Abstract
Objectives:
There is recent evidence that aldosterone play a role in the pathogenesis of cardiovascular disease in dialysis patients, which leads to the opportunity to block its actions for the benefit of these patients. In nondialytic chronic kidney disease, spironolactone was safe and effective in reducing left ventricular hypertrophy. However, routine use has been precluded in hemodialysis patients due to the risk of hyperkalemia. The aim of this study is to verify the safety and efficacy in regression of left ventricular hypertrophy with spironolactone in hemodialysis patients undergoing pharmacotherapeutic monitoring.
Methods:
We performed a controlled, randomized, double blind study evaluating 17 hemodialysis patients who received spironolactone at a dose of 12.5 mg titrated, in the second week, to 25 mg of spironolactone or placebo. The patients were treated for 6 months.
Results:
The groups were composed of eight patients (intervention) and nine patients (control). These groups did not differ in their baseline characteristics. The group receiving spironolactone had a left ventricular mass index reduction from 77 ± 14.6 g/m2.7 to 69 ± 10.5 g/m2.7, p < 0.04, whereas in placebo group there was an increase from 71 ± 14.2 g/m2.7 to 74 ± 17.4 g/m2.7. Systolic or diastolic blood pressure did not change during the study. Potassium did not differ statistically between groups in all instances.
Conclusion:
Spironolactone treatment in hemodialysis patients was secure and effective in regression of left ventricular hypertrophy, a major risk factor for cardiovascular events in these patients. This effect occurred in spite of blood pressure stability.
Trial registration:
ClinicalTrials.gov identifier NCT01128101
Introduction
Left ventricular hypertrophy (LVH) is a powerful predictor of mortality in patients with chronic kidney disease (CKD) [Martin et al. 2004] and also the most common cardiac abnormality [Foley et al. 1995; Zolty et al. 2008]. The regression of LVH has a positive effect on survival of hemodialysis patients [Fukuda et al. 2010]. However, the reduction of LVH in these patients is not easy to obtain because, apart from the difficulty in controlling its determinants (hypertension, volume overload and anemia) [Macedo and Oliveira, 2010], other factors may play an important role in the development of LVH such as activation of the renin–angiotensin–aldosterone system, oxidative stress, inflammation, growth factors [Becker et al. 2009] and inadequate elevated levels of aldosterone [Essick and Sam, 2011], namely, no suppression of aldosterone in spite of volume overload [Schrier, 2010].
In hypertensive patients and in the general population, the harmful effects of sodium overload are more evident with aldosterone excess [Acelajado et al. 2010]. Restricting salt intake would be much more logical to mitigate the deleterious effects on cardiac remodeling, but the difficulty in changing old habits makes this an unattainable target. Another possibility would be the utilization of mineralocorticoid receptor antagonists to block the harmful effects of excessive aldosterone levels [Lee et al. 2008].
Traditionally, the use of potassium-sparing diuretics in CKD patients has been avoided because of the high risk of hyperkalemia. Despite this, recent studies have shown the beneficial effect of spironolactone in the early stages of CKD (stages 2 and 3). Independently of its diuretic effect, the use of spironolactone at a dose of 25 mg/day showed a positive effect in reversing cardiac mass and cardiac mass index [Edwards et al. 2009]. Another study, which evaluated the efficacy and safety of spironolactone in hemodialysis patients with heart failure, showed that in addition to promoting regression of LVH, spironolactone was also able to reduce the risk of both cardiovascular morbidity and death among hemodialysis patients [Matsumoto et al. 2014].
Recently a systematic review analyzing six studies with mineralocorticoid receptor antagonists in hemodialysis patients confirmed the safety of the use of this drug class in relation to hyperkalemia. The authors suggested that hemodialysis patients are able to tolerate higher levels of potassium due to adaptive responses and because the hemodialysis process itself acts as a safety factor in maintaining kalemic levels [Chua et al. 2010].
No previous study had evaluated the safety and effectives of spironolactone to regress LVH specifically in hemodialysis patients with preserved systolic function. Therefore, the purpose of this work was to verify the safety and efficacy in reducing LVH with spironolactone in hemodialysis patients with pharmacotherapeutic monitoring.
Methods
Study design
The study constitutes a randomized, single-center, prospective, double-blind, placebo-controlled, interventional trial to evaluate the efficacy and safety of spironolactone in reducing the left ventricular mass of hemodialysis patients [ClinicalTrials.gov identifier: NCT01128101]. Patients were evaluated prior to inclusion, weekly for 4 sequential weeks, monthly thereafter, and at the end of study protocol. The protocol followed the 196/96 Resolution of National Health Council [Conselho Nacional de Saúde, 1996] and was approved by the local Ethics Committee Protocol (CEP 3439-2010). All patients gave their written informed consent. The spironolactone and placebo capsules were subjected to quality control testing for weight and the content [Ministério da Saúde, 2010].
Inclusion criteria
Patients on hemodialysis aged at least 18 years, who presented with a Left Ventricular Mass Index (LVMI) >51 g/m2.7, indexed for height, with stable antihypertensive treatment in the last 6 months.
Exclusion criteria
Dialysis dose measured by Kt/V <1.2; history or evidence of angina or myocardial infarction, heart failure, peripheral vascular disease, previous hyperkalemia, valvular heart disease, atrial fibrillation, hemoglobin <10g/dl, patients under treatment with spironolactone.
Fractionation of spironolactone and its placebo
Spironolactone used in this protocol was acquired in the market as Aldactone® 25 mg (reference product) [Congresso Nacional, 1999; Agência Nacional de Vigilância Sanitária, 2009] Laboratory Pfizer, lot AO26-1. The placebo tablets were produced at the Laboratory of Drugs, Faculty of Pharmaceutical Sciences, UNESP - Araraquara. Its composition consisted of colloidal silicon dioxide, corn starch, lactose monohydrate, microcrystalline cellulose and magnesium stearate, inert excipients in accordance with literature reference [Ferreira, 2010]. Both spironolactone or placebo tablets were broken with a tablet divider device and encapsulated in doses of 12.5 mg and 25 mg. All capsules tablets containing spironolactone or placebo at a dose of 12.5 mg were weighed, and 7.5% of the capsules tablets containing spironolactone or placebo at a dose of 25 mg were weighed. A criterion for approving the coefficient of variation in weight of less than 6% was adopted [Ministério da Saúde, 2010]. Capsules consisting of spironolactone and placebo were fractionated into bottles containing 30 capsules each, labeled and coded. Fractionation and coding were performed by an independent pharmacist’s research group.
Groups
Patients were divided into two groups: group A and group B (active drug and placebo, respectively) (Figure 1). Before inclusion, blood samples were collected for laboratory tests. Importantly, the patients had no changes in their prescriptions, except the addition of active drug or placebo, after clinical evaluation and laboratory tests performed before the second weekly session of hemodialysis. Patients began treatment after the next Sunday, initially at a dose of 12.5 mg/day; this dose remained for 2 weeks. Patient’s kalemic levels were measured before the second dialysis session of the week; those who were below 5.5 mEq/l had their doses titrated to 25 mg/day and underwent monitoring of their kalemic levels in the next two subsequent weeks, after which they were followed monthly. Patients who had serum potassium levels between 5.5 and 5.9 mEq/l were maintained at the dose of 12.5 mg/day. Patients whose potassium was ⩾6.0 mEq/ml were dropped from the study. All patients received pharmaceutical care and nutritional counseling additionally to that given routinely.
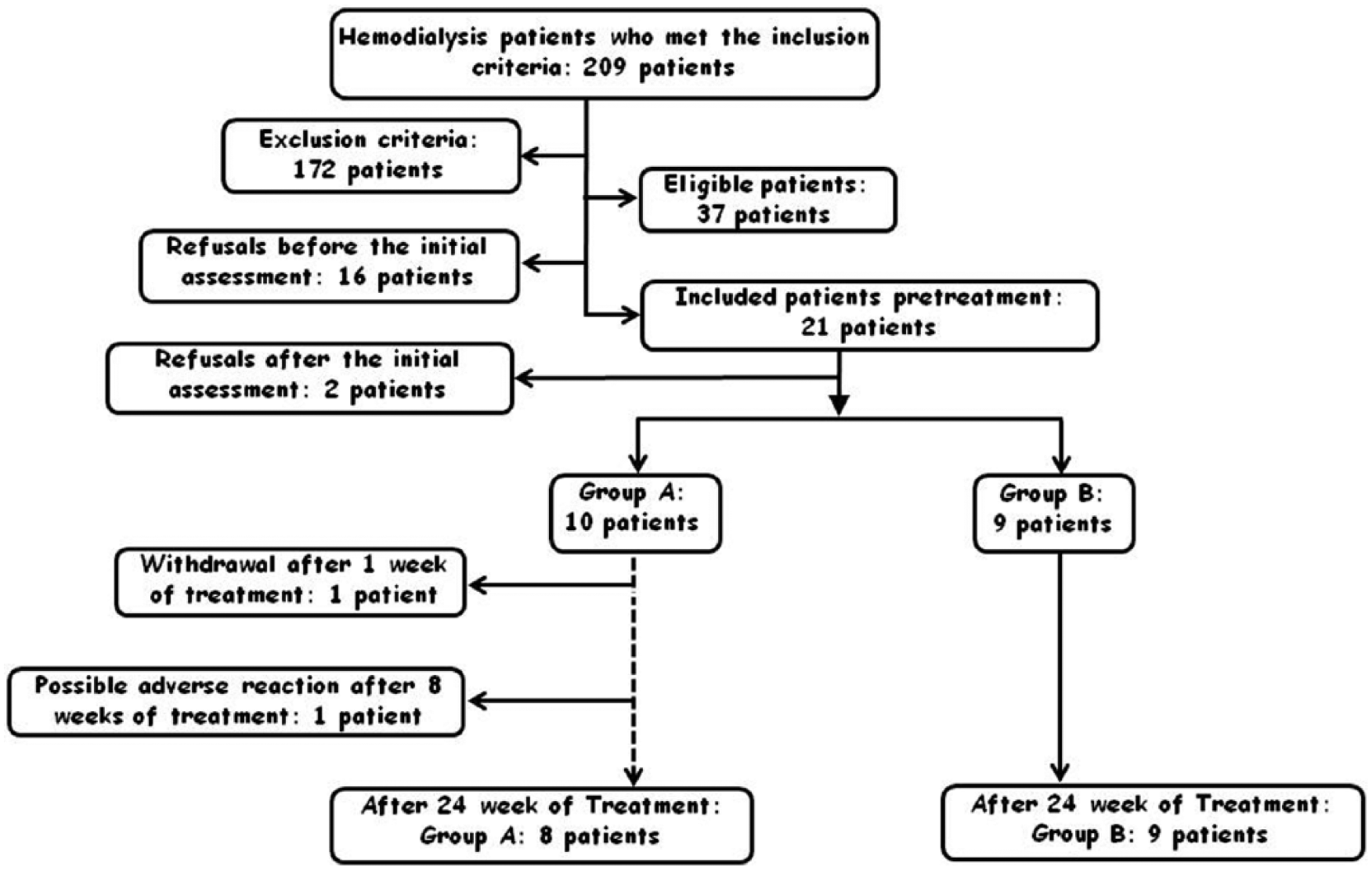
Flow chart of the selection process for inclusion and exclusion of patients in the clinical study.
Variables
Laboratory tests were performed according to standardized routine dialysis unit of the University Hospital of Botucatu, which follows the rules of operation of the hemodialysis established by the Brazilian Ministry of Health [Agência Nacional de Vigilância Sanitária, 2004]. Blood samples were collected immediately prior to the start of hemodialysis for routine hematology (red blood cells, white blood count and platelet count) and biochemical (urea, creatinine, potassium, calcium, phosphorus and alanine amino transferase) monthly. Quarterly measurements were made of levels of total proteins and fractions, alkaline phosphatase, transferrin, serum iron, ferritin, C-reactive protein, parathyroid hormone, lipid profile and, for diabetics, glycated hemoglobin. Monthly samples were collected pre and post dialysis for urea. The dose of dialysis was determined by the Daugirdas formula [Daugirdas, 1995]. Before starting treatment and after 24 weeks, blood samples for measurement of aldosterone were collected after the patient remained supine for 30 minutes (for the stabilization of plasma levels). The samples were centrifuged at 2500 revolutions per minute (rpm) and 4°C, and the plasmas were stored in Eppendorf tubes at −80°C for later analysis.
Echocardiography (ECHO)
Echocardiograms were performed at the Center for Diagnostic Imaging by a single examiner. The equipment used was a Vivid S6 GE (General Electric) equipped with a multifrequency ultrasonic transducer (2.0–3.5 MHz) and recording system. Images were obtained and analyzed according to the recommendations of the American Society of Echocardiography [Lang et al. 2005] on the interdialitic day [Martin et al. 2003]. The following data were recorded: heart rat; systolic and diastolic dimensions of the left ventricle; and the diastolic thickness of the posterior wall and septum.
These data were used to calculate ventricular mass, using the following formula:
where LVDD is the left ventricular diastolic dimension, and EDPW and EDS represent the thickness of the end-diastolic posterior wall and interventricular septum, respectively.
The left ventricular mass was normalized for height to the power 2.7 [Zoccali et al. 2001].
Ambulatory blood pressure monitoring
Ambulatory blood pressure monitoring (ABPM) was performed with a SpaceLabs 90202 monitor (SpaceLabs, Redmond, WA) in an inter-hemodialysis period that was 24 hours before the beginning of the next hemodialysis (24 hour ABPM). Examinations were considered valid if they fulfilled the following criteria [Sociedade Brasileira de Cardiologia et al. 2007]: minimum of 21 hours’ recording; and minimum number of valid measurements per hour, three during waking hours and two during sleep hours. The following parameters were considered for interpretation in the monitoring: blood pressure average over 24 hours; and magnitude of nocturnal drop for systolic, diastolic and pulse pressures.
Pulse wave velocity, augmentation index and central arterial pressure
These parameters were obtained by applanation tonometry using Sphygmocor® equipment (AtCor Medical, Sydney, Australia). The patient fasted on the day of examination and remained at rest for 5 minutes before the examination started. Analysis of the pulse wave velocity (PWV) was performed simultaneously with the verification of blood pressure. The readings were made by placing the forearm without arteriovenous fistula in support equipment; the radial artery was palpated and received the tonometer sensor. The shape of the central wave was generated by a validated transfer function. Speed and pulse waves were recorded for 10 seconds (after at least 20 recorded waveforms) at least twice, directly onto a laptop computer.
Bioelectrical impedance
We used the monofrequencial bioimpedance device (BIA) of Biodynamics®, model 450. The brand analysis of BIA is based on the measurement of total body resistance to the passage of an electric current of 800 μA and 50 kHz. The measurement was performed on the contralateral side of vascular access, with the patient in a supine position on a nonconductive surface; it was checked that their legs did not touch and that their arms did not touch their trunk. Weight, height, sex and age were recorded on the device. The values determined by the device are based on the calculation of equations proposed by Kushner and Schoeller [Kushner and Schoeller, 1986] and Cohn and colleagues [Cohn et al. 1985]. Resistance, reactance, phase angle, total body water, intracellular and extracellular water, lean mass and fat mass values were either determined or estimated.
Statistical analysis
The data were expressed as mean ± standard deviation for normally distributed variables and median (first and third quartiles) for the variables of nonparametric distribution. Clinical characteristics of patients were compared between groups by the ‘t’ test only at time zero. Frequencies were compared between groups using Fisher’s exact test. To evaluate the potassium levels, as well as for the analysis of the numerical variables that were evaluated during the study, we used two-way repeated measures analysis of variance when these variables had a normal distribution. For the others we used repeated measure design considering the effects of group, time and group versus time using a generalized linear model with gamma distribution followed by the multiple comparison tests for interaction group versus time. Data were expressed as mean ± standard deviation.
Results
The general characteristics of the patients are presented in Table 1. The groups did not differ in terms of age, sex, height and body weight. The groups were homogeneous with respect to duration of dialysis, body mass index, total body water, extracellular water, relationship between extracellular water and total body water, and use of angiotensin-converting enzyme inhibitors and/or beta-blockers.
Clinical characteristics of patients with chronic kidney disease on hemodialysis undergoing treatment with spironolactone or placebo for 6 months.

p, comparing all groups and moments.
ARB, angiotensin receptor blocker; ACEI, angiotensin-converting enzyme inhibitors; BMI, body mass index; ECW, extracellular water; TBW, total body water.
Initially, 10 patients were included in the spironolactone group and nine in the placebo group (Figure 1). Two patients from spironolactone group were withdrawn; one by abandonment and another by hyperkalemia not related to spironolactone (the decay to normal levels occurred only 3 months after the withdrawal of the drug).
Laboratory data are given in Table 2. Except for aldosterone, which decreased in the placebo group posterior to treatment, all laboratory variables tested were homogeneous between groups at baseline and moments within the groups.
Laboratory variables of patients with chronic kidney disease on hemodialysis undergoing treatment with spironolactone or placebo for 6 months.
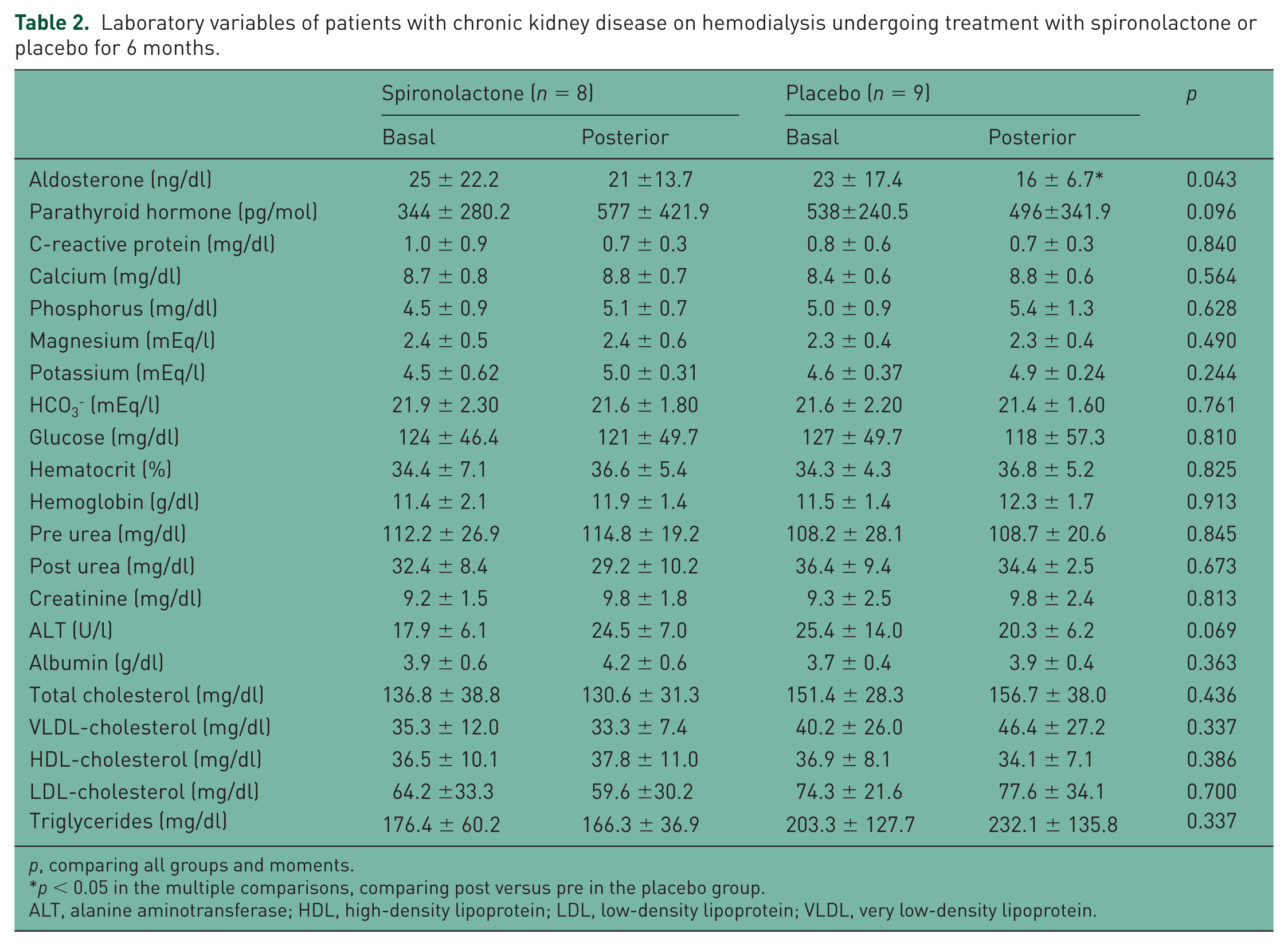
p, comparing all groups and moments.
p < 0.05 in the multiple comparisons, comparing post versus pre in the placebo group.
ALT, alanine aminotransferase; HDL, high-density lipoprotein; LDL, low-density lipoprotein; VLDL, very low-density lipoprotein.
Figure 2 shows the LVMI data for the spironolactone and placebo groups. The group receiving active spironolactone had a reduction in their LVMI from 77 ± 14.6 g/m2.7 to 69 ± 10.5 g/m2.7 (p = 0.039). In the placebo group, there was a different behavior with the LVMI, which increased from 71 ± 14.2 g/m2.7 to 74 ± 17.4 g/m2.7 (p > 0.05 ).

Left Ventricular Mass Index (g/m2.7) during 24 weeks of treatment with spironolactone or placebo.
The results of echocardiography, ABPM, PWV, augmentation index and central blood pressure are presented in Table 3. Left ventricular mass and posterior wall thickness decreased in the spironolactone group after treatment. When comparing pre and post treatment ABPM, pulse pressure in 24 hours and awake ABPM pulse pressure, there were a parallel decrease in both groups. The augmentation index was inferior in the placebo group in basal moment and maintained the same difference post treatment. All other parameters were homogeneous between groups and times.
Echocardiographic and ambulatory blood pressure monitoring data of patients with chronic kidney disease on hemodialysis undergoing treatment with spironolactone or placebo for 6 months.

p <0:05 in the multiple comparisons, comparing post versus pre in the spironolactone group.
Different superscript letters signify p < 0.05 in the multiple comparisons.
ABPM, ambulatory blood pressure monitoring; AI, augmentation index; DA, diastolic dimension of the atrium; DBP, diastolic blood pressure; EF, ejection fraction; LVDD, left ventricular diastolic dimensions; LVMI, Left Ventricular Mass Index; LVSD, left ventricular systolic dimension; IST, interventricular septum thickness; PP, pulse pressure; PWT, posterior wall thickness; PWV, pulse wave velocity; SBP, systolic blood pressure.
With regard to serum potassium levels (Table 2), the kalemic levels at the end of 24 weeks were not statistically different either between groups or between times in any evaluated moment (Figure 3).

Serum potassium levels during 24 weeks of treatment with spironolactone or placebo at the doses of 25 mg/day.
Discussion
The blockage of the aldosterone effect by spironolactone in LVH is a promising therapy for hemodialysis patients. Its use, however, has been precluded for fear of hyperkalemia [Chua et al. 2010]. The objective of this study was to determine whether the use of a mineralocorticoid receptor antagonist would reverse LVH in hemodialysis patients without heart failure. In the present study, spironolactone was effective in obtaining a favorable outcome.
Among the LVH promoting factors, aldosterone seems to have a direct trophic effect, in cardiomyocytes, additional to the hemodynamic effect [Becker et al. 2009; Fukuda et al. 2010; Schrier, 2010; Essick and Sam, 2011]. In the current study, the failure to obtain a statistically significant reduction in ABPM points in favor of this premise. The reduction of left ventricular posterior wall thickness, in the spironolactone group, corroborates this hypothesis. Moreover, importantly, the patients in this study were anuric, which would indicate that this effect on blood pressure could be distinguished from the diuretic effect of spironolactone.
In the genesis of uremic cardiomyopathy, the influence of endogenous cardioactive glycosides (endogenous ouabain and marinobufagenine), whose levels are elevated in renal failure, it has been postulated [Hamlyn et al. 1996; Bagrov and Shapiro, 2008]. The role of these glycosides in the pathogenesis of cardiomyocytes proliferation have been well investigated, but its effect in fibroblasts is less well known [Schoner and Scheiner-Bobis, 2008; Fedorova et al. 2010]. Moreover, canrenone, the active metabolite of spironolactone, adds a pharmacokinetic advantage by its ability to antagonize the binding of endogenous ouabain to Na+-K+-APAse in cardiac muscle [Finotti and Palatini, 1981].
To counteract the pathophysiological mechanisms triggered by these hormones in CKD, the use of a drug that blocks these deleterious effects on two fronts (aldosterone and endogenous ouabain effect) would be of great clinical importance. Moreover, a decrease of cytosolic intracellular concentrations of Na+ occurs with this blockade. Thus, the equilibrium is restored between the gradient of Na+/Ca++, reducing the influx of intracellular Ca++ and leading to a decrease in the vascular tonus [Blaustein et al. 2009]. Therefore, it is desirable to antagonize aldosterone through blockade of the mineralocorticoid receptor or to prevent the damaging effects of imbalance in sodium concentrations triggered via binding of endogenous cardioactive steroids to Na+-K+-APAse. Spironolactone therefore has a high potential for use in CKD because of these two mechanisms.
Spironolactone use promotes survival benefits in heart failure and potential renoprotection in CKD [Sica, 2007]. Left ventricular geometry is a good predictor of cardiovascular events, in either hypertension or CKD patients, and its regression showed a positive effect in prognosis of these patients [Levy et al. 1994; Zoccali et al. 2001]. Hence, LVH could be a surrogate endpoint in these patients predicting a better survival with this drug in dialysis patients. This premise is corroborated by the recent study by Matsumoto and colleagues [Matsumoto et al. 2014] which demonstrated a better survival with spironolactone use in dialysis patients.
Awake pulse pressure was reduced in both groups, in a parallel fashion; if cardiac mass decrease occurred only in the spironolactone group, it is improbable that pulse pressure reduction was the cause of mass regression. In the present work, although not statistically significant, the paradoxical relative rise in blood pressure during sleep in the intervention group should be noted. This may possibly be explained by the pharmacokinetics of spironolactone, whose halflife is approximately 10 hours, whereas the halflife of canrenone is 17 hours [Benet et al. 1995]. It is of note that our patients took the drug in a unique take in the morning. We can speculate that the use of this drug twice a day would prevent this phenomenon.
Regarding the statistically significant difference in the augmentation index between the groups, we emphasize that it occurred without a reduction in this index in both groups and that the spironolactone group showed the higher augmentation index, indicating an unfavorable clinical situation in relation to the placebo group, since the augmentation index indicates greater arterial stiffness [Taylor et al. 2014]. Thus, in spite of this unfavorable effect to cardiac mass regression in the spironolactone group, this group demonstrated a reduction in LVMI. This bias corroborates the conclusion rather than being against it.
The stable potassium behavior points favorably to the safety of spironolactone in hemodialysis patients. However, it is important to stress that these patients were submitted to rigorous pharmacotherapeutic monitoring throughout the treatment.
The homogeneity and stability of the laboratory data is an important finding, because these variables could influence cardiac structure. If, for example, parathyroid hormone or hemoglobin levels had dropped in the spironolactone group, this could explain the obtained results for LVMI, instead of a spironolactone effect on aldosterone. So we can say that these factors are not important in cardiac mass behavior because of the stability demonstrated in the laboratory data. Aldosterone also showed a drop in the placebo group post treatment. Thus, in spite of this favorable situation of cardiac mass regression in the placebo group, this group did not demonstrate any LVMI reduction. This bias also corroborates the conclusion rather than being against it.
A limitation in our study must be acknowledged. The results are related to a small number of patients; however, the representative status is evidenced by the uniformity in the characteristics of the groups. Undoubtedly, more studies should be conducted to confirm our results, but this work paves the way for multicenter projects with a larger number of hemodialysis patients. The MiREnDa study [Hammer et al. 2014], not yet completed, is a prospective, randomized, placebo-controlled, double-blind, multicenter study which presents a research proposal similar to our study; its authors hope to detect a difference in LVMI between the intervention group and the control group of 7 g/m2. Their results would to corroborate our findings.
Conclusion
The use of spironolactone in hemodialysis patients is effective in reversing LVH, a major risk factor for cardiovascular events in these patients. This reinforces the idea of the use of spironolactone therapy for antagonizing the harmful effects of aldosterone on the heart of these patients.
Footnotes
Acknowledgements
Thanks are given to those people who, without their cooperation, it would not have been possible to perform this study: the multidisciplinary staff of the Dialysis Unit, Hospital das Clinicas, UNESP – Botucatu, São Paulo; and the multidisciplinary staff of the Laboratory of Molecular Biology, Hospital das Clinicas, UNESP – Botucatu, São Paulo.
Funding
We thank FUNDUNESP (Foundation for the Development of UNESP, Process 0090910) and FAPESP (Foundation for Research Support of São Paulo, Process 2010/10439-1) for financial support.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.

