Abstract
Background:
Myocarditis is now one of the most fatal and morbid complications of COVID-19. Many scientists have recently concentrated on this problem.
Objectives:
This study assessed the effects of Remdesivir (RMS) and Tocilizumab (TCZ) in COVID-19 myocarditis.
Design:
Observational, cohort study.
Methods:
Patients with COVID-19 myocarditis were enrolled in the study and divided into three groups, TCZ-treated, RMS-treated, and Dexamethasone-treated patients. After 7 days of treatment, patients were reassessed for improvement.
Results:
TCZ significantly improved patients’ ejection fraction in 7 days, but it had limited efficacy. RMS improved inflammatory characteristics of the disease, but RMS-treated patients showed exacerbated cardiac function over 7 days, and the mortality rate with RMS was higher than TCZ. TCZ protects the heart by decreasing the miR-21 expression rate.
Conclusion:
Using Tocilizumab in early diagnosed COVID-19 myocarditis patients can save their cardiac function after hospitalization and decrease the mortality rate. miR-21 level determines the outcome and responsiveness of COVID-19 myocarditis to treatment.
Introduction
COVID-19 was declared a pandemic in 2020, and it has infected people almost in all countries worldwide. 1 COVID-19 Is defined with sets of clinical presentations caused by SARS-CoV-2. Most patient’s first manifestation is the respiratory symptom that can be as severe as acute respiratory distress syndrome. 2 Cardiovascular involvement of COVID-19 is crucial because it is significantly associated with a higher mortality rate in hospitalized patients.3,4 Myocarditis is now one of the most fatal and morbid complications of COVID-19 that many scientists focused on recently.5,6
Epigenetic factors affect the expression of genes, and they make patients susceptible or resistant to diseases. MicroRNAs (miRNAs) are new epigenetic factors that circulate in the blood and are secreted by almost all tissues in the body.7,8 miR-21 has been shown to increase in viral myocarditis. Decreasing the expression of miR-21 can protect myocytes in the myocarditis process.9,10
Tocilizumab (TCZ) is recently approved to be used for critically ill hospitalized COVID-19 patients. TCZ is a monoclonal antibody that antagonizes the interleukine-6 (IL-6) receptor.11,12
Remdesivir (RMS) is a viral RNA polymerase inhibitor approved by the FDA on 22 October, 2020, for treating ill COVID-19 patients. 13
We assessed the effects of these two drugs on the COVID-19 patients’ level of miR-21. The rate of COVID-19 myocarditis mortality in patients who had been treated with these drugs was assessed too.
Materials and methods
Patients
In this observational, cohort study, patients admitted to the cardiac care unit (CCU) from 1st October 2020 to 1st July 2021 in 12 hospitals participated. All methods were reviewed and approved by Zanjan University of Medical Science ethics committee (A-1-133-2). We used patients’ CCU documents for collecting clinical data (age, past medical history, triage unit electrocardiogram (EKG), troponin I (TnI), Creatine kinase-MB (CK-MB), treating drugs, C-reactive protein (hs-CRP), erythrocyte sedimentation rate (ESR), and Echocardiography). This study was approved by Zanjan University of Medical Science ethics committee. For assessing the miR-21 level, 500 µL of blood was obtained from the blood samples that CCU nurses collected for routine laboratorial tests. Informed consent was obtained from all patients to use their data. For healthy individuals control group, a questionnaire and a full description of the study were printed and given to the receptions of the study hospitals’ laboratories. After confirming the full healthy state of the individuals based on the questionnaire, a written informed consent was taken and the cost of the extra tests (not in their main prescription) was paid by the researchers. For COVID-myocarditis control group, patients who were admitted to emergency department unit with positive COVID-19 polymerase chain reaction (PCR) test and no sign of myocarditis were selected. ‘COVID-19 myocarditis’ group’s patients were the CCU admitted patients with myocarditis in hospitals which had no access to RMS and TCZ so they were not given neither of them but corticosteroids and conservative COVID treatments. After completing the data collection for COVID-19 patients, we included 20 healthy individuals as the control group, 20 mild COVID-19 patients as the COVID-control group, and 21 patients as the COVID-myocarditis control group. Control group individuals were healthy people who visited the laboratory for their routine checkup (other than COVID-19). Except for control and COVID-control patients, all other group’s patients were treated with almost the same conservative medications plus one or two shots of Dexamethasone. Conservative treatments were vitamin E, Zinc sulfate, vitamin D3, acetaminophen (IV or PO), ciprofloxacin 500 mg/daily, and doxycycline 100 mg/BID. Figure 1 shows multiple step selection of patients in details.

Hierarchical chart of participant’s enrollment.
Sample size
All eligible patients were included in the study.
Inclusion and exclusion criteria
Inclusion criteria were signed informed consent and age older than 40 years. Exclusion criteria were any chronic diseases history and any past long-term drug use history.
Myocarditis score
We used myocarditis score, consisting of chest pain, ESR >20 mm/h, C-reactive protein (hs-CRP) >3 mg/L, fever, left ventricle ejection fraction <50%, and troponin serum levels >3 ng/L to diagnose patients with myocarditis. All factors listed above were given 0 or 1 point. Patients with >4 points and diffuse T inversion in ECG were considered myocarditis. 14
RNA isolation
After extracting RNA from 200 µL of serum sample with RNase-Free DNase Set (Qiagen, Valencia, CA), 200 µL of serum sample was mixed with Buffer Reagents for Proteins Lysis (RPL); then it was mixed with Buffer Reagents for Proteins Precipitation (RPP). Isopropanol was added to the supernatant. The mixture was eluted with RNase-free water on the RNeasy UCP MinElute spin column. As an internal loading control, we used miR-942. 15
Real-time qPCR
Isolated total RNA from the previous step was reverse transcribed with SYBR® Green Quantitative RT-qPCR Kit (QR0100-1KT, Sigmaaldrich co, Germany). PCR was carried out with SYBR® Green Extract-N-Amp™ Tissue PCR Kit (XNATRG-1KT, Sigmaaldrich co, Germany). And then, we used the products to implement real-time qPCR with TB Green Premix Ex Taq II (Tli RNase H Plus) Kit (Cat No. RR820Q) and Applied BioSystem (ABI7300) (See Supplemental data for full detail). The primer used for hsa-miR-21-5p was MystiCq® microRNA qPCR Assay Primer hsa-miR-21-5p (Sigma-Aldrich-MIRAP00047) (Sequence: UAGCUUAUCAGACUGAUGUUGA), and for hsa-miR-942 was MystiCq® microRNA qPCR Assay Primer (Sigma-Aldrich-MIRAP00720-250RXN) (Sequence: UCUUCUCUGUUUUGGCCAUGUG). Melting curves were used for purity confirmation, and fluorescent signals were normalized with internal reference. Threshold cycles (CT) were set for the increasing phase of the PCR. ΔCT (ΔCT=CT (miR-21) – CT (miR-942)) method relative to miR-942 was used to show the results. The fold change of patients’ miR-21 expression in the second and eighth days was calculated with 2-ΔΔCt. ΔΔCT = ΔCT first day-ΔCT seventh day
Statistical analysis
Data were analyzed with International Business Machines Corporation (IBM corp.) (SPSS) V.21 software. Analyses of variance, repeated-measures analyses of variance, and paired T-test with Tukey post hoc test were used to calculate p-values. For graphs, we used GraphPad Software Inc. (GraphPad Prism) (version 9.0). All p < 0.05 were considered significant.
Results
Patients
A total of 2654 patients were admitted to CCU with prominent respiratory syndromes of COVID-19 and chest pain. All patients had increased serum levels of TnI and CK-MB. A total of 1567 had a positive PCR test for SARS-CoV, and 659 of them were treated with either TCZ or RMS for their COVID-19. Three hundred eighty-five patients fulfilled the myocarditis scores and were considered COVID-19 induced myocarditis. Two hundred one of three hundred eighty-five patients had a history of heart diseases, diabetes mellitus, and chronic uncontrolled hypertension before this episode of hospitalization; these patients were excluded from the study. One hundred forty-two patients were 40–60 years old, and they were enrolled in the study. Forty-one patients were treated with an 8 mg/kg IV infusion of TCZ on the second day of hospitalization, and 80 patients were treated with a 200 mg IV infusion of RMS on the second day of admission plus 100 mg IV daily infusion for four consecutive days. Last 21 patients were COVID-19 myocarditis patients who were treated with Dexamethasone and other mentioned conservative treatments (neither RMS nor TCZ had been prescribed for them). Totally 182 person were enrolled in the study. Eight patients in TCZ, twenty-two patients in RMS, and sixteen patients in COVID19-myocarditis control groups expired during hospitalization, their data are shown as TCZ expired (TCZ-E), RMS expired (RMS-E), and COVID19-myocarditis control expired groups (Figure 1). Patients’ non-laboratorial data are shown in Table 1.
Non-laboratorial information comparison between groups.

p < 0.05 control versus other.
p < 0.05 COVID-control versus TCZ, RMS, TCZ-E, and RMS-E.
RMS, Remdesivir; RMS-E, RMS expired; TCZ, Tocilizumab; TCZ-E, TCZ expired.
Laboratory and echocardiography
Change of ESR, hs-CRP, TnI, CK-MB, and ejection fraction (EF) for the control group from the first sample to the following week sample was not significant. ESR and CRP in the COVID-control group were significantly decreased by conservative therapy. ESR and hs-CRP were significantly decreased in both COVID-myocarditis groups (expired and survived). TnI and CK-MB were significantly increased in COVID-myocarditis-E patients. EF did not significantly change in COVID-myocarditis patients (expired and survived). ESR, hs-CRP, TnI, and CK-MB were significantly decreased with TCZ therapy in the TCZ group, and EF significantly increased since the second day of hospitalization. ESR, hs-CRP, TnI, CK-MB, and EF were significantly decreased by RMS therapy in 7 days. In TCZ-E patients, ESR, and CK-MB decrease was not significant, but hs-CRP were significantly lower on the eigth day of hospitalization than the first sample; EF in these patients was significantly diminished through the treatment course. TnI level in TCZ-E patients was significantly higher on day 8 compared to TCZ and RMS groups. RMS-E patients had significantly decreased ESR and hs-CRP levels.
TnI and CK-MB blood levels significantly increased on day 8. EF in this group did not significantly change in the first 8 days of the hospitalization. Post hoc analysis showed that the decrease of ESR, TnI, and CK-MB and increase of EF in the TCZ group were significantly higher than all other groups. Except for the significant decrease of hs-CRP in the RMS-E group compared to TCZ-E and COVID-myocarditis-E groups’ patients, the change of other variables was not significantly different between these three groups. First sample ESR and hs-CRP levels in the control group were significantly lower than other groups. TnI and CK-MB of the first sample were significantly lower in the control and COVID-control groups than in other groups. The first sample’s ESR levels were not significantly different between groups. The first sample’s hs-CRP level in control and COVID-control groups was not significantly different. However, it was significantly lower in these groups than in other groups. The hs-CRP levels of the first sample of TCZ-E, RMS-E, and COVID-myocarditis-E patients were not different, but they were significantly higher than those of the TCZ, RMS, and COVID-myocarditis groups, respectively. The first sample TnI in the control and COVID-control groups was not significantly different. However, it was significantly lower in these groups than in the other groups.
TnI levels in the first sample of TCZ-E, RMS, and COVID-myocarditis-E were not different. They were significantly higher than the TCZ, RMS, and COVID-myocarditis groups, respectively. However, it was significantly different between TCZ and RMS groups. The first sample’s CK-MB levels in the control and COVID-control groups were not significantly different. However, they were significantly lower in these groups than the other groups. The first sample’s CK-MB level of TCZ-E, RMS-E, and COVID-myocarditis-E patients was not significantly different. However, it was significantly higher than TCZ, RMS, and COVID-myocarditis groups, respectively; the first sample’s CK-MB difference between TCZ, RMS, and COVID-myocarditis groups was insignificant. There was no significant difference in the first calculated EF between control and COVID-control groups. However, it was significantly higher in these groups than the other groups. The first Ejection fraction percent (%EF) of TCZ-E, RMS-E, and COVID-myocarditis-E patients was not significantly different. First EF in TCZ, RMS, TCZ-E, COVID-myocarditis, and COVID-myocarditis-E was not significantly different. However, RMS-E patients’ first EF was significantly lower than that of the TCZ and RMS groups (Table 2).
ESR, hs-CRP, TnI, and EF comparison between groups.
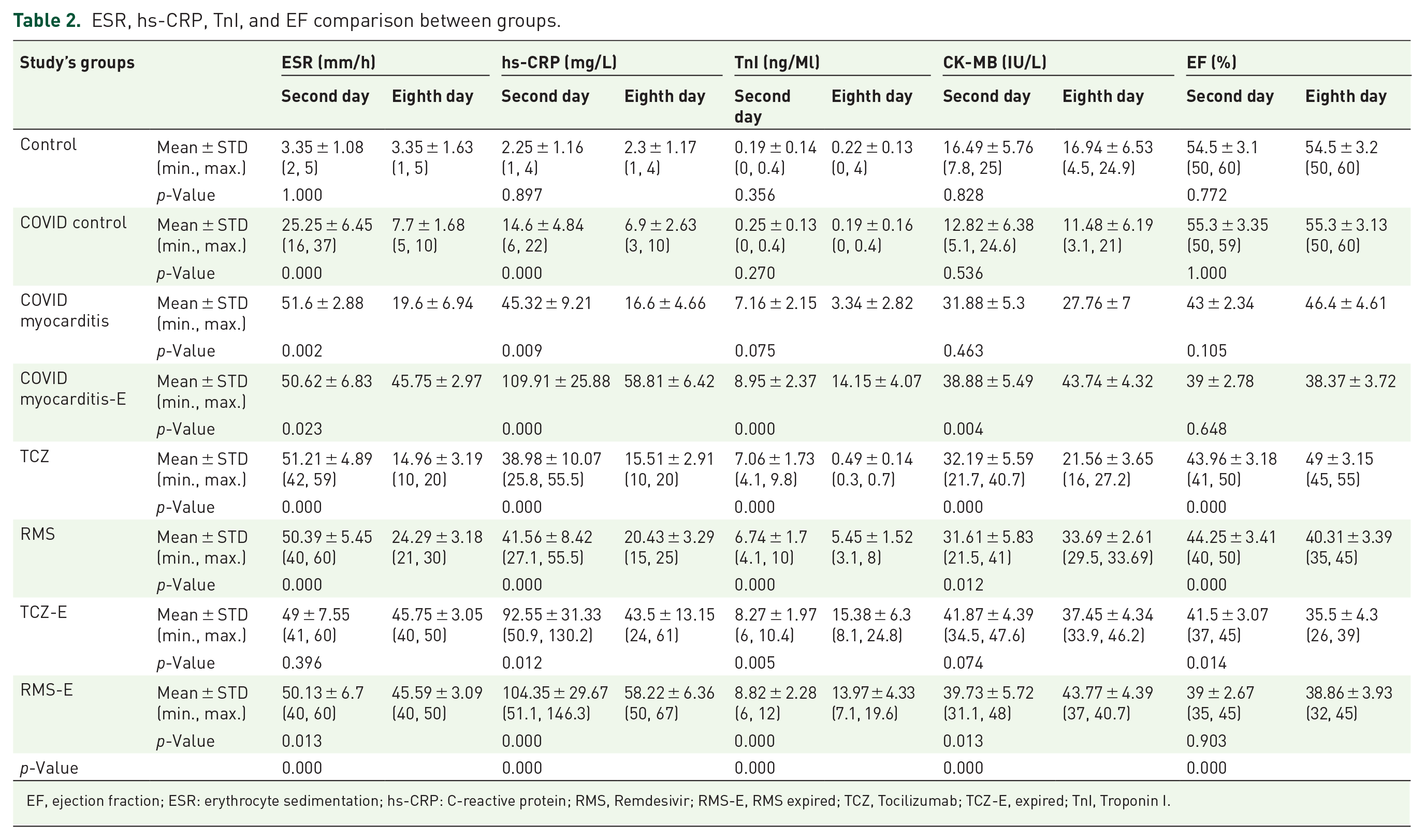
EF, ejection fraction; ESR: erythrocyte sedimentation; hs-CRP: C-reactive protein; RMS, Remdesivir; RMS-E, RMS expired; TCZ, Tocilizumab; TCZ-E, expired; TnI, Troponin I.
MiR-21 PCR results
Two samples were obtained from patients’ blood for miR-21 RT-qPCR, one on the second day of hospitalization, 1 h before TCZ or RMS infusion. The other was obtained on the eighth day of hospitalization (1 week after the first day of treatment). Six patients in the TCZ group, Twenty-three patients in RMS group, and sixteen patients in COVID-myocarditis expired during hospitalization. Second day’s miR-21 level in RMS, TCZ, TCZ-E, RMS-E, COVID-myocarditis, and COVID-myocarditis-E groups was significantly higher than the control and COVID-control groups. Second day’s miR-21 level in TCZ-E, RMS-E, and COVID-myocarditis-E was significantly higher than the TCZ, RMS, and COVID-myocarditis groups, respectively. miR-21 level on the eighth day in RMS-E, TCZ-E, TCZ, RMS, COVID-myocarditis-E, and COVID-myocarditis groups was significantly higher than the control groups. miR-21 level on the eighth day in TCZ patients was significantly lower than the RMS-E, TCZ-E, RMS, COVID-myocarditis, and COVID-myocarditis-E groups. miR-21 level on the eighth day in RMS-E, RMS, COVID-myocarditis, and COVID-myocarditis-E groups was significantly higher than the TCZ-E group. miR-21 level on the eighth day in COVID-myocarditis, and COVID-myocarditis-E groups was significantly higher than RMS-E group. miR-21 level on the eighth day in RMS-E and COVID-myocarditis-E patients was significantly higher than RMS and COVID-myocarditis groups, respectively. Figure 2 shows the comparison of miR-21 expression level between groups.

(a) Scatter-dot plot of the first sample of miR-21 expression level relative to miR-942; (b) mean + standard deviation bar box plot of the second first sample of miR-21 expression level relative to miR-942. *p < 0.05 versus control and COVID-control groups, #p < 0.05 RMS-E, COVID-myocarditis-E, and TCZ-E versus RMS, COVID-myocarditis, and TCZ, respectively, ¥p < 0.05 RMS-E and COVID-myocarditis-E versus TCZ-E. (c) Scatter-dot plot of the eighth first sample of miR-21 expression level relative to miR-942; (d) mean + standard deviation bar box plot of the eighth first sample of miR-21 expression level relative to miR-942. *p < 0.05 versus control and COVID-control groups, γp < 0.05 RMS-E, TCZ-E, RMS, COVID-myocarditis, and COVID-myocarditis-E versus TCZ, ɤp < 0.05 RMS-E, RMS, COVID-myocarditis, and COVID-myocarditis-E versus TCZ-E, ζp < 0.05 RMS-E and COVID-myocarditis-E versus other groups.
Analysis showed that the expired patients in TCZ group had significantly higher first sample miR-21 level than that of the survived TCZ-treated patients.
miR-21 expression level and laboratorial properties correlation
Table 3 shows correlation between admission miR-21 expression level and patients’ laboratorial data. Table 3 demonstrates if there is a relation between the first day’s miR-21 expression level and change of other laboratorial data of patients during the hospitalization time (without considering the treatment group). Patients with higher miR-21 expression level at admission had lesser decrease in hs-CRP, TnI, and CK-MB during hospitalization independent of the treatment type. This means patients with a higher miR-21 level at admission responded to treatment weaker than patients with a lower miR-21 expression level at their admission.
Correlation of admission miR-21 level and laboratorial properties’ changes of patients during hospitalization.
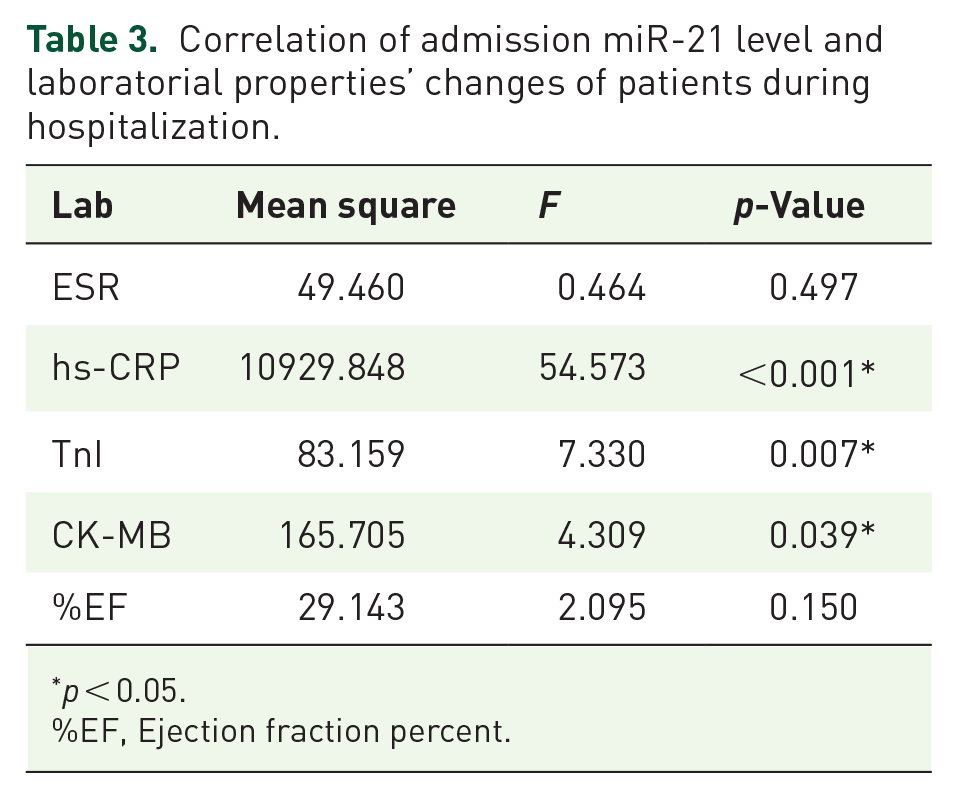
p < 0.05.
%EF, Ejection fraction percent.
Discussion
This cross-sectional study aimed to compare the efficacy of two of the most used drugs on treating COVID-19 induced myocarditis, TCZ and RMS. The other aim of this study was to determine the relation of miR-21 level and mortality rate in COVID-19 patients with myocarditis. The fundamental reason for proposing this hypothesis was that the miR-21 level increases in viral myocarditis and plays a role in its progression. 16 It also has been shown that the miR-21 level rises in critically ill COVID-19 patients. 17 This study is the first study that assessed the efficacy of two of the most used COVID-19 medications on myocarditis; this study also tried to find a link between this effect and the level of miR-21. Our study showed that gender had no potential relation to the rate of myocarditis occurrence and mortality. The first-day body temperature of patients was not different between the patients who survived myocarditis and those who expired. The pretreatment level of ESR and its change during the next 7 days showed that the first ESR level of patients with myocarditis was higher than those with mild COVID-19. The change of the ESR in the first 7 days after drug injection was significant in patients who survived in TCZ and RMS groups. 18 ESR level did not significantly decrease in patients who expired in the course of hospitalization and were injected by TCZ. It seems a higher ESR can be one of the factors associated with a higher probability of myocarditis occurrence. It showed that, nevertheless, the RMS is more effective in decreasing the ESR level. High-ESR level at the time of admission is strongly relative to COVID-19 myocarditis mortality rate during hospitalization.
TCZ and RMS showed almost the same efficacy in lowering the hs-CRP levels. Our study showed that the first sample’s hs-CRP level was not significantly different between healthy and mild COVID-19 patients. However, the patients who had developed myocarditis had higher hs-CRP on the first day of hospitalization. Expired patients either treated with TCZ or RMS had higher hs-CRP levels than those who survived. 19 Although the hs-CRP level was decreased in expired patients during 7 days of study, higher hs-CRP at admission is associated with a higher mortality rate of COVID-19 myocarditis patients.
TnI level in patients with myocarditis was higher than mild COVID-19 patients and healthy individuals. 20 Both RMS and TCZ decreased TnI levels, but the amount of decrease in patients who were treated with TCZ was significantly more than patients treated with RMS. Expired patients had a higher level of TnI in their first pretreatment sample, and TnI levels increased during the first 7 days after treatment. A Higher TnI level at admission is related to a higher mortality rate in COVID-19 myocarditis patients. 21
CK-MB level in patients with myocarditis was higher than mild COVID-19 patients and healthy participants. 22 TCZ treatment in both survived and expired patients decreased the level of CK-MB in 7 days. However, RMS could not reduce the CK-MB level in patients, and CK-MB increased in these patients in 7 days. Patients who expired in the hospital had significantly higher CK-MB levels than those who survived. TCZ showed notable efficacy on CK-MB level than RMS.
The %EF in patients with myocarditis is lower than patients and healthy individuals without myocarditis. Our study showed that TCZ significantly improved EF in COVID-19 myocarditis. However, in 7 days, the EFs of patients did not reach to normal range. On the other hand, RMS exacerbated the EF, and it had no positive effect on %EF in patients. The first %EF of myocarditis patients who expired in the hospital was not substantially lower than the that of the survived patients. TCZ also improves %EF in patients who expired in the hospitalization period. TCZ had a significant improving effect on %EF in COVID-19 myocarditis patients.
miR-21 is one of the cardiovascular microRNAs which upregulate in inflammatory heart diseases. 16 Like the previous studies, our study showed that the miR-21 level in patients with mild COVID-19 does not change significantly compared to healthy individuals. However, it excessively increases in COVID-19 myocarditis patients. 23 Targeting miR-21 can be a crucial approach to COVID-19 myocarditis patients. 24
IL-6 is an inflammatory factor in different situations like infections and cancer. One of the pathways of IL-6 in inducing inflammation is through STAT-3. 25 STAT-3 directly increase miR-21 expression rate. 26 Recent studies showed that STAT-3 is one of the main role players in cardiac hypertrophy and reduced cardiac contractility. 27 Inflammation and reduced myocardial contractility are the cornerstones of the myocarditis, in which it has been shown that miR-21 levels rise significantly. It seems that IL-6 can cause myocardial dysfunction in myocarditis status through IL-6 – STAT-3 – miR-21 pathway. Since TCZ is a strong inhibitor of IL-6 and its antagonist, higher efficacy of TCZ in controlling myocarditis patients can be attributed to blocking the IL-6 – STAT-3 – miR-21 pathway.
After 7 days of the TCZ infusion, the expression level of miR-21 decreased considerably. However, it did not decrease as much as to reach the control groups’ miR-21 expression level. Even in expired patients treated with TCZ, there was a significant decrease in miR-21 expression. RMS therapy decreased the expression of miR-21 level neither in survived nor in expired patients. the miR-21 level was significantly higher in patients who expired in hospital due to COVID-19-induced myocarditis.
miR-21 level in patients who were treated with drugs other than RMS and TCZ showed high level of miR-21. Considering the higher mortality rate of other treatments than TCZ (Survival rate of groups: TCZ = 80%, RM = 73%, COVID-myocarditis = 24%) group it can be concluded that sustained higher miR-21 level in patients with COVID-19 myocarditis can be a predictor of poor outcome. miR-21 can be considered as one of the markers of mortality probability determiner in COVID-19 myocarditis patients; it also can be a potential therapeutic target for COVID-19 myocarditis patients.
In independent analyses of the survived patients, results showed that a higher admission level of miR-21 significantly correlated to less improvement in %EF of the studied patients in TCZ groups patients who had more decrease in miR-21 level had a significantly higher increase in their %EF.
Our study had some limitations, including that we could not do this study as a randomized clinical trial with calculated sample size to match the patients in the best way. We tried to apply tight restrictions on patients’ selection to overcome this limitation. Nevertheless, conducting this study as a Randomized Clinical Trial (RCT) will help to eliminate most of heterogeneity of participants. Another limitation of our study was that we followed up the patients up to their discharge day. However, it would be better to follow them for a longer time to assess the long-term effects of these treatments on these patients. Our study had a small number of available cases for participating which was a limitation for our study.
Conclusion
In this study, our results showed that a higher level of ESR, hs-CRP, CK-MB, and TnI at admission was related to a higher mortality rate of myocarditis. Treating with TCZ showed significantly higher efficacy than RMS in COVID-19 myocarditis. It had good benefits on cardiac function by decreasing cardiac biomarkers and increasing %EF of the treated patients. The adverse effect of RMS on myocarditis could be due to its cumulative effect on miR-21 expression level, which is one of the most critical detrimental factors in myocarditis. Unlike RMS, TCZ protected the heart of COVID-19 myocarditis patients from collapsing and exacerbation by decreasing the expression of miR-21. Our study recommends evaluating ESR, hs-CRP, CK-MB, and TnI levels in moderate to severe COVID-19 patients with or without myocarditis at admission. We recommend using TCZ as soon as possible in moderate to severe COVID-19 patients with high ESR, hs-CRP, CK-MB, and TnI at admission to protect them from developing myocarditis. We also declare that using TCZ in COVID-19 patients and other routine treatments should be considered to save cardiac function. Since the TCZ decreases the miR-21 expression rate and miR-21 has been shown to play a pivotal role in all kinds of inflammatory myocarditis, it seems that using TCZ as an adjuvant medication in patients with this kind of myocarditis can be helpful. Although we did not use miR-21 level-based TCZ doses, it can be concluded that using the miR-21 level of admission day to adjust the TCZ dose in these patients can optimize drug efficacy and increase survival rates.
It is recommended that scientists do a similar study with a larger study population to determine a practical cutoff for the markers as mentioned earlier. These cutoffs would help to precisely define the best criteria for using TCZ in such patients. Designing a study that describes the proper doses of TCZ (other than 8 mg/kg that we used in this study, if it is possible) according to miR-21 level of the admission day can reveal a great approach for COVID-19 myocarditis patients.
Research in context
What are already known in this context?
It has been shown that miR-21 increases viral myocarditis.
Myocarditis is now one of the most fatal and morbid complications of COVID-19.
What is the main question?
Which one of Tocilizumab and Remdesivir drugs are better for COVID-19 myocarditis treatment and is there any relation between their effects and miRNA-21 level?
What are the new findings?
miRNA-21 increases in COVID-19 myocarditis patients.
Tocilizumab decreases miRNA-21 expression level.
Tocilizumab is more effective than Remdesivir in COVID-19 myocarditis patients.
What are the impacts on clinical practice?
COVID-19 patients diagnosed with concomitant myocarditis are the best candidates to use Tocilizumab with their routine COVID-19 medications. Tocilizumab can decrease the rate of mortality and morbidity of COVID-19 myocarditis in these patients.
Supplemental Material
sj-docx-1-tak-10.1177_17539447231182548 – Supplemental material for MiR-21 and Tocilizumab interactions improve COVID-19 myocarditis outcomes
Supplemental material, sj-docx-1-tak-10.1177_17539447231182548 for MiR-21 and Tocilizumab interactions improve COVID-19 myocarditis outcomes by Amir Hossein Heydari, Saeid Ghaffari, Zahra Khani, Sophia Heydari, Zakaria Eskandari and Mohammad Esmaeil Heidari in Therapeutic Advances in Cardiovascular Disease
Footnotes
Acknowledgements
We acknowledge CCU staffs of Zanjan Mousavi Hospital, Bahman Hospital, Tehran heart center, Tehran Rajaei heart center, Tehran Milad Hospital, Tehran Trita hospital, Tehran Erfan hospital, Tehran Kasra hospital, Tabriz Shahid Madani hospital, Tehran Najmieh hospital, Tehran baghiat-allah hospital, Tehran Army hospital, Tehran Jamaran hospital, Karaj Rajaei hospital, and Karaj Takhte Jamshid hospital. We acknowledge the staff of Cedars-Sinai Smidt Heart Institute and UCLA medical center.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
