Abstract
Objective:
Prosthetic heart valve implantation is commonly performed in patients that have valvular heart disease. Prior to clinical evaluation of newly developed prostheses, preclinical animal studies are performed for the assessment of both acute and chronic valvular function. Commonly, one size of valve is used in these preclinical studies, which can present difficulties with the implantation procedure and assessing valve function. Due to these potential problems, we developed a preoperative screening assessment in potential ovine candidates for prosthetic aortic valve implantation. By determining if there is a correlation between conscious and anesthetized echocardiographic examinations, an improvement in surgical confidence can predict that the animal is deemed a suitable candidate for a particular size of prosthetic valve for implantation prior to subjecting the animal to anesthesia and surgery.
Methods:
A total of 53 crossbred sheep (Ovis aries), male and female, 10–37 months of age, weighing between 41 and 77 kg, underwent conscious echocardiography and a subset of 29 of these animals underwent echocardiographic assessment under anesthesia for a preoperative valve size comparison in these animals prior to surgical prosthetic aortic valve implantation. Using 2D echocardiographic assessment, left ventricular outflow tract (LVOT) dimensions were assessed.
Results:
The mean paired difference between anesthetized and conscious LVOT diameter measurement was −0.87 mm (p = 0.0066, standard deviation 1.598, 95% confidence interval, −0.4796, −0.26378, n = 29).
Conclusion:
This pilot study evaluation revealed that conscious echocardiographic assessment can play a role preoperatively in selecting potential candidates for surgical prosthetic aortic valve implantation, thereby minimizing the potential in prosthetic-native annular mismatching, which can contribute to altered LVOT function.
Keywords
Introduction
Prosthetic valve implantation has been utilized as a treatment option in patients with significant valvular heart disease. Aortic stenosis is a common valvular disease in Western society with an estimated prevalence predicted to increase due to an aging population. Surgical prosthetic aortic valve replacement is a definitive therapy for patients demonstrating symptoms of left ventricular dysfunction and hemodynamically significant aortic valve stenosis [Bonow et al. 2006; Lung et al. 2003; Nkomo et al. 2006]. There are two types of prostheses that are commonly implanted: mechanical or bioprosthetic. Manufacturers of aortic valve prostheses continue to explore ways of improving existing valves, such as valve design and implantable materials [Sapirstein and Smith, 2001]. To evaluate these newly designed and developed prostheses, an ovine model is commonly utilized for preclinical evaluation prior to clinical studies [Bianco et al. 2009].
A range of valve sizes is available clinically for patients undergoing prosthetic aortic valve replacement, for which the correct size is selected according to the patient’s anatomy [Doty and Doty, 2012]. However, commonly in preclinical valve studies, a single size of the study prosthesis is common to reduce the variability in hemodynamic evaluation, as well as to financially limit production of test devices and to decrease the total number of animals to be evaluated. Obviously, this can present problems at the time of implantation and also in hemodynamic long-term follow-up evaluations due to a potential mismatching with the device size and the animal’s anatomy. Preoperative imaging techniques, such as echocardiography or computed tomography, can help screen and identify suitable surgical candidates and thereby minimize operative deaths and long-term morbidities and mortalities. Commonly these evaluation studies are performed only prior to surgical implantation, without assessing how the anatomy might change during anesthesia [Jilaihawi et al. 2013]. Due to the lack of this type of data, we have investigated what differences in left ventricular outflow tract (LVOT) anatomy might occur between preoperative and intraoperative evaluations. The findings of this investigation are reported in this paper.
Materials and Methods
A total of 53 crossbred adult sheep (Ovis aries), 10 to 37 months of age, weighing between 41 and 77 kg, were initially considered as surgical candidates for this study. All animals underwent conscious echocardiographic examination and a subset (n = 29) also underwent anesthetized echocardiography in the operating room for anatomic dimension comparison. Two sonographers participated in performing these examinations. The research study protocol was approved by the Physiological Research Laboratory Institutional Animal Care and Use Committee (Medtronic, Inc.). Animals were housed and received humane care in accordance with the Guide for Care and Use of Laboratory Animals [Institute for Laboratory Animal Research, 2011], and Animal Welfare Act as Amended [Animal Welfare Act as Amended, 2008]. A 21 mm aortic valve prosthesis was chosen by the sponsor of the study for implantation in each surgical candidate.
Accepted surgical candidates underwent a preoperative conscious transthoracic echocardiographic evaluation using a GE Vivid 7 Dimension or GE Vivid Q with GE M4S Matrix Array 1.7 to 4.0 MHz cardiac probe (GE Healthcare, Wauwatosa, WI, USA). Under manual restraint with the animal straddling a large bolster in a quadrupedal stance, the ultrasound probe with coupling gel (Aquasonic 100, Parker Laboratories, Fairfield, NJ, USA) was used to visualize the LVOT by placing the probe lateral to the sternum on the right anterior thorax between the third and fifth intercostal space. The image from this window bisects the long axis between both the aortic and mitral valves. The target image to measure the LVOT showed the left atrium with the midanterior (A2) and midposterior (P2) leaflets of the mitral valve, the left ventricle with posterior and septal walls, the aortic valve with the right and noncoronary cusps, and the right ventricular outflow tract (Figure 1). The echocardiographic acceptance criteria for LVOT dimensions at the time of the conscious examination was between 22 and 25 mm to avoid oversizing in accordance of the chosen 21 mm aortic valve for implantation. The target for the LVOT diameter at the intraoperative, anesthetized echocardiographic examination was set between 21 and 25 mm. The conscious and anesthetized LVOT dimensions were obtained during midsystole (two frames after complete aortic valve opening by visual assessment) just proximal to and parallel with the plane of the aortic valve, from the inner edge of the septal endocardial echo to the leading edge of the base of the anterior mitral leaflet.
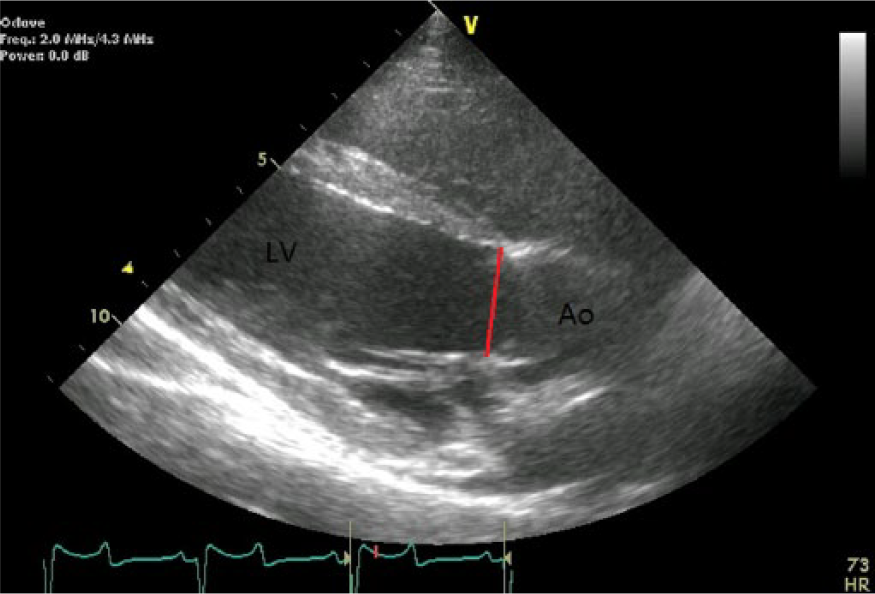
Representative image of the echocardiographic measurement of the left ventricular outflow track diameter. The line indicates the measurement from the inner edge of the septal endocardial echo to the leading edge of the base of the anterior mitral leaflet.
In the conscious examination, animals with an LVOT diameter of 22–25 mm (n = 29) were re-evaluated under general anesthesia just prior to aortic valve replacement surgery. The average number of days between the conscious evaluation and anesthetized evaluation was 34 days (21–87 days). For the anesthetized examination, each sheep was premedicated with morphine (0.05 mg/kg intramuscularly), induced for anesthesia with propofol (10 mg/kg intravenously), and maintained with isoflurane (1.5–2.5%) with mechanical ventilation assistance. In a right-lateral recumbency position on the operating-room table with a removable section in the operating table in order to access the chest wall, LVOT measurements were acquired in a right-parasternal long-axis view through the third to fifth intercostal spaces during an expiratory phase of the animal’s respiratory cycle. Two sonographers acquired both the conscious and anesthetized data and were blinded to any acquired previous data.
Statistics
Statistical data analyses were achieved using a computerized data analysis system (Minitab 16, Minitab Inc., State College, PA) using a paired t-test assessment.
Results
Of the 53 initially selected potential surgical candidates who underwent a conscious screening echocardiographic examination for the study, 32 animals were found to qualify within the protocol-established parameters (conscious LVOT dimensions of 22–25 mm). The values for the conscious LVOT and anesthetized LVOT dimensions for each animal are shown in Figure 2. Of the 32 that met the conscious LVOT diameter criteria, 29 underwent an anesthetized LVOT dimension assessment in the operating room prior to the surgical prosthetic valve implantation procedure. Only 3 of these 29 animals were found to be outside of the 21–25 mm target range, per the protocol (n = 1, 19.1 mm and n = 2, 26.7 mm and 25.2 mm). The mean dimension values for the conscious LVOT and anesthetized LVOT are presented in Table 1. The mean paired difference for the anesthetized and conscious LVOT diameter was −0.87 mm [p = 0.0066, standard deviation 1.598, 95% confidence interval (CI), −1.4796 to −0.26378, n = 29]. The difference plot for anesthetized and conscious LVOT dimensions is shown in Figure 3.

Individual values for the conscious left ventricular outflow track and anesthetized left ventricular outflow track (mm).
Preimplantation of valve diameter measurements in conscious and anesthetized adult sheep (n = 29, *p = 0.0066).
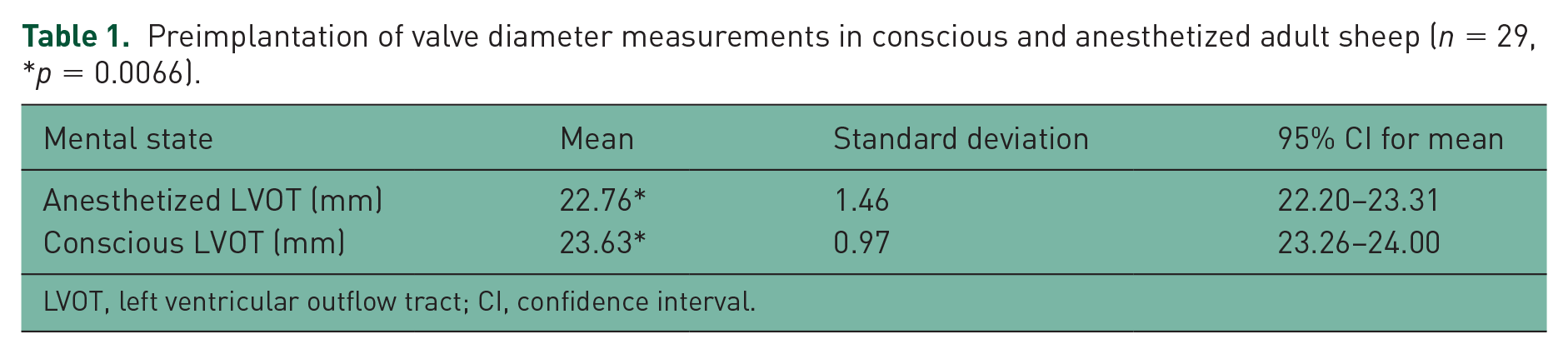
LVOT, left ventricular outflow tract; CI, confidence interval.

Difference plot for conscious and anesthetized left ventricular outflow track diameters.
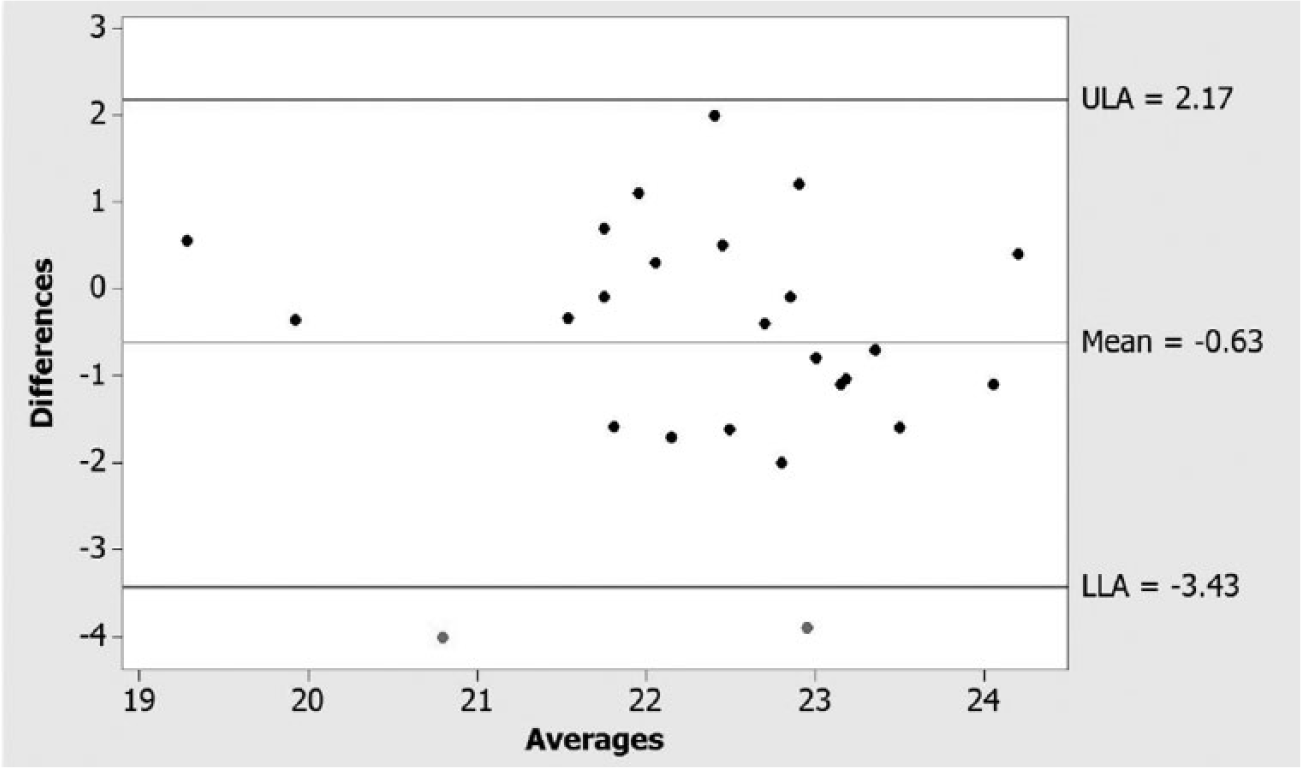
The paired results for the retrospective evaluation of interobserver variation are shown in Figure 4. The mean values from the animals examined by each sonographer were: 22.0 mm (standard deviation 1.309, 95% CI 21.503–22.583) and 22.7 mm (standard deviation 1.389, 95% CI 22.099–23.245). The mean paired difference was −0.63 mm (p = 0.038, standard deviation 1.43 mm, 95% CI −1.2 to −0.04).
Difference plot for retrospective sonographer image re-analysis.
Discussion
Preoperative anatomic evaluation of the LVOT measurements are important for determining correct annular size and LVOT acceptance for the implantation of a prosthetic aortic valve. Echocardiographic and MRI measurements have shown they are, and can be, most helpful in determining what acceptable prosthetic aortic valve size should be implanted, thereby minimizing the potential for size mismatching. We chose to use echocardiographic assessment preoperatively and intraoperatively prior to aortic valve implantation. Normal echocardiographic valvular measurements have been previously reported in sheep, which have included sedated 2D, m-mode and Doppler values [Poser et al. 2013], m-mode values in nonsedated animals [Moses and Ross, 1987], pulsed-wave Doppler-flow velocities and patterns [Kirberger and Van Den Berg, 1993], and left ventricular dimensions and indices and comparison with standardized human values [Locatelli et al. 2011]. Further, the employment of preoperative echocardiographic assessment in sheep has also been reported in pulmonary valve homograft implants and in chronic heart failure animal models [Hong et al. 2000; Borenstein et al. 2006].
Sheep were chosen for this preclinical study based on many factors, including a minimal age requirement. Our previously used standard for preoperative sheep selection involved an anesthetized echocardiographic assessment of the LVOT dimensions collected just prior to surgery that provided an additional data set for any prosthetic aortic valve implantation study. The potential benefit of performing this preoperative evaluation is helpful in assessing whether or not the animal’s anatomy would accommodate the chosen aortic valve size for implantation. If the animal’s anatomy is not appropriate for implant due to a discrepancy in aortic valve dimensions, as determined by this examination, the animal can be recovered from anesthesia without undergoing the surgical procedure. However, anesthesia can present risks. The animals that are anesthetized but found not appropriate for a surgical prosthetic valve implant incur stress from the anesthesia itself, as well as preoperative preparations and the resources expended for the delay in prosthesis-valve implantation. Due to these factors, we elected to refine the selection process and reduce the number of animals that need to at least undergo anesthesia for an echocardiographic examination. We hypothesized that the conscious echocardiographic values for the LVOT measurements may be similar to the anesthetized echocardiographic values in adult sheep. By investigating and determining if there is a correlation between conscious and anesthetized echocardiographic examinations, an improvement in surgical confidence that the animal is deemed a suitable candidate for a particular size of prosthetic valve for implantation prior to even subjecting the animal to anesthesia and surgery could be assumed.
Although conscious echocardiographic examination for LVOT dimensions cannot be used as a substitution for anesthetized echocardiographic LVOT assessment, it can be used as a technique refinement to preselect animals for anesthetized echocardiographic screening and potentially subsequent surgical prosthetic-valve implantation. We found a mean paired difference of −0.87 mm, with a 95% CI −1.4796 to −0.26378 (p = 0.0066), which is adequate for early and rapid determination of whether or not an animal is likely to have appropriately sized anatomy for further evaluation and surgical prosthetic-valve implantation. Three animals in the dataset had conscious and anesthetized LVOT differences outside of the upper and lower limits, according to the protocol criteria. The conscious examination values (conscious LVOT dimensions) tended to be larger than the anesthetized values. We elected, and continue, to set higher target values during conscious assessment (22–25 mm LVOT diameter for a 21 mm prosthetic-valve implant) as criteria for animals to be selected for anesthetized echocardiographic examination. The reasons for this difference were not apparent from the results of this preliminary study; however these differences may be related to animal noncompliance during the conscious echocardiographic examination or the noncircular nature of the aortic annulus and the single point measurement at each evaluation time point.
The acquired data from this study supports a larger aortic valve evaluation study to acquire anesthetized echocardiographic data on the animals that had conscious echocardiographic examination values outside of the target range. We recognize that this is a weakness of this evaluation as the animals outside of the target range during conscious examination may have a different correlation to the anesthetized echocardiographic examination. However we believe this design approach still has merit for the benefits in animal refinement by ensuring that animals presenting under anesthesia for anesthetized echocardiographic examination and surgery will have anatomy suitable for prosthetic aortic valve implantation. Of the population that underwent conscious echocardiographic evaluation, 60% (32 of 53 animals) were within the targeted size range. This is a refinement for our facility such that 40% of the animals have minimal manipulation (a single conscious echo examination) and resources expended (returned to farm-style housing). Of the sheep that met the conscious echocardiographic examination targets, 90% (26 of 29 animals) had anesthetized LVOT dimension values between of 21 to 25 mm and all (29 of 29 animals) could be implanted with the chosen study size of 21 mm aortic valve prosthesis.
Conclusion
In conclusion, conscious echocardiographic values for the LVOT diameter were similar to the anesthetized echocardiographic values for LVOT diameter in 90% of the surgical candidates and this assessment approach should be given consideration to help in identifying potential animals with appropriately sized anatomy prior to performing aortic valve implantation; thereby minimizing both operative mortalities and long-term morbidities and mortalities. Further, the potential financial benefits are obvious with the potential reduction in study mortalities, thereby decreasing the required number of study animals due to these avoidable deaths.
Limitations of the study
In this study, the interobserver measurement technique variability and the acquisition ability of the sonographers were not analyzed and are recognized limitations [Gottdiener et al. 2002]. Ideally, multiple data points of the LVOT diameter would have been acquired and averaged for each time point, as well as having both sonographers examine the same animal at the same time point until a statistically significant understanding of the differences between the two sonographers was obtained. The retrospective analysis of the intersonographer variation indicating a mean difference of 0.63 mm (p = 0.038, standard deviation 1.43 mm, 95% CI −1.2 to −0.04) does partially parse the contribution of multiple operators to the total variability. Retrospectively, unblinded review of the acquired datasets revealed limitations in the technical acquisition of data during the original examinations, such as not being aligned along the long axis of the valves, which may have influenced the results and reinforces the importance of consistency in the data acquisition. The limitations were not significant enough to warrant exclusion from the analysis.
Footnotes
Acknowledgements
The authors wish to thank Medtronic Structural Heart Disease Research for sponsoring this study and the Physiological Research Laboratories’ surgical staff for their superb support during this study.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
N. Barka, J. Bourgoin, and E. Grassl are employees of Physiological Research Laboratories, Medtronic PLC, and J. A. St. Cyr is a consultant for Medtronic PLC.
