Abstract
The burden of heart failure has long plagued the productive years of the population, with therapeutic advances in the timely treatment of ischemic heart disease decreasing its associated mortality. Angiotensin-converting enzyme inhibitors and β-blockers have impacted heart failure therapeutics in a revolutionary way. The importance of blockade of the renin–angiotensin system and adrenergic stimulation are fully accepted concepts that apply in young and old, symptomatic and asymptomatic, borderline low and very low Ejection Fraction (EF), left ventricular failure and biventricular failure. Despite several interventions, both pharmaceutical and device based for the treatment of ensuing heart failure, the incidence is increasing in large proportions. Newer molecules like sacubitril show more promise. Despite these novel therapies, several patients relentlessly progress to a stage of advanced heart failure. The use of left-ventricular-assist devices has variable clinical benefit, with some patients progressing to heart transplantation.
Introduction
The burden of heart failure has long plagued the productive years of the population. With therapeutic advances for the timely treatment of ischemic heart disease, its associated mortality has decreased considerably, with increasing morbidity of ensuing heart failure. 1
Two classes of drugs, namely angiotensin-converting enzyme inhibitors and β-blockers have impacted heart failure therapeutics in a revolutionary way. The importance of blockade of the renin–angiotensin system and adrenergic stimulation are fully accepted concepts that apply in young and old, symptomatic and asymptomatic, borderline low and very low Ejection Fraction (EF), left ventricular (LV) failure and biventricular failure. 1 Despite these and several other interventions, both pharmacological and device based for the treatment of ensuing heart failure, the incidence is increasing in large proportions. Newer molecules like sacubitril show promise in many patients. 2 Despite novel therapies, several patients relentlessly progress to a stage of advanced heart failure. The use of left-ventricular-assist devices (LVADs) has shown variable clinical benefit, with some patients progressing to heart transplantation. 3
Cardiac resynchronization therapy has shown benefit in specific populations addressing the substrate of electrical dyssynchrony. 4 With the increasing burden of ischemic cardiomyopathy and heart failure, heart transplantation cannot be accessible to large populations, mainly due to lack of donor availability. Therefore, palliative therapies tailored to specific populations should be the panacea for this increasing morbidity.
Role of randomized controlled trials
Several authors agree that most heart failure trials yield results that are disappointing or difficult to interpret. 1 Despite the seemingly neutral findings of the trial, need for further trials is the often-repeated stance.
When this is the situation in the development of molecular therapies, it is not very clear regarding devices for advanced heart failure. While LVAD’s efficacy has been established, other innovative devices like the acorn cap and myosplint devices have been disregarded due to lack of benefit, although the initial results were promising in small selected samples. 5
Surgical therapy for heart failure and ventricular arrhythmias
The palliative surgery based on Laplace’s law for ischemic cardiomyopathy with LV aneurysms has seen applicability in large populations. The results of surgical ventricular restoration have been varied, with several factors being responsible. Rigorous inclusion criteria for surgical inclusion are vital, as is the surgical technique. Large areas of akinesia/dyskinesia of at least 35% of the left ventricle with presence of contractile myocardium ensure surgical success.6,7 A scarred area of 35% leads to LV dilatation with an LV end-systolic volume (ESV) of 60 ml/m2. Severe pulmonary hypertension is a relative contraindication. This surgery is not applicable to dilated cardiomyopathy and does not have benefits when the akinetic area exceeds 50%. Smaller areas of akinesis of less than 25% may not qualify for this procedure. 7
In the largest randomized study comparing revascularization alone and revascularization with surgical ventricular restoration (SVR) conducted in patients with ischemic cardiomyopathy, there was no added clinical benefit of SVR.8,9 This study included 1000 patients and was studied worldwide. This trial was severely flawed from its design and inclusion criteria. A large proportion of patients (nearly 66%) did not have LV volume measurements at entry into the study. Consequently, the SVR procedure may have reflected a small LV plication or limited intracavitary reconstruction. A reduction in LV ESV of 40% is required for clinical success of SVR, whereas in this trial, a reduction of 19% was observed.8,9 SVR has also been performed in patients with ventricular tachyarrhythmias and LV aneurysms.10,11 Initially, the arrhythmic focus was identified by endocardial mapping. The endocardium was resected and LV endoaneurysmorrhaphy was performed. Ventricular tachyarrhythmias were inducible in very few patients (2 out of 25). There was a very low mortality rate both due to sudden cardiac death and congestive heart failure. Blind cryoablation during SVR done at the transition zone had excellent results with recurrence in 2 out of 31 patients, with one of them requiring Intra Cardiac Defibrillator (ICD) implantation.
Hemodynamic characteristics of ischemic cardiomyopathy with left ventricular aneurysms and implications on surgical ventricular restoration
LV aneurysm formation is dependent on a variety of factors, chief among which is the thickness of the involved myocardium. 12 In anterior aneurysms, the LV apex is maximally involved, as it is thinnest. This is also the reason LV aneurysms are a sequela of transmural myocardial infarctions rather than subendocardial infarctions. Small aneurysms of less than 11% of the left-ventricular end-diastolic volume (LVEDV) usually do not present with congestive heart failure. When the aneurysmal volume exceeds 15–20% of the LVEDV, it leads to increases in LV end-diastolic pressure, ultimately leading to congestive heart failure. The contractile remote myocardium dilates in order to maintain an effective forward stroke volume. This increases wall stress by the Laplace’s law. Although the remote contractile myocardial contraction contributed to systolic ejection, a marked delay was observed in time to peak contraction, while the peak contraction was at least a third less than normal. 13 This has been observed in cardiac magnetic resonance (CMR) studies 13 (Figure 1). The shape of aneurysms has been studied theoretically in a mathematical model, demonstrating a rectangular shape having an increased ratio of active to inactive muscle compared with a circular shape. 12 The hemodynamic characteristics of the LV aneurysm need to be understood in order to plan the surgical strategy.

Mean endocardium-averaged mean myocardial velocity (MMV) characteristics of normal subjects (pink) and patients (blue) are compared against the mean MMV of the aneurysm region alone (red) and that of viable/remote myocardium alone (green), along with standard deviations at each cardiac phase. Remote endocardium had delayed preoperative peak MMV in comparison with the expectation from the mean normal MMV, and the aneurysm was dyskinetic with the remote myocardium.
Physicians using the techniques of SVR have witnessed a steady evolution over the years, with greater understanding of the underlying pathological substrate. A mere reduction in LV volume may not be sufficient, while an optimal ellipsoid restoration of the LV may seem distant. Recent advances in cardiac imaging technology have enabled study of LV geometry and function.
The intraventricular patch geometry reflects the theoretical calculations in a mathematical model where the ratio of contractile to infracted muscle was greater with a rectangular aneurysm than a circular one. The patch mimics the aneurysm in this model. The remote or contractile myocardium demonstrated a delay in time to peak contraction with decrease in amplitude of contraction. The systolic contraction extended into early diastole, while the early diastolic filling was decreased. Following SVR using a rectangular patch, improvements were seen in the remote myocardium with earlier onset of systolic contraction and an increase in the amplitude of contraction. 13
Following SVR by circular endoventricular patch plasty, significant improvements were seen in the LV basal rotations, while there were no changes in the apical rotations. 14 This demonstrated a greater decrease in LV volume at the base than at the apex, resulting in a less ellipsoid LV configuration. Modifying the patch geometry to a narrow rectangular one, it was seen that there were significant improvements in both apical and basal LV rotations (Figure 2). 6 This was also demonstrated in a study by Cirillo et al., 15 where a narrow linear patch was used. It has been noted that in smaller LVs, use of a narrow patch may simulate linear repair, while it may be adequate in larger LVs. 16 Therefore, patch geometry also needs individualization for the underlying LV geometry for the success of SVR.
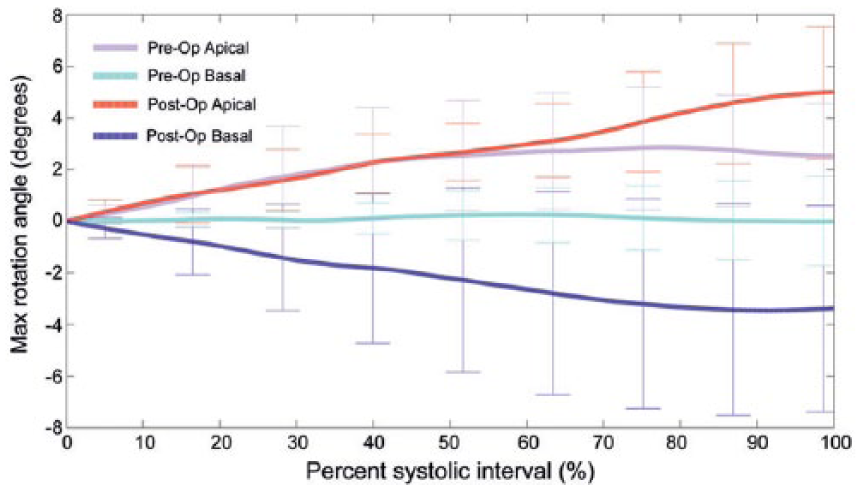
Maximum anticlockwise (positive), coded light pink, and clockwise (negative) rotation, coded light blue, of the apical and basal slice levels, respectively, during the systolic interval of the cardiac cycle preoperatively. The postoperative rotations are coded red for apical rotation and blue for basal rotation. [Here, anticlockwise direction (apex) is positive and clockwise direction (base) is negative]. Reproduced with permission from Adhyapak et al. 6
SVR is based on Laplace’s law of decreasing the LV radius, thereby decreasing wall stress. The success of SVR, though critically dependent on decreases in LVEDV, is further dependent on an optimal restoration of an ellipsoid LV. Restoration of LV rotation is essential for normalization of LV function, as discussed. Decreasing the LVEDV alone does not ensure optimal surgical results, the shape of the restored LV being dependent on the geometry of the endoventricular patch.
With limited therapeutic options for this heterogeneous population, caution should be exercised in interpreting RCT findings, and therapeutic innovations should be prudently individualized to each underlying substrate.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Note
Institutional Ethics committee approval was obtained for all our patients studied at our institution.
