Abstract
Percutaneous coronary intervention (PCI) of calcified coronary arteries is associated with poor outcomes. Poorly modified calcified lesion hinders the stent delivery, disrupts drug-carrying polymer, impairs drug elution kinetics and results in under-expanded stent (UES). UES is the most common cause of acute stent thrombosis and in-stent restenosis after PCI of calcified lesions. Angiography has poor sensitivity for recognition and quantification of coronary calcium, thereby mandating the use of intravascular imaging. Intravascular imaging, like intravascular ultrasound and optical coherence tomography, has the potential to accurately identify and quantify the coronary calcium and to guide appropriate modification device before stent placement. Available options for the modification of calcified plaque include modified balloons (cutting balloon, scoring balloon and high-pressure balloon), atherectomy devices (rotational atherectomy and orbital atherectomy) and laser atherectomy. Coronary intravascular lithotripsy (IVL) is the newest addition to the tool box for calcified plaque modification. It produces the acoustic shockwaves, which interact with the coronary calcium to cause multiplanar fractures. These calcium fractures increase the vessel compliance and result in desirable minimum stent areas. Coronary IVL has established its safety and efficacy for calcified lesion in series of Disrupt CAD trials. Its advantages over atherectomy devices include ease of use on workhorse wire, ability to modify deep calcium, no debris embolization causing slow flow or no-flow and minimal thermal injury. It is showing promising results in modification of difficult calcified lesion subsets such as calcified nodule, calcified left main bifurcation lesions and chronic total occlusion. In this review, authors will summarize the mechanism of action for IVL, its role in contemporary practice, evidence available for its use, its advantages over atherectomy devices and its imaging insight in different calcified lesion scenarios.
Plain language summary
Presence of coronary calcium during stenting is associated with the risk of stent under expansion. It’s imperative to adequately modify this coronary calcium before placing the stent. Till recent past, the most effective method for calcium modification is debulking it with rotational artherectomy, which is associated with the risk of coronary perforation, slow flow or abrupt vessel closure. Recently, a balloon-based lithotripsy device has established its safety and efficacy for treating such lesions. Coronary intravascular lithotripsy (IVL) is an easy to use calcium modification device and is associated with almost negligible complications, when compared with artherectomy devices. In this review, we will discuss the mechanism of IVL action and its use in different scenarios of calcified coronary artery disease.
Keywords
Introduction
Coronary calcification has been an ongoing challenge to interventionalists since the era of bare metal stents, primarily because of its association with worse clinical and procedural outcomes. 1 While the introduction of drug eluting stents led to improvements in the landscape of percutaneous coronary intervention (PCI), outcomes in calcified coronaries continue to be significantly inferior when compared with PCI in the absence of coronary calcification. The most common cause of this poor outcome is acute and chronic stent failure caused by UESs. Heavy calcification impedes stent delivery, disrupts drug-delivering polymer and subsequently impairs drug delivery dynamics.2,3 Hence, adequate calcified lesion preparation before stent delivery is of paramount importance for good short- and long-term outcomes.3,4
The challenge faced by most interventionalists is to properly identify and quantify the amount of calcium they are dealing with to facilitate complete revascularization. Unfortunately, coronary angiography has low to moderate sensitivity in detection and quantification of coronary calcium. It is here that the role of intravascular imaging becomes substantial. The remarkable accuracy of intravascular imaging techniques such as ‘Intra Vascular UltraSound’ (IVUS) and ‘Optical Coherence Tomography’ (OCT) to identify coronary calcium has been adequately validated.5–7 Both IVUS and OCT provide the interventionalists with a reliable tool in the form of calcium scores to identify calcified stenosis at risk for stent under expansion requiring adjunctive calcium modification before stent implantation.8,9
Calcium modification devices have come a long way since the introduction of PCI with the availability of modified balloons (high-pressure balloons, cutting and scoring balloons), atherectomy devices [rotational atherectomy (RA), orbital atherectomy (OA) and laser atherectomy] and more recently intravascular lithotripsy (IVL). While modified balloons can be used for lesions with less calcium burden, their role in severely calcified lesions is limited to adjunctive therapy. 10 RA and OA on the other hand are viable options for severely calcified coronaries, especially for balloon uncrossable lesions as debulking devices. They essentially act by preferentially ablating the hard calcium plaques while the elastic healthy tissue deflected away from the burr. Both RA and OA have been shown to improve clinical outcomes in patients with severely calcified lesions. However, these devices are associated with a significant learning curve and peri-procedural complications.4,11,12
IVL is the newest addition to the panel of modalities available for modification of severely calcified lesions and has shown promising results. Similar to renal calculi lithotripsy, this therapy utilizes pulsatile sonic pressure waves to preferentially target calcium. While it has been shown to be especially effective on circumferential calcium, its role in calcified nodules (CN) is also being explored. IVL has smaller profile than many available specialized balloons and atherectomy devices and is easier to use. Additionally, it is associated with very low risk of slow flow and coronary perforation as compare to atherectomy devices.13,14 The role of lithotripsy for coronary calcified lesions has been validated by the Disrupt CAD series of trials, with excellent safety and efficacy outcomes.15–18 In this review, we will discuss the role of coronary IVL in contemporary practice along with the unique advantages and opportunities it provides us in this present age of increasingly complex coronary interventions.
Intravascular lithotripsy
The Shockwave Medical Coronary Rx Intravascular Lithotripsy System (Shockwave Medical, Inc., Fremont, CA, USA) is one of the newest modalities to have joined the ranks of calcium modification techniques. IVL for coronary and peripheral artery diseases evolved from lithotripsy for renal and ureteral calculi.19,20 IVL uses acoustic pressure waves to interact with arterial calcification, producing micro and macro fractures in the calcified plaque allowing for increased vessel compliance and larger stent expansions. These acoustic pressure waves preferentially interact with calcified lesions which have a higher density, while sparing the normal soft tissue primarily because of differing acoustic impedance of calcified tissue and soft tissue (hard on hard and soft on soft).14,21
Mechanism of action
The coronary IVL system consists of a single use monorail catheter, a pulse generator and a connector. The monorail catheter has two sets of emitters at its tip covered by a semi-compliant balloon. Each pair of emitters is arranged on diametrically opposite sides of the balloon to produce spherical energy signals resulting in a peak acoustic pressure aligning with the location of the emitter. The balloon size ranges from a diameter of 2.5 to 4 mm and a constant length of 12 mm, while its entry profile ranges from 0.042 to 0.046 inches. The IVL emitters employ electric sparks to produce rapid expansion and collapse of vapor bubbles within the balloon that results in production of the acoustic pressure waves. These acoustic pressure ‘shockwaves’ travel through soft tissues with minimal effect because of similar acoustic impedance parameters of fluid in the IVL balloon and soft tissue. However, fracturing effects are seen when these waves encounter tissues with differing acoustic impedances, such as a transition from soft tissue to calcified tissue. As these waves are transmitted both circumferentially and transmurally in an unfocussed manner, they affect both superficial and deep lying calcium. The coronary IVL acoustic shockwaves produce relatively lower positive peak pressures (50 atm versus 1000 atm) with a negligible trailing peak negative pressure (−3 atm versus −150 atm) compared with renal IVL resulting in a tensile stress which remains well below the threshold associated with tissue damage. Thus, the leading edge of the shockwave imparts compressive stress to calcium once it is encountered and is the primary mechanism of calcium fracture while limiting potential for soft tissue injury. 22 The acoustic energy delivery of IVL is circumferential and is not affected by wire bias or device size, in contrast to other atherectomy technologies.
The IVL balloon is sized at 1:1 ratio with the reference diameter of the target vessel to allow for apposition to the vessel wall. It is taken across the calcified lesion and is connected with the pulse generator via the connector, followed by inflation of the balloon with a mixture of saline and contrast (in a 50/50 ratio) to a sub-nominal pressure of 4 atm, thus avoiding barotrauma.23,24 Each catheter emits a total of 80 shockwave pulses in 8 cycles (10 pulses in each cycle at a frequency of 1 pulse/s). At the end of each cycle, inflation pressure is increased to 6 atm (nominal pressure) followed by deflation of the balloon to allow distal perfusion and dissipation of heat. The balloon may be deflated after five pulses if prolonged inflation is causing hemodynamically significant ischemia as in the case of unprotected left main (LM) intervention. Recently, new IVL balloons with a maximum of 120 pulses have come into the market, thereby offering treatment of long calcified vessels with a single device. Bulkier crossing profile of the IVL balloon may sometimes hinder its delivery across tight lesions necessitating the use of pre IVL non-compliant (NC) balloon dilatation or use of guide extension.
Evidence
The coronary IVL device has received both FDA and CE Mark approval based on the Disrupt CAD series of trials which evaluated the safety and efficacy of coronary IVL. The Disrupt CAD I, II, III, and IV studies (Table 1) confirmed the safety and effectiveness of IVL for severely calcified lesions and showed high procedural success with minimal complications.15–18 A pooled analysis of all four Disrupt CAD trials 25 showed that IVL safely facilitated successful stent implantation with a high procedural success rate of 92.4% and freedom from major adverse cardiovascular events (MACE) at 30 days at a rate of 92.7%. Rates of serious post-IVL angiographic complications were 0.3% with no IVL associated abrupt closure or episodes of no-reflow and only 1 (0.3%) incidence of post stent perforation. These low rates of serious angiographic complications are consistent with IVL’s mechanism of action, which involves calcium fracture in situ without the generation of athero-embolic debris and/or significant heat energy as seen with atherectomy devices. By fracturing the calcific layer, IVL improves vessel compliance without the need for aggressive high-pressure balloon dilatation prior to stent delivery. It thus reduces barotrauma to the vessel and consequently the chance of severe dissections. The CAD Disrupt trials involved post-IVL dilatation prior to stent delivery at the operator’s discretion and was not used in most patients (83.2%). Nonetheless, stent delivery was successful in 99.5% of patients (Table 1).
Summary of Disrupt CAD trials.
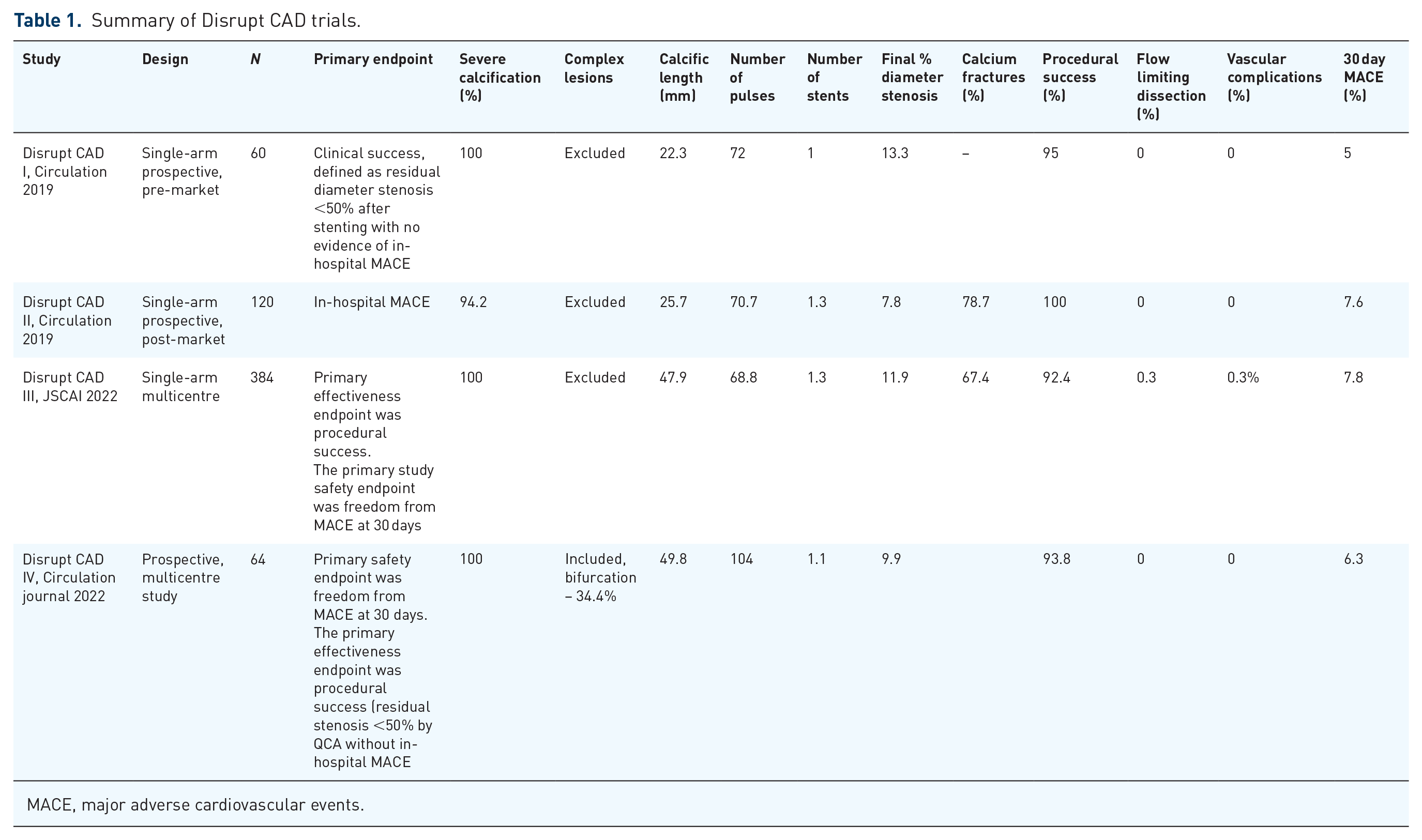
MACE, major adverse cardiovascular events.
One potential complication of IVL is ventricular ectopic activity or transient IVL-induced capture leading to ventricular ectopics known as shockotopics. These rates dependent and sustained ventricular captures are possible only when the acoustic pressure waves depolarize the cardiac tissue via the stretch-activated response and the native heart rate is lower than 60 beats/min. While this phenomenon was frequently seen in the Disrupt CAD III cohort (41.1% of cases), it did not result in sustained ventricular arrhythmias of any clinical consequence.15–17 This is probably due to the fact that the 8 μJ of mechanical energy applied over the IVL balloon is several orders of magnitude lower than the electric energy required for ventricular tachycardia or ventricular fibrillation induction (0.6–2.0 J).
Role of IVL in contemporary practice
Calcified coronary lesions cause significant hindrance to PCI and have conventionally been treated with high-pressure balloon expansion and atherectomy. Balloon-based technologies are dependent on high-pressure inflations for plaque modification thus causing barotrauma and increasing the risk of dissection, perforation and abrupt arterial closure. In contrast, IVL uses shockwaves delivered through a semi-compliant balloon inflated only to four atmospheric pressure and thus drastically reduces this risk of barotrauma. Athero-ablative technologies such as RA and OA rely on localized debulking of superficial calcium, which subjects the target vessels to thermal injury and vascular complications. These techniques also carry the risk of debris embolization, resulting in slow flow or no-reflow. Apart from breaking the calcium plaque, atherectomy devices have also been shown to reduce the calcium thickness which can then be followed by balloon dilatation to facilitate calcium fractures. OCT studies indicate that a calcium thickness of less than 0.24 mm predicts calcium fractures when treated with balloon dilatation. The ROTAXUS and PREPARE-CALC RCTs compared RA to balloon dilation prior to stenting and demonstrated higher success rates.26,27 However, vessel tortuosity is often a limiting factor in the use of atherectomy due to difficulty in advancing the wire and increased risk of guidewire bias, that is, deflection of guidewire from the central axis which can result in vessel dissection and perforation. This risk is also evident in smaller sized vessels. Wire bias can also result in eccentric cuts leading to incomplete calcium modification.
Coronary IVL is an operator friendly balloon-based therapy, where a semi-complaint balloon is used on the workhorse wire for delivering highly effective shockwaves to modify calcified lesions. It is unique in the sense that it fractures both superficial and deep calcium in situ and there is no debris embolization or thermal injury causing slow flow, no-reflow and vascular complications. While superficial calcium lesions are good targets for atherectomy and modified balloons, these modalities are often ineffective against deeper calcium, that is, calcium which is closer to the adventitia than to the lumen. 28 In a cadaveric study using micro-computed tomography (CT) and histopathology, calcium fractures were seen involving both deep and superficial layers of the vessels with the use of IVL. 14 Ali et al. 29 also noted calcium fractures in both superficial and deep calcium in an OCT analysis of coronary IVL. While circumferential calcium (presence of >270° of calcium) is most susceptible to the shock waves, as evidenced in the Disrupt CAD trials, there has also been increasing evidence supporting the use of IVL in CN. Pooled data from the Disrupt CAD I and II trials showed similar angiographic outcomes and complications between eccentric (nodular) and concentric lesions treated with coronary IVL. 30 In severely calcified lesions where operators are unable to cross the lesion with the IVL catheter or any balloon, atherectomy could be used to facilitate IVL use. This synergistic use has been demonstrated in several small studies and case reports, indicating that even the most severely calcified lesions could be adequately prepared with the right strategy.31,32 Figure 1 shows the proposed algorithm for the management of coronary calcified lesions and demonstrates that IVL can be employed as an effective strategy in most scenarios involving calcified lesions.

Proposed algorithm for approach to severely calcified coronary artery lesion.
Imaging insight of coronary IVL
Intravascular imaging has an integral role to play in the assessment and modification of calcified lesions. Angiography alone significantly underestimates the amount of calcium when compared with intra-coronary imaging with a sensitivity of around 40%. IVUS has been reported to have a sensitivity and specificity of 89–90% and 97–100% respectively when compared with histology. 31
The utility of IVUS is limited by its inability to assess the thickness and volume of the calcific sheet as the ultrasound beam does not penetrate calcium and thus is unable to picturize the distal borders of the calcium. It is in this aspect that the advent of intra-coronary OCT has further transformed the field in terms of even better characterization of calcified lesions, providing us the ability for detailed analysis of calcium thickness, length, arc and volume. And while these factors undoubtedly have immense prognostic implications in terms of procedural and clinical outcomes, they also offer an avenue to make informed decisions regarding plaque modification strategies. Although IVUS has a higher sensitivity of detecting calcium, OCT helps to identify deeper calcium often missed on angiography, and also helps to choose the optimal calcium modification strategy to be employed.6,33,34 Additionally, it is a useful tool to assess the impact of IVL shockwaves on the calcium layer. OCT sub-studies of IVL-based trials have shown that the primary mechanism of luminal gain following IVL treatment remains calcium fracture (Figure 2). These fractures are multiplanar, that is, circumferential and longitudinal, and the luminal gain observed is without the need for high-pressure balloon dilatation. By fracturing the calcium layer, IVL improves vessel compliance and facilitates fracture expansion following stent implantation resulting in improved stent expansion areas in calcified coronary arteries. This ultimately may favourably affect long-term target lesion failure rates. The Disrupt CAD III OCT sub-study has shown calcium fractures in 67.7% of lesions after IVL. An interesting observation is that the absence of fractures on OCT has not been shown to have any impact on stent areas and clinical outcomes. This has been postulated to be because of deep out of plane fractures or microfractures that are beyond the imaging depth and resolution of OCT. These IVL-induced microfractures have been confirmed on histology and micro-CT studies and help in achieving increased vessel compliance and stent areas. Therefore, the absence of fracture on imaging should not be taken as IVL failure. In patients with renal dysfunction, IVUS is the imaging modality of choice to prevent the extra contrast load. Recently, we have published the role of saline as a flushing media for zero contrast OCT in these patient subsets.35,36

Imaging insight of IVL effect on circumferential coronary calcium. (a–e) Pre-IVL OCT and IVUS images showing 360° arc of calcium (asterixis) with their respective lumen areas. (f–j) Post-IVL increase in lumen areas and fractures in calcium arc (marked by arrows). (k–o) Post-PCI final MSAs and widening of calcium fractures (asterixis).
Special considerations for IVL
CN have a unique plaque morphology that consists of nodular calcification with an overlying fibrous cap which may be disrupted or intact. CN are associated with acute coronary syndromes (ACS), worse outcomes after PCI and higher rates of stent failure.37–39 These are, hence, high-risk lesions that are particularly difficult to modify due to their eccentricity which often leads to suboptimal lesion preparation and stent under expansion. In the realm of coronary calcium, CNs have been associated with worse clinical outcomes and limited management options. While some operators have recommended a combination of stent-less PCI after atherectomy and drug-coated balloons for treating lesions with CNs, the outcomes remain significantly worse in these patients.40,41 The risk of wire bias associated with atherectomy is further increased in the presence of nodular calcium to the extent that the ablation burr may not even touch the site of interest. Not to mention there remains the risk of distal embolization after atherectomy, which may be amplified because of the unstable plaque composition of CNs. The heterogenous hardness and eccentric distribution of CNs also makes balloon-based modalities only partially effective. 41 Limited studies regarding the effect of IVL on CNs have shown promising results. Ali et al. 42 examined the safety and efficacy of IVL in CNs from the Disrupt CAD I-IV trials and found that it was a highly effective modality with similar procedural outcomes when compared to non-CN lesions in terms of residual area stenosis, stent expansion or acute gain. The potential of IVL in management of these lesions and the impact it has on long-term outcomes should be further explored in depth. In our experience, a greater number of shockwave pulses are required for modification of CNs as compare to circumferential calcium and the base of the CN is the most common site of visible facture on OCT (Figure 3).

IVL for coronary CN. (a–c) OCT pullback from LAD artery showing CN (asterixis) with their lumen areas. (c–e) Post-IVL OCT images at same anatomical level as that of pre-IVL showing increase in lumen areas and calcium fracture at apex (d) and at base (e, f) of the CN. (g–i) Post-PCI OCT cross sections with their respective MSAs.
LM bifurcation PCI
Coronary bifurcation lesions are a procedural challenge with lower rates of procedural success and higher restenosis rates. 43 LM bifurcation PCI is a high-risk complex intervention primarily because a large part of the myocardium is at risk. LM bifurcation lesions are usually associated with heavy calcifications. This often poses a significant challenge to device delivery and expansions given the complex anatomy. Heavily calcified bifurcation lesions also carry the risk of side branch (SB) occlusion during main branch (MB) stenting. 44 IVL has several features that theoretically sets it up for success in LM bifurcation lesions as compared to atherectomy devices. Its simplicity of use along with smaller profile makes crossing tight complex lesions easier. IVL can be used on any workhorse guidewire with another guidewire in the SB, thus ensuring that SB access is never lost and that the SB is protected from occlusion during plaque modification in the MB. In contrast, atherectomy devices require specialty wires and are accompanied with the risk of losing the SB, as no wire can be kept in the SB during atherectomy in the MB due to the threat of SB wire fracture and embolism. Another added benefit of IVL may be the effect of the shockwaves on any adjacent calcifications along the SB ostium, making the ostium more compliant for higher stent expansion areas. These advantages of IVL over atherectomy devices provide an avenue for safer intervention of a heavily calcified LM lesion (Figure 4). There have been scattered case reports employing the use of IVL in bifurcation and LM lesions, often in conjunction with RA. 45 Although CAD Disrupt I, II and III trials excluded patients with bifurcation lesions, CAD Disrupt IV trial enrolled 72 patients and bifurcations constituted 34.4% of the lesions. 17 A study by Leung et al. 46 in 2022 also explored the use of IVL and constituted 18% bifurcations. In a pooled analysis of all four CAD disrupt trials, significant difference in procedural success was observed in the bifurcation lesion sub-group (94.3% versus 88.9%). Validation of the safety and effectiveness of IVL in such lesions is a field of intrigue and interest that should be further explored.

Use of intravascular lithotripsy in calcified LMBD. (a) Coronary angiography showing critical distal LMBD. (b, c) OCT pullback from LM to LAD revealed deep eccentric calcium in L and B modes. Both LAD and LCx were wired and IVL balloon sized 3.5 × 12 mm2 was used to modify the deep calcium. (d) Post-IVL coronary angiography showed decrease in distal LM stenosis without slow flow, perforation, flow limiting dissection and LCx compromise. (e, f) Post-IVL OCT showed increase in distal LM luminal area (arrow) on L and B modes. A deep linear fracture was also seen in distal LM. (g) Post-PCI angiography showed significant reduction in distal LM residual stenosis without LCx compromise. (h, i) Post-PCI OCT showed increase in distal LM lumen area from 1.61 to 10.48 mm2.
The role of atherectomy is also being cautiously explored in bifurcation lesions, with Ito et al. demonstrating high success rates and low rates of complications with RA.47,48 Chambers et al. 49 showed similar 30-day MACE rates in bifurcation lesions treated with OA or RA compared to non-bifurcation lesions. However, given the high incidence of SB closure in bifurcation lesions and high risk of SB perforation associated with atherectomy devices, the use of these devices should be limited to experienced operators. 50
Stent failure
UES is associated with an increased risk of in-stent restenosis and stent thrombosis. Poorly modified calcium is usually the cause of UES. RA can be used to ablate the stent struts (intimal ablation) and fragment the underlying calcium, but it is associated with high risk of procedural complications. IVL is a potential solution in these cases where calcified lesion was the cause of under expansion. Case reports have suggested that despite adequate pre and post dilations if there is significant stent under expansion, IVL can be a useful strategy to facilitate complete stent expansion. 51 IVL can also be used as a bail out strategy if there remains significant under expansion even after aggressive post dilation with high-pressure OPN NC balloons. The SMILE registry included 39 cases of UESs in 34 patients. IVL was successful in 34 cases (87.1%), with significant improvement in stent diameters and minimum stent areas (MSAs) (p < 0.00001). Non-fatal peri-procedural ST-elevation myocardial infarction occurred in one case (2.5%) due to IVL balloon rupture. No cardiac death, target lesion revascularization or stent thrombosis occurred in-hospital and at 30-day follow-up. 52 The use of IVL for UES is an off-label indication, as there is a concern of shockwave-induced drug delivery polymer disruption of the stent. Therefore, it is always advisable to adequately modify the coronary calcium before putting the stent rather than after.
Chronic total occlusion
Chronic total occlusion (CTO) PCI is associated with a higher risk of complications when compared to non-CTO PCI, particularly so for complex lesions (e.g. heavily calcified lesions). There has been convincing evidence supporting the use of IVL in CTOs with excellent success rates demonstrated by Kosantinis et al. Of the 82 CTOs treated with lithotripsy, there were only 2 cases of coronary perforation that were managed conservatively. 53 Given that CTOs are often associated with heavily calcified and balloon undilatable lesions, IVL provides a unique opportunity to manage these lesions. Use of IVL for softening the proximal CTO cap by delivering the shockwave pulses in nearby SB is also being explored.
Conclusion
Shockwave IVL is the newest modality available for the management of severely calcified coronary arteries and has shown promising results in terms of procedural success and safety. Its ability to act on deep calcium, minimal effect on normal soft vascular tissue, SB protection during bifurcation PCI, minimal complication rates and the ease of use contribute to the growing interest in this technology. At present its cost, single available length of 12 mm, bulkier crossing profile and limited number of pulses are the major factors that may limit the use of IVL. Its role in CN, bifurcation lesions and ACS is being explored with great interest. This therapy has the potential to transform the way we manage severely calcified coronary lesions.
