Abstract
Being overweight or obese is associated with a higher individual risk of venous thromboembolism and poorer postprocedural outcomes after hip or knee replacement surgery. In addition, there is evidence that obesity represents a significant driving factor for the current and projected prevalence of atrial fibrillation. Rivaroxaban and other direct oral anticoagulants offer fixed-dose regimens for these indications. They do not require therapeutic drug monitoring or dose adjustment according to the weight of the patient. However, primary care physicians seem to be hesitant to accept the concept of a fixed-dose regimen for patients at extremes of weight, perhaps because of familiarity with weight-based dosing of other drugs including low molecular weight heparins. The main concerns related to unadjusted dosing are increased exposure in underweight patients leading to a risk of excessive bleeding and conversely to underanticoagulation of overweight patients. Rivaroxaban has shown similar efficacy and a similar or better safety profile compared with standard treatment for several venous and arterial indications, including venous thromboembolism, nonvalvular atrial fibrillation, and acute coronary syndrome. Prespecified subgroup analyses of patients stratified by weight or body mass index demonstrated outcomes that were consistent with the overall analysis and within each weight and body mass index group. The results suggest that standard-dose rivaroxaban can be safely prescribed in adult patients of all weights.
Introduction
Rivaroxaban and other direct oral anticoagulants (DOACs) were developed to overcome the limitations of the previous standard therapeutic approach with vitamin K antagonists (VKAs) and parenteral anticoagulants. DOACs have simplified patient management significantly by offering a fixed-dose regimen associated with fewer drug–drug and drug–food interactions compared with VKAs [Nutescu et al. 2011]. In addition, no routine anticoagulation monitoring is necessary with the newer agents, and rivaroxaban (as with apixaban) is given without the use of initial parenteral anticoagulation in the treatment and secondary prevention of venous thromboembolism (VTE) [comprising deep vein thrombosis (DVT) and pulmonary embolism (PE)] [Bayer Pharma AG, 2015; Nutescu et al. 2011]. Among the DOACs, rivaroxaban has the largest clinical study program, with more than 15 completed phase III clinical trials to date. Rivaroxaban was also the first DOAC to be approved for clinical use in the treatment of DVT and PE and secondary prevention of VTE in adults. Other indications for which rivaroxaban has received approval include the prevention of VTE in adult patients undergoing elective hip or knee replacement surgery, the prevention of stroke and systemic embolism in adult patients with nonvalvular atrial fibrillation (AF) with one or more risk factors, and the prevention of atherothrombotic events in adult patients with elevated cardiac biomarkers after an acute coronary syndrome (ACS). Rivaroxaban is approved for the latter indication in the European Union but not in the United States. Rivaroxaban has no indication in pediatric patients at present [Bayer Pharma AG, 2015; Janssen Pharmaceuticals Inc., 2015]. Rivaroxaban was assessed in large phase III clinical trials (RECORD1–4, EINSTEIN DVT/EINSTEIN PE, ROCKET AF, and ATLAS ACS 2 TIMI 51) and has, compared with the previous standard therapy, demonstrated a favorable benefit–risk profile throughout these trials [Eriksson et al. 2008; Kakkar et al. 2008; Lassen et al. 2008; Mega et al. 2012; Patel et al. 2011; The EINSTEIN Investigators, 2010; The EINSTEIN–PE Investigators, 2012; Turpie et al. 2009].
The measurement of bodyweight and/or body mass index (BMI) provides some information on the general health status of a patient. A BMI of 18.5–<25 kg/m2 is classified as normal, a value below 18.5 kg/m2 is classified as underweight, and a value of 25 kg/m2 or above is classified as overweight or obese (overweight, BMI 25–29 kg/m2; obese, BMI 30–39 kg/m2; morbidly obese, BMI ⩾ 40 kg/m2) [Centers for Disease Control and Prevention, 2015; Friedman et al. 2013]. The prescribing information for rivaroxaban defines under 50 kg and over 120 kg as extremes of bodyweight, without consideration of the patient’s height [Bayer Pharma AG, 2015].
There is a growing body of evidence suggesting an association between bodyweight and cardiovascular disease. Obesity was found to be an independent risk factor for both DVT and PE, particularly in women and in patients (men and women) aged under 40 years [Stein et al. 2005]. An increasing BMI has been linked to higher numbers of global total hip or knee replacement (THR or TKR) procedures which, in turn, increases the number of individuals at risk of VTE [Wendelboe et al. 2003]. In addition, patients who are morbidly obese have demonstrated poorer postprocedural outcomes after THR or TKR [Friedman et al. 2013]. The structural and functional abnormalities promoted by obesity and its associated cardiometabolic comorbidities (e.g. hypertension, vascular disease, pericardial fat, obstructive sleep apnea, insulin resistance) may represent some of the principal driving factors behind the current and projected AF epidemic [Abed and Wittert, 2013]. Paradoxically, recent epidemiological studies have shown that patients who are overweight or mildly obese tend to have better prognosis of cardiovascular disease than normal weight patients [Lavie et al. 2014]. Patients who are overweight or obese demonstrated lower mortality rates from chronic heart failure and acute VTE, as well as improved survival rates after stroke [Hainer and Aldhoon-Hainerova, 2013]. Several factors have been proposed as reasons for this unexpected observation, including greater metabolic reserves, lower prevalence of smoking, and younger age at presentation [Lavie et al. 2014]. However, this obesity paradox only seems to apply when patients with low fitness levels are compared and is not apparent in higher fitness populations, which have a generally good prognosis [Lavie et al. 2014].
Although being underweight has been identified as a risk factor for coronary heart disease [Suastika et al. 2012], there is, at present, no evidence to suggest that it may increase the risk of other thromboembolic disorders such as VTE or AF. Moreover, one case–control study has shown a significantly reduced risk of VTE in underweight subjects compared with those of a normal bodyweight [Delluc et al. 2009].
Because established anticoagulants such as low molecular weight heparin and fondaparinux require weight-adjusted dosing [Garcia et al. 2012], clinicians may be uncertain about whether this is also required for rivaroxaban and other DOACs when treating patients at extremes of weight. There is concern that patients who are underweight may have increased exposure to the DOAC via higher plasma concentrations with an increased risk of bleeding; conversely, patients who are overweight may be underanticoagulated and at greater risk of thromboembolic events. Given the strong link between obesity and cardiovascular conditions, many of the phase III studies conducted for rivaroxaban included prespecified subanalyses in which patients were stratified according to bodyweight or BMI [Mega et al. 2012; Patel et al. 2011; Prins et al. 2013; The EINSTEIN Investigators, 2010; The EINSTEIN–PE Investigators, 2012; Turpie et al. 2011]. The safety and efficacy profile of rivaroxaban in the total population analysis was maintained across prespecified subgroups classified according to weight or BMI [Mega et al. 2012; Patel et al. 2011; Prins et al. 2013; The EINSTEIN Investigators, 2010; The EINSTEIN–PE Investigators, 2012; Turpie et al. 2011].
This article summarizes the current knowledge of the role of bodyweight on the safety and efficacy of rivaroxaban from phase I–III studies involving more than 50,000 patients [Mega et al. 2012; Patel et al. 2011; Prins et al. 2013; Turpie et al. 2011].
Pharmacological parameters for rivaroxaban
Phase I and II clinical trials have established predictable pharmacokinetic (PK) and pharmaco-dynamic (PD) properties of rivaroxaban in patients over a wide range of bodyweights [Kreutz, 2014]. In brief, results of the phase I studies demonstrated that rivaroxaban is well tolerated in healthy human subjects, with predictable plasma concentration profiles and dose-dependent inhibition of factor Xa after single and multiple doses. They further showed an elimination half life of 5–9 h in young individuals (20–45 years) and 11–13 h in older subjects (60–76 years), and no relevant drug accumulation with twice-daily dosing [Bayer Pharma AG, 2015; Kubitza et al. 2005, 2008]. Rivaroxaban has two elimination pathways: one third of the drug is eliminated as unchanged parent drug via the urine, whereas two thirds is metabolized by the liver, half of which is then excreted renally and half via the hepatobiliary route [Kreutz, 2014].
A dose of rivaroxaban 10 mg was studied in a randomized, single-blind, placebo-controlled study that compared subjects with extremes of bodyweight [>120 kg (n = 12) or ⩽50 kg (n = 12)] with normal-weight patients (70–80 kg; n = 12) [Kubitza et al. 2007]. The maximum plasma concentration (Cmax) was significantly increased by 24% in the ⩽50 kg weight group compared with the normal-weight group (Figure 1(a)). However, no significant difference was observed between the Cmax values for the >120 kg and 70–80 kg groups, and the bioavailability of rivaroxaban was similar in all three weight categories as indicated by the area under the curve of plasma concentrations (Figure 1(a)). The time to Cmax of rivaroxaban was similar across the three weight groups; however, a nonsignificant increase in half life was seen in subjects weighing ⩽50 kg [Kubitza et al. 2007]. Pharmacodynamic measures revealed a similar degree of inhibition of factor Xa activity by rivaroxaban in all weight groups and no inhibition in subjects receiving placebo (Figure 1(b)). The maximum PD effect (Emax) occurred 3–4 h after administration of rivaroxaban and no significant differences in Emax between the groups was demonstrated. The PK and PD assessment indicated that rivaroxaban was equally well tolerated in subjects of all weight classes [Kubitza et al. 2007]. The limited effect of bodyweight on the PK profile of rivaroxaban may be explained by the low volume of distribution of the drug [Kubitza et al. 2007]. The tissue affinity of rivaroxaban is moderate, with no known irreversible organ-specific interactions but extensive binding to plasma proteins (~90%) [Weinz et al. 2005]. The distribution of rivaroxaban is, therefore, mainly expected in the interstitial space and vascular bed, matrices that are unlike to vary significantly between subjects with different bodyweights [Kubitza et al. 2007].
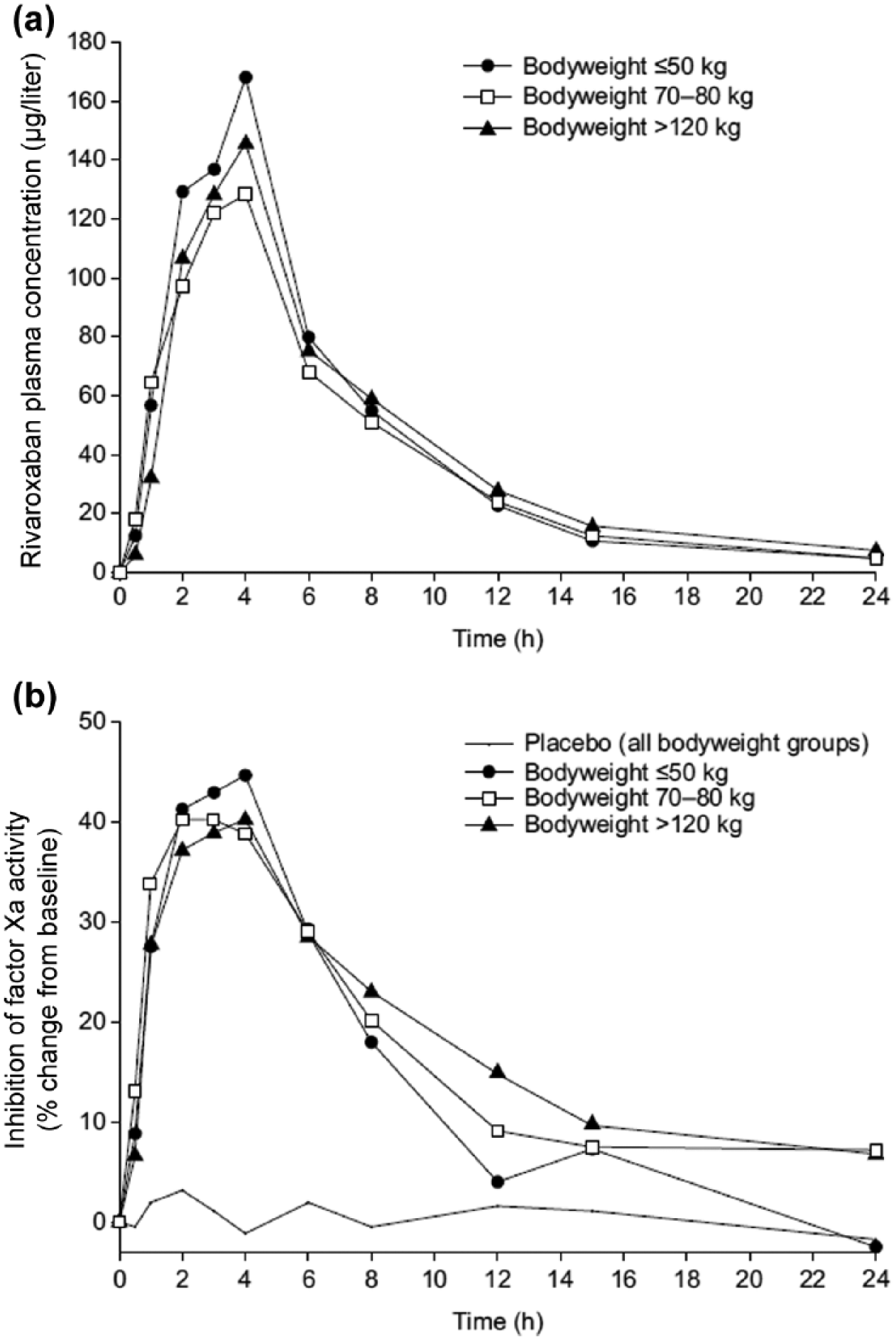
Mean plasma concentrations of rivaroxaban (a) and median inhibition of factor Xa activity (b) after administration of a 10 mg single dose to healthy male and female subjects in three weight categories (n = 12 per group) [Kubitza et al. 2007].
Rivaroxaban: phase III clinical trial outcomes
Efficacy and safety outcomes of rivaroxaban versus standard therapy have been stratified by bodyweight and/or BMI in phase III clinical studies evaluating the following indications: VTE prevention after THR or TKR surgery, treatment and secondary prevention of VTE, prevention of stroke and systemic embolism in patients with nonvalvular AF with one or more risk factors, and the prevention of atherothrombotic events in adult patients with elevated cardiac biomarkers after ACS (Tables 1 and 2) [Mega et al. 2012; Patel et al. 2011; Prins et al. 2013; The EINSTEIN Investigators, 2010; The EINSTEIN–PE Investigators, 2012; Turpie et al. 2011].
Prespecified subgroup analysis of weight and body mass index on efficacy and safety from the rivaroxaban phase III clinical trials for venous indications.
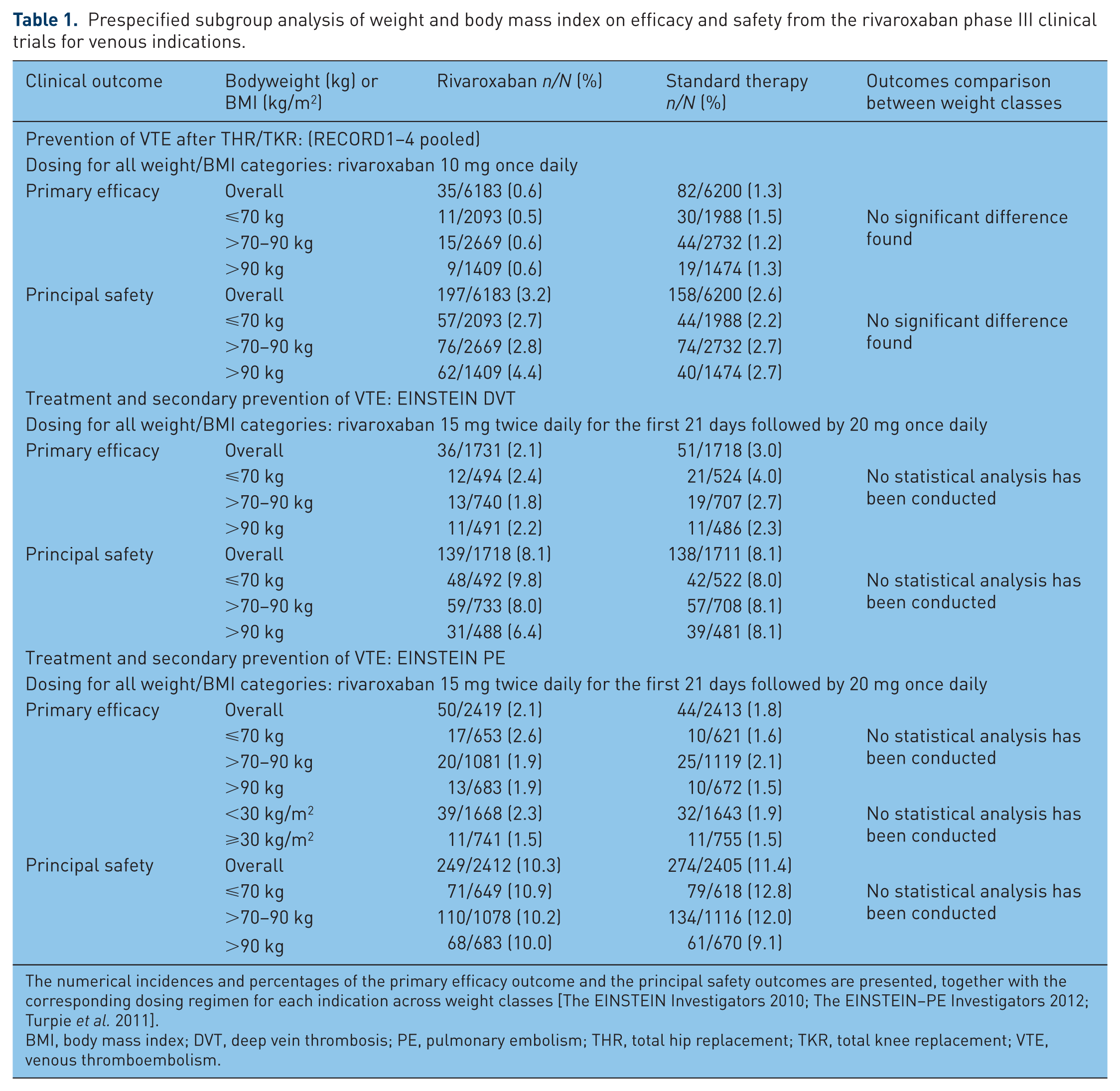
The numerical incidences and percentages of the primary efficacy outcome and the principal safety outcomes are presented, together with the corresponding dosing regimen for each indication across weight classes [The EINSTEIN Investigators 2010; The EINSTEIN–PE Investigators 2012; Turpie et al. 2011].
BMI, body mass index; DVT, deep vein thrombosis; PE, pulmonary embolism; THR, total hip replacement; TKR, total knee replacement; VTE, venous thromboembolism.
Prespecified subgroup analysis of weight and body mass index on efficacy and safety from the rivaroxaban phase III clinical trials for arterial indications.

The numerical incidences and percentages of the primary efficacy outcome and the principal safety outcomes are presented, together with the corresponding dosing regimen for each indication across weight classes [Patel et al. 2011; Mega et al. 2012].
ACS, acute coronary syndrome; AF, atrial fibrillation; ASA, acetylsalicylic acid; BMI, body mass index.
Four phase III trials (RECORD1–4) were conducted to assess rivaroxaban versus enoxaparin for the prevention of VTE after THR or TKR. Each of the RECORD studies demonstrated superiority of rivaroxaban over enoxaparin for reductions in the primary efficacy endpoint (the composite of symptomatic and asymptomatic DVT, nonfatal PE, and all-cause mortality) and a similar profile for the main safety outcome (major bleeding) [Eriksson et al. 2008; Kakkar et al. 2008; Lassen et al. 2008; Turpie et al. 2009]. A pooled analysis of the four RECORD trials showed consistent reductions in symptomatic VTE and all-cause mortality in rivaroxaban-treated patients compared with enoxaparin, irrespective of patient weight (subgroups included ⩽70 kg, 71–90 kg, >90 kg) (Table 1) [Turpie et al. 2011]. A similar incidence of major and clinically relevant non-major bleeding was seen in rivaroxaban-treated patients compared with enoxaparin-treated patients across weight classes [Turpie et al. 2011]. Comparing the same outcomes within the rivaroxaban treatment group, no significant differences were demonstrated across the weight classes. However, there was a numerical increase in major plus clinically relevant nonmajor bleeding in the >90 kg weight class compared with >70–90 kg and ⩽70 kg (4.4% versus 2.8% versus 2.7%; p = 0.246) (Table 1) [Turpie et al. 2011].
A retrospective analysis of the data from the RECORD program assessed complication rates in patients who were morbidly obese (⩾40 kg/m2) compared with patients who were not (<40 kg/m2) after THR or TKR after anticoagulation treatment with rivaroxaban or enoxaparin [Friedman et al. 2013]. No increased risk of VTE or bleeding was observed in patients who were morbidly obese; however, the condition was associated with an increased risk of early postoperative complications, including wound inflammation or infection, erythema, diarrhea, and gastrointestinal or abdominal pain [Friedman et al. 2013].
The EINSTEIN DVT and EINSTEIN PE clinical trials assessed the efficacy and safety of rivaroxaban against enoxaparin/VKA for the treatment and secondary prevention of VTE, with recurrent VTE as the primary efficacy outcome and clinically relevant bleeding (composite of major and clinically relevant nonmajor bleeding) as the principal safety outcome [The EINSTEIN Investigators, 2010; The EINSTEIN–PE Investigators, 2012]. Rivaroxaban demonstrated similar efficacy and safety versus standard therapy in both trials, but with significantly lower rates of major bleeding in EINSTEIN PE [The EINSTEIN Investigators, 2010; The EINSTEIN–PE Investigators, 2012]. Prespecified subgroup analyses for EINSTEIN DVT and EINSTEIN PE confirmed similar efficacy and safety profiles for rivaroxaban and enoxaparin/VKA for different weight classes (⩽70 kg, 71–90 kg, >90 kg) (Table 1). Classification according to BMI [1496 patients with obesity versus 3311 patients who were not obese (<30 kg/m2)] was also conducted to allow for assessment of efficacy across the treatment groups in the EINSTEIN PE study, and the results were consistent with those gained when patients were classified according to weight. Comparing different weight and BMI categories within the rivaroxaban treatment group did not reveal any differences in outcomes [The EINSTEIN Investigators, 2010; The EINSTEIN–PE Investigators, 2012].
Pooled analysis of the EINSTEIN DVT and EINSTEIN PE data confirmed the findings from the EINSTEIN PE trial, with similar efficacy compared with standard therapy but significantly lower rates of major bleeding (absolute risk reduction 0.8% in favor of rivaroxaban) [Prins et al. 2013]. In addition, the pooled analysis has allowed for increased statistical power across subgroup analyses and has enabled assessment of the treatment effect in a small number of patients from both treatment arms with bodyweight ⩽50 kg (n = 107) [Prins et al. 2013]. Similar efficacy of rivaroxaban and enoxaparin/VKA was shown; however, safety outcomes were non-estimable owing to small group sizes [Prins et al. 2013]. Assessing a larger group of frail patients [defined as one or more of the following criteria: age >75 years, calculated creatinine clearance <50 ml/min or low bodyweight (⩽50 kg)] as part of the pooled analysis permitted estimation of efficacy and safety outcomes. Significantly lower rates of major bleeding and an improved net clinical benefit compared with enoxaparin/VKA were observed in frail patients receiving rivaroxaban. Comparing frail patients with nonfrail patients within the rivaroxaban treatment group demonstrated higher rates of VTE recurrence in frail versus nonfrail patients (2.7% versus 1.9%) and higher incidences of major bleeding in frail versus nonfrail patients (1.3% versus 0.9%) [Prins et al. 2013]. The ongoing EINSTEIN CHOICE phase III study aims to assess reduced-dose (10 mg once daily) and standard-dose (20 mg once daily) rivaroxaban versus acetylsalicylic acid (ASA; 100 mg once daily) in the long-term secondary prevention of symptomatic VTE. Many clinicians may be particularly concerned about using a rivaroxaban dose as low as 10 mg in patients who are overweight. Consequently, EINSTEIN CHOICE data in patients who are overweight will be very important and may also offer some insight into efficacy and safety of a lower dose of rivaroxa-ban in underweight patients [ClinicalTrials.gov identifier: NCT02064439]. The estimated enrollment is 2850 patients.
Rivaroxaban was assessed against warfarin for the prevention of stroke and systemic embolism in patients with nonvalvular AF in the ROCKET AF clinical trial. The primary efficacy endpoint was the composite of stroke and systemic embolism, whereas the principal safety endpoint was the composite of major and nonmajor clinically relevant bleeding [Patel et al. 2011]. A similar efficacy and safety profile was demonstrated for rivaroxaban compared with warfarin, although rivaroxaban was associated with less-frequent intracranial and fatal bleeding events [Patel et al. 2011]. Prespecified subgroup analysis of patients stratified by bodyweight (⩽70 kg, 71–90 kg, >90 kg) and BMI (⩽25 kg/m2, 25 – ⩽35 kg/m2, >35 kg/m2) was consistent with the findings from the total population analysis. No significant differences in clinical outcomes were observed between the different weight and BMI groups within the rivaroxaban treatment group (Table 2) [Patel et al. 2011].
The ATLAS ACS 2 TIMI 51 trial was conducted to evaluate rivaroxaban together with standard antiplatelet therapy (ASA alone or ASA with clopidogrel or ticlopidine) versus standard antiplatelet therapy alone, for the prevention of atherothrombotic events in adult patients with elevated cardiac biomarkers after an ACS [Mega et al. 2012]. Rivaroxaban achieved a significant reduction of the primary efficacy (composite) endpoint of death from cardiovascular causes, myocardial infarction, or stroke compared with antiplatelet therapy alone. As expected, rivaroxaban treatment versus placebo was associated with increased rates of major bleeding not related to coronary artery bypass grafting and intracranial hemorrhage, but not increased rates of fatal bleeding. A prespecified subgroup analysis was conducted for different bodyweight classes (<60 kg, 60–89 kg, ⩾90 kg) and was consistent with the overall analysis for the primary efficacy endpoint and major bleeding outcomes. Similar rates of clinical outcomes were also seen comparing the different weight classes within the rivaroxaban treatment group (Table 2) [Mega et al. 2012].
The results of the phase II–III rivaroxaban clinical study program have led to clear guidance on rivaroxaban use across different weight classes: the EU Summary of Product Characteristics and US Prescribing Information do not recommend dose adjustment of rivaroxaban for patients at extremes of bodyweight (defined as <50 kg and >120 kg) [Bayer Pharma AG, 2015; Janssen Pharmaceuticals Inc., 2015].
Real-world evidence
Real-life evidence of the influence of bodyweight on rivaroxaban effectiveness in unselected patient populations is limited at present. The multicenter, prospective, observational, noninterventional XALIA study investigated the effectiveness and safety of rivaroxaban versus standard therapy in patients with DVT and, after approval in the PE indication, in patients with DVT plus concomitant PE [Ageno et al. 2015]. The study cohort included 5142 adult patients with objectively confirmed acute DVT and an indication for anticoagulation treatment for at least 3 months. Rivaroxaban-treated patients showed a lower frequency of major bleeding (0.8% versus 2.1%; p = 0.44) and recurrent VTE (1.4% versus 2.3%; p = 0.72), and all-cause mortality (0.4% versus 3.4%; p = 0.074) and a similar incidence of treatment-emergent adverse events (36% versus 37.5%), compared with standard therapy. Subgroup analyses conducted according to bodyweight (⩽70 kg, >70 – <90 kg, ⩽90 kg) demonstrated no significant differences in the rates of recurrent VTE or major bleeding, except in the middle weight group (>70–<90 kg) in which numerically lower rates of major bleeding were observed in the rivaroxaban group [Ageno et al. 2015]. Further data may be expected from ongoing registries such as the Dresden NOAC Registry, in which the management and safety of peri-interventional DOAC use is evaluated in a cohort of patients from daily care across different DOAC indications [Beyer-Westendorf et al. 2014]. The ORBIT-AF registry aims to generate information on the treatment of patients with AF in the community practice setting [Piccini et al. 2011]. Factors such as stroke prevention, transitions to new therapies, rate and rhythm control, and clinical- and patient-centered outcomes are evaluated with the goal to increase quality of AF care and to improve patient outcomes. Finally, the international GARFIELD-AF registry includes patients with newly diagnosed AF and at least one additional risk factor for stroke [Kakkar et al. 2012].
Case studies have emerged in which patients who are obese and morbidly obese have developed stroke or PE during long-term anticoagulation with dabigatran [Breuer et al. 2013; Rafferty et al. 2013]. These findings have led to concern among physicians that DOACs may fail to provide adequate protection in this patient population. Indeed, in a patient who is morbidly obese and has had an ischemic stroke and is receiving dabigatran, the Cmax of dabigatran was lower than the 25th percentile of the therapeutic trough level [Breuer et al. 2013]. In another case, a 67-year-old male patient with obesity (BMI 39.6 kg/m2) presented with ischemic stroke and previously unknown AF. He was started on dabigatran 10 days after the event [Safouris et al. 2014]. Monitoring revealed that the patient failed to reach the 25th percentile level for Cmax and was below the 25th percentile of the therapeutic trough level most of the time, further indicating that dabigatran could be at subtherapeutic levels in patients who are obese or morbidly obese. Dabigatran was substituted with rivaroxaban, which reached peak and trough levels consistent with effective anticoagulation. A higher proportion of dabigatran is renally excreted compared with rivaroxaban (~80% for dabigatran and ~35% for rivaroxaban) and the observed glomerular filtration rate in patients who are obese is higher than in those who are not obese. Therefore, it has been suggested that the subtherapeutic drug levels were caused by increased drug clearance. In addition, phase II studies of rivaroxaban demonstrated clinical efficacy with very low trough levels (hence the licensing of a once-daily regimen) [Safouris et al. 2014].
Although scarce, evidence from plasma levels of rivaroxaban suggests that efficacy and safety are maintained, even in patients who are morbidly obese: one patient with a BMI of 61 kg/m2 who presented with an unstable international normalized ratio and a high risk of VTE after recent bariatric surgery was switched from VKA to standard-dose rivaroxaban [Mahlmaan et al. 2013]. Rivaroxaban is usually absorbed rapidly in the upper gastrointestinal tract; however, this process did not seem to be significantly impaired by bariatric surgery, as indicated by a peak concentration observed in the plasma after 3 h, which was within the expected range of published data [Mahlmaan et al. 2013].
Conclusion
There is still caution among primary care physicians with regard to prescribing standard-dose rivaroxaban in patients who are over- or underweight, owing to the perception that the patient may be underanticoagulated or may be over-exposed to the anticoagulant. Few studies exist that are dedicated to the use of rivaroxaban in patient populations at extremes of weight. However, within the large phase III studies, prespecified subgroup analyses stratified by weight and/or BMI have been undertaken for each approved indication of rivaroxaban. Efficacy and safety outcomes are consistent with the overall analysis of each trial (rivaroxaban versus comparator) and they are also similar between different-weighted rivaroxaban users. Moreover, case studies suggest that the bioavailability of rivaroxaban is not impaired in patients who are obese or morbidly obese. Available PK and PD data suggest that standard-dose rivaroxaban can be used safely in patients of all weight ranges.
Footnotes
Acknowledgements
Concept and outline, interpretation of the data, drafting, critical review, and approval of the submission draft were the responsibility of the author. The author would like to acknowledge Jasmina Saric, who provided editorial support.
Funding
Funding for editorial support was provided by Bayer HealthCare Pharmaceuticals.
Conflict of interest statement
Speaker bureau for Bayer, BMS, Pfizer. Scientific advisory boards for Bayer and Leo pharmaceuticals. Travel support from Bayer and Pfizer.
