Abstract
Objectives:
The aim of this retrospective study was to describe demographic and clinical characteristics of patients with diastolic dysfunction heart failure (DHF) in a safety-net institution.
Methods:
Descriptive statistics were used to summarize characteristics for adults with DHF hospitalized for acute decompensated heart failure (ADHF) between 1 January 2009 and 30 June 2010.
Results:
Of 483 patients with an ADHF-related admission, only 83 (17.2%) had pure DHF. Mean age was 59.6 ± 13.1 years old, with nearly equal proportions of male and female patients. Hypertension (89.2%), coronary artery disease (74.7%), and diabetes (63.9%) comorbidities were most common. Diuretics (91.6%), angiotensin converting enzyme inhibitors or angiotensin receptor blockers (69.9%), and β blockers (60.3%) were most frequently prescribed on discharge.
Conclusion:
In our population, DHF is more prevalent in younger patients of any ethnicity, especially Hispanic/Latino and black ethnicities, compared with nationwide estimates. Our patients with DHF had higher ejection fractions and were prescribed higher rates of traditional heart failure medications.
Introduction
Approximately 50% of patients with heart failure have diastolic dysfunction heart failure (DHF), but it is neither easily diagnosed nor documented. Patients with DHF may manifest typical symptoms of systolic heart failure (SHF), including dyspnea, fluid retention, and fatigue despite a normal or preserved left ventricular ejection fraction (LVEF) greater than 40% as per Heart Failure Society of America (HFSA) 2010 guidelines or at least 50% as per American College of Cardiology Foundation/American Heart Association (ACCF/AHA) 2013 guidelines [HFSA, 2010; Yancy et al. 2013]. The diagnosis of DHF is made based on clinical signs and symptoms, preserved LVEF (variably defined as >40–50%), imaging, and evidence of abnormal relaxation, filling, or stiffness [HFSA, 2010]. Diastolic dysfunction is likely the result of slow or incomplete filling of the left ventricle in combination with an inability of myofibrils to return to their resting length rapidly or completely, thus resulting in impaired relaxation [Verma and Solomon, 2009]. Based on Medicare payment data from 1992 to 1998, the estimated 5-year cost of managing diastolic dysfunction, or preserved LVEF, heart failure for an individual patient was more than $40,000 [Liao et al. 2006].
Currently, there is a lack of studies specifically targeting the diagnosis, management, and treatment of patients with DHF. The HFSA Comprehensive Heart Failure Practice Guideline contains predominantly level C evidence for the recommendations in the diagnosis, management, and treatment of patients with DHF [HFSA, 2010] whereas the ACCF/AHA primarily contains level IIa/IIb recommendations for the treatment of patients with preserved LVEF heart failure [Yancy et al. 2013]. Only a small number of studies and registries have previously identified and characterized this patient population (Table 1) [Fonarow et al. 2004; 2007; Adams et al. 2005; Rathore et al. 2003; 2005; 2006; Havranek et al. 2002; Tsuchihahi-Makaya et al. 2009]. Data from four large-scale registries, including the Organized Program to Initiate Lifesaving Treatment in Hospitalized Patients with Heart Failure (OPTIMIZE-HF), the Acute Decompensated Heart Failure national Registry (ADHERE), the National Heart Failure Project (NHF), and the Japanese Cardiac Registry of Heart Failure in Cardiology (JCARE-CARD), have demonstrated that, in general, patients with DHF are more likely to be older (>65 years of age), female, frequently have comorbid medical conditions [especially hypertension (HTN) or coronary artery disease (CAD)], and are less likely to be treated with traditional medications indicated for the treatment of SHF [Fonarow et al. 2004; 2007; Adams et al. 2005]; [Rathore et al. 2003; 2005; 2006; Havranek et al. 2002; Tsuchihahi-Makaya et al. 2009].
Demographic characteristics of heart failure registries.*

ACEI, angiotensin converting enzyme inhibitor; ADHERE, Acute Decompensated Heart Failure National Registry; ARB, angiotensin receptor blocker; JCARE-CARD, Japanese Cardiac Registry of Heart Failure in Cardiology; NHF, National Heart Failure Project; OPTIMIZE-HF, Organized Program to Initiate Lifesaving Treatment in Hospitalized Patients with Heart Failure.
Current treatment with diuretics, β blockers, angiotensin converting enzyme inhibitors (ACEIs), angiotensin receptor blockers (ARBs) and other medications in the DHF population is based on the presence of, and indication for, comorbid medical conditions, including diabetes mellitus (DM), HTN, and CAD [HFSA, 2010; Yancy et al. 2013; Fonarow et al. 2004]. The purpose of this study was to establish demographic and clinical characteristics of the DHF patient population in a safety-net health system.
Methods
This was a retrospective observational study designed to characterize the DHF patient population in a safety-net health system; to determine the rates of readmission at 30, 90, and 180 days post hospital discharge; and to determine common medication therapies used in the treatment of these patients. This study was conducted at a large, urban, safety-net health system in Denver, CO, USA. Approval by the Colorado Multiple Institutional Review Board was obtained prior to data collection and analysis.
Electronic medical records for patients with a hospital admission for acute DHF (ADHF) between 1 January 2009 and 30 June 2010 were identified and then further classified with DHF by International Classification of Diseases ninth revision (ICD-9) code (428.30–428.33). There was a small cohort of patients (N = 22) who had multiple hospital admissions during this time frame; only demographic data from their index hospitalization were included in this study. Adult patients aged 18–89 years old with a hospitalization for ADHF between the aforementioned 18-month time period met criteria for study inclusion. Women who were pregnant or patients who were involved in one or more investigational drug studies were excluded.
Data collection
Demographic data including age, sex, ethnicity, height, weight, and length of stay were collected from Lifetime Clinical Record (Siemens, Malvern, PA, USA) and Enterprise Data Management (MultiTech Solutions, Glen Allen, VA, USA) systems. Initial laboratory values from the index hospital admission including complete blood count, basic metabolic panel, serum calcium, serum magnesium, serum lipid, serum creatinine, thyroid stimulating hormone, hemoglobin A1C, iron studies, and troponin levels in addition to vital signs, electrocardiogram, and echocardiogram (to obtain LVEF%) documentation were collected and recorded. Patient charts were reviewed for documentation of cardiac arrhythmias, New York Heart Association Classification, and HFSA disease staging.
Concomitant disease states were identified by ICD-9, Clinical Modification diagnosis codes. The concomitant disease states include HTN (ICD-9 401.9), atrial fibrillation (ICD-9 427.31), CAD (ICD-9 414.xx), human immunodeficiency virus (ICD-9 042), hyperlipidemia (ICD-9 272.4), DM (ICD-9 250.xx), chronic obstructive pulmonary disease (ICD-9 490.xx-496.xx), hepatitis C virus (ICD-9 070.xx), and history of venous thromboembolism (ICD-9 415.xx, 453.xx) were collected for each patient. Medication therapies and regimens for DHF were collected for each patient based on the medication list at the time of hospital discharge.
Statistical analysis
Descriptive statistics were used to summarize patient and hospitalization characteristics. χ2 was used to evaluate variables of age, sex, comorbidities, medication therapies in patients readmitted versus not readmitted and number of readmissions. An a priori level for statistical significance was defined as p less than 0.05.
Results
Primary outcome
There were 483 patients identified by the ICD-9 code for ADHF with hospital admission between 1 January 2009 and 30 June 2010. In that cohort, 255 patients had SHF, 123 had DHF, and 105 had heart failure that was not classified as either systolic or diastolic dysfunction in nature. Of the 123 patients noted to have DHF, only 83 (67.5%) were found to have pure diastolic DHF (ICD-9 428.30–428.33) (Figure 1). The remaining 40 patients had mixed systolic and diastolic dysfunction. The results are reported here for the 83 unique patients with pure diastolic DHF.
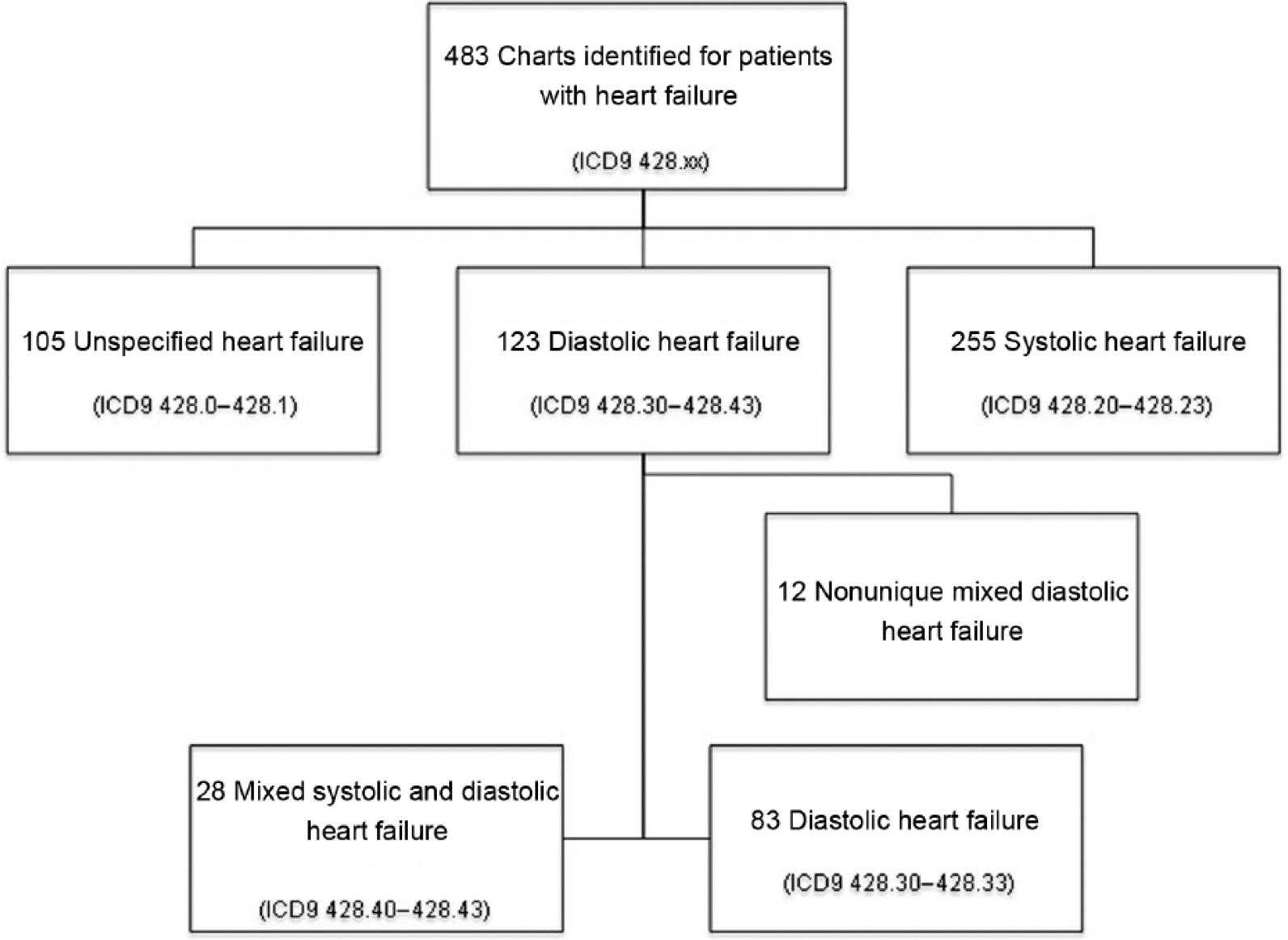
Selection of Study Population ICD9 = International Classification of Diseases, 9th revision.
In the DHF cohort (N = 83), the mean patient age was 59.6 ± 13.1 years. The male to female ratio was approximately 50:50. The mean ejection fraction (EF) was 61.9% ± 17.9%; see Table 2 for demographic data for all study patients. HTN was the most common comorbidity (89.2%), followed by CAD (74.7%), and DM (63.9%). Table 3 summarizes the comorbid medical conditions for all study patients. Of note, patients with DHF and multiple hospital readmissions were more likely to have CAD and HTN.
Demographic data of patients with diastolic heart failure.

Concurrent comorbid medical conditions.

AIDS, acquired immunodeficiency syndrome; CAD, coronary artery disease; COPD, chronic obstructive pulmonary disease; HIV: human immunodeficiency virus; VTE, venous thromboembolism.
Secondary outcomes
For the 83 patients with DHF, the mean length of hospital stay was 5.8 ± 10.4 days. In the subset of patients with multiple readmissions (N = 22), the mean length of hospital stay was 5.6 ± 4.2 days. Of the patients readmitted, seven were readmitted within 30 days, 7 were readmitted in greater than 30 days but less than 90 days, 5 were readmitted in greater than 90 days but less than 180 days of their index hospitalization, and 3 were readmitted in greater than 180 days.
Patients with DHF were most frequently discharged on a diuretic (91.6%), followed by an ACEI or ARB (69.9%) and a β blocker (60.3%). Of the patients receiving diuretic therapy, furosemide was most commonly prescribed (80.7%) compared with thiazide-type diuretics (9.6%), and spironolactone (1.2%). Medications and doses that patients were taking at hospital discharge are summarized in Table 4.
Medications at discharge in patients with diastolic heart failure (N = 83).

Discussion
Diastolic dysfunction has been estimated to be present in about half of all patients with heart failure; however, in our cohort of 483 patients admitted for ADHF, 25% had any type of diastolic dysfunction (including mixed systolic and diastolic), and only 17% of patients had pure diastolic dysfunction. The low incidence of DHF in our cohort could be due to the difficulties associated with diagnosing diastolic dysfunction, lack of symptoms that are characteristic of DHF, lack of understanding or experience in treating DHF, or a combination of these and other factors. While all patients were initially identified by ICD-9 code for heart failure, there may be variability in ICD-9 coding, especially for DHF. Reliability testing was not performed and the ICD-9 standard coding process was not evaluated. Additionally, patients with admissions or multiple readmissions due to other comorbid conditions may not have been accurately coded for DHF.
The average age for our cohort is younger than the median age reported by the larger DHF registries. The average age for our cohort was 59.6 ± 13.1 years, whereas the average age within the four registries was 75 years. Our study found that men and women were both equally affected by DHF versus the registries and previous studies that identified DHF as occurring more commonly in women and is most likely related to the older age reported in the registries. Our cohort was composed of 40.9% Hispanic/Latino patients, whereas the previously published registries and studies have described the DHF patient population as predominantly white. However, the ethnicity percentages reported by our study correlates with the percentage breakdown of ethnicities seen in the general population within our large, urban, safety-net health system.
Additionally, our study cohort of patients with DHF was found to have a higher EF (61.9% ± 17.9) compared with findings from the registries (47.6%). This difference may be attributed to the use of the Teichholz’s formula to estimate the EF in a majority of our patients, as well as the younger average age of our patients.
No difference was seen in the mean length of hospital stay between patients readmitted versus patients not readmitted. This adds to the support that the readmissions were not related to being discharged too quickly resulting in a cause of the hospitalization following the initial admission, but this cannot be confirmed based on the retrospective nature of this analysis. In addition, almost 70% of the readmissions occurred greater than 30 days from the initial discharge for ADHF, which supports the fact that patients were being discharged in a stable condition and then decompensated more than 1 month after their hospital discharge. Based on the length of hospital stay date there is a need to evaluate other potential factors contributing to the stability of these patients (e.g. medications).
Chronic medication regimens for our patient population differ in comparison to registry and study data in that our patients were more likely to be prescribed a diuretic (92% versus 74%), ACEI/ARB (70% versus 64%), or β blocker (60% versus 40%). While evidence exists to support the use of certain medications in SHF (e.g. β blockers), evidence for the use of specific medications in DHF has yet to be substantiated in clinical trials. When evaluating specific doses of heart failure medications prescribed in this population there is quite a large range. The β blocker doses on average of carvedilol and metoprolol succinate were below the target doses shown to reduce cardiovascular events in the SHF population and only one-third of the β blocker use was with these specific β blockers with known benefit. ACEI/ARB and diuretic dose ranges were comparable to previous registry data reporting on average doses utilized in the heart failure population. Our study cohort appears more likely to receive treatment according to HFSA guidelines and ACCF/AHA guidelines for treatment recommendations for SHF.
Based on the findings of this analysis there is a less clear approach to the optimal medication management of patients with DHF compared with SHF. It is clear that the approach needs to factor in comorbidity management as concurrent disease states can contribute to the stability or lack of stability resulting in risk of hospitalization and decompensation. A focus should be placed on hypertension management as this is a major risk factor for developing DHF, especially if chronically uncontrolled. Using proven risk reduction medications in SHF in patients with DHF appears to be safe, but is lacking the efficacy data seen in the SHF population for the use of angiotensin-inhibiting agents, β blockers, and aldosterone antagonists. These agents are reasonable options to help manage HTN in the DHF population, but have not been proven to reduce morbidity and mortality in a clinical trial at this point. The same principles of diuretic use hold true for patients with SHF and DHF as these medications should be used only when they are needed to acutely remove excess fluid and to chronically prevent fluid accumulation in patients with known decompensation of diuretics. Continued research in the optimal medication management of patients with DHF is warranted.
Study limitations
As a retrospective observational cohort study, this study is limited by its small sample size and retrospective study design. Patients were identified by the ICD-9 code for heart failure and first hospital admission for ADHF within a small 18-month time frame and then further selected by diagnosis code for DHF. ICD-9 codes are not always all inclusive and may be inaccurate or lack reliability and validity due to variability in diagnosing or coding conditions. Some data were missing, which may adversely affect overall results based on omissions of data.
Echocardiographic monitoring for EF% using Teichholz’s formula was recorded when available; however, not every patient received a Teichholz’s formula estimate and not all patients had an echocardiogram performed during their ADHF hospital admission that occurred during the specified study time period. The Teichholz’s formula measurement may not be the standard of practice at every institution, but patients in our study frequently had their EF calculated with Teichholz’s formula. We did not perform an internal validation to ensure that the ICD-9 code for DHF was matched with a formal diagnosis of DHF based on HFSA recommendations, and symptoms and imaging. Overall, documentation of NHYA classification or ACCF/AHA staging of HF in the medical record were underreported.
Conclusion
Compared with previous study and registry data, the results of our retrospective cohort study demonstrate that in our patient population, DHF is more prevalent in younger patients of any ethnicity, especially patients of Hispanic/Latino and black ethnicities. Additionally, our patients have a higher EF and are more likely to be treated with medications recommended in the management of both SHF and DHF, especially diuretics, β blockers, and angiotensin-inhibiting agents.
Footnotes
Acknowledgements
During the time this project was conducted, Dr Wobeter was a PGY1 Pharmacy Practice Resident at Denver Health Medical Center in Denver, CO, USA.
Disclosures
All co-authors have contributed substantially to this research and manuscript and approved as submitted. None of the authors have conflicts of interest to disclose regarding the content of this manuscript.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
