Abstract
Objectives:
The newborn circulating, cardiac and renal renin–angiotensin systems (RASs) are essential for blood pressure control, and for cardiac and renal development. If cardiac and renal RASs are immature this may contribute to cardiovascular compromise in preterm infants. This study measured mRNA expression of cardiac and renal RAS components in preterm, glucocorticoid (GC) exposed preterm, and term piglets.
Methods:
Renal and cardiac RAS mRNA levels were measured using real-time polymerase chain reaction (PCR). Genes studied were: (pro)renin receptor, renin, angiotensinogen, angiotensin converting enzyme (ACE), ACE2, angiotensin type 1 receptor (AT1R) and angiotensin type 2 receptor (AT2R).
Results:
All the genes studied were expressed in the kidney; neither renin nor AT2R mRNA were detected in the heart. There were no gestational changes in (pro)renin receptor, renin, ACE or AT1R mRNA levels. Right ventricular angiotensinogen mRNA levels in females were lower in preterm animals than at term, and GC exposure increased levels in male piglets. Renal angiotensinogen mRNA levels in female term piglets were lower than females from both preterm groups, and lower than male term piglets. Left ventricular ACE2 mRNA expression was lower in GC treated preterm piglets. Renal AT2R mRNA abundance was highest in GC treated preterm piglets, and the AT1R/AT2R ratio was increased at term.
Conclusions:
Preterm cardiac and renal RAS mRNA levels were similar to term piglets, suggesting that immaturity of these RASs does not contribute to preterm cardiovascular compromise. Since preterm expression of both renal and cardiac angiotensin II-AT1R is similar to term animals, cardiovascular dysfunction in the sick preterm human neonate might be effectively treated by agents acting on their RASs.
Keywords
Introduction
Preterm infants frequently suffer from cardiovascular compromise which is characterized by hypotension and/or low systemic flow [Osborn, 2005]. This occurs about 6–24 hours after birth; most commonly in male infants that have not been treated antenatally by maternally administered glucocorticoids (GCs) [Moise et al. 1995]. Treatment of poor cardiovascular function is important because it is associated with poor neurodevelopmental outcome. However, treatment is difficult and almost half of affected infants have a poor response to the commonly used inotropes dopamine and dobutamine [Osborn et al. 2007; 2008; 2010].
Previously we have shown that the preterm pig heart studied at 91 days gestation (developmentally equivalent to 23–25 weeks human gestation, [Eiby et al. 2013]) (term 115 days) is structurally immature relative to the term heart, in that it has a smaller proportion of terminally differentiated ventricular cardiomyocytes, a high level of proliferative activity and a low level of apoptosis [Kim et al. 2014a]. These structural changes are associated with an inability to maintain aortic blood flow when afterload is increased or preload is reduced [Eiby et al. 2012; Kim et al. 2014a] and therefore could contribute to cardiovascular compromise. In addition, the preterm pig has a lower level of expression of cardiac β-adrenoceptors [Kim et al. 2014], potentially limiting its response to both endogenous and exogenous catecholamines. This could partially explain the poor response of many neonates to the inotropes, dopamine and dobutamine [Osborn et al. 2010]. The renin–angiotensin system (RAS), if it is expressed in the preterm infant, therefore might be a better target for treatments to improve cardiovascular function because it may have beneficial effects on cardiac and renal development as well as positive effects on cardiovascular homeostasis.
The circulating RAS plays a key role in cardiovascular and fluid and electrolyte homeostasis. It has been known for many years that the circulating RAS plays a more significant role than the sympathetic nervous system in maintaining blood pressure in altricial neonates because the autonomic nervous system is immature at birth [Gootman et al. 1992; Yu and Lumbers, 2002]. As well, tissue RASs are essential for normal organ growth and development [Beinlich et al. 1991; Tufro-Mcreddie et al. 1995; Choi et al. 2002]. An intact renal renin-angiotensin system is essential for normal renal development [Tufro-Mcreddie et al. 1995] while the intracardiac RAS influences cardiomyocyte growth and organization [Sadoshima and Izumo, 1993].
Active renin, which is able to generate angiotensin I (Ang I) from angiotensinogen, is only secreted by the kidney. Ang I can also be formed from angiotensinogen when inactive prorenin binds to a (pro)renin receptor exposing its catalytic site. The prorenin–(pro)renin receptor also has actions independent from the formation of Ang I [Nguyen et al. 2002] in the kidney and heart. Ang I is mainly converted to angiotensin II (Ang II) by angiotensin converting enzyme (ACE).
Ang II acts via two major pathways. The actions of Ang II acting via its type 1 receptor (AT1R) include vasoconstriction, hypertension, enhanced efferent sympathetic nerve activity and aldosterone release [De Gasparo et al. 2000]. This RAS pathway is also proinflammatory and proliferative [De Gasparo et al. 2000]. Normal growth of the neonatal pig heart and of the neonatal rat heart is disrupted by ACE blockade [Beinlich et al. 1991; Choi et al. 2002] as is normal growth of the kidney. (Pro)renin may also induce cardiac hypertrophy via prorenin receptor in an angiotensin independent manner [Saris et al. 2006].
Ang II also acts on an angiotensin type II receptor (AT2R), as does angiotensin-(1-7) (Ang-(1-7)). Ang-(1-7) is mainly produced by ACE2 from Ang II, and acts via the Mas receptor (MasR). These two pathways (i.e. Ang II/AT2R and Ang-(1-7)/MasR) largely oppose the effects of the Ang II–AT1R interaction. Their actions include vasodilation, hypotension, natriuresis, anti-inflammatory and proapoptotic effects [Santos et al. 2013]. Thus there are two major and opposing arms in the RAS cascade that could, through their very different actions, influence the circulatory status of the preterm neonate, and the growth and development of the kidney and heart.
Antenatal administration of GCs improves lung function of preterm infants by stimulating surfactant production and structural maturation [Robertson, 1993]. We have recently shown that maternal betamethasone treatment matured cardiac structure [Kim et al. 2014b] and improved aortic blood flow in the isolated preterm piglet heart [Eiby et al. 2012]. There is a powerful interaction in the preterm animal between GCs and RAS, with GCs not only improving the capacity of the sympathetic nervous system to release renin [Segar et al. 1998], but also enhancing the pressor activity of Ang II [Tangalakis et al. 1992]. In addition, both AT1R and AT2R have GC response elements in their promoter regions [Xue et al. 2014]. The interactions in the neonate of antenatal GCs with the cardiac and renal RASs and those pathways that are associated with Ang-(1-7) production are unknown.
To determine which components of the cardiac and renal RASs are present in the preterm neonate and may be suitable as a novel therapeutic target to support preterm cardiovascular function, we examined the expression of RAS genes in the preterm piglet heart and kidney at 91 days and at term. The effects of maternally administered GCs on the expression of cardiac and renal RAS genes, and potential sex-specific effects were also measured.
Materials and methods
The project was approved by the University of Queensland Animal Ethics Committee (AEC Approval Number: UQCCR/999/08) and conforms to the Australian Code of Practice for the Care and Use of Animals for Scientific Purposes (7th edition, 2004) and the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication No. 85-23, revised 1996). All surgery was performed under anesthesia and all efforts were made to minimize suffering.
Animals
Large White X Landrace piglets were delivered by caesarean section at two ages, preterm piglets delivered at 91 days (term is 115 days) and term piglets delivered 2 days before the expected farrowing date. An additional group of preterm piglets was exposed to maternally administered GCs (preterm + GC, betamethasone, 0.19 mg/kg bodyweight, applied intramuscularly (i.m.); Celestone Chronodose, Schering-Plough, USA) given 48 and 24 hours before delivery. The timing and dose/kg are equivalent to that given to women presenting with threatened preterm labor [Eiby et al. 2013].
Caesarean delivery was performed as described previously [Eiby et al. 2013]. Piglets were sequentially removed from the uterus and anaesthetized with ~5 mg/kg propofol (Provive 1%; AFT Pharmaceuticals, New Zealand) via an umbilical vein. The umbilical cord was clamped and cut, and the piglet was immediately weighed and sexed. The chest and abdominal cavity were opened and the heart and kidneys rapidly excised. The excised heart was weighed, dissected and placed into modified Krebs solution [Kim et al. 2014a, 2014b]. A small (~ 0.5 cm × 0.5 cm) block of myocardium extending from epicardium to endocardium was obtained at the corner of interventricular and atrioventricular grooves [posterior for left ventricle (LV) and anterior for right ventricle (RV)] of each ventricular free wall. The heart tissue block and a kidney were snap frozen in liquid nitrogen and crushed into fine power. After all the piglets were delivered, the sow was euthanized by intravenous (IV) injection of pentobarbital sodium (60 ml; Lethabarb, Virbac, Australia).
Quantitative real-time reverse transcription coupled polymerase chain reaction
An aqueous solution containing total RNA was separated from crushed tissue (25~50 mg) using Qiazol (Qiagen, Germany) according to the manufacturer’s instructions. Samples were then further purified using an RNeasy mini RNA extraction kit (Qiagen, Germany) with an on-column DNase I treatment using a DNase free RNase kit (Qiagen, Germany). The enriched mRNA was then subject to an additional DNase I treatment using a Turbo DNase free kit (Life Technologies, Ambion) to minimize genomic DNA content. The integrity and concentration of each RNA sample was confirmed before and after the additional DNase treatment using 1% agarose gel electrophoresis and absorbance spectrometry (NanoDrop1000, Thermo Fisher Scientific Inc.). Each mRNA sample was spiked with 1 × 107 copies of Alien RNA (StrataGen Systems, La Jolla, CA, USA) per microgram total RNA which served as the reference RNA for internal standardization [Gilsbach et al. 2006]. Enriched RNA from each sample was then reverse transcribed using a high capacity RNA-to-cDNA kit (Life Technologies, Applied Biosystems).
Real-time polymerase chain reaction (PCR) was performed in a Mastercycler® ep Realplex2 (Eppendorf South Pacific, Germany) using custom designed primers for renin (REN), ACE type 2, a homolog of ACE (ACE2) [Vickers et al. 2002], and the two angiotensin II receptors (type 1, AGTR1 and type II, AGTR2; Table 1) and primer sequences published previously [for the (pro)renin receptor (ATP6AP2) for angiotensinogen (AGT) and ACE, see Table 1]. The custom primer pairs were designed using Primer Express software (Applied Biosystems). Every primer pair was subjected to a validation experiment where the efficiency of the target amplification and the efficiency of the reference (Alien mRNA) amplification were tested for equality. Each reaction contained 12.5 μl of Power SYBR green PCR master mix (Applied Biosystems) and primers (listed in Table 1), with a final volume of 25 μl per reaction. The data obtained were analyzed using the comparative CT method (2-ΔΔCT method) as described previously [Livak and Schmittgen, 2001; Schmittgen and Livak, 2008].
Primer pairs used for real-time PCR.

Fwd/S, forward/sense; Rev/AS, reverse/antisense; PCR, polymerase chain reaction.
Briefly, the amount of mRNA for each gene was normalized to the internal standard (Alien) and then expressed relative to a sample obtained from pooled male and female term kidneys (2-ΔΔCT). Dissociation curves for validation of homogeneity of amplification products were generated for all reactions and no-template (for confirming the absence of non-specific amplification products due to primer interactions), and no-enzyme control samples (for testing contamination of genomic DNA) were included in all assays. The predicted sizes of the PCR products were verified by agarose gel electrophoresis (data not shown). Intra-assay coefficient of variation (%) for each gene was (pro)renin receptor (ATP6AP2): 8.73 ± 1.8, REN: 2.17 ± 0.77, AGT: 2.20 ± 0.32, ACE: 4.68 ± 0.70, ACE2: 7.18 ± 1.9, AGTR1: 12.6 ± 2.0 and AGTR2: 9.74 ± 3.2 [mean ± standard error of the mean (SEM)].
Data analysis
Data were analyzed using SPSS (version 21, IBM, Chicago, USA). Homogeneity of variance and normality of data were assessed using Levene and Shapiro–Wilk statistics. Where necessary, data were then transformed using either logarithmic (ATP6AP2, ACE, ACE2, AGTR1 in the LV; REN and ACE2 in the kidney) or square root (AGTR1 in the kidney) functions. Differences in both morphology and mRNA expression levels between groups (untreated preterm, GC treated preterm and term), and between males and females within a group, were detected using three-way analysis of variances (ANOVAs) (where group and sex were fixed factors and litter was a random factor nested in group). Differences in mRNA expression levels between tissues (LV, RV and kidney) were detected using repeat measures ANOVAs (where tissue and sex were fixed factors and litter was a random factor) separately for untreated preterm, maternal GC treated preterm and term groups. For all analyses, significant differences are reported only where these existed independently of litter effects.
Within each group, Spearman’s ranked-order correlation tests were used to analyze relationships between genes within tissues. Results are expressed as median and interquartile range (IQR). Statistical significance was set at p < 0.05 unless specified otherwise.
Results
Animals and RAS mRNA detected
For each group, untreated preterm, GC treated preterm and term piglets, a total of 12 piglets (6 males, 6 females) from 3 litters were studied except in untreated preterm piglets, n = 11 (6 males, 5 females) for RV; GC treated preterm piglets, n = 8 (4 males, 4 females) for kidney; and term piglets, n = 11 (5 males, 6 females) for both ventricles, n = 8 (4 males, 4 females) for kidney.
Previously we have described the effects of gestation on body size and organ weights [Kim et al. 2014]. Table 2 reports body, heart and kidney weights, and these organ weights relative to body weight.
Body and organ weights of piglets.

Preterm piglets (91 days), with and without maternal glucocorticoids (GC) treatment, and term piglets (113 days, term is 115 days).
Data are mean (standard deviation). n is 12 (6 male and 6 female) for all groups, except for kidney values where n = 11 for the untreated preterm group and n = 8 for both the GC treated preterm and term groups.
Kidneys weight and Kidneys:body weight values are for the combined left and right kidney weights.
indicates a significant increase in the term group compared with both preterm groups.
GC, glucocorticoid.
All RAS genes studied [(pro)renin receptor, renin, angiotensinogen, ACE, ACE2, AT1R and AT2R] were expressed in all kidneys (Table 3). In piglet hearts, neither renin nor AT2R mRNA were detected; however, all the other RAS genes were detected in piglet hearts.
mRNA abundances of RAS genes in the left ventricle (LV), right ventricle (RV) and kidney; measured in untreated preterm piglets (91 days gestation), maternal glucocorticoid (GC) treated preterm piglets (91 days gestation) and term piglets (115 days is term).

Genes: (Pro)renin receptor (ATP6AP2), renin (REN), angiotensinogen (AGT), angiotensin converting enzyme types 1 (ACE) and 2 (ACE2), angiotensin II receptors type 1 (AGTR1) and type II (AGTR2).
Values are median (interquartile range) fold changes relative to term kidney.
n is 12 (6 male and 6 female) for all tissues in all 3 groups except: untreated preterm piglets, n = 11 for RV; GC treated preterm piglets, n = 8 for kidney; and term piglets, n = 11 for LV and RV, n = 8 for kidney.
Indicates the GC treated preterm group is different to the other two groups.
Indicates the term group is different to the other two groups.
Different to kidney in GC treated preterm and term groups only.
Different to kidney in all groups.
Different to kidney in GC treated preterm group only.
LV, left ventricle; ND, not detected; RAS, renin–angiotensin systems; RV, right ventricle.
Effects of gestational age, GC treatment and sex on RAS mRNA expression
(Pro)renin receptor, renin, ACE and AT1R mRNA levels were unaffected by gestational age, GC treatment or sex. The RAS genes affected by gestational age, GC treatment or sex were angiotensinogen, cardiac ACE2 and renal AT2R.
There was an interaction between group and sex on angiotensinogen expression in the RV such that levels in preterm females appeared lower than in term animals, and GC exposure appeared to increase levels in male piglets (Figure 1, g*s: p = 0.028). There was an interaction between group and sex on renal angiotensinogen expression such that levels in females from both preterm groups appeared higher than term females, and levels in male term piglets appeared higher than in term females (Figure 2, g*s: p = 0.046). There were no effects or interactions between gestation and sex on left ventricular angiotensinogen.

Expression of angiotensinogen (AGT) mRNA in the right ventricle of female (F-dots) and male (M) piglets from the three groups: untreated preterm, maternal glucocorticoid (GC) treated preterm and term piglets.

Renal angiotensinogen (AGT) mRNA levels in female (F-dots) and male (M) piglets from the three groups: untreated preterm, maternal glucocorticoid (GC) treated preterm and term piglets.
Left ventricular ACE2 mRNA was lower in the GC treated preterm animals compared with both untreated preterm (p = 0.016) and term (p < 0.001) animals (Figure 3). There was no effect of sex. There were no effects or interactions between gestation and sex on right ventricular or renal ACE2.
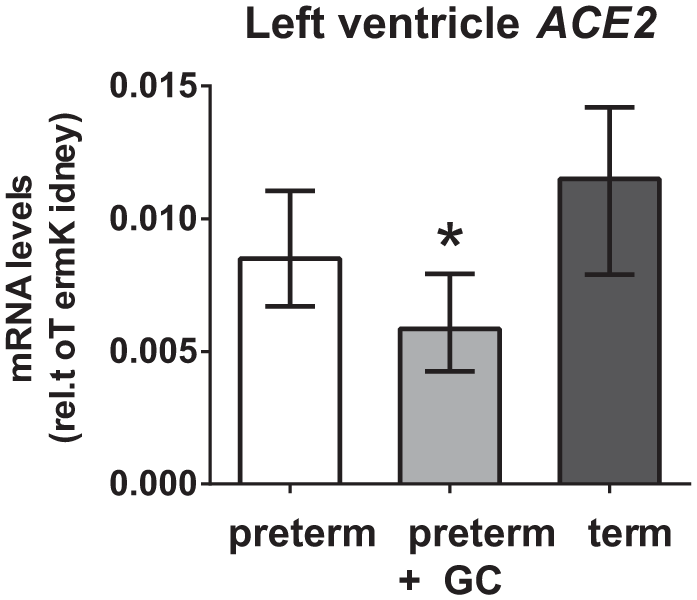
Left ventricular angiotensin converting enzyme 2 (ACE2) mRNA levels of untreated preterm, maternal glucocorticoid (GC) treated preterm and term piglets (male and female combined).
Renal AT2R mRNA was higher in the GC treated preterm group compared with both the untreated preterm (p < 0.001) and term (p < 0.001) groups (Figure 4a). There were no effects of sex, or interactions between gestation and sex. AT2R mRNA was not detected in cardiac tissue.
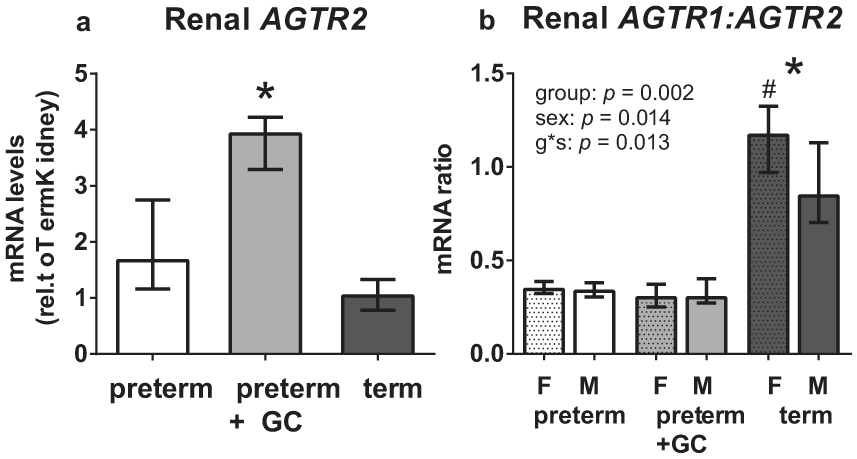
Renal mRNA levels of (a) AT2 receptor (AGTR2) (male and female combined), and (b) the AT1R/AT2R ratio (AGTR1:AGTR2) in females (F-dots) and males (M), for untreated preterm (6F + 6M), maternal glucocorticoid (GC) treated preterm (4F + 4M) and term (4F + 4M) piglets.
The AT1R/AT2R ratio in the kidney was lower in both preterm groups than in the term group, and was not affected by GC treatment (p < 0.001). The significant interaction between group and sex showed that this gestational effect was stronger in females than in males (p = 0.013) (Figure 4b).
Differences in RAS gene expression between tissue types
Right and left ventricular mRNA levels were similar for all the RAS genes expressed in the myocardium (Table 3). Angiotensinogen mRNA levels were higher in the RV than in the kidney in all groups, but LV expression was higher than kidney only in GC treated preterm and term animals (Table 3). For all groups, ACE and ACE2 expression was lower in the RV than in kidney, but only in GC treated preterm animals were (pro)renin receptor and AT1R levels lower in the RV than kidney (Table 3). For all piglet groups, (pro)renin receptor, ACE, ACE2 and AT1R mRNA levels were lower in the LV than in the kidney (Table 3).
Patterns of expression of RAS genes
To see if renin secretion is controlled constitutively or via physiological control mechanisms, we correlated renin mRNA abundances in kidney with abundances of other RAS genes, separately in each group. In untreated preterm kidney, renin expression was correlated with (pro)renin receptor (rho = 0.89, p < 0.001), ACE (rho = 0.72, p = 0.008), AT1R (rho = 0.85, p < 0.001) and AT2R (rho = 0.90, p < 0.001) expression. These correlations were not present in GC treated preterm or term kidneys.
Discussion
The aim of this study was to measure the expression of the cardiac and renal RASs in the preterm and term piglet to determine if these systems have a similar level of maturity in preterm and term piglets. The study also examined the effects of GCs on the expression of these RAS genes to determine if these could account for the known beneficial effects of maternal antenatal GCs on preterm cardiovascular function. The preterm pig was studied at 91 days gestation – an age when cardiovascular development is equivalent to a 23–25 week human preterm neonate [Eiby et al. 2013; Kim et al. 2014].
As there were few gestational changes in RAS mRNA levels, it would seem that the cardiac and renal RAS mRNA levels are as mature at 91 days gestation as they are at term. Therefore, it is not surprising that antenatal treatment with GCs had only minor effects on expression of RAS genes in the preterm piglet. Significantly, in terms of circulatory homeostasis neither gestation nor GC treatment affected the expression of renin in the kidney.
Release of active renin into the circulation regulates the activity of the circulating RAS which is essential to maintain newborn cardiovascular function. In the newborn rabbit, the circulating RAS is active at birth and levels of Ang II-like activity in newborn rabbits are similar to or higher than adult levels [Broughton Pipkin et al. 1971]. In neonatal rabbits, the ability to maintain blood pressure during hemorrhage depended on the presence of the kidneys; only later in life did the response to hemorrhage become dependent upon the sympathetic nervous system [Mott, 1969]. Therefore, in neonates, the RAS plays a more crucial role in cardiovascular control than the sympathetic nervous system. In piglets [Gootman et al. 1992; Dong et al. 2011] and probably also human infants, since sympathetic control of the cardiovascular system is immature neonates may depend more upon the activity of the renal RAS than the sympathetic system for maintaining cardiovascular function.
Just as renin is the key component of the RAS in regulation of day-to-day production of Ang II, so renal AT1Rs may be critical in maintaining blood pressure in the very preterm neonate. It has become apparent that renal AT1Rs, in particular, play a major role in maintaining blood pressure [Coffman and Crowley, 2008; Gurley et al. 2011]. In mice, ‘knockout’ of the renal AT1R has the same blood pressure lowering effect as ‘knocking’ out all the AT1Rs in the systemic vasculature, while ‘knocking out’ both kidney and systemic AT1Rs causes even more profound hypotension. When Ang II is infused into these mice, in which only systemic AT1Rs were knocked out and the renal ATIRs were intact, blood pressure increased to levels similar to those seen in wildtype mice. However in ATIR renal knockouts (in which systemic ATIRs were intact), Ang II failed to increase blood pressure. These findings show that the component of renal vascular tone that is regulated by Ang II–ATIR interactions plays a significant role in maintenance of blood pressure. This could be particularly important for survival of preterm animals because neural control of the circulation is immature [Gootman et al. 1992; Segar et al. 1998]. Since expression of both renal renin and renal and cardiac AT1R mRNA were similar in preterm and term piglets, their poor cardiovascular function [Eiby et al. 2012, 2014] is not due to a lack of mRNA expression of either of these genes.
Ang II interacting with its AT2R, and Ang-(1-7) interacting with both the AT2R and its G-protein coupled Mas receptor, act synergistically to oppose the actions of Ang II mediated via its AT1R. The renal AT2R stimulates nitric oxide (NO) production and guanosine monophosphate (cGMP), a vasodilator– natriuretic pathway, and thus opposes the pressor actions of the AT1R [Carey et al. 2000]. Furthermore, cardiac AT2Rs oppose recovery of mechanical function after ischemia-reperfusion while cardiac AT1Rs enhance it [Ford et al. 1996]. Thus the higher level of expression of renal AT2R and a lower AT1R/AT2R ratio in the untreated preterm piglet (Figure 4) may adversely affect cardiovascular function. This low renal AT1R/AT2R ratio found in preterm piglets is similar to that found in fetal sheep; thus it is very likely that similar ontogenetic changes occur in the human kidney [Robillard et al. 1995]. This altered pattern of renal expression of Ang II receptor subtypes and presumably proteins (personal communication, E.R. Lumbers) in preterm neonates may offset the pressor effect of Ang II–AT1R interactions [Coffman and Crowley, 2008; Gurley et al. 2011] and so compromise preterm blood pressure. Indeed in young rats the pressor effects of Ang II are decreased because of the higher renal expression of AT2R [Brown et al. 2014].
Antenatal GC treatment was associated with a marked increase in renal AT2R expression (Figure 2a), although overall there was no effect of GC treatment on the ratio of expression of the two Ang II receptors (Figure 2b). Whether or not this means that GC treated neonates are more likely to have a smaller blood pressure response to Ang II, and more likely to have a natriuresis, remains to be investigated. Interestingly, GC treated preterm piglets compared with untreated preterm and term piglets have a greater and more sustained increase in skin blood flow in response to hypoxia, suggesting a weaker vasoconstrictor response to Ang II [Eiby et al. 2014].
The conversion of Ang II to Ang-(1-7) by ACE2 is a major pathway for production of Ang-(1-7). The expression of renal ACE2 was unaffected by gestation, sex or GC treatment, although this was not the case for cardiac ACE2; left ventricular ACE2 expression was decreased in GC treated preterm piglets. A reduction in the proapototic Ang II/ACE2/Ang-(1-7)/MasR pathway in GC exposed neonates could promote cardiac growth and could contribute to the better cardiovascular function observed in GC exposed piglets and infants [Moise et al. 1995; Eiby et al. 2012, 2014].
If a tissue RAS is to affect cellular function, the effective production of Ang peptides, which requires the expression of a number of genes (renin, angiotensinogen, ACE, ACE2) may well depend on a coordinated pattern of expression of these genes. In addition there needs to be concomitant expression of appropriate receptors. The renal RAS is essential for normal renal development; blocking its activity by blocking formation of Ang II or its interaction with the AT1R causes gross structural defects [Tufro-Mcreddie et al. 1995]. So perhaps it was not surprising that we found a number of significant correlations between levels of expression of renin and other RAS genes in the preterm kidney. The loss of this pattern of coordinated expression in the term kidney may represent increasing influence of physiological control systems, such as the renal baroreceptor, macula densa and sympathetic stimulation nerves, on renin expression and secretion [Henrich et al. 1979]. GC treatment also resulted in loss of these correlated patterns of renal RAS gene expression. This could be the result of maturation of pathways that regulate renin release into the circulation; however, it could have effects on continuing renal development if switched on too early.
We were not able to detect either renin or AT2R mRNA in the preterm or term pig heart (Table 3). Renin can, however, be taken up by the heart from the circulation and kidney, or its activity might be supplemented by other myocardial proteases [Hackenthal et al. 1978; Katwa et al. 1997]. In addition, circulating (pro)renin and renin can bind to the (pro)renin receptor. This interaction can, through angiotensin independent pathways (HSP27/p38 MAPK), affect β-actin filament dynamics within cardiac myocytes. This may cause cardiac hypertrophy as is seen in animal models that over express prorenin [Saris et al. 2006]. Such an action of prorenin and renin released from the kidney and acting via the (pro)renin receptor in the neonatal heart might be crucial in establishing effective cardiac function within the first days of life.
The lack of cardiac AT2R mRNA expression in the piglet contrasts with findings in a fetal ovine receptor binding study and adult pig heart. Fetal sheep myocardium (mean gestational age of 118/150 days) had significant populations of angiotensin receptors and the major subtype was the AT2R [Burrell et al. 2001]. Although our finding of significant levels of AT2R mRNA in the kidney suggests that our primers are effective, in later experiments we redesigned our primers and retested piglet hearts. Again we found only very low expression of AT2R, so we have concluded, that unlike the fetal sheep, AT2R expression is low at the gestational ages studied. Expression may be different at an older gestation when the maturity of the piglet heart will be more similar to the fetal sheep. The absence of cardiac AT2R expression at this stage of gestation in the piglet may potentiate the proliferative and pro-angiogenic arm of the RAS, providing a reserve of myocytes and maximal capillary density to support rapid fetal growth [Kajstura et al. 1998; Jonker et al. 2007]. There are no data on levels of AT2R expression in human infant myocardium.
There were differences in the levels of expression of RAS genes between heart and kidney. Overall, most RAS genes were expressed at higher levels in the kidney than in either ventricle, and differences were most marked between kidney and LV. Angiotensinogen mRNA abundance was the exception where levels were higher in both ventricles than in the kidney. Myocardial angiotensinogen may play an important role in cardiac growth. Since cardiac angiotensinogen is a potential substrate for circulating renin taken up by the heart or for other proteases such as chymase [Urata et al. 1993], and Ang II is a peptide that promotes cardiac hypertrophy [Booz and Baker, 1996; Sampaio et al. 2003], cardiac maturation of the heart could be influenced by the cardiac levels of angiotensinogen. Results suggest that levels of expression of angiotensinogen are influenced by cardiac load, as levels were higher in the RV (where the load is greater in utero) than the LV, and increased with gestational age as workload increased. GC exposure had a maturing effect on angiotensinogen expression in the preterm RV but only in male piglets. This may be due to higher levels of 11β-hydroxysteroid dehydrogenase type 2 in female placentas reducing their exposure to GC compared with the male fetus [Stark et al. 2009]. This relationship was not present in the LV. In the fetal sheep, high doses of cortisol infused between 128 and 130 days gestation (term 150 days) were associated with increased expression of angiotensinogen both ventricles [Lumbers et al. 2005]; this effect was dose dependent. GCs and other steroids induce hepatic angiotensinogen production, so it was surprising that antenatal GCs had no effect on left ventricular or renal angiotensinogen mRNA abundance.
A limitation of the study was that only mRNA expression was measured. The poor quality or lack of specific antibodies for RAS components limits the ability to measure protein levels.
Currently, treatment options for preterm infants with hypotension or low systemic flow are limited to inotropes that are β-adrenoceptor agonists. Expression of cardiac β1-adrenoceptor subtype mRNA is low in preterm piglets compared with term piglets [Kim et al. 2014b]. Thus adrenergic inotrope therapy in preterm neonates is often ineffective [Osborn et al. 2010]. Perhaps angiotensin could be a more effective inotrope because, even in the preterm, neonate cardiac AT1R expression is present. Ang II is pressor in both fetal sheep [Tangalakis et al. 1992] and young lambs [Chappellaz and Smith, 2005]. In fetal sheep however, the pressor response will be enhanced by AT1 receptors on the umbilical arteries. AT1R is the only Ang II receptor subtype expressed on the umbilical arteries, in contrast to the systemic arteries where AT2R dominate and the density of AT1R increases with gestation [Burrell et al. 2001]. Following preterm birth and removal of the extracorporeal blood vessels, AT2Rs will induce vasodilatation, potentially compromising preterm cardiovascular function. It is possible that blocking AT2Rs would enhance the pressor effects of Ang II [Brown et al. 2014]. Interestingly, in the fetal sheep, intravenous infusions of Ang-(1-7) have no effect on blood pressure [Moritz et al. 2001]. The potential roles of angiotensin peptides as pressor agents need to be investigated in the preterm neonate.
Conclusion
mRNA levels for renin and other RAS genes in the kidneys and hearts of preterm piglets are similar to term piglets. Therefore treatment with antenatal GCs did not profoundly affect levels of mRNA levels for these RAS genes. There were few sex specific effects on renal or cardiac RAS mRNA levels. Since the mRNA expression of cardiac and renal AT1R were not different in preterm compared with term animals and not adversely affected by GCs, Ang II peptides may offer a novel form of inotropic support.
Footnotes
Acknowledgements
We wish to thank the veterinary team of Dr Helen Keates and Dr Ranald Cameron.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
Funding
This work was supported by the National Health and Medical Research Council of Australia [grant number 569635].
