Abstract
Objective:
Early injection of atropine during dobutamine stress echocardiography (DSE) has been demonstrated in retrospective analyses to reduce the duration and dose of dobutamine infusion, while preserving a similar diagnostic accuracy with a lower incidence of adverse effects. This study explores the safety of using atropine as a start drug before dobutamine infusion (ADSE protocol) in comparison with the conventional protocol (DASE protocol) in older patients undergoing DSE for ischemia evaluation.
Methods:
One hundred consecutive older patients were prospectively enrolled. When eligible, they were randomly assigned to undergo either the DASE protocol (group A, 50 patients) or the ADSE protocol (group B, 50 patients) when atropine (1.0 mg) was first administered 3 min before dobutamine infusion followed by 0.5 mg increments (maximum 1.0 mg) thereafter. Patients were monitored for adverse drug effects. Test duration was calculated.
Results:
The mean age of the whole study cohort was 67.8 ± 4.3 years and 58 (58%) were men. Patients in group A had longer test duration (21.8 ± 1.3 versus 13.7 ± 0.77 min, p < 0.001) and higher mean dobutamine infusion rate (39 ± 8.2 versus 28.2 ± 9.5 μg/kg/min, p < 0.001). The two groups received a similar total dose of atropine. Group A patients showed significantly higher incidence of extrasystoles, nonsustained ventricular tachycardia and severe hypotension (p < 0.05).
Conclusion:
In older patients undergoing DSE, using atropine as a start drug, that is, adopting the ADSE protocol, is associated with shorter test duration, lower mean dobutamine infusion rate and consequently fewer adverse effects.
Introduction
Dobutamine stress echocardiography (DSE) is widely used for the diagnosis of coronary artery disease (CAD) [Płońska et al. 2004]. As a result of advanced treatment options, and the ensuing increase in life expectancy, diagnostic procedures for CAD detection have been increasingly used in the older population. Due to the obvious limitations of exercise testing in older patients, DSE had become widely used in the evaluation of such patients [Poldermans et al. 1994]. Safety of DSE for ischemia detection has been extensively evaluated. The most frequent side effects are hypotension and arrhythmias [Ling et al. 1996; Hepner et al. 1997; Pingitore et al. 1996]. Recent modifications in the DSE protocols include earlier injection of atropine in patients with poor chronotropic response to dobutamine [Lewandowski et al. 1998]. This strategy has been shown to be safe and effective in reducing the test duration and adverse effects, maintaining similar diagnostic accuracy for detecting angiographically significant CAD as that with the conventional protocol [Tsutsui et al. 2004]. It is hypothesized that reducing test duration and drug adverse effects, especially hypotension and arrhythmias, would be of utmost importance for older patients undergoing DSE for evaluation of coronary ischemia. Thus, the author prospectively sought to explore the safety of starting the stress echocardiography test using atropine solely, followed by dobutamine administration, that is, atropine DSE (ADSE), in achieving target heart rate compared with the conventional dobutamine atropine stress echocardiography (DASE) protocol. The study was carried out in a series of older patients referred for evaluation of suspected CAD.
Methods
Study design and data collection
One hundred consecutive older patients (above 60 years) were prospectively enrolled in this study. They were referred to our stress echocardiography lab between March 2011 and July 2013. Patients were considered eligible for inclusion if they had symptoms suggestive of myocardial ischemia and if they showed normal resting left ventricle ejection fraction. Exclusion criteria included patients with prior history of unstable angina or myocardial infarction (MI), those with prior percutaneous coronary intervention (PCI) or coronary artery bypass graft (CABG) surgery, those with significant valvular disease, congenital heart disease or any myocardial disease apart from ischemia. Also, those with contraindications to dobutamine infusion (for example, with a history of complex ventricular arrhythmias or uncontrolled hypertension with blood pressure >180/110 mmHg), those with contraindications to atropine (for example, with a history of narrow-angle glaucoma or obstructive uropathy) and patients with limited life expectancy due to coexistent disease (for example, malignancy) were excluded. All included patients stopped β blocker and calcium antagonist therapies 48 h before the DSE test, while nitrate therapy was stopped 24 h before the test. Before inclusion, informed written consent was obtained from each patient and the study protocol was reviewed and approved by our local institutional human research committee, as it conforms to the ethical guidelines of the 1975 Declaration of Helsinki, as revised in 2008.
Definition of risk factors of CAD
The presence of hypertension was defined as systolic blood pressure at least 140 mmHg or diastolic blood pressure at least 90 mmHg, previously recorded by repeated noninvasive office measurements, which led to lifestyle modification or intake of antihypertensive drug therapy [Mancia et al. 2013]. The presence of diabetes mellitus was defined as fasting plasma glucose at least 126 mg/dl or 2 h post glucose load at least 200 mg/dl, or specific antidiabetic drug therapy [Genuth et al. 2003]. Dyslipidemia was defined as low-density lipoprotein cholesterol greater than 100 mg/dl, serum triglycerides greater than 150mg/dl or high-density lipoprotein cholesterol less than 40 mg/dl (<50 mg/dl in women) [Enkhmaa et al. 2005].
Baseline echocardiographic assessment
Assessment of regional and global left ventricular systolic functions was performed in all patients by trans thoracic echocardiography using a General Electric Vivid 7 cardiac ultrasound machine (General Electric, Horten, Norway), equipped with harmonic imaging capabilities. A 2.5 MHz phased array probe was used to obtain standard two-dimensional, M-mode and Doppler images. Patients were examined in the left lateral recumbent position using standard parasternal and apical views. Images were digitized in cine-loop format, and saved for subsequent playback and analysis. Being blinded to the study randomization process, all views were analyzed by a single echocardiographer employing the software program of the echocardiography machine. Regional wall motion was assessed according to the standard 17-segment model as recommended by the American Society of Echocardiography [Cerqueira et al. 2002]. Regional wall motion was visually assessed for each segment individually, considering both endocardial excursion and systolic thickening. Each segment was graded according to the semiquantitative scoring system described by Knudsen and colleagues [Knudsen et al. 1998]. Segments with poorly defined endocardial borders for 50% or more of their length were considered nonvisualized and assigned a score of 0 [Chaudhry et al. 2000]. Wall thickening was assessed at a distance of at least 1 cm from the adjacent segment to minimize the effect of tethering [Meluzín et al. 1998].
Stress echocardiography protocols
Once eligible, patients were randomly assigned in 1:1 fashion according to a computer-generated random series of numbers. Randomization was performed by block randomization (blocks of 10 patients) to undergo one of the following two stress echocardiography protocols.
Conventional DASE protocol (50 patients, group A)
Dobutamine (Mylan, Mylan S.A.S, France) was administered by intravenous infusion starting at a dose of 5 μg/kg/min and raised incrementally every 3 min up to a maximum of 40 μg/kg/min or until a study endpoint was reached. In patients not achieving 85% of their age-predicted maximal heart rate at the end of the final stage, atropine was administered intravenously in 0.5 mg increments at 1 min intervals up to a maximum dose of 2.0 mg, while dobutamine infusion was continued.
ADSE protocol (50 patients, group B)
The test protocol was started by intravenous administration of 1.0 mg atropine, followed by starting the intravenous dobutamine infusion (as mentioned in the conventional protocol), 3 min later. A dose of 0.5 mg atropine was administered with a dobutamine infusion rate of 10 μg/kg/min and finally a similar dose was given with a dobutamine infusion rate of 20μg/kg/min, if required. Dobutamine infusion was continued up to a maximum of 40 μg/kg/min or until a test endpoint was reached.
Test termination endpoints
Endpoints for terminating the test included attainment of the maximum dose of dobutamine or atropine; achievement of target heart rate (>85% of age-predicted maximal heart rate); echocardiographic detection of wall motion abnormality not present at baseline or worsening of previously existing wall motion abnormality; symptoms judged to be unacceptable by the attending cardiologist; serious arrhythmia detected by electrocardiogram (ECG) monitoring, ST segment elevation greater than 0.1 mV at 80 ms from the J point, systolic blood pressure greater than 200 mmHg or diastolic blood pressure greater than 110 mmHg, or a decrease in systolic blood pressure greater than 30 mmHg from the baseline [Lewandowski et al. 1998]. Standard views were recorded at baseline, at the end of each stage of dobutamine infusion, as well as during recovery. Visual assessment of wall motion and systolic thickening was performed as before. The test was considered positive on detecting wall motion abnormality not present at baseline, or worsening of previously existing wall motion abnormality in two contagious left ventricular segments belonging to the same blood supply territory.
Patient monitoring
All patients had continuous heart rate, ECG and pulse oximetry monitoring. Heart rate and blood pressure readings were recorded at baseline, at the end of each stage of dobutamine infusion and during recovery. Mild hypotension was defined by a drop in systolic or diastolic blood pressure by up to 20 mmHg, while severe hypotension was defined by a drop in systolic or diastolic blood pressure by more than 20 mmHg. Hypertension was defined by an increase in systolic or diastolic blood pressure by more than 20 mmHg. A 12-lead ECG was recorded at baseline and during recovery. Patients were asked at the end of the test about any symptoms or adverse drug reactions. Test duration was recorded from the onset of dobutamine infusion until the end of the recovery period. The peak dose of dobutamine infusion at which the test was terminated was recorded for each patient. Moreover, the amount of intravenous atropine was quantified for each patient.
Statistics
All continuous variables were statistically described in terms of mean ± standard deviation (±SD). Categorical variables were described with absolute and relative (percentage) frequencies. Comparison of continuous variables between the studied groups was done using Student’s t test. For comparing categorical data, Pearson Chi square test or Fisher exact test (according to the expected cell count) were performed. p Values were used to describe significance (P >0.05= non-significant, P <0.05=significant, P <0.001= highly significant). All statistical calculations were done using Statistical Package for Social Sciences (SPSS for Windows) software (version 15.0, SPSS Inc., Chicago, IL, USA).
Results
Baseline clinical characteristics
A total of 100 older patients were prospectively enrolled in the current study: 50 patients were randomly assigned to undergo the conventional DASE protocol (group A), and 50 were randomly assigned to undergo the ADSE protocol (group B). The mean age of the whole study cohort was 67.8 ± 4.3 years and 58 (58%) were male patients. The two groups were matched regarding age, sex and risk factors for CAD. These data are summarized in Table 1.
Baseline characteristics of the two studied groups.

All variables, except age (mean ± SD), are presented as number (percentage).
Student’s t test and Pearson Chi-Square test.
CAD, coronary artery disease; SD, standard deviation.
Test protocol data
There was no recorded significant difference between both studied groups regarding echocardiographic data, both at rest and peak stress (Table 2). Analysis of intraobserver variability revealed a close correlation between repeated measurements of regional wall motion by the single operator, with a correlation coefficient r = 0.94. Patients belonging to group A had longer test duration (21.8 ± 1.3 min versus 13.7 ± 0.77 min, p < 0.001). All patients achieved their target heart rate. For group A, 6 (12%) patients achieved target heart rate at a dobutamine infusion rate of 30 μg/kg/min, while the remaining 44 (88%) patients achieved it at a dobutamine infusion rate of 40 μg/kg/min with or without intravenous atropine. Meanwhile, in group B, 15 (30%) patients achieved their target heart rate at a dobutamine infusion rate of 20 μg/kg/min, 25 (50%) patients achieved it at a dobutamine infusion rate of 30 μg/kg/min, and 10 (20%) patients achieved it at a dobutamine infusion rate of 40 μg/kg/min. The mean dobutamine infusion rate at which the target heart rate was achieved was calculated for both groups and was recorded to be significantly higher in group A (39 ± 8.2 versus 28.2 ± 9.5 μg/kg/min, p < 0.001). There was no significant difference in mean intravenous atropine dose administered for groups A and B (1.01 ± 0.2 versus 0.97 ± 0.3 mg respectively, p > 0.05).
Echocardiographic data of the two studied groups at rest and at peak stress.

All variables are presented as mean ± SD.
Student’s t test.
LVEDD, left ventricle end diastolic dimension; LV EF, left ventricle ejection fraction; LVESD, left ventricle end systolic dimension; WMSI, wall motion score index.
Hemodynamic data during stress testing
Table 3 shows hemodynamic data recorded during the two stress protocols. No significant difference was recorded for heart rate, systolic and diastolic blood pressure both at baseline and at peak stress on comparing both groups.
Hemodynamic data of the two studied groups at rest and at peak stress.

All variables are presented as mean ± SD.
Student’s t test.
BP, blood pressure; bpm, beats per minute.
Safety of stress echocardiography protocols
The DASE protocol showed positive results for coronary ischemia in 44 (88%) patients, while the ADSE protocol showed positive results in 42 (84%) patients. The reported adverse events for both test protocols are summarized in Table 4. Patients belonging to group A showed higher incidence of ventricular extrasystoles, atrial extrasystoles, nonsustained ventricular tachycardia, nausea, headache and severe hypotension. There were no recorded cases of sustained ventricular tachycardia, ventricular fibrillation, syncope or death in group A or group B during or immediately after the test. Moreover, group A showed a higher incidence of atrial fibrillation, supraventricular tachycardia, symptomatic bradycardia, chest pain and hypertension, yet did not reach statistical significance.
Adverse outcomes among the two studied groups.
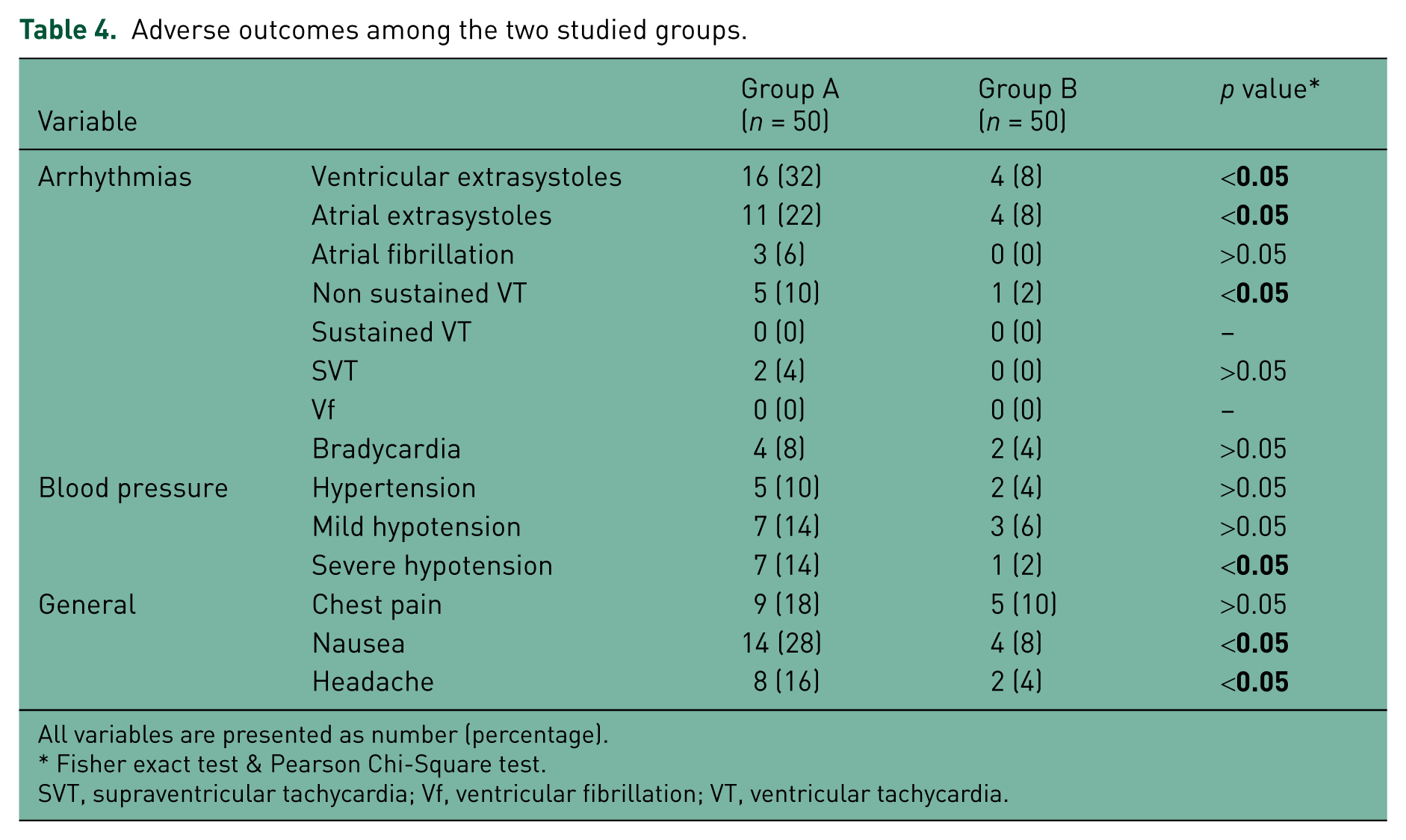
All variables are presented as number (percentage).
Fisher exact test & Pearson Chi-Square test.
SVT, supraventricular tachycardia; Vf, ventricular fibrillation; VT, ventricular tachycardia.
Discussion
Currently, DSE is widely approved for the detection of CAD [Segar et al. 1992; Baptista et al. 1994; Pellikka et al. 1995], for risk stratification after MI [Salustri et al. 1994] and for prediction of perioperative and late cardiac events in patients scheduled for major surgery [Poldermans et al. 1997]. However, in addition to being time consuming, DSE has often been limited by patient intolerance; chiefly related to drug adverse effects and test duration [Secknus and Marwick, 1997]. Furthermore, heart rate response might be inadequate in older patients and in patients receiving β-blocker therapy [Chen et al. 1996]. In this case, larger doses of dobutamine and longer test durations are needed, besides the addition of atropine to achieve the target heart rate [McNeill et al. 1992; Sawada, 2000]. In turn, this would further increase patients’ intolerance. The current study tested the hypothesis that starting the stress echocardiography test with intravenous atropine would reduce dobutamine dose and test duration, and consequently improve the tolerance of older patients with no history of previous MI, PCI or CABG surgery.
Mechanisms of drug action
At low doses (up to 10 µg/kg/min), dobutamine induces a marked inotropic effect, probably mediated by a combined β1 and α1 adrenoreceptor stimulation, augmenting myocardial contractility without a significant increase in heart rate [Lewandowski et al. 1998]. At higher doses of dobutamine infusion (20–40 µg/kg/min), heart rate progressively increases through its action on β2 adrenoreceptors. Systemic blood pressure modestly increases as a result of an increase in cardiac output and a decrease in systemic vascular resistance; α1 adrenoreceptor mediated vasoconstriction is balanced by the β2 adrenoreceptor mediated vasodilator effect. Consequent upon these hemodynamic changes, myocardial oxygen demand increases. Yet, in myocardial segments supplied by a coronary artery with significant stenosis, this increased demand cannot be met with a parallel increase in myocardial blood flow. Hence, regional myocardial ischemia develops and manifests as regional wall motion abnormalities, detected by two-dimensional echocardiography. Atropine, instead, acts as a reversible competitive antagonist to acetylcholine action at muscarinic receptors in the sinoatrial node, resulting in a positive chronotropic effect. Indirectly, an inotropic effect would also be expected through the force–frequency relationship; probably mediated by increased availability of cytosolic calcium for actin–myosin interaction [Sawada et al. 1991].
Consequences of early atropine administration
The current study results showed that early atropine administration significantly decreased the test duration by almost 40%. Also, a larger number of patients reached their target heart rate at lower dobutamine infusion rates. This mounted to a significantly lower mean dobutamine infusion rate at which target heart rate was reached. The two protocols yielded positive results in nearly the same percentage of patients. Hence, yielding similar diagnostic outcome at a lower level of pharmacological stress. This adds to the safety profile of the ADSE protocol. This might be due to a modest increase in coronary blood flow with higher doses of dobutamine infusion, balancing myocardial oxygen demand and delaying the appearance of myocardial wall motion abnormalities.
Safety of atropine dobutamine stress protocol
Although a similar percentage of patients showed positive results for myocardial ischemia in both groups, patients undergoing the DASE protocol showed a higher incidence of adverse effects, including arrhythmias, pointing to the arrhythmogenic potential of dobutamine [Lewandowski et al. 1998]. Interestingly, all episodes of nonsustained ventricular tachycardia recorded in this group of patients were recorded in the recovery period. This highlights the importance of continuous monitoring of this period.
Comparison with other studies
Previous studies were able to demonstrate minor reductions in both dobutamine dose and test duration with early administration of atropine during DSE stress testing (11% and 8% in the study by Lewandowski and colleagues, 14% and 15% in the study by Tsutsui and colleagues) [Lewandowski et al. 1998; Tsutsui et al. 2004]. They used higher total doses of atropine (compared with the present study) and eventually they showed a similar rate of adverse effects in comparison to the conventional DSE protocol. Another randomized trial by Camarozano and colleagues reported reductions in both dobutamine dose and test duration with the early atropine protocol, showing a uniform side-effect profile [Camarozano et al. 2006]. In comparison, the current study demonstrated that early atropine administration in the course of stress echocardiography resulted in an almost 28% reduction in mean dobutamine infusion rate and an almost 40% reduction in the test duration, with no need for higher doses of atropine and with an obvious reduction in adverse effects. In agreement with the results of the current study, Abdel-Salam and Nammas reported the safety of early atropine administration in older patients undergoing stress echocardiography [Abdel-Salam and Nammas, 2010]. It is worth mentioning that none of the previous studies used atropine as a start drug before dobutamine infusion. Rather, they started intravenous atropine administration after proceeding with dobutamine infusion. So, to the best of the author’s knowledge, the current study presents the first research on the safety and tolerability of early administration of intravenous atropine as a start drug before dobutamine infusion in older patients undergoing stress echocardiography. Finally, the fairly high rates of positive test results in the DASE group (88%) and the ADSE group (84%) are in agreement with the sensitivity rates reported in validated articles in the literature [Geleijnse et al. 2009; Elhendy et al. 1996] which described a sensitivity range of 74–86%.
Clinical implications
An increasing number of older patients with known or suspected CAD are currently being evaluated by DSE. This population of patients is much more susceptible to complications of DSE protocols, primarily due to adverse drug reactions (dobutamine and atropine). The performance of DSE with a significantly lower dose of dobutamine, shorter test duration, with almost the same total dose of atropine and with an essentially lower side-effect profile would be an appealing option. All these advantages were clearly offered by the ADSE protocol, as shown in the present study. Hence, at least from the ‘safety’ point of view, the author can recommend the ADSE protocol to be the protocol of choice for older patients undergoing DSE for evaluation of suspected CAD.
Limitations of the study
The study data presented only apply for patients defined by inclusion and exclusion criteria. This is a single-center study with a relatively small sample size, a fact that makes it difficult to generalize the results to all older patients undergoing DSE. Larger multicenter trials are needed to confirm the current results. Furthermore, the test duration was calculated from the onset of atropine intake or dobutamine infusion until the end of the recovery time, which might be difficult to demarcate in some cases. However, being subjective in nature, it is difficult to delineate precisely the end of the recovery time. Another limitation is the lack of quantitative methods of measuring systolic left ventricular wall thickening. Instead, the echocardiographer adopted visual assessment only. Finally, the sensitivity, specificity and diagnostic accuracy of the ADSE protocol were not validated against ‘gold standard’ evaluation by invasive coronary angiography, as this evaluation was outside the scope of the study.
Conclusion
In older patients undergoing DSE, using atropine as a start drug (i.e. adopting the ADSE protocol) is associated with shorter test duration, lower mean dobutamine infusion rate for target heart rate achievement and consequently fewer adverse effects.
Footnotes
Acknowledgements
The author would like to express his gratitude to the medical and nursing staff of the stress echocardiography lab, Cardiology Department, Ain Shams University for their cooperation in accomplishing this work.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
