Abstract
Purpose:
Baroreflex activation therapy has favorable effects in heart failure patients. We report the results of a single-center study of baroreflex activation therapy in heart failure with reduced ejection fraction including cardiopulmonary exercise testing for the first time to show the effect on exercise capacity.
Methods:
A total of 17 patients were treated with baroreflex activation therapy. Eligibility criteria were the New York Heart Association class ⩾III and ejection fraction ⩽35% on guideline-directed medical and device therapy. The New York Heart Association class, quality of life, and 6-min hall walk distance were assessed in all patients. Twelve patients underwent cardiopulmonary exercise testing before and 8.9 ± 6.4 months after initiation of baroreflex activation therapy.
Results:
The New York Heart Association class and 6-min hall walk distance improved after baroreflex activation therapy, while quality of life remained stable. Weight-adapted peak oxygen uptake increased significantly from 10.1 (8.2–12.9) ml/min/kg to 12.1 (10.4–14.6) ml/min/kg (p = 0.041). Maximal heart rate was stable. Maximal oxygen pulse increased from 9.7 (5.5–11.3) to 9.9 (7.1–12.1) ml/heartbeat (p = 0.047) in 10 patients with low maximal oxygen pulse at baseline (<16.5 ml/heartbeat). There was no significant change in maximal oxygen pulse in the whole cohort. Ventilatory efficiency remained stable.
Conclusion:
Weight-adapted peak oxygen uptake improved after baroreflex activation therapy, pointing to an enhanced exercise capacity. Ventilatory efficiency and heart rate did not change, while oxygen pulse increased in patients with low oxygen pulse at baseline, indicating an improvement in circulatory efficiency, that is, a beneficial effect on stroke volume and peripheral oxygen extraction.
Introduction
The imbalance of the autonomic nervous system – that is, an activation of the sympathetic and inhibition of the parasympathetic component – promotes the development and progression of heart failure with reduced ejection fraction (HFrEF). 1 The high sympathetic tone leads to vasoconstriction, reduced left ventricular stroke volume, adverse remodeling, and salt and water retention. 2 Reduced cardiac output leads, in turn, to further sympathetic activation, so patients with chronic HFrEF are trapped in a vicious circle. 3 β-blockers and inhibitors of the renin–angiotensin–aldosterone system interrupt this vicious circle, and thereby have beneficial effects on morbidity and mortality of patients with HFrEF.4,5
Baroreflex activation therapy (BAT) – that is, electrical stimulation of baroreceptors by a subclavicular pulse generator connected to a carotid sinus lead – causes a central inhibition of sympathetic tone, as evidenced by a reduction of muscle sympathetic nerve activity (MSNA) in patients with HFrEF. 6 Thereby, BAT may interrupt the vicious circle one step earlier than drugs do. 3 The first randomized, controlled clinical trial of BAT involving 146 patients with HFrEF has shown an acceptable safety and a statistically significant enhancement in the New York Heart Association (NYHA) class, quality-of-life score, 6-min hall walk distance (6MHW), and N-terminal pro-brain natriuretic peptide (NTproBNP). Left ventricular ejection fraction (LVEF) was numerically enhanced under BAT. Preliminary results of a larger, still ongoing trial confirmed the initial findings and pointed toward a reduction of cardiovascular events under BAT. 7
These trials did not include any data on enhanced testing of cardiopulmonary function, which may add substantial information on the mode of action of BAT and provide further objective evidence of its efficacy. We hypothesized that BAT would improve peak oxygen uptake, which is a key parameter of cardiopulmonary function. Here, we report the results of a single-center, open-label study of BAT in HFrEF, which includes cardiopulmonary function testing for the first time.
Methods
Eligibility criteria, implantation procedure, and therapy
Initial eligibility criteria for BAT were NYHA class ⩾III and ejection fraction ⩽35% on stable guideline-directed medical and device therapy. Reversible causes of heart failure like untreated valvular heart disease or acute myocarditis were exclusion criteria.
The Barostim neo™ system (CVRx Inc., Minneapolis, MN, USA) and the implantation procedure have been described in detail before.2,8 Briefly, the basic principle of BAT is to stimulate baroreceptors by a pulse generator connected to a small unipolar electrode, which is sutured onto the carotid sinus wall. The lead is tunneled subcutaneously to the pulse generator implanted in the subclavicular region, similar to a pacemaker. BAT was activated within 2 weeks after implantation. Electrical stimulation intensity was titrated over a series of follow-up visits up to the threshold of side effects. The patients’ maximum tolerable dose differed.
Endpoints and examinations
A total of 17 patients with HFrEF were treated with BAT at our center. Patients were enrolled in a multicenter registry (ClinicalTrials.Gov Identifier: NCT02880618) or a multicenter randomized trial [Health Outcomes Prospective Evaluation for Heart Failure (HOPE-4-HF), ClinicalTrials.Gov Identifier: NCT01720160]. Endpoints collected in the registry and randomized trial were NYHA class, 6MHW, LVEF, Minnesota Living with Heart Failure Questionnaire (MLHFQ) score and serum biomarkers as NTproBNP, serum creatinine, and estimated glomerular filtration rate (eGFR) at 6- and 12-month follow-up. Although patients were enrolled in these multicenter studies, all parameters included in the present analysis were not derived from the study databases, but from a retrospective evaluation of our local clinical database. The sponsor of the above-mentioned multicenter studies approved the publication of our local data.
As part of the routine heart failure investigations at our institution, which was not included in the registry or randomized trial, 12 patients at our center underwent cardiopulmonary exercise testing (CPET) before and after initiation of BAT. Moreover, metanephrines and normetanephrines were analyzed. As these parameters were not part of the registry or the randomized trial, the time-points of baseline and follow-up examinations were more variable, and data were not available for all 17 patients at all time-points.
The first activation took place on 2 September 2011. The first baseline value was measured on 10 August 2011, and the final follow-up was on 6 May 2017. The protocols of the prospective registry and randomized trial conformed to the Declaration of Helsinki and were approved by the local ethics committee and regulatory authorities. Additional local investigations were also approved by the local ethics committee (no. 15-344). Patients provided written informed consent for all investigations before enrollment.
CPET protocol
Relevant parameters of exercise capacity as well as circulatory and ventilatory efficiency were analyzed by CPET, performed with incremental workload (10–20 watts/minute) up to maximal individual tolerance. Key parameters included peak oxygen uptake (peak VO2), maximal oxygen pulse, defined as the amount of oxygen uptake per heartbeat (peak VO2/peak heart rate), ventilatory efficiency, expressed as the relationship between minute ventilation and carbon dioxide output (VE/VCO2-slope), and end-tidal CO2 partial pressure (PETCO2). General reproducibility of peak VO2 measured in our laboratory was confirmed by repeated CPET in six patients with HFrEF not treated by BAT. Peak VO2 in these six control patients was 14.7 ± 4.2 ml/min/kg at the first test and 14.5 ± 3.0 ml/min/kg at the second test (p = 0.74, interval between tests 7–24 months). The detailed protocol is included in the Supplemental Methods.
Statistics
Data were analyzed descriptively, and median [interquartile range (IQR)] or count (percentage) as well as the number of available patients (n) is presented. Differences between pre- and post-BAT treatment measurements were analyzed using the Wilcoxon signed-rank tests and test of marginal homogeneity. Resulting two-sided p values are presented and considered statistically significant if ⩽0.05. CPET parameters were retrospectively collected only once before and after the implementation of BAT. All other parameters were collected retrospectively 6 and 12 months after activation of BAT. As the time spans for each outcome variable differed slightly, the time-points are simplified in the results as first (~6 months) and second (~12 months) follow-up for better readability, and the exact time spans are provided as mean value ± standard deviation in Supplemental Table 1. Analyses were performed in SPPS Statistics 25 (IBM Corp., Armonk, NY, USA).
Owing to the retrospective design, data were not available for all patients at all time-points. Median (IQR) values presented in the article refer only to paired data sets. Because of this form of data presentation, the baseline values for each follow-up differ. Median (IQR) values of all available data from all patients at a given time-point are presented in Supplemental Table 2.
Results
Baseline characteristics
A total of 17 patients with HFrEF were treated with BAT at our center. A total of 16 (94.1%) patients were in NYHA class III before activation of BAT and one (5.9%) in NYHA class IV. All patients were on stable guideline-directed medical and device therapy before establishing BAT. The details on baseline characteristics, medication, and device treatment are presented in Table 1.
Baseline characteristics.

ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker; CRT-D, cardiac resynchronization therapy–defibrillator; DBP, diastolic blood pressure; ICD, implantable cardioverter-defibrillator; LVEF, left ventricular ejection fraction; MRA, mineralocorticoid-receptor antagonist; NYHA, New York Heart Association; SBP, systolic blood pressure.
Values are presented as median (IQR) or count (%). Age and BMI are presented as mean value ± standard deviation.
As sacubitril/valsartan was not yet available; none of the patients were treated with this drug.
CPET
Twelve of 17 (70.6%) patients underwent CPET examination before and after activation of BAT (see Table 2). Baseline CPET examination was 0.7 ± 0.6 months before, and follow-up was 8.9 ± 6.4 months after BAT activation.
Differences in CPET parameters before and after activation of BAT.

BAT, baroreflex activation therapy; BP, blood pressure; bpm, beats per minute; peak VO2, maximal oxygen consumption; PETCO2, partial pressure of end-tidal carbon dioxide; VE/VCO2, minute ventilation/carbon dioxide production.
Peak VO2 was 0.9 (0.57–1.6) l/min at baseline and 1.2 (0.8–1.5) l/min after BAT (p = 0.071). Weight-adapted peak VO2 increased significantly from 10.1 (8.2–12.9) ml/min/kg before BAT to 12.1 (10.4–14.6) ml/min/kg after BAT (p = 0.041). Body weight did not change with 92.5 (85.5–115.3) kg at baseline and 91.0 (82.8–111.8) kg at follow-up (p = 0.779).
Maximal heart rate was stable with 105 (88–114.3) bpm at baseline and 114 (88.3–120.8) bpm at follow-up (p = 0.385). Peak oxygen pulse did not change with 10.2 (5.7–13.0) ml/heartbeat at baseline and 10.3 (7.8–15.8) ml/heartbeat at follow-up in the whole cohort (p = 0.084). When excluding two patients with a high maximal oxygen pulse (>16.5 ml/heartbeat) already at baseline, the remaining cohort showed a significant improvement of peak oxygen pulse from 9.7 (5.5–11.3) ml/heartbeat to 9.9 (7.1–12.1) ml/heartbeat (n = 10, p = 0.047; see Figure 1); mean values increased from 8.7 to 10.5 ml/heartbeat. Peak circulatory power increased numerically from 1268.8 (837.5–1638.7) mmHg × mlO2 × min−1 × kg−1 to 1512 (1207.9–1952.1) mmHg × mlO2 × min−1 × kg−1 (p = 0.214, n = 9). There was no significant difference in maximal workload and maximal blood pressure under treatment with BAT.

CPET data. Baseline and follow-up for each individual patient as well as median are presented. Nine patients achieved an improvement in both weight-adapted peak oxygen uptake and maximal oxygen pulse under BAT. Three patients showed a deterioration of the above-mentioned parameters at the follow-up examination. Weight-adapted peak oxygen uptake increased significantly from 10.1 (8.2–12.9) ml/min/kg before BAT to 12.1 (10.4–14.6) ml/min/kg under BAT [p = 0.041, (a) individual data (left) and box plots (right)]. Peak oxygen pulse did not change significantly in the whole cohort. When excluding two patients with a high maximal oxygen pulse (>16.5 ml/heartbeat) already at baseline, the remaining collective showed a significant improvement of peak oxygen pulse from 9.7 (5.5–11.3) ml/heartbeat to 9.9 (7.1–12.1) ml/heartbeat [p = 0.047, (b)]. Mean values (marked with ×) increased from 8.7 to 10.5 ml/heartbeat.
VE/VCO2-slope remained stable with 37.5 (28.9–52.9) before and 37.8 (35–44.5) after treatment with BAT (p = 0.583). PETCO2 was stable with 33.6 (28.6–36.3) mmHg before BAT and 32.2 (30.3–35.9) mmHg after BAT (p = 0.937).
We performed another analysis at a submaximal workload of 15 watts as data were available for each patient at this workload. Weight-adapted VO2 showed an increase of borderline significance at submaximal workload from 5.5 (4.5–6.2) ml/min/kg at baseline to 6.4 (5.5–7.1) ml/min/kg at follow-up (p = 0.050; see Supplemental Table 3). No significant changes were found for the other parameters at submaximal workload.
Effect of BAT on symptoms, 6MHW, LVEF, and laboratory values
At first follow-up, seven patients had improved from NYHA class III to II and one patient from NYHA class IV to III. Thus, at first follow-up, there was a significant improvement with seven patients being in NYHA class II (43.8%) and nine in class III (56.3%; n = 16, p = 0.005). At second follow-up, four patients were in class II (30.8%) and nine patients in class III (69.2%; n = 13, p = 0.052 versus baseline).
Further efficacy endpoints and laboratory values are presented in detail in Table 3 (6 months follow-up) and Table 4 (12 months follow-up). There was a significant improvement of 6MHW from 355 (286.8–412.5) m at baseline to 395 (350.3–452.5) m at second follow-up (n = 6, p = 0.046). No effect on quality of life assessed by MLHFQ score was noticed. LVEF was increased from 29.5 (19.5–30)% at baseline to 30.5 (27.8–35.5)% at second follow-up (n = 8, p = 0.025).
Efficacy endpoints and laboratory values at 6-month follow-up after BAT activation, only paired values.
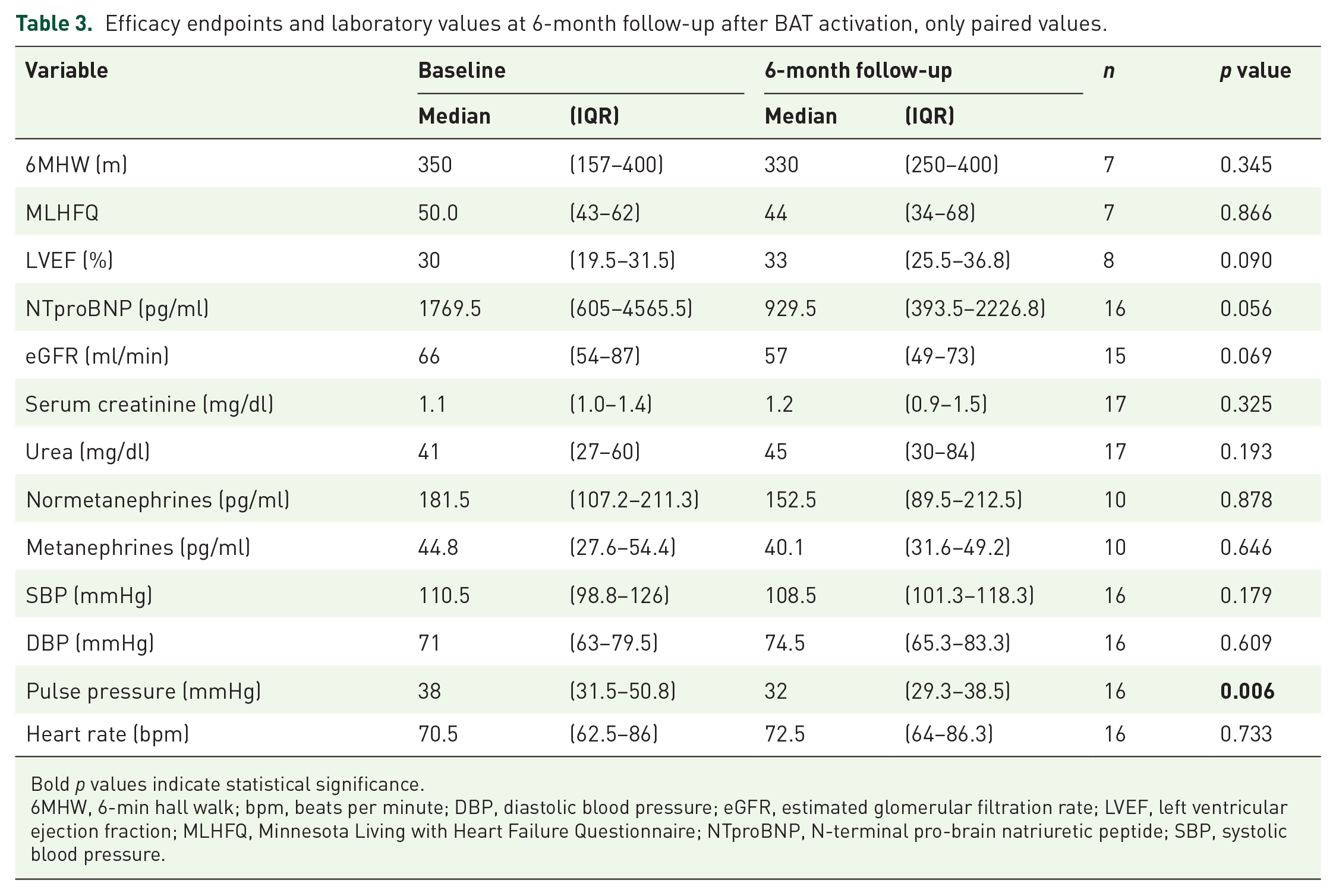
Bold p values indicate statistical significance.
6MHW, 6-min hall walk; bpm, beats per minute; DBP, diastolic blood pressure; eGFR, estimated glomerular filtration rate; LVEF, left ventricular ejection fraction; MLHFQ, Minnesota Living with Heart Failure Questionnaire; NTproBNP, N-terminal pro-brain natriuretic peptide; SBP, systolic blood pressure.
Efficacy endpoints and laboratory values at 12-month follow-up after BAT activation, only paired values.
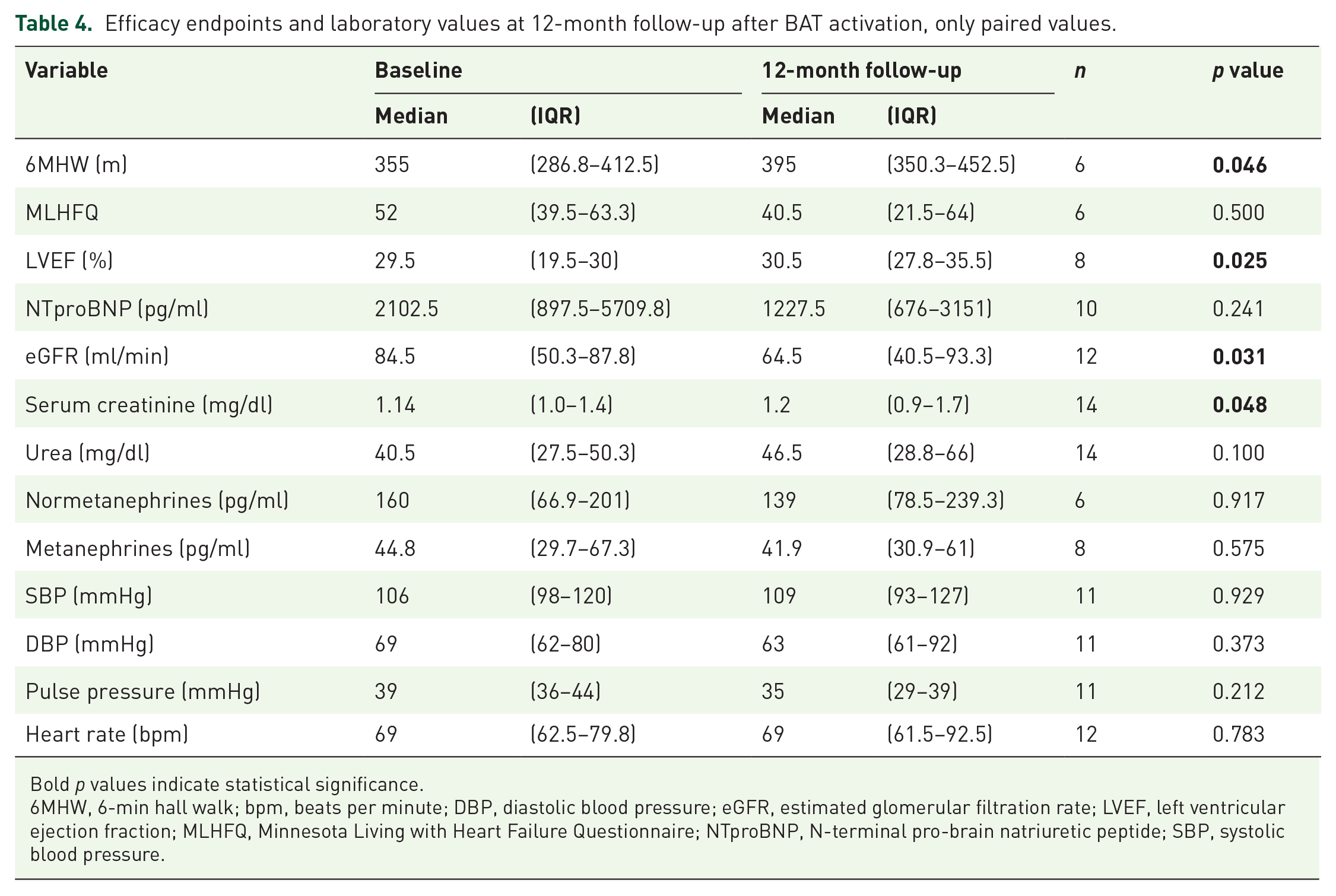
Bold p values indicate statistical significance.
6MHW, 6-min hall walk; bpm, beats per minute; DBP, diastolic blood pressure; eGFR, estimated glomerular filtration rate; LVEF, left ventricular ejection fraction; MLHFQ, Minnesota Living with Heart Failure Questionnaire; NTproBNP, N-terminal pro-brain natriuretic peptide; SBP, systolic blood pressure.
There was no significant change in NTproBNP. eGFR decreased significantly from 84.5 (50.3–87.8) ml/min at baseline to 64.5 (40.5–93.3) ml/min at second follow-up (n = 12, p = 0.031), with a corresponding increase in serum creatinine. Normetanephrine and metanephrine levels were stable.
Safety
Systolic and diastolic blood pressure as well as heart rate remained stable (see Tables 3 and 4). In seven of 17 patients, follow-up was limited by clinical events. One BAT device had to be explanted because of a local infection after 9 months. Three patients died 3, 15, and 16 months after BAT had been established; there was no association to BAT. One patient demanded deactivation of BAT 26 months after implantation, because of pain in the left arm, which was probably not related to BAT, as far as our experience goes. Two patients underwent heart transplantation 6 months and 3 years after BAT.
CPET proved to be safe. All patients tolerated the procedure well, and no adverse events occurred.
Discussion
In this single-center, open-label study on HFrEF patients treated by BAT, we present first data on cardiopulmonary function assessed by CPET, which point toward an improved exercise capacity and circulatory efficiency.
The most important novel finding of this study was that weight-adapted peak VO2 was improved significantly in HFrEF patients under BAT, reflecting the enhanced exercise capacity and heart failure symptoms shown in the present and previous trials. Weight-adapted peak VO2 is a prognostic indicator in HFrEF.9,10 Keteyian et al. 11 found that a peak VO2 of 9.2 ml/min/kg was associated with 10% 1 year all-cause mortality in patients with HFrEF, whereas a peak VO2 of 16.9 ml/min/kg carried only a 3% risk. In this study, BAT improved peak VO2 from ~10 to ~12 ml/min/kg in severely compromised patients. According to the data of Keteyian et al., 11 this may potentially translate into a reduction in 1-year all-cause mortality from ~13% to ~9% for men and from ~7% to ~3% for women.
Patients with HFrEF have an autonomic disbalance with high sympathetic tone 12 and reduced baroreflex sensitivity,13,14 which poses a high risk of adverse events and makes the autonomic system a promising therapeutic target in HFrEF. 12 Gronda et al. 6 have demonstrated by recordings of MSNA that sympathetic nerve traffic decreases under BAT. While MSNA decreases in healthy people during exercise, it increases in patients with HFrEF. 15 An inverse relationship was observed between MSNA and peak VO2 in patients with HFrEF. 16 Exercise training was able to reduce MSNA at rest and under mild exercise and to improve peak VO2. 15 There may be a causal link between MSNA reduction and improved exercise performance and peak VO2, as reductions in muscle blood flow due to high sympathetic tone may constrain exercise performance. 15 This may explain the improved peak VO2 observed here under BAT.
Resetting of peripheral baroreceptors as well as a central resetting of baroreceptor input has been described. 17 Electrical stimulation of baroreceptors would not be expected to have a sustained benefit if resetting was complete. Thus, the sustained reduction in MSNA reported by Gronda et al. up to 6 months after initiation of BAT and the sustained benefit on peak VO2 and other exercise parameters reported by the present and previous studies2,6,7,18 argue against a complete resetting at the level of the baroreceptors and the central nervous system. On the other side, resetting may at least partly explain the variable response of patients to BAT.
Oxygen pulse rose numerically in the whole cohort, but this did not reach statistical significance. Two patients had a high oxygen pulse (>16.5 ml/heartbeat) already at baseline, which may have made a further increase difficult to achieve. In the 10 patients with low oxygen pulse at baseline, a significant improvement was observed. As heart rate was not increased but even numerically decreased in this study, the improvement of oxygen uptake and oxygen pulse indicates an improvement in circulatory efficiency under BAT, that is, a beneficial effect on stroke volume and peripheral oxygen extraction, based on the Fick Equation (VO2 = heart rate × stroke volume × arteriovenous oxygen content difference).9,19
BAT had a neutral effect on other CPET parameters. VE/VCO2-slope stayed stable on a high level, so ventilatory efficiency was not influenced by BAT. High VE/VCO2-slope values indicate a higher risk of hospitalization and death. 20 PETCO2, which was stable under BAT on a low level <33 mmHg, is also associated with higher mortality. 21 These observations emphasize the severity of heart failure in our cohort.
NTproBNP dropped numerically, but this did not reach statistical significance, which may be explained by the low patient number. In the Baroreflex activation therapy for heart failure (BEAT-HF) trial, a significant drop of NTproBNP was only found in patients with initial NTproBNP <1600 pg/ml. In this population, which was defined as intended use population during the course of the study, a significant 21% decrease of NTproBNP was observed after 6 months. 7 Taking into account only patients with a baseline NTproBNP of <1600 pg/ml, BAT resulted in a 41% decrease of NTproBNP after 6 months in this study, but this did also not reach statistical significance [602 (210–992) pg/ml at baseline to 354 (226–897) pg/ml at follow-up; n = 7, p = 0.236]. A post hoc analysis of the BAT-HF trial investigated the response to BAT depending on sex. While baseline values of NTproBNP did not differ in women and men, only women showed a significant decrease in NTproBNP under BAT. 22 Only 17.6% of our cohort were female, preventing a subgroup analysis.
As norepinephrine and epinephrine correlate with sympathetic activity,23,24 the concentration of their metabolites normetanephrines and metanephrines is considered as markers of sympathetic activity. These markers were stable after BAT, which is in contrast to the decrease of norepinephrine observed in dogs with pacing-induced heart failure treated with BAT 25 and the reduction in MSNA reported by Gronda et al. 6 It is probable that the high standard deviation of lab values and low patient number of the present trial impaired the validity of this analysis.
Limitations
Because of the low number of patients enrolled and the retrospective evaluation of the database, the findings of this trial must be interpreted very carefully and must be regarded as hypothesis generating. This major limitation of this study is related to the novelty of the treatment and the low number of patients treated with BAT worldwide.
Another important limitation is that patients and investigators were unblinded, and there was no control group, which could have led to an overestimation of the observed effects. On the other side, our findings on CPET are plausible and in line with previous observations from other HFrEF trials.11,26–29
A total of 23.5% of patients enrolled were treated with cardiac resynchronization therapy (CRT) before BAT. Studies have shown that CRT has a positive effect on the autonomic imbalance, which may lead to a better response to BAT in patients without CRT. 30 This may have diminished effects of BAT observed in this study.
Patients were not treated with an angiotensin receptor–neprilysin inhibitor (ARNI) or sodium-glucose co-transporter-2 (SGLT2) inhibitors, which were not available at the time of data collection. It is speculative and should be the scope of future trials on BAT (and other established device therapies), whether device-related beneficial effects are preserved in patients with current state-of-the-art medical therapy.
Conclusion
Weight-adapted peak oxygen uptake improved after BAT, pointing to an enhanced exercise capacity. Ventilatory efficiency and heart rate did not change, while oxygen pulse increased in patients with low oxygen pulse at baseline, indicating an improvement in circulatory efficiency, that is, a beneficial effect on stroke volume and peripheral oxygen extraction.
Supplemental Material
sj-docx-1-tak-10.1177_17539447221131203 – Supplemental material for Cardiopulmonary function during exercise in heart failure with reduced ejection fraction following baroreflex activation therapy
Supplemental material, sj-docx-1-tak-10.1177_17539447221131203 for Cardiopulmonary function during exercise in heart failure with reduced ejection fraction following baroreflex activation therapy by Pia I. Nottebohm, Daniel Dumitrescu, Stefanie Hamacher, Christopher Hohmann, Navid Madershahian, Stephan Baldus, Hannes Reuter and Marcel Halbach in Therapeutic Advances in Cardiovascular Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
