Abstract
Background:
Management of high blood pressure (BP) typically requires adherence to medication regimes. However, it is known that the COVID-19 pandemic both interrupted access to some routine prescriptions and changed some patient health behaviours.
Aim:
This study, therefore, retrospectively investigated prescription reimbursement of cardiovascular (CVD) medicines as a proxy measure for patient adherence and access to medicines during the pandemic.
Methods:
A cohort study of all primary care patients in England prescribed CVD medicines. The exposure was to the global pandemic. Prescriptions were compared before and after the pandemic’s onset. Statistical variation was the outcome of interest.
Results:
Descriptive statistics show changes to monthly prescriptions, with wide confidence intervals indicating varying underlying practice. Analysis of variance reveals statistically significant differences for bendroflumethiazide, potassium-sparing diuretics, nicorandil, ezetimibe, ivabradine, ranolazine, colesevelam and midodrine. After the pandemic began (March–October 2020), negative parameters are observed for ACE inhibitors, beta-blockers, calcium channel blockers, statins, antiplatelet, antithrombotics, ARBs, loop diuretics, doxazosin, bendroflumethiazide, nitrates and indapamide, indicating decelerating monthly prescription items (statistically significant declines of calcium channel blockers, antithrombotic, adrenoreceptor blockers and diuretics) of CVD medicines within the general population. Many data points are not statistically significant, but fluctuations remain clinically important for the large population of patients taking these medications.
Conclusion:
A concerning decline in uptake of CVD therapies for chronic heart disease was observed. Accessible screening and treatment alongside financial relief on prescription levies are needed. A video abstract is (4 min 51 s) available: https://bit.ly/39gvEHi
Keywords
Introduction
High blood pressure (BP) is a leading risk factor for cardiovascular disease (CVD). 1 Coronary heart disease (CHD), heart failure, atrial fibrillation, aortic valvular disease, sudden cardiac death (SCD), sick sinus syndrome (SSS), left ventricular hypertrophy and abdominal aortic aneurysms are all associated with hypertension, especially in men. 2 In the United Kingdom, age-standardised CVD, CHD and stroke death rates have declined since 1979. 3 CHD prevalence has remained constant at around 3% in England and 4% in Scotland, Wales and Northern Ireland with declining CVD mortality in men and women, while hospital admissions show increased admissions for men. 3
Importance of hypertension control
The 2019 Guidelines 4 by the American College of Cardiology/American Heart Association suggest that clinicians should evaluate the social determinants of health that affect individuals to inform treatment decisions and that the target BP should generally be <130/80 mmHg. The prevention of hypertension and lowering BP helps to reduce the risk of developing heart disease. 2 There is clear evidence that aspirin, statins and pharmacological treatment of hypertension, β-blockers, and angiotensin-converting enzyme (ACE) inhibitors or angiotensin receptor blockers (ARBs) improve clinical outcomes in individuals with stable CVD.5–7 Underuse of these agents results in preventable complications and fatalities from CVD. Globally, it is estimated that 62% of cerebrovascular and 49% of ischaemic heart disease might be attributable to suboptimal BP control. 8 All of these categories of medicines appear on the essential medicines list, 9 that which every country should be able to offer its citizens in all healthcare settings. Individual-level access to these medicines can be influenced by universal health coverage, healthcare availability, medication availability, out-of-pocket expenses, complexity of medical regimens, use of acceptable formulations, patient self-efficacy and understanding, patient non-adherence (intentional and non-intentional), physician performance incentives and quality assessment, and the increasing role of nonphysician healthcare workers. These forces all apply in England where the National Health Service (NHS) provides universal care, funded through taxation.
Factors affecting primary care access, prescription refill and adherence (intentional or unintentional)
During the pandemic, critically ill COVID-19 patients have been intensively studied for CVD effects. A study observed myocardial injury, BP dysregulation, arrhythmia and variable cardiac function in severe and critical cases.10(p19) Hospitalised COVID-19 patients with clinical manifestation of acute cardiovascular events show an almost fivefold increased mortality. 11 Other early studies established that the main receptor for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) was the ACE2 receptor, which is also the main cell entry receptor for SARS-CoV-2.12,13 It was assumed that ACE2 levels correlate with a susceptibility to SARS-CoV-2 infection,14–18 indicating the potential for patients normally taking ACE inhibitors (ACEIs) and ARBs to being more susceptible to the virus. 17
Ambulatory non-COVID cardiac patients, generally based in the community, have also experienced social restrictions including lockdown. A rapid review 19 found that in such patients, physical activity decreased, and sedentary behaviour increased among all age groups during the COVID-19 lockdown, adults consumed more alcohol, dietary quality worsened and food intake increased along with weight gain. An Italian survey (n = 3533) found weight gain to be common (48.6%). A Moroccan study 20 of 100 patients identified sedentary behaviour, weight gain, increased systolic blood pressure (SBP) ⩾140 mmHg, increased caloric and salt intake, worsening heart failure symptoms, with more patients experiencing lower limb oedema, patients with valvular pathology presenting with worsening dyspnoea and an incidence of 10% of cardiovascular events after 60 days of the first lockdown.
Not much is known about rates of cardiovascular events in ambulatory patients during the COVID-19 pandemic, nor rates of CVD drug adherence during the lockdown. However, recent evidence is emerging from the United Kingdom that plausibly connects the onset and course of the pandemic to temporal changes in drug series21–24 [for gonadorelins, calcineurin inhibitors, asthma and chronic obstructive pulmonary disease (COPD) medicines, and anticoagulants], trends that are being observed globally, providing an early signal for some form of system fragility or system failure with reduced prescription drug coverage.25,26
Normal clinical practice
Patients with long-term routine chronic conditions such as hypertension and related cardiovascular conditions are on routine repeat medication prescriptions in normal clinical practice. Patients are initially diagnosed in primary care, may have a complicating event that admits them to hospital within secondary care and are discharged back to the community after medicine optimization and a degree of patient education. Within primary care settings, patients routinely request prescribed medication on a monthly basis (~28-day cycle), which are dispensed by community pharmacies. Early prescriptions or multi-month prescriptions are normally not issued due to budget impacts. All these activities are normally carried out by hand, by the patient, on written requests.
During the pandemic, these services were interrupted because face-to-face appointments were suspended and access to primary care was prioritised for urgent patients, with growing waiting lists for planned NHS treatment. The British and Irish Hypertension Society did make a public announcement that ‘All hypertensive patients should be strongly reassured that continuing their current medications is both safe and desirable’, 27 and the Chairman of Blood Pressure UK 28 advised people taking ACEIs or ARBs to ‘keep taking your blood pressure medicines as prescribed’ (6 January 2021). However, analysis of monthly data releases by NHS England highlights the huge pressures being placed on backlogs across the NHS even today, including on operations, cancer waiting lists, general practitioner (GP) referrals and Accident and Emergency (A&E) waiting times as noted by the British Medical Association. 29 This meant that patients could not get their routine prescriptions, routinely. They had to fill out online or telephone requests (with long wait queues) for their medication, 30 which quickly became chaotic 29 for patients and providers.31–33
A web-based survey in 39 countries (38 in Europe) found that in 35 (90%) countries face-to-face primary care and out-patient consultations were reduced during the COVID-19 pandemic. Electronic prescriptions were available in 36 (92%) countries. Online ordering and home delivery of prescription medication (avoiding pharmacy visits) were available in 18 (46%) and 26 (67%) countries, respectively. In 20 (51%) countries, respondents were unaware of any national guidelines regarding maintaining medication availability for non-communicable diseases, nor advice for patients on how to ensure access to medication and adherence during the pandemic. 34 In conditions such as seizures and epilepsy, where drug therapy is the main form of control, adherence to medication was poor. 35 Collectively, this builds a picture of non-adherence to pharmacotherapy that may be intentional or unintentional. This study aims to investigate prescription reimbursement data of CVD medicines as a proxy measure for rates of adherence and access to essential medicines.
Material and methods
Study design
This was a retrospective cohort study of all patients in England within primary care who were prescribed CVD medicines. The exposure was to the global pandemic. We compared prescription claims data in England before and after the pandemic’s onset. Statistical variation was the outcome of interest.
Data source
The ‘English Prescribing Dataset’ (EPD), 36 provided by the NHS Business Services Authority (NHSBSA), supplied anonymised prescription data in England covered by Open Government Licence (OGL) which is not linked to other datasets. All prescription data processed across primary care within the NHS are included in this study. The data include processing period (month, year) and aggregated total dosage quantities issued against each clinical commissioning groups (CCGs). Tablets are counted at single doses. Data from January 2019 to October 2020 were examined with March 2020 as the interrupt point: January 2019 to February 2020 (14 months before the pandemic) and March to October 2020 (8 months after its onset). The EPD does not provide demographic or individual-level data. The United Kingdom’s Central Alerting System (https://www.cas.mhra.gov.uk/SearchAlerts.aspx) was interrogated for medicine shortages.
Changes in the population
In 2019, there were 712,680 live births in the United Kingdom (731,213 in 2018) and 604,707 deaths (616,014 in 2018), 37 with a net growth of 107,973. Provisional statistics put 608,016 deaths in England and Wales in 2020. 38 The number of births for the first three quarters in England and Wales for 2020 was 464,437 (481,767 in 2019). 39 Extrapolating provisional estimates (birth 580,546, death 760,020, net 179,474) gives a net decline of approximately 180,000. While mortality may be higher for these patients, no data currently exist to quantify these claims.40,41
Outcome measures
The primary outcome was the total quantity of each medicine. These were total quantities per month of individual medicines, including branded and generics. To assess changes, a rolling continuous period from January 2019 to October 2020 was identified. Formulations included tablets, capsules, oral solutions and liquids (see Supplemental 1). A 10% sampling validation was conducted against https://openprescribing.net/. Findings are presented according to the REporting of studies Conducted using Observational Routinely-collected Data (RECORD) statement. 42
Statistical analysis
We extracted all prescription data on all items (including dressings/bandages etc.), dispensed across England within the specified time period, and examined 387,288,884 rows of data (528.8 GB) against inclusion criteria. Of these, only 169,094 rows were related to ACEIs, alpha-adrenoceptor blockers, ARBs, antiarrhythmics, antiplatelet, antithrombotics, beta-blockers, CCBs and statins, which were identified and analysed in this publication. Analysis of variance (ANOVA) test was used to analyse the differences between the ‘before’ and ‘after’ dataset means. An interrupted time-series (ITS) design43–45 at 95% confidence level was used, which provides powerful evidence of causal effects because it controls for secular trends in study outcomes. The Cochrane Resources 43 form the ‘benchmark’ methodology for this study, and others have previously used the Box–Jenkins techniques21,22,23,25,26,46–50 described here. A commonly used time-series modelling framework [autoregressive integrated moving average (ARIMA)] to analyse the monthly total quantity of prescription data from the EPD was employed. ARIMA is a flexible modelling construct, allowing lagged (auto)correlations and seasonal differences to be modelled while controlling for confounding. Such analysis measures, if a natural event like the pandemic causes abrupt changes in the level or the pre-existing trends (slope) of study outcomes and is appropriate for examining the impact of natural events at a population level. A ‘step change’ or a ‘change in trend’ was assessed before and after March 2020. Autocorrelation and the influence of seasonality were assessed by including lag terms in sensitivity analysis (see Supplemental 3). Ethical approval was not required for this database study. Patients or members of public were not involved in the design, conduct, reporting or dissemination of this research. A few models were created for sensitivity analysis with no transformation because prescription data are not random and should be stable; a 1-month autocorrelation better reflects routine clinical practice of issuing a prescription and having it generally dispensed within 1 month, hence the ARIMA model (1,0,0) (0,0,0). The model allows for correlation with the previous month’s prescription volumes.
Results
The ‘categories’ model includes ACEIs, alpha-adrenoceptor blockers, ARBs, antiarrhythmics, antiplatelet, antithrombotics, beta-blockers, CCBs and statins. Detailed constituent of each category is presented in Supplemental 1 and summarised in Table 1, below.
Average monthly descriptive statistics of total product quantities before and after the onset of the pandemic: mean, SD, and 95% LCI, UCI of the mean in millions of doses.
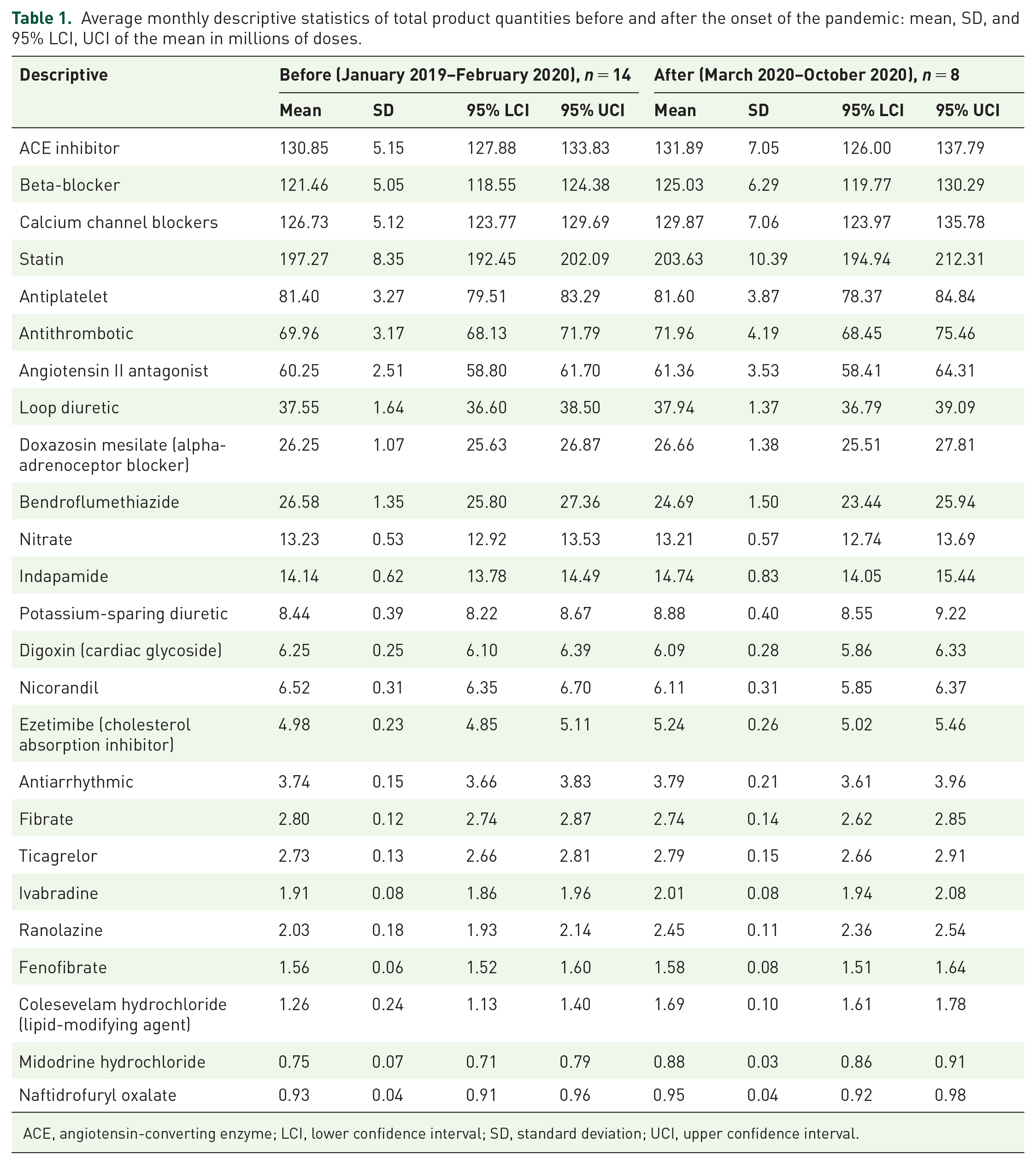
ACE, angiotensin-converting enzyme; LCI, lower confidence interval; SD, standard deviation; UCI, upper confidence interval.
Examining the descriptive statistics before and after the onset of the pandemic shows that there are changes to the monthly prescription volumes; however, these are accompanied with wide standard deviations and confidence intervals after the pandemic indicating varying underlying practice.
ANOVA reveals statistically significant differences between the before and after dataset for bendroflumethiazide (F = 9.184, p = 0.007), potassium-sparing diuretics (F = 6.443, p = .020), nicorandil (F = 9.249, p = 0.006), ezetimibe (F = 6.048, p = 0.023), ivabradine (F = 7.181, p = 0.014), ranolazine (F = 35.861, p = 0.000007), colesevelam (F = 23.433, p = 0.000099) and midodrine (F = 25.114, p = 0.000067). Caution must be exercised when comparing categories of medicines against high-volume individual drugs, for example, ACEIs versus ivabradine, but numbers are accurate.
Interrupted time series
The ITS analysis was more cautious and only analysed the first 12 high-volume items. For Syntax, see Supplemental 4. Table 2 is segmented into three datasets: before, after and at the interrupt point at self.
ARIMA model (1,0,0) (0,0,0) for ACE inhibitors, beta-blockers, calcium channel blockers, statins, antiplatelet, antithrombotics, angiotensin II antagonists, loop diuretics, doxazosin, bendroflumethiazide, nitrates and indapamide in millions of doses.

ACE, angiotensin-converting enzyme; ARIMA, autoregressive integrated moving average; LCI, lower confidence interval; UCI, upper confidence interval.
Table 2 demonstrates that before the pandemic, monthly expected growth rates of all medicines were positive except for bendroflumethiazide, with statin use accelerating fastest, for example, prescribed at 1.08 million doses month on month. This may reflect the steady growth rate of CVD within the general population in the prior 14 months of data. The accelerating growth rate of beta-blockers, calcium channel blockers, statins, antithrombotic, ARBs/ARBs, doxazosin and indapamide was statistically significant, with statistically significant deceleration in bendroflumethiazide.
After the pandemic began (period March 20–October 20), negative parameters are observed across the board representing decelerating monthly prescription items (e.g. ACEIs declined by 1.02 million items on average in the months after March 2020) with statistically significant declines of calcium channel blockers, antithrombotic, adrenoreceptor brokers such as doxazosin and diuretics such as indapamide. This reflects the decelerating use of cardiovascular medicines within the general population in the subsequent 8 months (March 2020–October 2020).
At the interrupt point, that is, March 2020, there was lower than expected issuance of ACEIs, ARBs and beta-blockers, but a larger than anticipated jump for antithrombotic, calcium channel blockers and statins. This supports the evidence that in the uncertainty of March 2020, long prescriptions were issued (e.g. 6 months of medicine were issued instead of the 1- or 2-month prescriptions that are standard practice); however, this is modest. Figure 1 illustrates the trends observed in the pre- and post-March 2020 periods.

ITS analysis using ARIMA (1,0,0) (0,0,0) for ACE inhibitor, beta-blocker, calcium channel blockers, statin, antiplatelet, antithrombotic, angiotensin-II antagonist, loop diuretic, doxazosin, bendroflumethiazide, nitrate and indapamide.
The standardised beta-regression coefficient in the 8 months after the pandemic was positive (3.805) for ACEIs, negative (−0.3594) for antithrombotics, positive (0.5374) for loop diuretics, positive (1.0394) for doxazosin, negative (−3.1874) for bendroflumethiazide, negative (−1.3564) for nitrates and negative (−0.9254) for indapamide. All the negative values above show a declining prescription volume for that category. In the 8 months after the pandemic, there was statistically significant change in linearity detected for calcium channel blockers, antithrombotics, doxazosin and indapamide, highlighting these categories of medicines as being underused compared with what would be expected. There is no significant step change. This can be visually examined in Figure 1.
Detailed total quantities are presented in Supplemental 1. Budget impact analysis has also been conducted, and details are presented in Supplemental 2.
Many data points are not statistically significant, but it should be made clear that the volumes remain clinically absolutely important for the large population of patients who take these medications.
Changes in bendroflumethiazide may be consequent to the Scandinavian study. 51
Discussion
Summary
We asked the question: Did the pandemic change the utilisation of medicines intended to treat CVD? We found yes, indeed it did. But since this study did not directly measure adherence, observed changes are a proxy to adherence rates and could indicate changing adherence over time. Results show a concerning decline in use of prescribed therapies for long-term chronic cardiovascular conditions with changes in clinical practice as a direct result of the pandemic. Given the scale and extent of the observed changes, this analysis of prescription reimbursement patterns provides an early signal of significant negative changes to medicine use by patients, which may be associated with disease progression. To put this in context, in the United Kingdom during 2020 there were 79,660 deaths from ischaemic heart disease and cerebrovascular diseases combined, compared with 69,101 from COVID-19. 52 This may also be the case in other advanced high-income Organisation for Economic Co-operation and Development (OECD) nations because of the similar patterns of prevalence observed historically.
Results show a concerning decline in use of prescribed therapies for long-term chronic cardiovascular conditions with changes in clinical practice as a direct result of the pandemic. As a result, the primary message is that undesirable advances in patients’ diseases may relate to a drop in prescription coverage. While we cannot be sure that the observed declining prescription coverage is directly the cause of mortality, we note that Wadhera et al. 58 demonstrate, in their recent study, that the incidence of patients’ diseases increased from 2019 to 2020 in the United States. They show an increase in deaths caused by ischemic heart disease and hypertensive diseases in regions of the United States during early 2020 (397,042 cardiovascular deaths from 1 January 2020 to 2 June 2020 with ratio of the relative change in deaths per 100,000 in 2020 versus 2019: 1.11, 95% confidence interval: 1.04–1.18) and lends credibility to a causal link.
Some possible explanatory factors for the results are patient factors (e.g. difficulty in accessing services, changes in health priorities 54 including lifestyle and adherence changes, possibly difficulty of paying for prescriptions) and system factors (e.g. telehealth-5855 and reduced opportunity for follow-up in-person with patients in which medications can be discussed, perhaps concern about some drugs interacting with COVID, perhaps focus on other health areas for overworked professionals during pandemic,56,57 etc.).
Patient presentations are more likely to appear as escalating or uncontrolled BP and increased number of undiagnosed and diagnosed people with hypertension, which could be detected via BP screening. This trajectory is also supported by reduced use of warfarin and direct-acting oral anticoagulants (DOACs) during the pandemic,24,58,59 which is different to the prior long-term trend and hopefully is temporary. The second- and third-order derivative outcomes because of such changes remain to be seen in the clinical data and wider epidemiological findings. Medicines management guidance in some instances, for example, anticoagulants,60,61 advised clinical changes to prescriptions that would reduce the burden of monitoring, selecting agents with relatively lower adverse effects, for example, protect the kidneys.
It must be made clear that the changes observed are not consequent to deprescribing or rationalisation of drug therapy and are likely to be subsequent to either mass abstention or lack of access to timely prescriptions. Deprescribing, or the intentional discontinuation or dose-reduction of medications, is an approach to reduce harms associated with inappropriate medication use. Some reductions could be theoretically a result of deprescribing, but what is likely happening is a passive process of disengagement rather than planned deprescribing. Evidence 62 suggests that clinicians recognise the importance of deprescribing cardiometabolic medication when appropriate, but cavate it as a medical decision that can only be made in close cooperation with the patient within a multidisciplinary approach, with appropriate monitoring making it a time- and person-intensive process, resources in short supply over the last 2 years. Informal discussions with other clinical staff support this. Arguments for or against ACEIs and ARBs have been previously raised, but the data support the deceleration of most medication taking, suggesting that patients did not stop taking the medication as a direct consequence of concerns about susceptibility of ACEIs and ARBs in severity of potential COVID-19.
Conversely, it could also be an indication that during the early phases of the pandemic, care providers were streamlining care closer to guidelines and the evidence base, in line with the anticoagulation literature indicating reduced use of warfarin and switching to DOACs during the pandemic in response to national guidelines.24,58,59 In 2011, the The Prospective Urban Rural Epidemiology (PURE) Study concluded that approaches are needed to improve the long-term use of basic, inexpensive and effective drugs, 8 and this remains the case today.
National and international programmes
High BP is a major risk factor for the development of CVD; conversely, management of high BP usually with pharmacotherapy will reduce disease burden and complications. The national CVD prevention programme 63 has been set up in 2019 to develop targeted interventions to optimise care by maximising diagnosis and treatment to minimise both individual risk factors and population risk to deliver commitments set out in the 2019 long-term plan. 64 In England, since October 2021, all adults above 40 years of age can access a health check 65 from convenient locations such as community pharmacies because they have been shown to be efficacious 66 and cost-effective. 67 Such screening in pharmacies, specifically, can be done faster and cheaper, increasing screening rates to detect CVD progression, something that should be promoted nationally and internationally given this study’s findings. International guidelines may also evolve in line with emerging evidence but need to remain harmonised to prevent the varied clinical practice.
Individual financial risks and vulnerabilities
Consideration should also be given to financial pressures that patients may be experiencing. The NHS provides subsidise medication, but a flat fee of approximately £10 is levied against each item on a prescription which can be off-putting for people in a precarious financial position perhaps heightened by the pandemic. Policy consideration should be given to suspend the NHS prescription fee levied during the pandemic. This is likely to encourage those people who have foregone therapy to become adherent again and may attempt to ‘level up’ some of the emerging health inequalities.
Public perception
Risks remain with public perceptions of the NHS and the ‘value for money’ argument, if services remain out of reach and delayed, being beyond ‘reasonably valued’, although the evidence of patient disengagement, especially for cardiovascular conditions, is currently weak and more evidence needs to be collected. There is some initial emerging evidence of disengagement from healthcare services including for mental health and cardiovascular and cerebrovascular disease68,69 and potentially an unintended move towards privatisation. There is out-of-pocket spending on health too. This stands at weekly household expenditure (£) on health in United Kingdom (financial year ending 2020), at £4.20 (Index of Multiple Deprivation 2019 Decile 1 – lowest 10%), £5.40, £5.00, £5.80, £8.10, £7.70, £9.00, £12.20, £9.20, £15.30 (Decile 10 – highest 10%), averaging £8.20 per week by household expenditure by gross income decile group. 70 This represents 2% of total expenditure for Deciles 1 and 2, and 1% of total expenditure for the rest. This represents a weekly spend of £10.00 for ages 50–64, £9.70 for ages 65–74 and £10.50 for ages 75 and over, disproportionately affecting older adults in income poverty. These numbers are likely to rise further in line with accelerating global and domestic inflation. To remain relevant, the NHS must rapidly reorient services that serve patients. Telehealth, mobile health and especially support for rural and vulnerable populations need to be done in different ways for the NHS to remain sustainable and reliably meet patient needs. Policy consideration must be given to resuming systematic efforts towards active patient assessments. Current healthcare services and staff are acknowledged to be operating at maximum capacity with risk of burnout71–73 and attrition from their professions. Hence, these policy decisions need to be more robust and go beyond simply asking staff to work longer and harder.
Strengths and limitations
There are several strengths of this study, primarily its originality and attempt to bring analytic rigour to not only understanding the likely impacts on patients of the pandemic but also providing early warning signals for the management of CVD medication use in the longer term. The results of this study are timely, rooted in real-world epidemiological evidence, adding new knowledge on the evidence base of general health in England to ground future practice and guidance. This provides evidence for actionable policy change to improve patient health and limit the negative impact of the pandemic. Limitations include not being able to characterise the causal reasons or effect size of confounders.
Future studies
In 2016, Bhatnagar et al. 3 highlight that improvements in the burden of CVD had not occurred equally between the four constituent countries of the United Kingdom or between men and women. While the present study reports data across England, it cannot comment on other nations or differences by gender. However, it generates early warning signals that Wales, Northern Ireland and Scotland populations may perform worse as historically compared with England, with men at higher risk of disease complications and hospitalisations. There is a concern that life expectancy may fall and health inequality along lines of deprivation may become entrenched; however, these are hypotheses for further investigations. So many rapid changes could have created a multi-tiered system of care with variation along geographic and deprivation lines, where some patients would have received careful attention and others could have had amendments that may not be suitable. Future studies are needed to conduct patient-level prescription analysis to investigate whether reductions are as a result of reduced initiation or maintenance (e.g. How many ‘new’ versus ‘old’ patients), rates of disease complications, gender difference and postcode analysis of deprivation.
Recommendations
Patients detected at risk of CVD via screening should not wait to see a physician or specialist and should be immediately initiated on an antihypertensive medication if the reviewing nurse or pharmacist is an independent or supplementary non-medical prescriber. To protect health, patient should be encouraged to return for a rapid review to ensure any incidental adverse reactions are detected and managed with follow-up by a specialist.
Conclusion
Results show a concerning decline in uptake of prescribed therapies for long-term chronic cardiovascular conditions with changes in clinical practice as a direct result of the pandemic. There is a concern that life expectancy may fall and health inequality along lines of deprivation may become entrenched. Consideration should be given to financial relief on prescription levies, accessible, accelerated screening and treatment. A video abstract is (4 min 51 s) of this article is available at https://bit.ly/39gvEHi
Supplemental Material
sj-docx-5-tak-10.1177_17539447221137170 – Supplemental material for Impact of the COVID-19 pandemic on cardiovascular heart disease medication use: time-series analysis of England’s prescription data during the COVID-19 pandemic (January 2019 to October 2020)
Supplemental material, sj-docx-5-tak-10.1177_17539447221137170 for Impact of the COVID-19 pandemic on cardiovascular heart disease medication use: time-series analysis of England’s prescription data during the COVID-19 pandemic (January 2019 to October 2020) by Ravina Barrett and James Hodgkinson in Therapeutic Advances in Cardiovascular Disease
Supplemental Material
sj-xls-4-tak-10.1177_17539447221137170 – Supplemental material for Impact of the COVID-19 pandemic on cardiovascular heart disease medication use: time-series analysis of England’s prescription data during the COVID-19 pandemic (January 2019 to October 2020)
Supplemental material, sj-xls-4-tak-10.1177_17539447221137170 for Impact of the COVID-19 pandemic on cardiovascular heart disease medication use: time-series analysis of England’s prescription data during the COVID-19 pandemic (January 2019 to October 2020) by Ravina Barrett and James Hodgkinson in Therapeutic Advances in Cardiovascular Disease
Supplemental Material
sj-xlsx-1-tak-10.1177_17539447221137170 – Supplemental material for Impact of the COVID-19 pandemic on cardiovascular heart disease medication use: time-series analysis of England’s prescription data during the COVID-19 pandemic (January 2019 to October 2020)
Supplemental material, sj-xlsx-1-tak-10.1177_17539447221137170 for Impact of the COVID-19 pandemic on cardiovascular heart disease medication use: time-series analysis of England’s prescription data during the COVID-19 pandemic (January 2019 to October 2020) by Ravina Barrett and James Hodgkinson in Therapeutic Advances in Cardiovascular Disease
Supplemental Material
sj-xlsx-2-tak-10.1177_17539447221137170 – Supplemental material for Impact of the COVID-19 pandemic on cardiovascular heart disease medication use: time-series analysis of England’s prescription data during the COVID-19 pandemic (January 2019 to October 2020)
Supplemental material, sj-xlsx-2-tak-10.1177_17539447221137170 for Impact of the COVID-19 pandemic on cardiovascular heart disease medication use: time-series analysis of England’s prescription data during the COVID-19 pandemic (January 2019 to October 2020) by Ravina Barrett and James Hodgkinson in Therapeutic Advances in Cardiovascular Disease
Supplemental Material
sj-xlsx-3-tak-10.1177_17539447221137170 – Supplemental material for Impact of the COVID-19 pandemic on cardiovascular heart disease medication use: time-series analysis of England’s prescription data during the COVID-19 pandemic (January 2019 to October 2020)
Supplemental material, sj-xlsx-3-tak-10.1177_17539447221137170 for Impact of the COVID-19 pandemic on cardiovascular heart disease medication use: time-series analysis of England’s prescription data during the COVID-19 pandemic (January 2019 to October 2020) by Ravina Barrett and James Hodgkinson in Therapeutic Advances in Cardiovascular Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
