is needed to clarify the extent of specificity and consistency of physiological responses in positive emotions.
Moderators of Autonomic Nervous System Reactivity in Positive Emotions
The list of potential moderators for ANS reactivity in positive emotions encompasses characteristics of the sampled individuals and the methods used, as well as the year of publication. Age and sex are individual characteristics, which are viewed as potential moderators of ANS reactivity to positive emotions. Women are generally viewed as being more emotionally reactive than men (Bradley et al., 2001), yet recent meta-analyses do not necessarily support the notion, showing no differences between men and women in response to positive emotions (Joseph et al., 2020; Siegel et al., 2018). Similarly, older individuals are expected to experience less intense emotions than younger individuals (Steenhaut et al., 2018; Charles & Carstensen, 2008). However, a meta-analysis did not support this view (Lench et al., 2011).
Several study characteristics, such as elicitation method, time interval for baseline and emotion elicitation, number of emotions induced in the experiment, number of studied ANS parameters, sample size, and study quality have been proposed as potential moderators of ANS reactivity to positive emotions (Foster et al., 2003; McGinley & Friedman, 2017; Lench et al., 2011; Levenson, 2014; Siegel et al., 2018). Results from a recent meta-analysis suggest that the most effective methods to elicit positive emotions are watching films, reading stories, and watching pictures of facial expressions (Joseph et al., 2020). However, other meta-analyses suggest no differences between the methods (Siegel et al., 2018; Lench et al., 2011). Moreover, a prior meta-analysis found that duration of emotion elicitation was the only significant moderator of ANS reactivity to happiness, showing that studies eliciting happiness for shorter than one minute had larger ANS reactivity, compared with studies with longer induction intervals (Siegel et al., 2018).
Finally, the reported strength of ANS reactivity to positive emotions might also be affected by the publication year. “Decline effect” (Schooler, 2011) or the “law of initial results” (Ioannidis, 2005) propose that strength of the effect sizes within a specific paradigm decline over time. This trend can be explained by the increasing number of rigorous studies with larger samples, leading to regression to the mean over time. The significant influence of the publication year can also be interpreted as an indicator of bias in the existing literature.
Overview of the Present Investigation
The main goal of the present study was to synthesize findings from past research, in which positive emotions were elicited experimentally, and their physiological component was measured. Experimental manipulation of emotions was important to ascertain causality from evocative stimulus to the physiological response. Next, to determine whether meta-analytic findings support the generalizability or differentiation hypothesis for physiological responses in discrete positive emotions, we focused our analysis on three aspects, namely consistency, specificity, and possible moderators of ANS reactivity to positive emotions.
First, we tested whether discrete positive emotions elicit consistent physiological responses. For this, we calculated pooled mean effect sizes for pairs of an ANS reactivity measure and a discrete positive emotion. Second, we examined whether the physiological responses were specific for discrete emotions by comparing ANS reactivity measures between discrete positive emotions. Finally, we investigated the role of moderators, i.e., participants’ characteristics (age, sex) and methodological factors, that may explain whether and why the physiological response does not occur in some contexts or under specific circumstances.
Based on theoretical models, we distinguished three groups of hypotheses: 1) we expected that discrete positive emotions would elicit consistent ANS reactivity: weak but significant reactivity of measures associated with PNS activity and lack of reactivity of measures associated with SNS reactivity; 2) we expected differences between some discrete positive emotions in ANS reactivity; and 3) we expected that study moderators would influence ANS reactivity to positive emotions. We believe that examining these effects will be important for moving forward with more empirically based theories of positive emotions. The present investigation is the first meta-analytic review of ANS reactivity in discrete positive emotions. The novelty of this review stems from contrasting a broad range of positive emotions against each other, rather than focusing only on happiness (Cacioppo et al., 2000; Lench et al., 2011; Siegel et al., 2018).
Methods
Selection of Studies
We performed a systematic literature search using PsycINFO, PubMed, and Google Scholar, covering the period from 1872 (earliest available articles) to October 2019. We used the following terms: [“positive emotion” OR amusement OR “anticipatory pleasure” OR “attachment love” OR awe OR contentment OR craving OR enthusiasm OR excitement OR gratitude OR happiness OR joy OR liking OR “nurturant love” OR pride OR “sexual desire”] AND [“autonomic nervous system” OR cardiovascular OR cardiac OR heart OR respiration OR respiratory OR electrodermal OR “skin conductance” OR psychophysiology OR physiology]. We also cross-checked the references in the studies that we retrieved. We contacted 139 authors who had published papers on ANS activity of positive emotions and asked for any unpublished material. Of 98 authors who responded to the request (71%), none reported any unpublished material.
We selected potentially eligible studies in two phases. First, the first author (MB) scrutinized the titles and abstracts. If the material was relevant to the subject of this meta-analysis (MB) screened the full-text articles. All of the studies identified as potentially eligible during the first selection phase were reassessed in the second selection phase. The inclusion criteria for eligible studies were: 1) at least one of the 11 discrete positive emotions was manipulated; 2) in healthy individuals; and 3) ANS reactivity was measured both during a baseline or a neutral condition and during emotional responding. The exclusion criteria were: 1) studies involved emotion regulation rather than spontaneous responses to stimuli; 2) no discrete emotion was targeted and reported, e.g., only ratings regarding valence and/or arousal were reported; or 3) the study design involved additional manipulations that affected physiological or emotional responses (e.g., exercising before emotion manipulation or priming during a cognitive task). Finally, we included studies that allowed us to calculate effect sizes based on available data. Figure 1 summarizes the search procedure. Table S1 (supplementary materials) presents the studies included in this meta-analysis with several study characteristics.
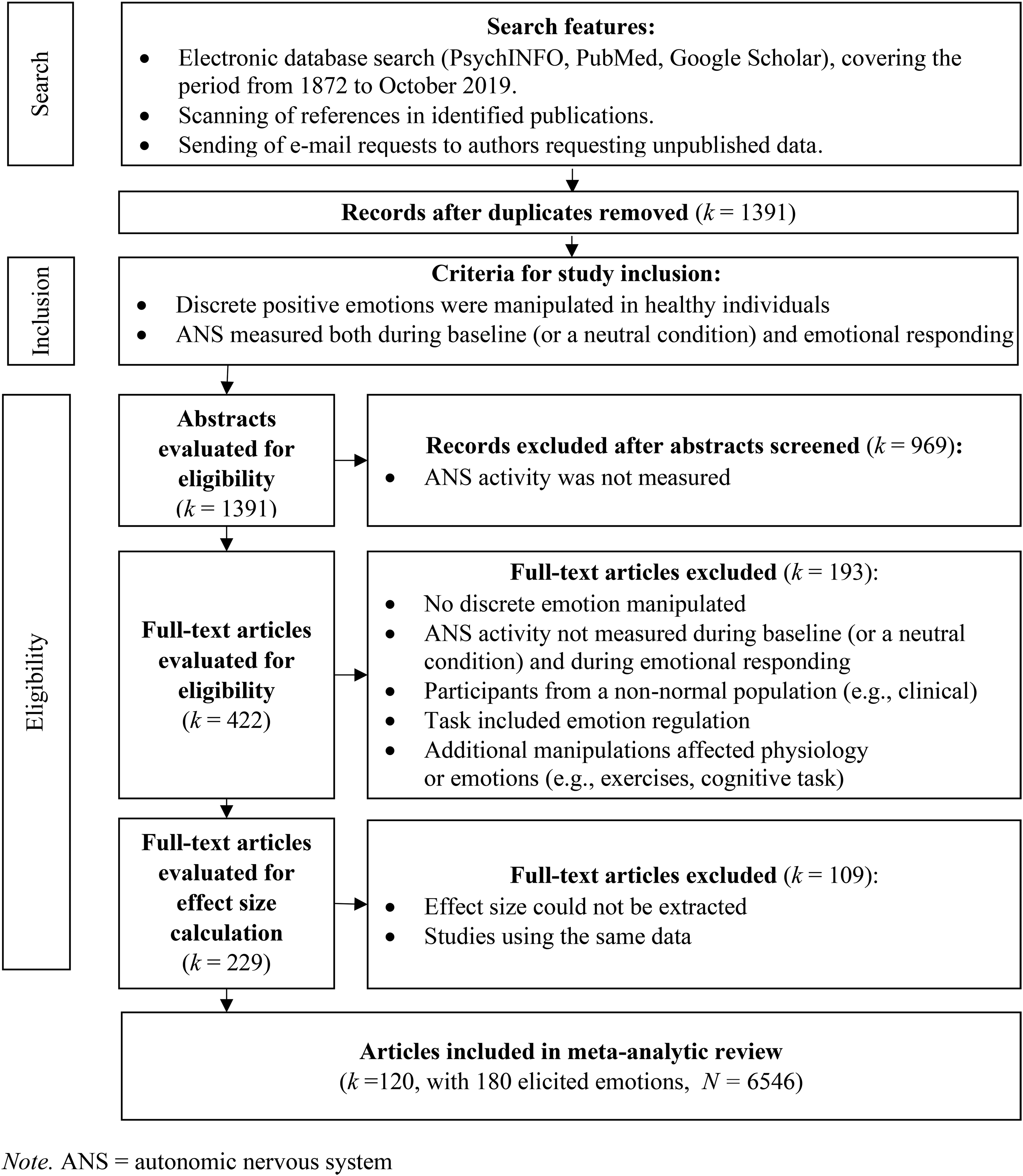
Flow Diagram of the Search Procedure.
Data Extraction
Coding
Based on methodological considerations (Levenson, 2014) and results from previous meta-analyses on ANS reactivity and emotions (Lench et al., 2011; Siegel et al., 2018), the following sample characteristics were coded: 1) mean age of the sampled participants (in years); and 2) sex (percentage of females). As for study characteristics, we coded: 1) type of ANS measure (for a complete list and descriptions of ANS reactivity indices, see Table 2); 2) type of experimental manipulation method, i.e., autobiographical recall, behavioral manipulation, film, imagination, music, picture presentation, priming, reading text, Velten-like statements, and VR; 3) baseline duration (length of the time interval that was used to calculate the physiological levels for baselines, reported in seconds); 4) emotion elicitation duration (length of the time interval that was used to calculate the physiological levels for emotion elicitation, reported in seconds); 5) type of physiological comparison (baseline or elicitation of neutral state); 6) number of emotions induced in the experiment; 7) the scope of ANS measures (number of studied parameters); 8) the positive emotion that was studied (categorized as amusement, attachment love, awe, contentment, excitement, gratitude, joy, craving, nurturant love, pride, and sexual desire); 9) sociality (whether participants were alone during the experimental task); 10) task type (whether participants were asked to passively watch or listen to the stimuli, or were asked to actively create the actions that elicited emotions); 11) task relevance (whether the stimuli were self-relevant to participant or standardized, universal stimuli for all participants); 12) video recording (whether participants were being video recorded during the experimental task); 13) sample size; 14) study quality (0-3); and 15) publication year. The study quality index comprises scores from three criteria: a) provision of exclusion criteria related to physiological activity, e.g., BMI, physical health, drug use (yes = 1, no = 0); b) report on artifacts, outliers, and missing data (yes = 1, no = 0); and c) the presence of a manipulation check (score from 0 to 1). We evaluated the presence of a manipulation check by averaging three manipulation check parameters, including the presence of: a) general manipulation check (e.g., an increase of positive valence; yes = 1, no = 0); b) within original study-specific manipulation check (e.g., increase of happiness after watching amusing video; yes = 1, no = 0), c) manipulation check for one of the 11 discrete positive emotions included in the current Meta-Analytic Review (e.g., increase of amusement after watching amusing video; yes = 1, no = 0).
Positive emotion label designation
When coding for the emotion label, we compared the emotion label designated by the primary study authors with the list of discrete positive emotions described in Table 1. In 86 cases (48%), we relabeled the original names according to information in the Methods section of the primary study. The most common reasons for relabeling were: 1) the stimulus was assigned a broad category (e.g., pictures eliciting positive affect) although a specific positive emotion was elicited (e.g., a cheerful, smiling face was presented to elicit joy; Anttonen et al., 2009); 2) the stimulus was assigned to evoke an emotion labeled as happiness although a more specific discrete emotion was elicited (e.g., a funny film elicited amusement; Kring & Gordon, 1998); 3) the emotion label was not provided because the authors conceptualized the situation in terms of the stimulus (e.g., erotic pictures, Bradley et al., 2017; delicious food, De Wijk et al., 2012; sports pictures; Gomez & Danuser, 2010).
Data clustering
We binned the emotion elicitation methods into seven categories: autobiographical recall, behavioral manipulation (real-life or laboratory scenarios, e.g., real food exposure), films, imagery (Velten and imagination), music, pictures, and reading text. We clustered timing moderators into three categories: short < 30 s, moderate 31 s - 180 s, and long > 180 s. We binned HRV measures (similar indices calculated by different computational methods, e.g., HF HRV, RMSSD, and SD1) because they covered the same physiological phenomenon, namely respiratory sinus arrhythmia (Ciccone et al., 2017; Shaffer & Ginsberg, 2017). For the same reason, we binned LF HRV, SDNN, and SD2 (Guzik et al., 2007). Finally, we multiplied the effect sizes for the IBI by – 1, so that higher numbers reflect higher cardiac activity, as the effect size for HR.
Effect size extraction
To calculate effect sizes, we computed the difference in autonomic levels between the baseline (or neutral condition) and emotion elicitation periods. For most studies, the authors reported means and standard deviations of the levels of baseline and emotion elicitation. For studies reporting other metrics (e.g., correlation or regression coefficients), we sent requests to the authors to provide us with the means of the relevant periods. Of the 139 authors we contacted, 98 responded to our inquiry (71%), and of those, 38 were able to send us the requested data (27%). Sixty authors responded negatively to our request (39%) and indicated, for instance, that they did no longer have access to the data used or that they did not have the time to perform the required analyses. In some cases, we were able to extract data from clearly labeled graphs using grid lines in GNU Image Manipulation Program (GIMP) (data from 16 articles representing 120 effect sizes or 17% of the final pool of effect sizes). We used Cohen’s d as the common effect size measure. We interpreted the magnitude of the effect sizes using conventional standards (small, d = 0.20; medium, d = 0.50; large, d = 0.80; Cohen, 1992). Table S2 and Table S3 presents the final numbers of extracted effect sizes for each discrete emotion category and physiological measure.
Meta-Analytic Procedures
To run meta-analytic procedures, we used R (R Development Core Team, 2017) with packages metafor (Viechtbauer, 2010) and dmetar (Harrer et al., 2019) following meta-analysis recommendations (Assink & Wibbelink, 2016; Harrer et al., 2019; Viechtbauer, 2010). We used the random-effects model because the diversity of the outcomes examined in each of the included studies was high, and considerable heterogeneity was likely (Siegel et al., 2018). Several theorists have advocated for adopting random-effects models for meta-analysis as these models are optimal in permitting the generalization of corrected effect sizes to the population (Field & Gillett, 2010; Hunter & Schmidt, 2000; Schmidt & Hunter, 2014). We used restricted maximum likelihood estimation to estimate the pooled mean effect sizes.
Most of the included studies provided multiple effect sizes of ANS reactivity for one or more positive emotions. To account for dependency between effect sizes, we used a three-level meta-analytic technique (Assink & Wibbelink, 2016; Viechtbauer, 2010), in which three sources of variance are considered: variance in effect sizes extracted from different studies (i.e., between-study variance) at level three of the model, variance in effect sizes extracted from the same study (i.e., within-study variance) at level two of the model, and sampling variance of the extracted effect sizes at level one of the model (Cheung, 2014; Hox, 2002; Van den Noortgate et al., 2013, 2014).
Coding bias
Before examining the degree to which physiological responding differentiated among positive emotions, we checked whether two aspects of our coding procedure would bias the results. To test this, we ran moderator analyses, in which we compared mean ANS reactivity between 1) included studies where we kept the original emotion label and studies where we renamed the emotion label; and 2) included studies calculating the ANS reactivity from comparison to baseline versus studies calculating the ANS reactivity from comparison to neutral conditions. As for the latter, we did this because the previous meta-analysis on physiological responses to affective stimuli revealed that effect sizes differ between comparisons to baseline with no stimuli versus comparisons to neutral stimuli (Siegel et al., 2018). Therefore, we built a three-level meta-analytic model to estimate the mean effect of the general ANS reactivity by pooling the effect sizes across all ANS reactivity measures. Next, we tested emotion label (original label vs. changed label) and type of comparison (baseline vs. neutral comparisons) as moderators of the general ANS reactivity. Significant results of these moderator analyses would indicate that there was a difference between the examined groups.
Magnitude and consistency of ANS reactivity to positive emotions
We aimed to examine a pooled mean effect size for each pair of an ANS measure (e.g., HR, SBP, SCL) and a discrete positive emotion (e.g., amusement, awe, joy), resulting in 231 potential meta-analyses (21 ANS measures x 11 positive emotions). However, we only ran a meta-analysis for a pair (e.g., HR reactivity to amusement) if at least two effect sizes were available for that comparison. Following the example of a pair of HR reactivity and amusement, the three-level meta-analytic model accounted for variance in HR reactivity extracted from different studies that elicited amusement (i.e., between-study variance) at level three of the model, variance in HR reactivity extracted from the same study that elicited amusement more than one time, e.g., using two elicitation methods (i.e., within-study variance) at level two of the model, and sampling variance of the extracted HR reactivity at level one of the model.
We also calculated the mean ANS reactivity for groups of measures, including cardiac, vascular, respiratory, and electrodermal measures. Thus, we grouped the reactivity of individual measures (e.g., SCL, SCA, SCR) into broader categories (e.g., electrodermal measures). This resulted in 44 additional possible meta-analyses (four groups x 11 positive emotions). For cardiac measures, we followed the approach presented in the cardiac autonomic regulatory capacity models (Berntson, 2019). In this way, higher cardiac reactivity would reflect higher levels of overall ANS control (total PNS and SNS activity that reflects its coactivity; Berntson, 2019). Thus, because increased sympathetic activation is associated with shortened PEP and PTT values, those measures were multiplied by −1.
We interpret the results considering two parameters, namely, ANS reactivity magnitude (significant vs. non-significant) and its consistency (no/low heterogeneity vs. high heterogeneity), which can be described in a 2 × 2 matrix of possible outcomes. Heterogeneous effect sizes may indicate that: a) the average ANS measure reactivity is not consistent for a given emotion in the population; b) the average ANS measure reactivity is moderated by different types of characteristics (e.g., elicitation method); or c) the size of the effect reflects real, contextual changes in ANS reactivity that are consistent with the generalizability hypothesis.
We checked whether the calculated mean effect sizes were consistent (homogeneous) with two parameters. First, we calculated the Q, which rejects the null hypothesis of homogeneity and indicates that the true effect size varies study-to-study or within the study, suggesting possible methodological or population differences that may introduce variation between or within studies. Second, we calculated the I2, which is less biased by the number of studies included in a meta-analysis compared to the Q and can be compared across meta-analyses with different sample sizes and different types of studies (Higgins et al., 2003). Moreover, the I2 allows for examining the amount of variance at levels two (variance in effect sizes extracted from the same study) and three (variance between studies) of the meta-analytic models. The sum of the I2 at levels two and three indicates the amount of variability with the value of 0% indicating an absence of dispersion, and larger values indicating the following levels of heterogeneity: 0-40% = might not be important; 30-60% = may represent moderate heterogeneity; 50-90% = may represent substantial heterogeneity; 75-100% = represents considerable heterogeneity (Deeks et al., 2021). For three-level models, we examined the significance of the variance at levels two (variance in effect sizes extracted from the same study) and three (variance between studies) by calculating two separate one-tailed log-likelihood-ratio tests. In these tests, the deviance of the full model was compared to the deviance of the model, excluding one of the variance parameters.
To determine whether our results support the hypothesis that discrete positive emotions would elicit weak but significant PNS reactivity and no SNS reactivity, we compared the number of significant and nonsignificant mean effect sizes with χ2 goodness of fit tests. A significant test result rejects the null hypothesis of homogenous distribution of effect sizes and would indicate that one group of results was significantly larger.
Finally, we performed a publication bias analysis to investigate whether null or weak results were likely to be systematically suppressed from publication (Schmidt & Hunter, 2014). This type of analysis is vital because studies with non-significant or negative results are less likely to be published in peer-reviewed journals (Borenstein et al., 2011). We tested for publication bias using an adapted version of the Egger's regression test and the Begg and Mazumdar’s rank-order correlation test (Assink et al., 2018; Assink et al., 2019; Begg & Mazumdar, 1994; Egger et al., 1997; Sterne et al., 2000). In the adapted Egger's regression test, we regressed the effect sizes on standard errors in a three-level meta-analytic model. Contrary to the classical Egger’s test, this adapted test accounted for effect size dependency. Significant positive values of both tests indicate that smaller studies with less precision are associated with larger effects, suggesting that publication bias is present (Begg & Mazumdar, 1994; Egger et al., 1997; Sterne et al., 2000).
Specificity of univariate ANS reactivity to positive emotions
After examining the magnitude of ANS reactivity to positive emotions, we tested whether univariate ANS reactivity is specific for single emotions or whether it is a common response to all positive emotions. We ran 21 single three-level meta-analyses for each ANS reactivity measure individually, with emotion category as the moderator of the mean effect. In comparison to the previous model, we nested specific univariate ANS measure reactivity to all emotions at level 3 because this allowed us to examine differences between positive emotions (as moderation factor) without violating the statistical assumption of effect size independence. The univariate ANS reactivity to a specific positive emotion was considered as significantly different from other emotions if the confidence intervals of the difference between effect sizes in subgroups did not include zero. To account for Type I error for multiple comparisons (e.g., difference in HR reactivity between amusement and awe, amusement and attachment love, amusement and contentment), that is frequent in meta-analyses (Cafri et al., 2010), we adjusted probability values using the false discovery rate (FDR) formula (Benjamini & Hochberg, 1995). This resulted in adjusting confidence intervals to balance Type I and Type II error.
Moderator analyses
Finally, to examine factors that may influence general ANS reactivity to positive emotions, we ran 165 three-level meta-analyses (i.e., 11 positive emotions x 15 moderators). In comparison to the first model, which was used to examine the magnitude of ANS reactivity to positive emotions, we nested all ANS measures reactivity at level two because we focused our analysis on the factors/context (e.g., elicitation method) that may explain variability in ANS reactivity (all measures) to positive emotions rather than the variability of specific ANS reactivity measure (e.g., HR). We determined whether ANS reactivity to discrete emotions differentiates within the factor by interpreting results of an omnibus test, in which significant F values indicate that there are differences in ANS reactivity within the factor (moderator). We considered the ANS reactivity as significantly different in one subgroup from other subgroups (e.g., ANS reactivity to amusement elicited with films would be different from ANS reactivity to amusement elicited with imagery or autobiographical recall) if confidence intervals of the difference between effect sizes in a given moderator subgroup did not include zero. In this procedure, the confidence intervals were adjusted using the false discovery rate for multiple comparisons.
Results
Descriptive Analyses
We included 120 articles with 180 elicited emotions presenting 686 effect sizes obtained from 6,546 participants with their age ranging from 8.1 to 71.0 years (M = 23.54 years, SD = 9.21). Based on the average age, we divided the studies into groups examining minors (ages 8-17, k = 5, n = 146), young adults (ages 18–35 years; k = 92, n = 5037), middle-aged adults (ages 36–55 years, k = 4, n = 352), and older adults (aged older than 55 years, k = 2, n = 89). Of all participants, 62% were female. The mean duration of physiological recording was 110.63 (SD = 125.69), and the median time was 60 s. The included studies were published between 1969 to 2020, with the median publication year of 2011. There was an exponentially growing trend in the number of publications per year, starting from three articles in the first 21 years 1969 −1990, followed by an increasing number of publications in successive five-year intervals, namely four articles in years 1991-1995, 12 articles in years 1996-2000, 16 articles in years 2001-2005, 22 articles in years 2006-2010, 28 articles in years 2011-2015, 35 articles in years 2016-2020.
The most frequently studied emotion was amusement (193 effect sizes; 28.1%), then excitement (106; 15.5%), contentment (90; 13.1%), joy (70; 10.2%), pride (47; 6.9%), craving (43; 6.3%), sexual desire (41; 6.0%), attachment love (34; 5.0%), awe (31; 4.5%), nurturant love (20; 2.9%), and gratitude (11; 1.6%). The most frequent method of emotion elicitation was presenting film clips to participants (318 effect sizes; 46.4%), then picture presentation (122; 17.8%), behavioral manipulation (84, 12.2%), autobiographical recall (57; 8.3%), imagination (39; 5.7%), music (39, 5.7%), and reading a text (25; 3.6%). The most frequent physiological measure was HR (149 effect sizes; 21.7%), then SCL (80; 11.7%), RSA (62; 9.0%), RR (48; 7.0%), DBP (46; 6.7%), SBP (45; 6.6%), PEP (32; 4.7%), SCR (23; 3.4%), CO (22; 3.2%), MAP (21; 3.1%), Vt (21; 3.1%), SV (20; 2.9%), TPR (20, 2.9%), SCA (17; 2.5%), skin temperature (15; 2.2%), LF HRV (15; 2.2%), FPA (12; 1.7%), Ti (11; 1.6%), PTT (10; 1.5%), Te (10; 1.5%), LF/HF HRV (5; 0.7%), and FPV (2; 0.3%).
Most researchers checked the intensity/arousal after emotion manipulation (163, 90.6%), provided exclusion criteria (103, 57.2%), and reported artifacts and missing data treatments (112, 62.2%). Participants were usually not video recorded during the experiment (148, 82.2%), in comparison to being recorded (27, 15.0%) and being recorded with a hidden camera (5, 2.8%). Participants were usually alone during the emotion manipulation (60, 33.3%), in comparison to being with the experimenter (17, 9.4%) or being with loved ones (2, 1.1%). However, in most studies (100, 55.6%), we were unable to determine whether participants were alone. We classified most tasks used for emotion manipulation as passive (126, 70.0%) and not self-relevant or universal (135, 75.0%).
Coding Bias
As the moderator analysis revealed, we found that the strength of general ANS reactivity to positive emotions was not affected by comparison type, F (1, 681) = 1.49, p = .22, (baseline comparison: d = 0.12, 95% CI [0.04, 0.20], p = .005; neutral comparison: d = 0.19, 95% CI [0.10, 0.28], p < .001); nor by our labeling procedure, F (1, 681) = 0.57, p = .45, (studies with original label: d = 0.17, 95% CI [0.09, 0.24], p < .001; studies with changed label: d = 0.13, 95% CI [0.06, 0.21], p < .001).
ANS Reactivity within Positive Emotions
Individual ANS measures
In total, we estimated 122 mean effects for pairs of a discrete emotion and an individual ANS measure, implying that for each pair, we found at least two effect sizes that accounted for a single ANS reactivity to a discrete positive emotion (e.g., HR reactivity to amusement). We considered 68 of these pairs to be sufficiently powered (k > 3) to interpret the results (see Table 3; For all results, see Table S4 in the supplementary materials). Of 40 consistent (homogenous) effects, four effects (10%) indicated a significant, consistent ANS increase to a discrete positive emotion (see Table 3). Amusement and joy resulted in small-to-large increases of at least one index of ANS reactivity, i.e., DBP and SCR in amusement, SBP and SCA in joy (Table 3). Further, 36 (90%) indicated nonsignificant ANS reactivity (Table 3). Specifically, attachment love, awe, contentment, craving, excitement, nurturant love, pride, and sexual desire did not produce any significant homogenous ANS reactivity.
Summary of meta-analytic review.
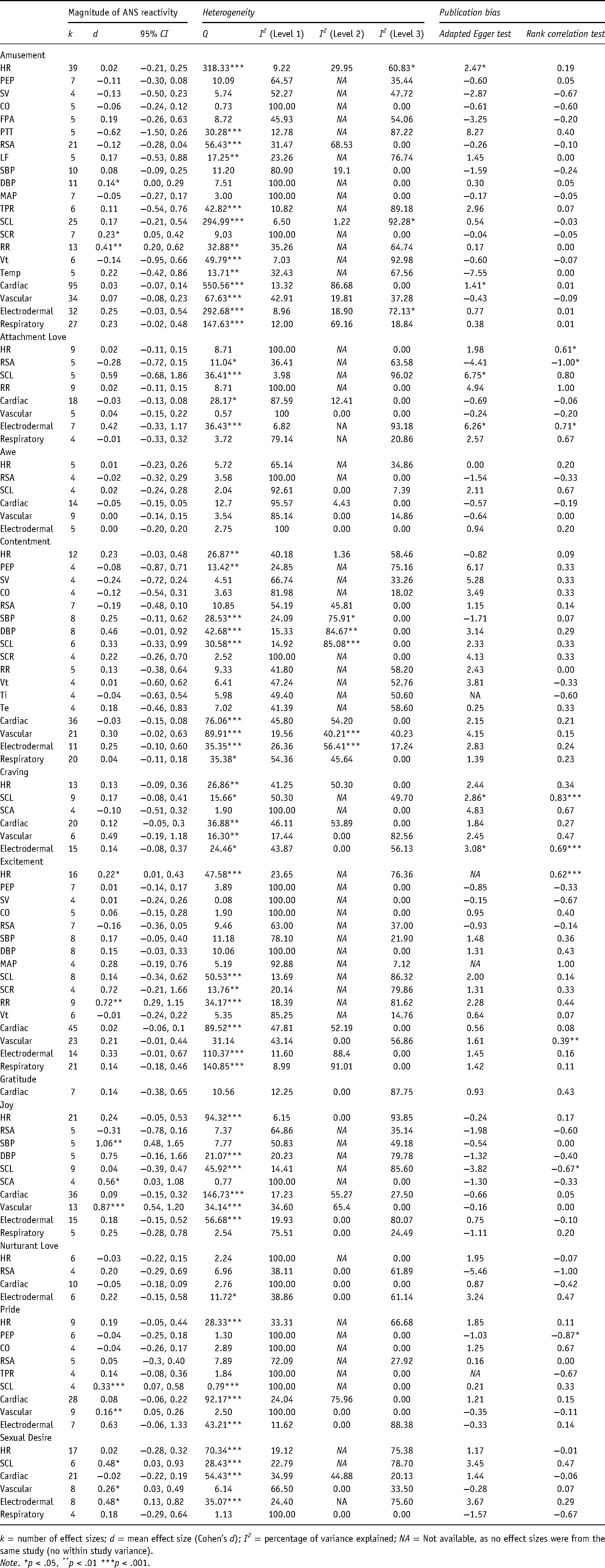
k = number of effect sizes; d = mean effect size (Cohen’s d); I2 = percentage of variance explained; NA = Not available, as no effect sizes were from the same study (no within study variance).
Note. *p < .05, **p < .01 ***p < .001.
The remaining 28 sufficiently powered effect sizes (41%) were inconsistent (heterogenous), indicating that each of these mean effects does not represent one individual population effect. Of these heterogeneous effects, five effect sizes significantly deviated from zero. That is, we observed increases in: RR to amusement, HR and RR to excitement, SCL to pride, and SCL to sexual desire (Table 3). A better estimation of the true magnitude of these effects would require accounting for additional moderators that might further differentiate these effects.
In summary, these results indicate that there were fewer consistent and significant effects (increases of ANS reactivity measure) (6%) relative to effects that were inconsistent or nonsignificant (94%), χ2 (1, N = 68) = 52.94, p < .001. Moreover, we found that the difference between the number of inconsistent effects and consistent effects was nonsignificant, χ2 (1, N = 68) = 2.11, p = .15. A forest plot (Figure 2) presents the mean physiological reactivity for each discrete positive emotion.
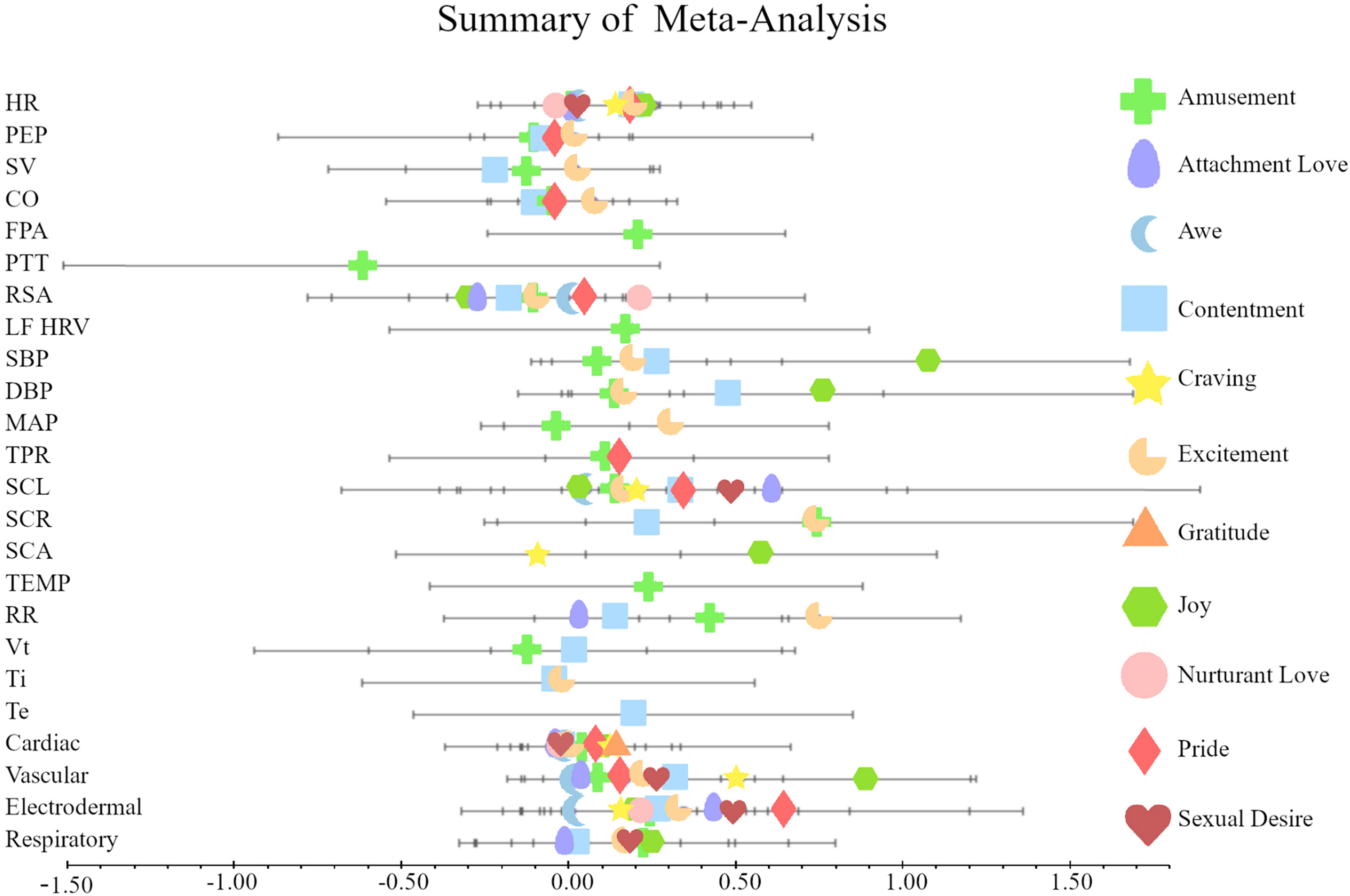
Mean effects (Cohen’s d) for each of the 11 discrete positive emotions with symbols representing the mean effect sizes and horizontal lines representing 95% confidence intervals.
We found evidence that the distribution of effects was asymmetrical in eight pairs of ANS reactivity and emotion. Based on the adapted Egger’s regression test we found an asymmetrical distribution of effects in pairs of HR and amusement, β = 2.47, p = .02, SCL and attachment love, β = 6.75, p = .04, and SCL and craving, β = 2.86, p = .01. Further, based on the rank order correlation test we found an asymmetrical distribution of effects in pairs of HR and attachment love, τ = .61, p = .03, RSA and attachment love, τ = −1.00, p = .02, HR and excitement, τ = .62, p < .001, SCL and joy, τ = -.67, p = .01, PEP and pride, τ = -.87, p = .02, and SCL and craving, τ = .83, p < .001. This indicates that despite possible bias in some studies (12%), the majority of studies (88%) were not likely to be biased, χ2 (1, N = 68) = 39.77, p < .001.
Types of ANS measures
Furthermore, we estimated 39 mean effects for pairs of a discrete emotion and composites of ANS measures. We considered 36 of these pairs to be sufficiently powered (k > 3) to interpret the results (see Table 3; For all results, see Table S4 in the supplementary materials).
Of 12 consistent (homogenous) effects, two effects (17%) indicated a significant, consistent ANS increase to a discrete positive emotion (see Table 3). Pride and sexual desire resulted in small-to-medium increases in vascular activity (Table 3). Further, 10 (83%) indicated nonsignificant ANS reactivity (Table 3). The remaining 24 sufficiently powered effect sizes (67%) were inconsistent (heterogenous). Of these heterogeneous effects, two effect sizes significantly deviated from zero. That is, we observed increases in: vascular measures to joy and electrodermal to sexual desire (Table 3). A better estimation of the true magnitude of these effects would require accounting for additional moderators that might further differentiate these effects.
In summary, these results indicate that there were fewer consistent and significant effects (increases of ANS reactivity measure; 6%) relative to effects that were inconsistent or nonsignificant (94%), χ2 (1, N = 36) = 28.44, p < .001. Moreover, we found that the difference between the number of inconsistent effects and consistent effects was significant χ2 (1, N = 36) = 4.00, p = .046. A forest plot (Figure 2) presents the mean physiological reactivity for each discrete positive emotion.
We found evidence that the distribution of effects was asymmetrical in three pairs of ANS reactivity and emotion. Based on the adapted Egger’s regression test we found an asymmetrical distribution of effects in pairs of electrodermal measures and attachment love, β = 6.26, p = .02, electrodermal measures and craving, β = 3.08, p < .01, and cardiac measures and amusement, β = 1.41, p = .03. Further, based on the rank order correlation test we found an asymmetrical distribution of effects in pairs of electrodermal measures and attachment love, τ = .71, p = .03, electrodermal measures and craving, τ = .69, p < .001, vascular measures and excitement, τ = .39, p < .01. This indicates that despite possible bias in some studies (11%), the majority of studies (89%) were not likely to be biased, χ2 (1, N = 36) = 21.78, p < .001.
Specificity of Univariate ANS Reactivity
Within each ANS measure (e.g., HR, SBP, SCL), we compared mean effect sizes between positive emotions (e.g., amusement vs. awe, amusement vs. joy, awe vs. joy) to determine whether ANS reactivity was specific to a discrete emotion. In total, we performed 152 comparisons between pairs of ANS reactivity to a positive emotion. The total number of comparisons was based on the number of emotions for which we were able to calculate the pooled mean effect sizes of ANS reactivity (e.g., for HR, we calculated mean effect sizes for ten discrete emotions, resulting in 45 comparisons between emotions, whereas for MAP we calculated mean effect sizes for two discrete emotions resulting in one comparison between emotions). We found only six significant differences in ANS reactivity between the emotions.
Specifically, we found a higher SBP reactivity in joy compared to amusement, Δd = 0.93, 98% CI [0.43, 1.43], contentment, Δd = 0.75, 95% CI [0.33, 1.16], excitement, Δd = 0.75, 97% CI [0.29, 1.21], DBP reactivity in joy compared to amusement, Δd = 0.59, 98% CI [0.02, 1.15], and a higher RR reactivity in excitement compared to attachment love, Δd = 0.48, 98% CI [0.04, 0.92], and to contentment, Δd = 0.46, 97% CI [0.05, 0.87], as indicated by the CIs, which in the case of SBP, DBP, and RR were adjusted for probability values with a FDR for three comparisons. In summary, these results indicate that there was far less (4%) specific ANS reactivity than nonspecific ANS reactivity (96%), χ2 (1, N = 152) = 128.947, p < .001. Thus, the present results do not suggest the robust specificity of univariate ANS reactivity to discrete positive emotions. However, four significant differences emerged when controlling for the false discovery rate, indicating that some exceptions exist.
Moderator Analyses
We found that the strength of ANS reactivity to positive emotions was not affected by most variables that were tested as potential moderators. After adjusting for FDR (e.g., for amusement for 15 comparisons), we observed that only in response to attachment love and craving was the strength of ANS reactivity moderated by study characteristics (see Table 4 for the results of the omnibus test of the moderator analyses; for a full description of all results of the moderator analyses see Table S5 in the supplementary materials). We found that studies using behavioral methods to elicit craving (i.e., exposure of real food) produced stronger effects than studies showing films, Δd = 0.56, 98% CI [0.16, 0.96], pictures, Δd = 0.41, 97% CI [0.10, 0.72], and studies using imagery of food, Δd = 0.77, 95% CI [0.06, 1.47]. Moreover, we found a positive association between the percentage of female participants and the size of the ANS reactivity, β = .27, 95% CI [.08, 0.46], p = .007, indicating that more female-dominated studies reported stronger effects than more male-dominated studies. We also found that the experimental method affected ANS reactivity to attachment love. Thus, elicitation of attachment love by reading text produced stronger responses than showing pictures, Δd = 0.63, 98.5% CI [0.28, 0.97], and showing films, Δd = 0.74, 98% CI [0.35, 1.14]. However, this difference was based on comparing single studies since only one study elicited attachment love with text reading.
Results of the moderator analyses for ANS reactivity to positive emotions.
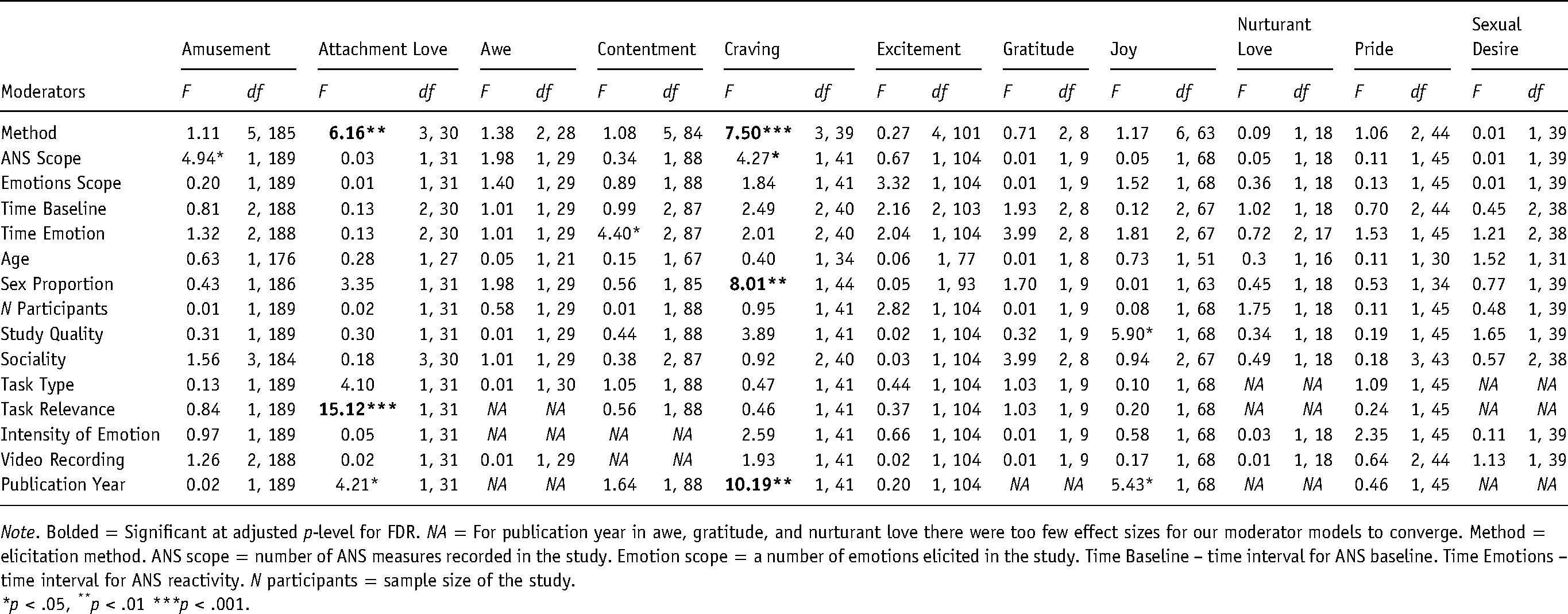
Note. Bolded = Significant at adjusted p-level for FDR. NA = For publication year in awe, gratitude, and nurturant love there were too few effect sizes for our moderator models to converge. Method = elicitation method. ANS scope = number of ANS measures recorded in the study. Emotion scope = a number of emotions elicited in the study. Time Baseline – time interval for ANS baseline. Time Emotions – time interval for ANS reactivity. N participants = sample size of the study.
*p < .05, **p < .01 ***p < .001.
Discussion
In this quantitative review, we aimed to inspect, evaluate, and synthesize (to the extent possible) findings from past research that measured a physiological component of positive emotions. We found the available data to be quantitatively imbalanced, with many studies focused on some positive emotions and physiological signals and few studies focused on other positive emotions and physiological signals. Furthermore, we found high variability in methods used for emotion elicitation and data collection. Recognizing that the empirical evidence might be insufficient to test some effects, we aimed to use all available empirical data and stringent criteria for multiple hypothesis testing to examine whether the currently available empirical findings allow us to conclude that positive emotions elicit ANS reactivity. We also explored whether the ANS reactivity is specific to discrete emotions (in terms of patterns and magnitudes) or—alternatively—whether similar ANS reactivity accompanies all positive emotions. Finally, we tested participant characteristics and methodological factors as moderators of the ANS reactivity to discrete positive emotions. One main and three secondary findings emerged.
Based on univariate analyses, most discrete positive emotions elicited no or weak ANS reactivity. Moreover, half of the effect sizes in ANS responses were highly inconsistent, suggesting that other significant physiological variability sources exist. We also found that similarities outweighed differences in ANS responses during positive emotions. This contrasts the literature suggesting a stronger physiological differentiation among discrete positive emotions (Kreibig, 2010; Shiota et al., 2017). Finally, we found few moderating effects of study or participant characteristics. Thus, the current empirical material supports the view that positive emotions produce no or only a weak and nonspecific ANS response relative to baseline and neutral conditions (Cacioppo et al., 2000; Lench et al., 2011; Siegel et al., 2018).
However, we emphasize that these conclusions must be considered tentative because they are based upon imbalanced and incomplete data and one type of analysis (univariate, not multivariate). This suggests the need for more systematic research on the physiology of positive emotions that will fill existing gaps and provide material for future robust evaluation of positive emotions and ANS activity. For instance, impedance cardiography that is often applied to the study of stress and negative emotions is relatively underrepresented in the study of positive emotions. Moreover, the psychophysiological study of amusement is greatly overrepresented relative to gratitude, pride, or love. Finally, we advocate more multivariate sampling and analysis of emotional responses in positive emotions.
Positive Emotions and ANS Reactivity: The State-Of-The-Art
We based this review on the most extensive collection of available studies, which produced over 686 effect sizes derived from 6,546 participants. However, in the coding process, we observed substantial variability across this large number of studies, which resulted in the collection of a widely dispersed dataset. The studies were conducted in different settings. For instance, the laboratories used various equipment, procedures, and data cleaning and analysis techniques. Most studies examined only reactivity related to two emotions using 2-3 ANS measures with a single elicitation method. Furthermore, when coding the possible moderators, in many cases, we were unable to determine whether participants were alone during the experimental task or the experimenter stayed in the room after placing the physiological sensors. We found that only 44% of studies explicitly stated that they video recorded participants during the experiments. In this way, our meta-analysis supports the message from the recent comment on the current state of the science of positive emotion (Shiota, 2017) - although the field made incredible progress in the last decades, affective scientists are still far away from the promise of this field being fully realized. This also includes the call for more detailed reporting of procedures (e.g., were participants explicitly observed) and data use (e.g., duplicate datasets).
The data collected so far on positive emotion is insufficient to strongly support the ANS specificity versus similarity for a wide range of positive emotions presented in our investigation. Our analyses were challenging due to a widely dispersed dataset with small numbers of studies per emotion and per ANS measure. Moreover, some comparisons and analyses were performed on a relatively small number of effect sizes. Although the field of affective science struggles to just-decide-already whether specificity or similarity of ANS emotion-related reactivity is the ground truth, with this review, we observed that the current state-of-the-art is not sufficient to address this expectation for definite conclusions. While reviewing hundreds of studies, we also observed that researchers moved quickly to asking complex questions related to functions of targeted emotion without addressing more basic questions, e.g., which ANS parameters are adequate for studying a specific positive emotion. We suggest that a return to more basic questions might advance the field of psychophysiology of emotions. It would be beneficial for the field of positive emotions to further examine ANS reactivity, in particular, to positive emotions that have not been explored yet, such as hope or schadenfreude, using multiple ANS measures. We advocate that the field of positive emotions would benefit from greater integration and uniform standards/rigor for emotion elicitation and data curation, analysis, and reports.
Do Positive Emotions Produce Robust Changes in ANS Reactivity?
We found that the set of inspected positive emotions produce no or weak increases in ANS reactivity in both SNS and PNS. Our findings are consistent with models of positive emotions that assumed that positive emotions do not generate independent sympathetic responses (Fredrickson & Levenson, 1998; Fredrickson et al., 2000; Fredrickson, 2013; Folkman, 2008; Levenson, 1988, 1999). Moreover, we did not observe increased PNS reactivity in positive emotions, as suggested by the polyvagal theory (Porges, 2011). One explanation for these null results is that large differences across experiments might be responsible for the responding range. Figure 2 presents that even the mean effects interpreted as medium sizes had wide confidence intervals that prevented them from being significant. These findings are consistent regardless of focusing only on separate ANS measures, measures that had more than ten studies, or measures aggregated into broader categories.
Our findings match previous meta-analyses focused on happiness that concluded that happiness produces weak ANS reactivity and that this reactivity is not different from neutral conditions (Lench et al., 2011; Siegel et al., 2018). Our findings do not support conclusions from qualitative reviews in which some positive emotions such as contentment and love decreased cardiovascular or electrodermal activity (Kreibig, 2010). However, more than half of the physiological responses’ directions in the qualitative review were based on fewer than three studies, suggesting that these findings were preliminary (Kreibig, 2010). With additional studies that were published over the last decade, we found support for the previously found directions of the electrodermal reactivity (increases) to positive emotions. We also found support for increased ANS activity to joy and amusement (Kreibig, 2010).
Are ANS Reactivity Patterns Specific to Particular Positive Emotions?
The main goal was to provide a quantitative review of the body of research related to ANS reactivity and positive emotions. However, we also evaluated the specificity or generality of ANS reactivity to discrete emotions. The basic expectation in this meta-analysis was that discrete positive emotions produce specific adaptive changes in physiology (Ekman & Cordaro, 2011; Levenson, 2011; Panksepp & Watt, 2011; Kreibig, 2010).
We found that similarities outweighed differences in ANS responses during positive emotions. This finding is consistent with the models that view ANS reactivity to emotion as context-sensitive and not discrete-emotion-sensitive (Barrett, 2013, 2017; Quigley & Barrett, 2014). Thus, the ANS reactivity is not random but is specific and supports actions in the specific context, which could vary for the same discrete emotion (Barrett, 2006; Barrett & Russell, 2015; Quigley, & Barrett, 2014). Theorists suggest that multiple distinct, context-sensitive physiological responses to discrete emotion are possible, as long as both serve the same adaptive function, e.g., freezing versus fleeing from a threat in fear (Ekman, 1992).
However, the ANS reactivity is only one component of emotional responding. Thus, major judgments about the structure of emotions should be interpreted along with affective and behavioral responses and should not be based solely on any one component.
Shared ANS reactivity to positive emotions might be related to common neural origin from a highly conserved circuit of neural structures, namely the mesolimbic pathway, often called the “reward system” (see Shiota et al., 2017 for discussion). The activation along the mesolimbic pathway has been linked to a wide range of stimuli associated with the family of positive emotions, including delicious foods (Berridge, 1996), monetary incentives (Knutson et al., 2001), babies (Glocker et al., 2009), loved ones (Bartels & Zeki, 2004), humor (Mobbs et al., 2003), and favorite music (Blood & Zatorre, 2001). It may explain the mechanism by which the discrete positive emotions share some overlapping properties that might be further differentiated depending on the conditions in which positive emotions are activated. Overlapping properties of positive emotions and continuous gradients between discrete emotion categories have been found in recent large-scale investigations (Cowen & Keltner, 2017). That study has shown that emotions were more precisely conceptualized in terms of continuous categories, rather than discrete emotions, showing smooth gradients between emotions, such as from calmness to aesthetic appreciation to awe (Cowen & Keltner, 2017).
Supporting the dimensionality of emotions, we found differences along the dimension of approach motivation (Gable & Harmon-Jones, 2010; Harmon-Jones et al., 2013). Positive emotions characterized by strong approach tendencies, such as joy and excitement, were accompanied by a higher sympathetic reactivity (e.g., DBP, MAP) than low-approach positive emotions like amusement. Our investigation may serve future studies to conceptualize positive emotions in terms of physiological arousal starting from the least arousing and ending with the most arousing positive emotions, namely awe, attachment love, gratitude, nurturant love, contentment, excitement, amusement, pride, craving, sexual desire, and joy.
Are There Moderators of ANS Reactivity to Positive Emotions?
We investigated several moderators that we thought might influence physiological responsiveness to emotions, but most did not moderate the observed effects. Only in craving did we observe a significant moderating effect of the elicitation method on the physiological response. We observed that behavioral methods, namely, exposure to food, produced stronger ANS reactivity than pictures, films, and imagery. This observation indicates the advantage of using active rather than passive emotion elicitation methods. Furthermore, in line with Lench and colleagues (2011), we found that reactions to craving were stronger when the proportion of women in samples increased.
Contrary to our expectations, we found no influence of several continuous variables on physiological reactivity to positive emotions, including age, sex proportion, participant number, and study quality (Kret & De Gelder, 2012; Stevens and Hamann, 2012; Mill et al., 2009; Sullivan et al., 2007). Like the previous meta-analysis of Lench and colleagues (2011), we found no evidence that the participants’ age influenced the degree of emotional reactivity. However, this may be due to imbalanced age distribution (skewed young), meaning we were underpowered to detect age differences. A thorough examination of how age influences the emotional experience's intensity requires additional research with older participants. In line with previous meta-analyses, we found no sex differences in response to positive emotions (Joseph et al., 2020; Siegel et al., 2018). Finally, although we found indications of publication bias for some pairs of ANS reactivity and positive emotion, we conclude that most mean effect sizes seem to be robust and unlikely to be an artifact of systematic error.
Limitations and Future Directions
First, as we emphasize throughout the paper, the conclusions we present are provisional and contingent upon current data availability. More definitive conclusions will await additional research, particularly on under-researched positive emotions and measures.
Second, in this project, we used a univariate approach to analyze the mean ANS reactivity to discrete positive emotions in a series of meta-analyses. Although available multivariate meta-analytic approaches (Riley et al., 2017) would provide a better fit to the characteristic of emotions (Kragel & LaBar, 2013; Stephens et al., 2010), several factors militated against using a multivariate approach. For instance, a multivariate meta-analysis requires a correlation matrix between the ANS measures. This was not possible to obtain because only 7 out of 128 articles included in our investigation reported correlations between some ANS measures. Along similar lines, for many analyses (e.g., amusement), two or more ANS measures were never observed jointly in the same study. Moreover, a previous meta-analysis found that multivariate pattern classifiers did not provide strong evidence of a consistent multivariate pattern for any emotion category (Siegel et al., 2018). Of note, the multivariate and univariate models produce similar point estimates, but the multivariate approach usually provides more precise estimates. Thus, the benefits of a multivariate meta-analysis are small (Riley et al., 2017). The advantages of using multivariate meta-analysis of multiple outcomes are greatest when the magnitude of correlation among outcomes is large, which was not the case for most of our analyses. In conclusion, our approach can produce statistically valid results for each pair of positive emotion and ANS reactivity measures (Pustejovsky & Tipton, 2021). Future studies might collect multiple physiological measures when studying ANS reactivity to emotions (Cacioppo et al., 2000) to provide data that allows for robust multivariate analyses.
Third, we used univariate statistics, which disrupt the physiological response's continuity and treat the entire emotion manipulation as a separate piece. Although univariate methods have historically dominated the literature (see Cacioppo et al., 2000; Kreibig, 2010, for the reviews), future reviews may use multivariate approach data to replicate our findings.
Fourth, during the coding, we relabeled examined emotions in many studies. However, we found no differences in effect sizes extracted from studies with the original emotion label and effect sizes extracted from studies for which we renamed the emotion label. Moreover, the conclusions that come from our literature research stress the importance of using precise terminology in emotion-related literature. The overview of existing empirical and theoretical models indicates not only a variety of discrete positive emotions but also a variety of terms used to describe them. For instance, researchers used different labels for emotions elicited by funny situations, such as amusement (Kreibig et al., 2013), happiness (Kring & Gordon, 1998), or mirth (Foster et al., 2003). The heterogeneity of labels suggests problems with discrete emotions’ construct validity and measurement invariance. Future research would benefit from a more uniform nomenclature and definitions accepted by researchers within affective science.
Fifth, most of the theoretical and empirical models conceptualize individual positive emotions without clearly addressing how different positive emotions might be interrelated (e.g., Cowen and Keltner, 2017; Ekman and Cordaro, 2011; Tong, 2015; Weidman & Tracy, 2020, with the exception of Kreibig, 2014; and Shiota et al., 2017). It might be useful to group discrete emotions into families or clusters based on their similarities. For instance, joy and excitement are similar emotions associated with progress in achieving one's goals, but excitement has an anticipatory response compared to joy that brings well-being and good fortune after an event (Lazarus, 1991; Shiota et al., 2017; Smith & Kirby, 2010). Future studies may focus on examining similarities rather than differences between positive emotions. Researchers should also balance between generalization and differentiation in studying emotions.
A sixth limitation resulted from including studies that examined physiological reactivity from baseline or neutral conditions. The results produced by these studies differ due to differences in the design of these studies. Some research used neutral movies as a baseline (e.g., De Wied et al., 2009), whereas other studies used neutral videos in the experiment (e.g., Codispoti et al., 2008). We followed the theoretical premise that both baselines and neutral conditions should be emotionally impartial. A moderation analysis showed that the type of comparison used in primary studies had no effect on the size of the physiological reactivity. These results allowed us to examine further hypotheses, but both decisions of including different types of comparison and relabeling the emotion categories may have produced bias in the results of our meta-analysis. Given the considerable increase in psychophysiological research on emotions in recent years, future meta-analytic work would provide empirical support for the emotional impartiality of baseline and neutral conditions.
Seventh, we found substantial research methodology variability in primary studies. We tested whether the study quality moderated the effect sizes by assessing the presence of exclusion criteria, manipulation check procedures, and protocols for reporting or handling artifacts and missing data. We found those three measures to be objective indicators of study quality. However, the study quality had no effect on the size of physiological reactivity. More studies that include multiple ANS measures and multiple discrete positive emotions with diverse samples are required to strengthen broad inferences about ANS responses to positive emotions. Future studies might also examine how emotions differ in ANS reactivity rather than asking whether emotions generally differ physiologically (e.g., Berntson et al., 1991; Stemmler, 1992). ANS reactivity produces the optimal bodily milieu to provide physiological support for behaviors associated with discrete emotion (Levenson, 2014). This requires unique configurations of multiple physiological responses rather than a single unique physiological change (Levenson, 2014). Single ANS measures might not be sufficient, given that most physiological measures used in the emotion-related literature constitute the physiological outcome of emotion-related states, showing a one-to-many relation between the physiological measure and emotions (see Cacioppo et al., 2000; Richter & Slade, 2017 for the discussion). Groups of emotions may lead to similar general activation that occurs in response to an upcoming action (Brehm, 1999; Fredrickson & Levenson, 1998; Frijda, 1987). For instance, excitement, craving, or sexual desire prepare the organism to “be ready for action,” and they produce similar sympathetic activation. However, more specific activity might be observed in targeted organs. For instance, craving might be observed in the gastrointestinal tract, sexual arousal in the genital system, and excitement in the locomotor system (Levenson, 2011, 2014).
Eighth, most of the ANS variables included in our meta-analysis are blends of SNS and PNS activation (e.g., HR). The measures more specifically related to PNS or SNS measures (e.g., RSA or PEP) were not broadly assessed across the positive emotions. Thus, we could not fully address whether positive emotions produce pure SNS or PNS activity but rather the co-product of one of the two systems.
Nineth, we believe that our meta-analysis opened the discussion for methodological issues in the psychophysiology of emotions that would be worth testing empirically. For instance, for moderators, we focused on the length of the time interval used to calculate the physiological levels for baselines and emotion manipulations. Although we did not find effects of the time interval on mean ANS reactivity to positive emotions, we believe that comparing different time intervals of the same physiological measure is problematic. Similarly, researchers usually used the same time intervals to present the reactivity of all ANS measures despite differences across the family of ANS variables. Thus, scientists tended to sacrifice the specificity of particular ANS measures for the sake of a uniform data analysis strategy.
Conclusions
This meta-analytic review addresses the empirical evidence for how discrete positive emotions influence autonomic nervous system reactivity. This review's novelty stems from the description of state-of-the-art scientific coverage of ANS in positive emotions and (as much as possible) contrasting a broad range of positive emotions with one another rather than focusing only on happiness. The available data was widely dispersed and suggested many limitations and gaps in the state-of-the-art. Our provisional findings indicated that when analyzed in univariate terms, all positive emotions produce a similar physiological pattern, namely no or weak increases in ANS activity to positive emotions studied to date. However, the limitations of the state-of-the-art identified in our investigation suggest caution in drawing strong conclusions based on the available evidence.
Supplemental Material
sj-docx-1-emr-10.1177_17540739211073084 - Supplemental material for Autonomic Nervous System Activity During Positive Emotions: A Meta-Analytic Review
Supplemental material, sj-docx-1-emr-10.1177_17540739211073084 for Autonomic Nervous System Activity During Positive Emotions: A Meta-Analytic Review by Maciej Behnke, Sylvia D. Kreibig, Lukasz D. Kaczmarek, Mark Assink and James J. Gross in Emotion Review
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Narodowe Centrum Nauki (grant number UMO-2017/25/N/HS6/00814, UMO-2019/32/T/HS6/00039).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
