Abstract
Introduction:
Hypertrophic cardiomyopathy (HCM) patients with left ventricular (LV) mid-cavity obstruction (LVMCO) often experience severe drug-refractory symptoms thought to be related to intraventricular obstruction. We tested whether ventricular pacing, guided by invasive haemodynamic assessment, reduced LVMCO and improved refractory symptoms.
Methods:
Between December 2008 and December 2017, 16 HCM patients with severe refractory symptoms and LVMCO underwent device implantation with haemodynamic pacing study to assess the effect on invasively defined LVMCO gradients. The effect on the gradient of atrioventricular (AV) synchronous pacing from sites including right ventricular (RV) apex and middle cardiac vein (MCV) was retrospectively assessed.
Results:
Invasive haemodynamic data were available in 14 of 16 patients. Mean pre-treatment intracavitary gradient was 77 ± 22 mmHg (in sinus rhythm) versus 21 ± 21 mmHg during pacing from optimal ventricular site (95% CI: −70.86 to −40.57, p < 0.0001). Optimal pacing site was distal MCV in 12/16 (86%), RV apex in 1/16 and via epicardial LV lead in 1/16. Pre-pacing Doppler-derived gradients were significantly higher than at follow-up (47 ± 15 versus 24 ± 16 mmHg, 95% CI: −37.19 to −13.73, p < 0.001). Median baseline NYHA class was 3, which had improved by ⩾1 NYHA class in 13 of 16 patients at 1-year post-procedure (p < 0.001). The mean follow-up duration was 4.6 ± 2.7 years with the following outcomes: 8/16 (50%) had continued symptomatic improvement, 4/16 had symptomatic decline and 4/16 died. Contributors to symptomatic decline included chronic atrial fibrillation (AF) (n = 5), phrenic nerve stimulation (n = 3) and ventricular ectopy (n = 1).
Conclusion:
In drug-refractory symptomatic LVMCO, distal ventricular pacing can reduce intracavitary obstruction and may provide long-term symptomatic relief in patients with limited treatment options. A haemodynamic pacing study is an effective strategy for identifying optimal pacing site and configuration.
Introduction
Hypertrophic cardiomyopathy (HCM) is the most common heritable cardiac disease affecting 1/500 people. 1 Many patients remain asymptomatic, while others complain of fatigue, dyspnoea, chest pain, palpitations and syncope. 2 The main causes of such symptoms are thought to be related to obstruction of blood flow within the left ventricle (LV), diastolic dysfunction, microvascular ischaemia, and abnormal heart rhythms such as atrial fibrillation (AF) or ventricular tachycardia (VT). 3
Partial or complete cavity obliteration at the level of the mid-cavity can result in LV mid-cavity obstruction (LVMCO) and occurs in only up to 10% of HCM patients. Two discrete chambers are formed during LV contraction, with a pool of blood trapped at the apex and abnormally elevated intracavitary systolic pressures. 4 The resulting increased wall stress may be associated with both increased metabolic demands 5 and reduced regional myocardial perfusion and ischaemia. 6 Patients with LVMCO may develop large apical aneurysms and have associated life-threatening complications such as thrombo-embolic stroke 7 and monomorphic VT. 8 LVMCO is also associated with significant morbidity. In one study, approximately 50% of patients reported severe, medically refractory symptoms such as chest pain, breathlessness, dizziness, and poor exercise capacity. 9
Medical therapy is the first-line treatment in HCM, but different invasive therapies exist for those suitable and with symptoms refractory to pharmacological therapy. 2 In left ventricular outflow tract obstruction (LVOTO), invasive therapy includes percutaneous alcohol septal ablation (ASA) or surgical myectomy.
There is limited evidence supporting the routine use of either ASA or surgical myectomy in the management of symptomatic LVMCO. The insertion of an implantable cardioverter defibrillator (ICD) for primary or secondary prevention of sudden cardiac death (SCD) is considered for these patients who fulfil criteria. 2 Dual-chamber pacing from the right ventricular (RV) apex has been shown to reduce LVMCO gradients and improve reported New York Heart Association (NYHA) class in 14 patients (5 of whom had concomitant LVOTO). 10 The authors determined that correct placement site of the ventricular lead is important for the success of pacing therapy, with maximum results obtained when the ventricular lead was positioned as distal as possible in the RV apex. 10 Pacing from the LV via a surgically implanted epicardial lead has reduced intracavity gradients and improved symptoms in a small number of patients with LVMCO, with the justification to proceed with limited thoracotomy provided by testing LV versus RV endocardial pacing during haemodynamic pacing study. 11
As there is no standard approach for pacing in this group of patients, we sought to determine optimal ventricular pacing location for gradient reduction and symptomatic improvement, with lead implantation guided by invasive haemodynamic assessment, in patients with primary device indications whose optimal medical therapy had failed.
Methods
Study design and overview
This was a retrospective, single-centre observational study evaluating the feasibility of distal ventricular pacing for relief of LVMCO gradients and improvement in symptoms via clinically reported NYHA class. This followed standard clinical practice at our centre, which includes an assessment of pacing for severely symptomatic patients with LVMCO on optimal medical therapy.
Study population
Between December 2008 and December 2017, 16 HCM patients with severe symptoms (⩾NYHA class II–III) refractory to optimised medical therapy (calcium channel blockers, beta blockers or disopyramide) and isolated LVMCO were listed for device implantation. Sample size was determined by inclusion of all eligible consecutive patients undergoing the procedure, limiting selection bias. Baseline characteristics are shown in Table 1. European Society of Cardiology guidelines on SCD risk were implemented at our centre approximately half-way through this consecutive series of patients, in accordance with publication timing. 12 Thirteen patients had indications for primary prevention of SCD based on one or more established risk factors, plus the risk modifiers of the presence of myocardial late gadolinium enhancement (LGE) and apical aneurysm. 3 One patient had device indications due to bradycardia secondary to medical therapy, and in the remaining two, the sole indication was for the relief of obstructive physiology and associated symptoms. All patients underwent thorough clinical assessment during routine follow-up in the Inherited Cardiovascular Conditions Clinic. This included symptom and exercise tolerance assessment, 12-lead electrocardiography, a 24-h Holter monitor, transthoracic echocardiography (TTE), cardiovascular magnetic resonance imaging (CMR) and work-up for implantable device with invasive haemodynamic assessment in a specialist combined heart muscle electrophysiology clinic.
Baseline characteristics.
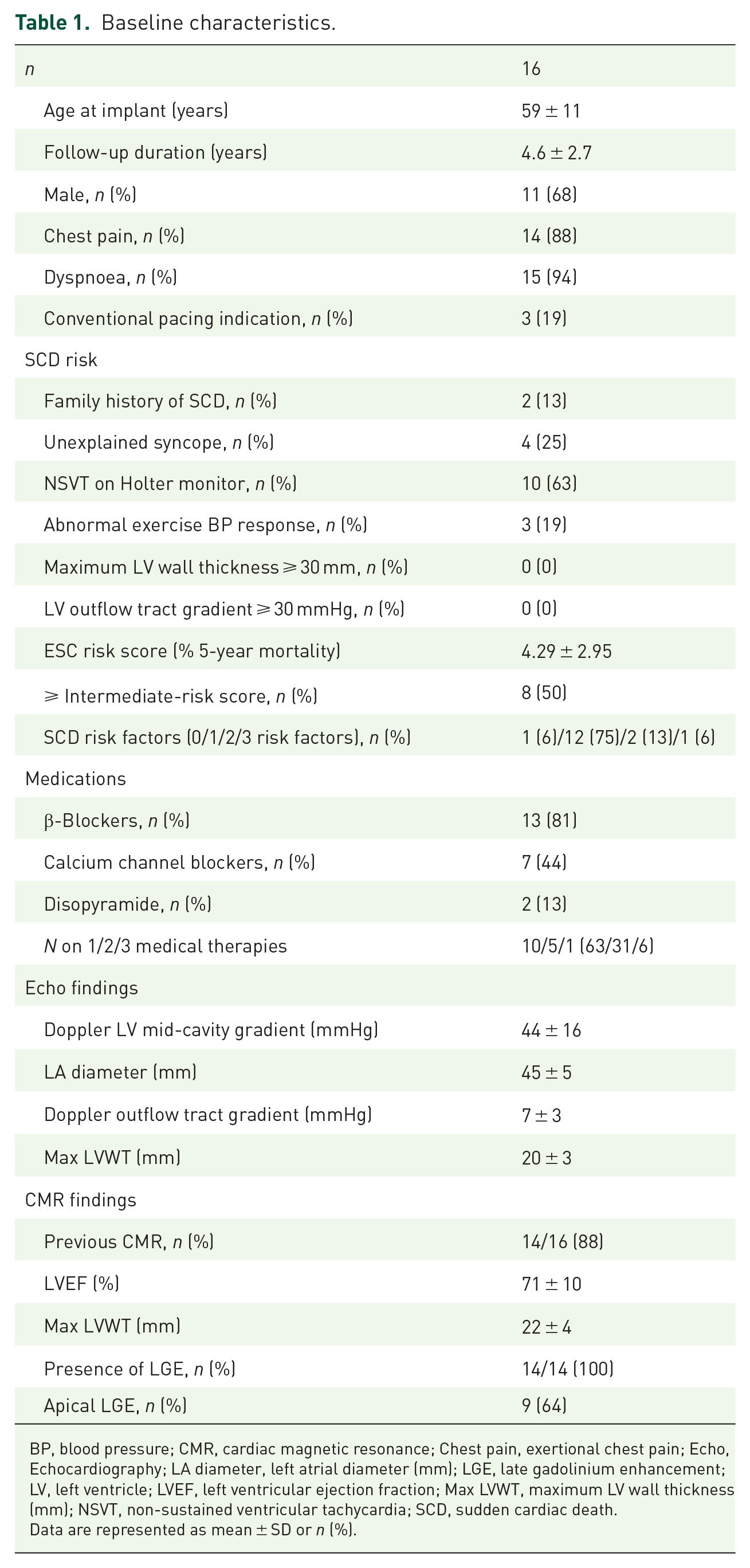
BP, blood pressure; CMR, cardiac magnetic resonance; Chest pain, exertional chest pain; Echo, Echocardiography; LA diameter, left atrial diameter (mm); LGE, late gadolinium enhancement; LV, left ventricle; LVEF, left ventricular ejection fraction; Max LVWT, maximum LV wall thickness (mm); NSVT, non-sustained ventricular tachycardia; SCD, sudden cardiac death.
Data are represented as mean ± SD or n (%).
Implant procedure
Thirteen ICD and three pacemaker implant procedures were performed with either conscious sedation or general anaesthesia, depending on standard practice at the time. Vascular access was obtained from the left subclavian vein in 15 (94%) of 16 cases with one axillary vein access for device lead implantation. Access was made via right femoral artery (RFA) with a 7-Fr (Fr) side-arm sheath for LV haemodynamic assessment. Active leads were first positioned in the right atrium and RV apex. The coronary sinus was engaged with a Medtronic Attain deflectable guiding catheter (Medtronic, USA) using X-ray contrast medium and a Terumo guide wire (Terumo Europe). At this point, 5000 IU of heparin was administered intravenously and a 6-Fr end-hole pigtail catheter (Cordis, USA) was advanced from the RFA into the LV over a 150-cm, 0.035-inch (0.89 mm) J-tipped guide wire. The end-hole pigtail was advanced into the LV apical aneurysm and simultaneous pressures recorded from here and the side arm of the 7-Fr femoral arterial sheath. An activated clotting time of 200–250 s was maintained for as long as a catheter or wire was in the LV apex. The peak-to-peak difference between the two pressures was taken as the magnitude of the LVMCO. If a spontaneous resting gradient was absent, it was provoked to steady state with an isoprenaline infusion. 13
Balloon occlusion angiography of the LV coronary venous system was performed in orthogonal projections with simultaneous contrast injection through the pigtail catheter opacifying the LV aneurysm. Particular attention was given to courses of the venous tributaries relative to the LV aneurysm. Atrio-ventricular (AV)-synchronous pacing with short AV delay was performed producing a clear change in QRS morphology and duration, confirming obligatory ventricular pacing. LV pacing was achieved with a standard, passive coronary venous pacing lead. Preservation of cardiac output during pacing was inferred by stable femoral artery systolic pressure and, where intra-operative transoesophageal echocardiography was available, by avoidance of mitral inflow truncation. Optimal pacing setting was chosen after stepwise assessment of the effect on obstructive gradient, clear change in surface electrocardiogram (ECG) QRS morphology/duration, preservation of cardiac output and satisfactory pacing parameters.
Patient follow-up
Patients’ functional and symptomatic status was determined by retrospective review of clinic notes at two time periods: 1-year post-procedure and maximal available follow-up. The secondary outcome of NYHA class was the most consistently reported symptom parameter. Echocardiography reports and serial device follow-up reports were reviewed for key parameters and evidence of cardiac arrhythmia and subsequent therapies.
Statistical analysis
Complete case analysis was performed, with continuous variables presented as mean ± SD and categorical variables as percentages. Within-group comparisons were performed using paired t-test. Wilcoxon signed rank test was used to assess categorical data. P values are two-sided with a value of <0.05 considered to indicate statistical significance. Statistical analyses were performed using GraphPad Prism version 8.0 (GraphPad Software, San Diego, CA, USA).
Results
Study patients
The study population consisted of 16 patients on maximal medical therapy with LVMCO (68% male) who underwent cardiac device implantation guided by haemodynamic testing (Table 1) between December 2008 and December 2017. Mean age at implant was 59 ± 11 years. Of the 16 patients, 13 (81%) were referred for primary (12/13, 92%) or secondary (1/13, 8%) prevention ICD. Of the 16 patients, 14 (88%) and 15 (94%) reported chest pain and dyspnoea, respectively, prior to device implantation. One-year survival was 100%, and after a mean follow-up duration of 4.6 ± 2.7 years, survival was 75%.
Acute effects of ventricular pacing on left ventricular haemodynamics
Invasive haemodynamic data were available in 14 of 16 patients (Table 2). One patient’s implant data were incomplete due to complications and are described later. Of those 14, 10 patients had resting obstruction documented by simultaneous LV apical and arterial pressure traces, with 4 patients requiring gradient provocation via isoprenaline infusion (mean dose, 3 µg/min). The mean pre-treatment invasive gradient was 77 ± 22 mmHg during sinus rhythm, versus 21 ± 21 mmHg during pacing from the optimal ventricular site [95% confidence interval (CI): −70.86 to −40.57, p < 0.0001)] (Figure 1). In 12 (86%) of 14 patients, this was from the distal MCV. In two patients without invasive data documented, the distal MCV was also used for LV lead implantation with gradients assessed using post-procedure echocardiography. In 1 of 14 patients, extensive apical LV fibrosis precluded distal LV lead placement via MCV, and therefore, the RV apex was chosen as the optimal pacing site. In 1 of 14 patients, it proved impossible to deliver a lead with stable or satisfactory pacing parameters to the MCV and RV apical pacing had no effect on the LVMCO. This patient had a dual-chamber ICD implanted initially followed by a surgical, epicardial LV apical lead, and the effect on the LVMCO was assessed by peri-operative echocardiography.
Haemodynamic pacing study and follow-up.
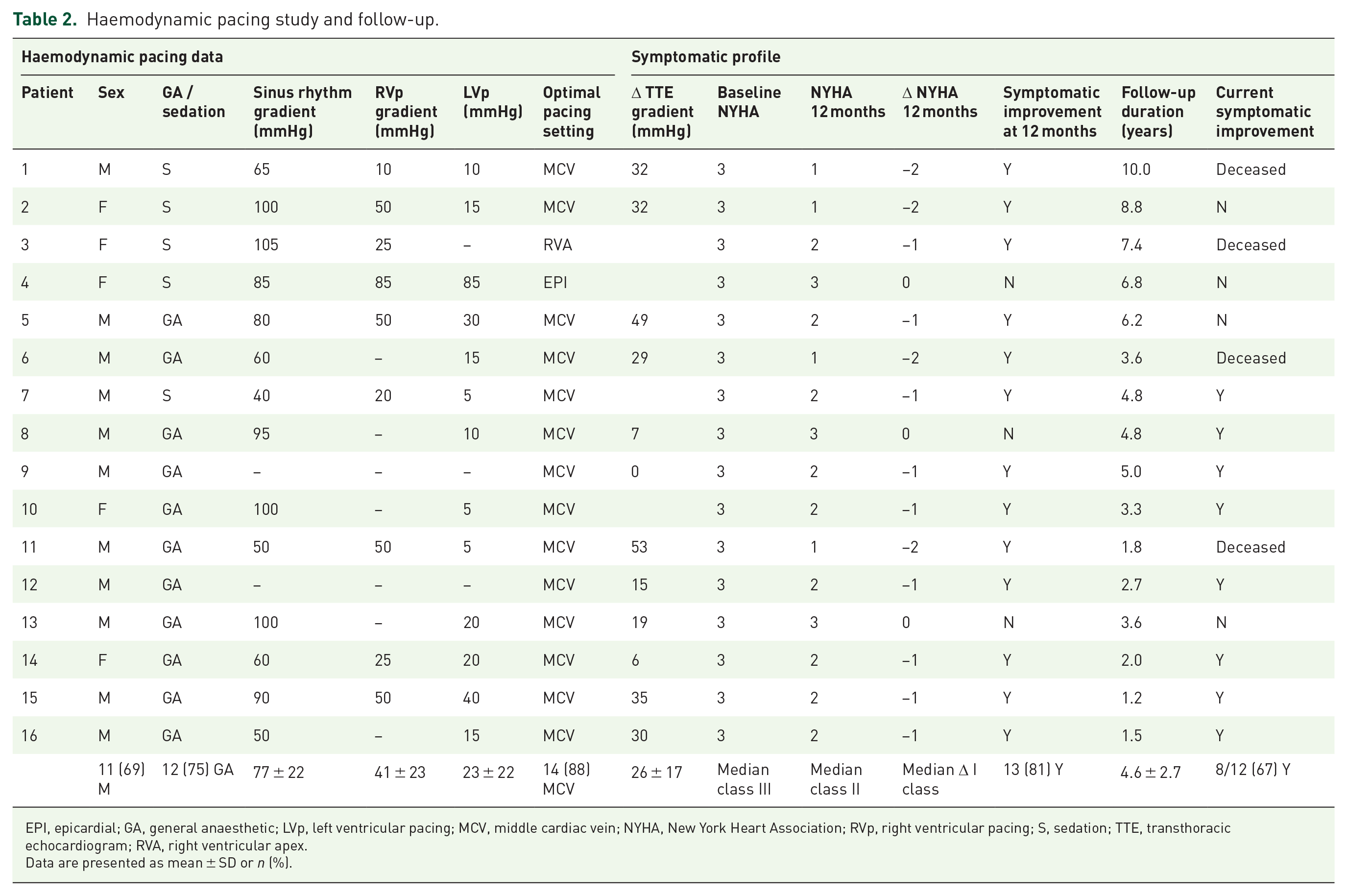
EPI, epicardial; GA, general anaesthetic; LVp, left ventricular pacing; MCV, middle cardiac vein; NYHA, New York Heart Association; RVp, right ventricular pacing; S, sedation; TTE, transthoracic echocardiogram; RVA, right ventricular apex.
Data are presented as mean ± SD or n (%).

Intracavity gradients during sinus rhythm and ventricular pacing in the 14 patients with available invasive haemodynamic pacing study data (a). Symptom classification of the whole cohort (n = 16) from pre-implant to latest follow-up (b).
Echocardiography-derived gradients
Paired pre- and post-procedural echocardiography data were available in 12 of 16 patients. Pre-pacing Doppler-derived gradients were significantly higher than those recorded at follow-up (47 ± 15 versus 24 ± 16 mmHg, p = 0.0005, 95% CI: −36 to −14). Median time to echocardiogram post device implant was 0.44 years [interquartile range (IQR), 0.32–0.98]. While endocardial definition in the retrospectively reviewed imaging was poor, no significant changes in visually estimated LV systolic function were described between the pre- and post-device echocardiograms. Differences in LV internal dimension (LVIDD) were small (1.2 ± 1.5 mm) as was degree of mitral regurgitation (MR) (1/12 patients had worsening MR by one category from trivial to mild).
Symptomatic profile
The median NYHA class prior to device implantation was 3.0 (IQR 0) and improved to 2.0 (IQR 0.75, p < 0.001) at 1-year post implant (Table 2 and Figure 1). Of the 16 patients, 13 (81%) reported reduction in NYHA class at 1 year with 3 (19%) of 16 describing no change in symptomatic status. After a mean follow-up duration of 4.6 ± 2.7 years, 8 (50%) of 16 patients had maintained improvement. Two of 13 patients with initial improvement subsequently declined in status, coinciding with concomitant development of AF and worsening VE burden, and 4 of 13 with initial improvement died. Of the three patients with no improvement in status at 1 year, 1 of 3 improved NYHA class at end of follow-up and 2 of 3 remained with unchanged symptoms.
The mean baseline gradient in responders (defined as any symptomatic improvement) was lower than non-responders (defined as no symptomatic improvement), although this did not meet significance (73 ± 22 mmHg versus 95 ± 9 mmHg, 95% CI: −6.87 to 52.82, p = 0.12). Possible explanations for loss of symptomatic improvement during follow-up include new post-procedure permanent AF (n = 5) and a high burden of ventricular ectopy (n = 1), with subsequent reduction of ventricular pacing. At the end of the follow-up period, there were four deaths which were attributable to congestive heart failure (n = 2), renal failure (n = 1) and pneumonia (n = 1).
Device follow-up
Five patients developed AF during follow-up necessitating anticoagulation. AV nodal ablation was undertaken in three patients in whom rate control was inadequate with medical therapy (in two cases after unsuccessful cardioversion/AF ablation) (Table 3). Ten patients experienced VT (seven non-sustained). Appropriate device therapy was delivered in three patients with ICDs for sustained VT [two anti-tachycardia pacing (ATP) and one shock]. Two of the patients who received appropriate therapy were in the intermediate-risk category for SCD and one had been considered in the low-risk category according to the SCD risk calculator, 12 but fulfilled American Heart Association (AHA) criteria due to non-sustained ventricular tachycardia (NSVT), severe hypertrophy and LGE on CMR. 3 One patient’s device delivered inappropriate therapy due to oversensing from a Sprint Fidelis shock lead (Medtronic, USA).
Complications and device follow-up.
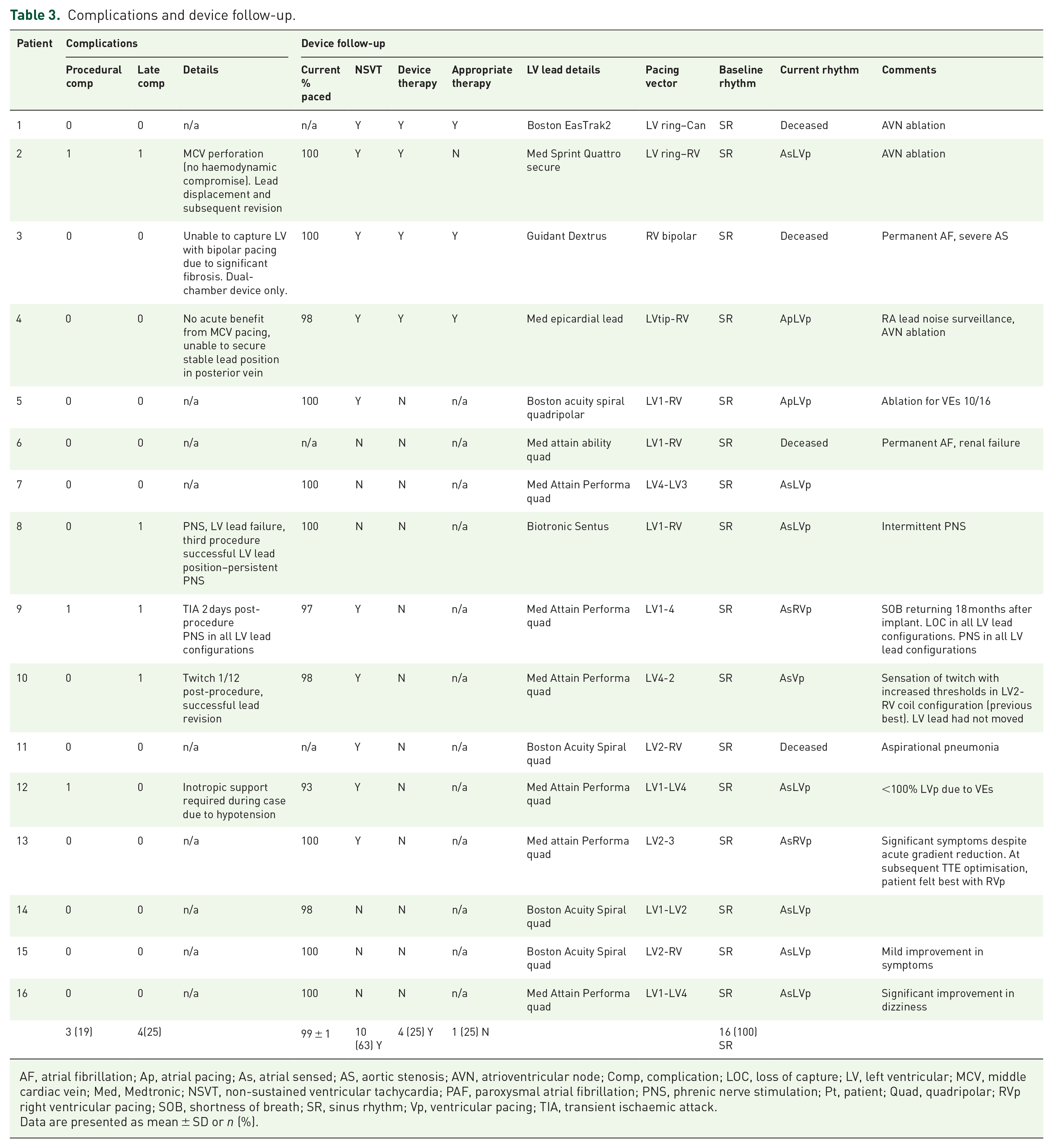
AF, atrial fibrillation; Ap, atrial pacing; As, atrial sensed; AS, aortic stenosis; AVN, atrioventricular node; Comp, complication; LOC, loss of capture; LV, left ventricular; MCV, middle cardiac vein; Med, Medtronic; NSVT, non-sustained ventricular tachycardia; PAF, paroxysmal atrial fibrillation; PNS, phrenic nerve stimulation; Pt, patient; Quad, quadripolar; RVp right ventricular pacing; SOB, shortness of breath; SR, sinus rhythm; Vp, ventricular pacing; TIA, transient ischaemic attack.
Data are presented as mean ± SD or n (%).
Complications
Procedural complications
All 16 subjects underwent successful device implantation, of whom 13 had uncomplicated insertion (81%) (Table 3). Two of 16 referred for biventricular pacemaker insertion had failure of LV lead placement, resulting in one dual-chamber and one surgical LV lead implanted system instead. Three procedural complications occurred: perforation of cardiac vein without overt haemodynamic compromise (1/16), transient ischaemic attack 2 days after procedure (1/16) and intra-procedural hypotensive episode requiring inotropic support during rapid sequence induction of general anaesthesia (1/16).
Late complications
Four patients suffered late complications. Phrenic nerve stimulation (PNS) occurred in three patients (one tolerated symptoms and two were unable to tolerate symptoms resulting in lead explanation/switching off LV pacing). LV lead fracture occurred in 1 of 15 patients in whom LV leads were eventually implanted; this was managed by an initial failed attempt at lead revision (and lead explant) after 6 months and successful revision after another 26 months. Intermittent PNS was detected in this subject after revision, but symptoms were manageable. LV lead displacement occurred in 1 of 15 patients who later underwent lead repositioning.
Discussion
This study demonstrates the feasibility of distal ventricular pacing at reducing LVMCO in patients with HCM and medically refractory symptoms. Historically, symptomatic management had reached its limits once maximal therapy was achieved, but a significant proportion of patients were left with unacceptable symptoms. Using techniques common to standard cardiac resynchronisation procedures, we found that symptoms were improved in 81% of patients at short-term follow-up, with average symptomatic improvement akin to a whole NYHA class. Acute gradient reduction was optimal most commonly from the MCV; however, this incurred a high proportion of lead complications. Post-procedural gradient reduction was confirmed with echocardiography in 12 of 16 patients who had paired echocardiography data. This subset of patients with LVMCO phenotype and apical aneurysm represent a unique and high-risk group, 14 the morphologic and symptomatic features requiring innovative approaches in an attempt to improve quality of life. Current data do not support the utility of this treatment in patients with other HCM phenotypes, and this treatment has only been considered in patients with isolated LVMCO and severe drug-refractory symptoms.
Feasibility, proposed mechanisms and relation to published data
This technique is highly sensitive to spatial location, with differences in obstructive gradient of significant magnitude between pacing sites separated only by short distances, multisite pacing allowing a greater number of sites to be examined. We attempted several pacing sites to determine the best strategy to reduce LVMCO. Early strategies included pacing from the RV outflow tract (RVOT) and lateral cardiac veins. However, this approach increased operating times without obvious benefit to LV haemodynamics, and we subsequently used only the RV apex and MCV to minimise procedure times and infection risk. LV apical fibrosis is well recognised in this form of HCM and may prevent electrical capture or require significantly greater voltages accelerating battery depletion.
Notably, occurrence of PNS is common in heart failure patients undergoing cardiac resynchronisation therapy, with reported prevalence of 15–37% 15 in keeping with our findings (20%).
The additive benefit of pacing from the MCV over the RV may be due to the more apical location of the LV lead, the importance of which has been previously described in LVOTO 16 and LVMCO. 10 In LVOTO, this is purported to be due to the apical location giving a higher degree of altered septal movement compared with a high septal location that may preserved normal contraction, 16 with similar findings in an LVMCO cohort. 10
We hypothesise that the gradient reduction and symptomatic effects are achieved by inducing the maximum degree of contractile dyssynchrony within the muscular ring that causes division of the LV cavity. This may prevent or delay the development of obstructive physiology (cavity obliteration) and result in reduced volume of the discrete apical chamber prior to development of obstruction. The resulting lower pressure within the LV aneurysm sac could mitigate apical ischaemia (Figure 2), thereby reducing symptoms of breathlessness and angina.
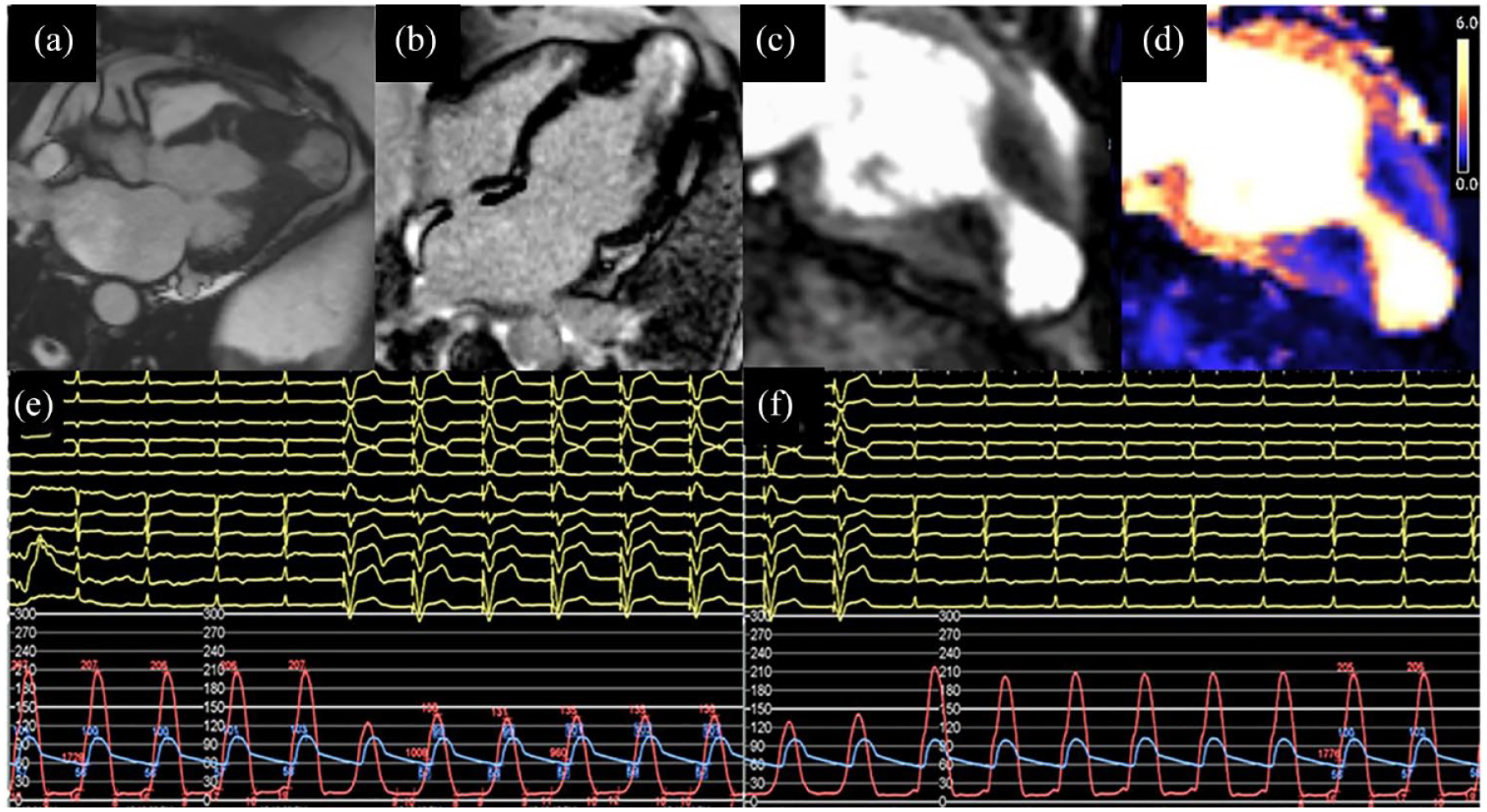
Illustrative CMR images showing large apical aneurysm in three-chamber view (a), late enhancement of the apex (b), perfusion abnormality at point of mid-cavity muscular apposition (c) and quantitative perfusion mapping in the same view (d). Haemodynamic pacing study with LV pacing onset, reduction in mid-cavity gradient and maintenance of aortic pressure (e) and offset (f), with immediate return of intracavity gradient.
Using LV pressure–volume loops, pacing-induced dyssynchrony has been demonstrated to reduce severity of cavity obliteration and magnitude of intraventricular gradients.5,17 The authors concluded that pacing diminishes isovolumic work at the distal regions of mid-systolic cavity compression 5 and undertook a small blinded cross-over study reporting beneficial effects of pacing on exercise capacity and symptoms. 17 Similar mechanisms may be responsible for acute LVMCO gradient reduction seen in our patients with distal ventricular pacing.
There is a paucity of data assessing pacemaker therapy in LVMCO. In two patients with ICDs in situ and LVMCO, RV apical pacing reduced Doppler-defined gradients and improved NYHA class. 9 In a larger cohort of patients, RV apical pacing acutely reduced invasively defined LVMCO gradients and improved NYHA class, with a trend towards increased exercise tolerance. 10 Honda et al. 11 performed invasive haemodynamic pacing studies during cardiac catheterisation in six patients with severe refractory symptoms and intracavity obstruction (four LVMCO and two LVOTO). The effects of RV apical and LV apical (endocardial) pacing on intracavity pressure gradients were assessed before implanting epicardial LV leads via limited thoracotomy in all six patients. Improvements in NYHA class were reported in all patients, and repeat invasive haemodynamic study after 3 months of LV pacing demonstrated a maintained reduction in gradients. In the absence of repeated invasive haemodynamic studies in our patients, follow-up assessment of gradients by echocardiography demonstrated significantly lower post versus pre-procedural gradients. However, Doppler echocardiography has significant shortcomings for assessment of obstructive gradients in this population compared with the more common outflow tract obstruction due to Doppler’s inability to measure continuing increases in velocity (and therefore pressure) across an obliterative mid-cavity. 18
Symptomatic improvement
While almost two-thirds of patients reported symptomatic improvement, the variability in achieving and maintaining that improvement may be partially explained by arrhythmia-induced reduction in ventricular pacing percentages from development of AF (all patients were in sinus rhythm at implant), high burden of ventricular ectopy or occurrence of PNS precluding use of distal ventricular pacing. LVMCO is associated with a high burden of atrial and ventricular arrhythmias, 8 and for a technique that relies on maintenance of sinus rhythm, it is therefore expected that this cohort would not maintain 100% benefit for the duration of follow-up.
In addition, factors other than magnitude of intracavitary obstruction contribute to symptoms in this heterogeneous condition. Notably, one patient described no improvement in symptoms with distal ventricular pacing despite significant reduction in LVMCO gradient (100 mmHg in sinus rhythm to 20 mmHg with LV pacing). The two other patients without improvement at 12 months experienced poor acute gradient reduction and post-implant complication (persistent twitch), respectively, which may have contributed to non-responder status.
Responders to pacing therapy have often been those with the most severe functional limitation at baseline that is refractory to optimised medical therapy. 19 In our cohort of patients with highly symptomatic LVMCO refractory to medical therapy, all patients were NYHA class III prior to intervention and hence were considered for the haemodynamic pacing procedure. Recent calls for pacing therapy to be re-instated as a first-line treatment in obstructive HCM remain debatable, 20 but may be relevant for a sub-group of highly symptomatic patients in sinus rhythm with LVMCO and limited treatment options.
Placebo effect is an often-cited criticism of pacing therapy for obstructive HCM. Promoted as evidence of underperformance compared with surgical myectomy or ASA, this fails to recognise the potential placebo effect of undergoing invasive interventions.
Study limitations
There are several limitations to this study. First, this retrospective proof-of-concept study collected clinically reported data that were available. While attribution of NYHA class may be considered subjective, it is a commonly used tool in clinical practice. In future studies, patient-reported outcome measures via validated questionnaires should be used to aid objectivity. Patients were un-blinded to their device settings, so placebo effect may contribute to reported symptomatic improvements, and we therefore interpret these results with a degree of caution. It is noteworthy that one patient with significant symptomatic improvement following pacing experienced symptomatic deterioration after LV lead displacement. Improvement of symptoms following LV lead repositioning indicates a genuine physiological response.
Another limitation is lack of repeat invasive haemodynamic assessment after a period of treatment to determine whether the symptomatic improvements correlated with ongoing reduction in invasively defined intracavity gradients; however, serial echocardiography was available in the majority of patients.
Conclusion
In drug-refractory symptomatic LVMCO, distal ventricular pacing can reduce intracavitary obstruction and may provide long-term symptomatic relief in patients with limited treatment options. A haemodynamic pacing study is an effective strategy for identifying optimal pacing site and configuration. The incidence of major intra-procedural complications was low, although the late complication rate largely driven by PNS and lead instability indicates further refinement of the technique is required.
