Abstract
Objective:
EXCITE (clinical EXperienCe of amlodIpine and valsarTan in hypErtension) evaluated the real-life effectiveness, safety, and tolerability of single-pill combinations (SPCs) of amlodipine/valsartan (Aml/Val) and amlodipine/valsartan/hydrochlorothiazide (Aml/Val/HCTZ) in patients with hypertension from 13 countries in the Middle East and Asia. Here, we present data from Pakistan.
Methods:
This was a 26-week observational, multicenter, prospective, open-label study. At week 26, change from baseline in mean sitting systolic (msSBP) and diastolic blood pressure (msDBP) and the proportion of patients achieving BP goal (SBP/DBP <140/90 mmHg; <130/80 mmHg in patients with diabetes) and response rates (SBP <140 mmHg [130 mmHg for patients with diabetes] or reduction of ≥20 mmHg; DBP <90 mmHg [80 mmHg for patients with diabetes] or reduction of ≥10 mmHg), were evaluated. Incidence of adverse events (AEs) and serious AEs (SAEs) was recorded as safety variables. Subjective assessment of effectiveness, compliance and tolerability was done by the physician.
Results:
A total of 500 patients with hypertension (mean age of 48 years) were prescribed Aml/Val (n = 471, 94%) or Aml/Val/HCTZ (n = 29, 6%); 439 (87.8%) patients completed the study. At week 26, the mean BP decreased from 153.4/91.1 mmHg at baseline to 128.9/78.4 mmHg in the Aml/Val cohort (−24.5/−12.7 mmHg; p < 0.0001) and from 171.6/99.3 mmHg at baseline to 127.7/77.4 mmHg (−43.9/−21.9 mmHg; p < 0.0001) in the Aml/Val/HCTZ cohort. BP goals were achieved by 57% and 55.2% of patients in the Aml/Val and Aml/Val/HCTZ cohorts, respectively. A total of 40 (8%) patients reported at least one AE during the study period. Most common AEs included nausea (1.6%), headache (1.2%), vomiting (1.2%), and edema (1.2%). Most patients in Aml/Val cohort and all patients in Aml/Val/HCTZ cohort rated the effectiveness, compliance and tolerability as ‘good’ or ‘very good’.
Conclusions:
Aml/Val with or without HCTZ in a SPC was effective and well-tolerated for BP reduction in this cohort of patients with hypertension from Pakistan.
Keywords
Introduction
High blood pressure (BP) is a major risk factor contributing to the global burden of disease and is a leading cause of death worldwide [Lim et al. 2012; Mancia et al. 2013]. The worldwide prevalence of hypertension is predicted to increase by 60% to a total of 1.56 billion by the year 2025. This rise can be chiefly attributed to an expected increase in the number of people with hypertension in the economically developing regions [Ibrahim and Damasceno, 2012; Kearney et al. 2005; Lim et al. 2012]. According to the World Health Organization (WHO) estimates, nearly 1.5 million people die every year in South-East Asia due to high BP or hypertension making it the single most important risk factor for noncommunicable diseases such as heart attack and stroke [WHO Regional Office for South-East Asia, 2011]. The South Asian regions of the world are currently facing a challenge of increased cardiovascular risk due to inadequate management of hypertension. BP control rates reported in developing countries are as low as 6% and 8% in India and China, respectively [Mohan and Campbell, 2009]. In a survey conducted by the National Health Survey of Pakistan (NHSP) from 1990 to 1994, ≤3% of hypertensive patients had their BP controlled to ≤140/90 mmHg, and ≥70% of all hypertensive patients (85% in rural areas) in Pakistan were unaware of the underlying disease [Pakistan Medical Research Council, 1998]. Similar findings have been reported in primary healthcare settings of Pakistan with barely 6% hypertension control rates [Saleheen et al. 2010]. Moreover, developing Asian countries are underrepresented in most hypertension clinical trials and very few trials are conducted specifically in these populations [Chazova et al. 2011].
As per the Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation and Treatment of High Blood Pressure (JNC7) recommendations and National Guidelines For Detection, Prevention, Control and Management of Hypertension in Pakistan, combination therapy may be considered as the ‘initial’ treatment for patients with stage 2 (≥160/≥100 mmHg) hypertension (or in patients with BP more than 20/10 mmHg above goal) [Chobanian et al. 2003; The Collaborative Group of Pakistan Hypertension League and Pakistan Cardiac Society, 2009]. Availability of two or more anti-hypertensive medications as a single-pill combination (SPC) will help to achieve BP control and aid in treatment compliance [Chobanian et al. 2003].
Amlodipine/valsartan (Aml/Val) and amlodipine/valsartan/hydrochlorothiazide (Aml/Val/HCTZ) are SPC agents approved for the treatment of hypertension in Pakistan since 2008 and 2011, respectively. In clinical trials, both SPCs have shown to be well tolerated and provide effective BP lowering [Calhoun et al. 2009a; Smith et al. 2007]. However, there is a large gap in the literature on the real-life effectiveness of these combination therapies in developing countries including Pakistan.
The EXCITE (clinical EXperienCe of amlodIpine and valsarTan in hypErtension) study was performed as separate studies in a real-world setting in developing countries including Egypt, Indonesia, Hong Kong, Middle Eastern countries (Bahrain, Kuwait, Lebanon, Oman, Qatar, United Arab Emirates), Pakistan, The Philippines, South Korea, and Taiwan. Overall pooled data and data at the individual country level were analyzed to provide a comprehensive overview of the therapies. The interim results of the EXCITE pooled data showed that in patients with hypertension, SPC treatment with Aml/Val and Aml/Val/HCTZ provided statistically significant and relevant reductions in BP from baseline [Shete et al. 2013]. This manuscript presents the data on real-life effectiveness, safety, and tolerability of the SPC of Aml/Val and Aml/Val/HCTZ in patients with hypertension from Pakistan.
Methods
Study population
The study population comprised male and female patients aged ≥18 years with an established diagnosis of hypertension, who were prescribed SPC of Aml/Val or Aml/Val/HCTZ as per the prescribing information. Patients provided the written informed consent for data collection. Patients were excluded for contraindications mentioned in the local Pakistan summary of product characteristics for both products. Patients with known hypersensitivity to any of the drug components or excipients and patients with diabetes taking aliskiren were excluded from study participation. Patients with anuria or hypersensitivity to sulfonamide-derived drugs were specifically excluded from the Aml/Val/HCTZ cohort.
Study design
This was an observational, multicenter, prospective, open-label study of patients with hypertension from Pakistan conducted from July 2010 to September 2012, in 35 medical centers from 7 cities. The therapy was prescribed in terms of the marketing authorization as per the definition of noninterventional trials. The assignment of patients to therapy was decided within the current practice and medical indication, and was separated from the decision to include the patient in the study. The study was carried out according to the Good Clinical Practice guidelines and was in compliance with the Declaration of Helsinki (2002) of the World Medical Association. The study protocol was approved by the institutional review board or ethical review committee of each study center.
During the observational period of 26 ± 8 weeks, the data were obtained from at least two routine examinations in each subject at baseline (visit 1), optional week 13 (visit 2) and week 26 (visit 3). No additional diagnostic or monitoring procedures were employed beyond usual care, and all data collected for the study originated from routine clinical practice. Patient demographics and baseline data including history of hypertension, risk factors, laboratory evaluations (urea, creatinine, sodium, potassium, glucose, HbA1c and assessment of albuminuria) vital signs (heart rate, body weight), medical history, and antihypertensive and other concomitant medications were recorded at study entry. Based on the prescribing patterns of the physicians, the two treatment cohorts were Aml/Val (dosages 5/80, 5/160, 10/160, and 5/320 mg) or Aml/Val/HCTZ (dosages 5/160/12.5, 10/160/12.5, and 5/160/25 mg).
Efficacy assessments
The means of three sitting BP values were recorded. The efficacy assessments at the end of study included: (a) change in the mean sitting systolic blood pressure (msSBP) and mean sitting diastolic blood pressure (msDBP) from baseline to week 26 ± 8; (b) proportion of patients achieving BP goal according to the WHO criteria (SBP/DBP <140/90 mmHg for patients without diabetes, and SBP/DBP <130/80 mmHg for patients with diabetes); (c) proportion of patients who achieved SBP response (SBP <140 mmHg or reduction of ≥20 mmHg); and (d) proportion of patients who achieved DBP response (DBP <90 mmHg or reduction of ≥10 mmHg). The efficacy variables were also analyzed in subgroups based on diabetic/nondiabetic status, age (<65 years and ≥65 years), and body mass index (BMI) categories (<30 kg/m2 and ≥30 kg/m2). Subjective assessment of therapy effectiveness was rated by treating physicians as ‘below average’, ‘average’, ‘good’, and ‘very good’ at the end of the study. Investigators entered the information in the Novartis case report forms.
Safety and tolerability assessments
Safety assessments included the monitoring and recording of incidence of adverse events (AEs), serious AEs (SAEs), and the incidence and intensity of edema. At the end of the study, subjective assessment of compliance and tolerability was rated by treating physician as ‘below average’, ‘average’, ‘good’, and ‘very good’.
Statistical analysis
The effectiveness and safety analysis was carried out on the full analysis set (FAS) population consisting of patients who participated in the study after providing informed consent. Data management was performed by Biometrical Practice AG, Basel, Switzerland (BIOP) using version 3.1.2 of Open Clinica, whereas statistical analysis was conducted by BIOP using version 9.2 of the SAS statistical package. Paired t-test was used to assess the change in BP from baseline to the end of study. For patients who discontinued before week 26, the last available post-baseline value was carried forward. Changes from baseline were analyzed separately by treatment cohort and dosage group.
Results
Patient disposition and characteristics
Of the 500 patients who participated in the study, SPC of Aml/Val was prescribed to 471 (94%) patients and SPC of Aml/Val/HCTZ was prescribed to 29 (6%) of patients. Of these, 439 (88%) patients completed the 26-week study period (Figure 1). Patient demographics and baseline characteristics are presented in Table 1. Overall, the mean age was 48 years, 25 patients (5%) were ≥65 years and the majority of patients were Asians (99%). The mean duration of hypertension was 5.4 years, 99 patients (19.8%) were obese (BMI ≥30 kg/m2) with a mean body weight of 75.8 kg (167.1 lb), and 83 patients (16.6%) had diabetes. At baseline, mean sitting BP was 153.4/91.1 mmHg in the Aml/Val cohort and 171.6/99.3 mmHg in the Aml/Val/HCTZ cohort. Most patients (98%) were on prior antihypertensive treatment (drug therapy or nondrug therapy) before participating in the study. Prior to study participation, most patients were prescribed angiotensin-converting enzyme (ACE) inhibitors (21.6%), angiotensin II antagonists (9.6%), selective beta blockers (23.8%), and calcium channel blockers (CCBs, 22.2%).

Patient disposition.
Patient demographics and baseline characteristics.
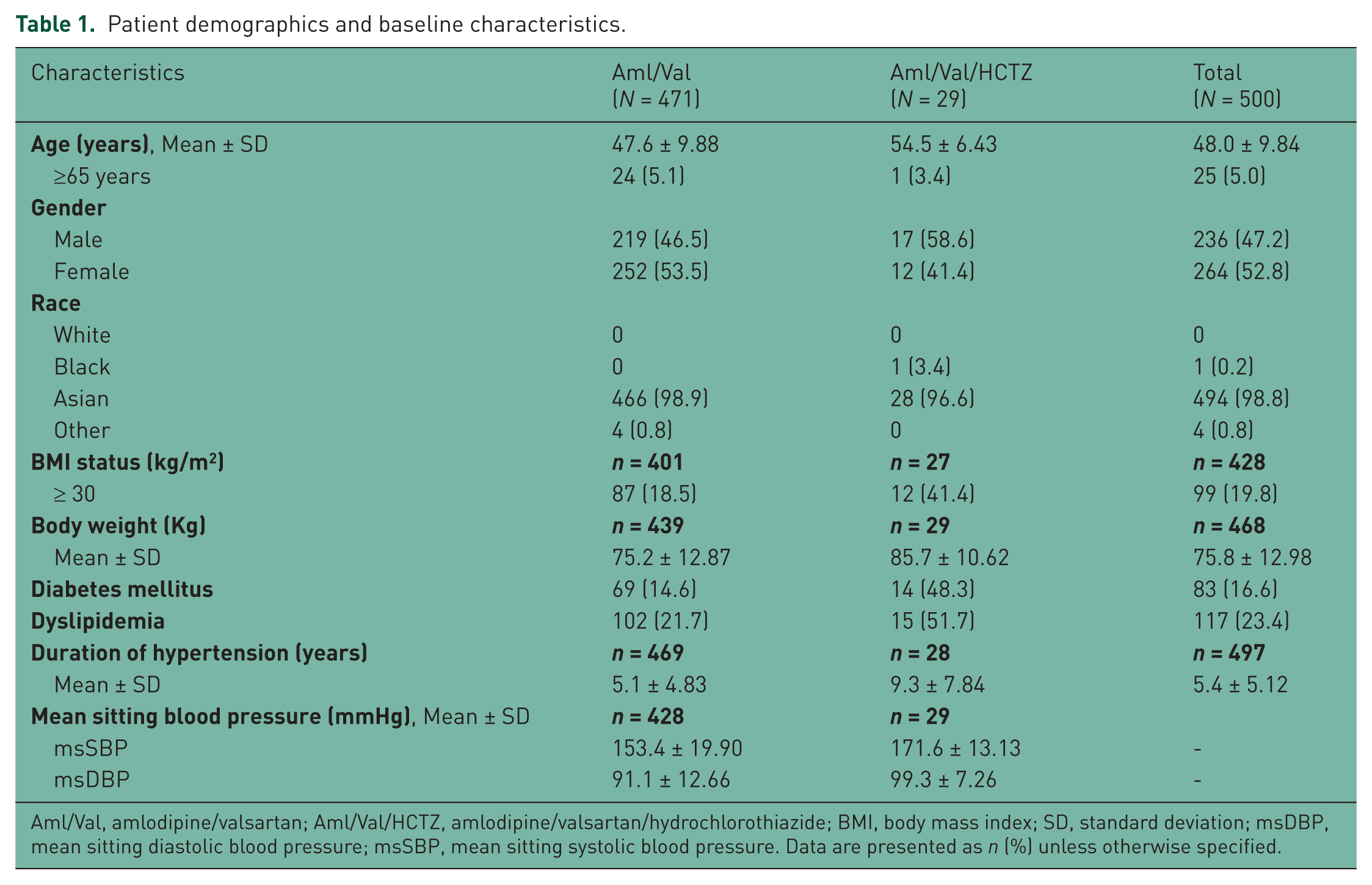
Aml/Val, amlodipine/valsartan; Aml/Val/HCTZ, amlodipine/valsartan/hydrochlorothiazide; BMI, body mass index; SD, standard deviation; msDBP, mean sitting diastolic blood pressure; msSBP, mean sitting systolic blood pressure. Data are presented as n (%) unless otherwise specified.
The most common reason for changing treatment to receive SPC of Aml/Val or Aml/Val/HCTZ was unsatisfactory BP control with prior treatment, as reported by 240 patients (51%) in the Aml/Val cohort and 24 patients (82.8%) in the Aml/Val/HCTZ cohort. Patients on Aml/Val were prescribed doses of 5/80 mg (n = 231, 49%), 5/160 mg (n = 159, 34%), 10/160 mg (n = 80, 17%), and 5/320 mg (n = 1, 0.2%). Patients in the Aml/Val/HCTZ cohort were prescribed doses of 5/160/12.5 mg (n = 23, 79%), 10/160/12.5 mg (n = 5, 17%), and 5/160/25 mg (n = 1, 3.4%). During the study period, 9.2% of patients received concomitant antihypertensive medications at baseline which reduced to 4.4% at the end of study. Among these, selective beta blockers were the most commonly prescribed medication.
Efficacy assessments
Change in mean sitting BP
The primary endpoint of this study was the change in msSBP and msDBP from baseline to week 26. Statistically significant decreases in msSBP and msDBP were observed for both the treatment cohorts (p < 0.0001 versus baseline; Figure 2). The Aml/Val cohort showed significant mean [95% confidence interval (CI)] reductions (p < 0.0001) from baseline to the endpoint in msSBP (−24.5 mmHg [−26.54, −22.50]) and msDBP (−12.7 mmHg [−13.84, −11.54]). The Aml/Val/HCTZ cohort also showed significant mean [95% CI] reductions (p < 0.0001) from baseline to the endpoint in msSBP (−43.9 mmHg [−49.66, −38.13]) and msDBP (−21.9 mmHg [−25.86, −17.93]); see Figure 2. A generally similar and consistent reduction in msSBP and msDBP was observed in difficult-to-treat subgroups such as in elderly patients (≥65 years), obese patients (BMI ≥30 kg/m2), or in patients with diabetes (Figure 3). The changes in msSBP and msDBP from baseline in the Aml/Val and Aml/Val/HCTZ dosages categories are presented in Table 2.

Change in mean sitting blood pressure from baseline to week 26 endpoint.

Change in msSBP and msDBP from baseline to week 26 by select subgroups: (A) Aml/Val cohort and (B) Aml/Val/HCTZ cohort.
Mean change in blood pressure from baseline to week 26 by treatment dosages.
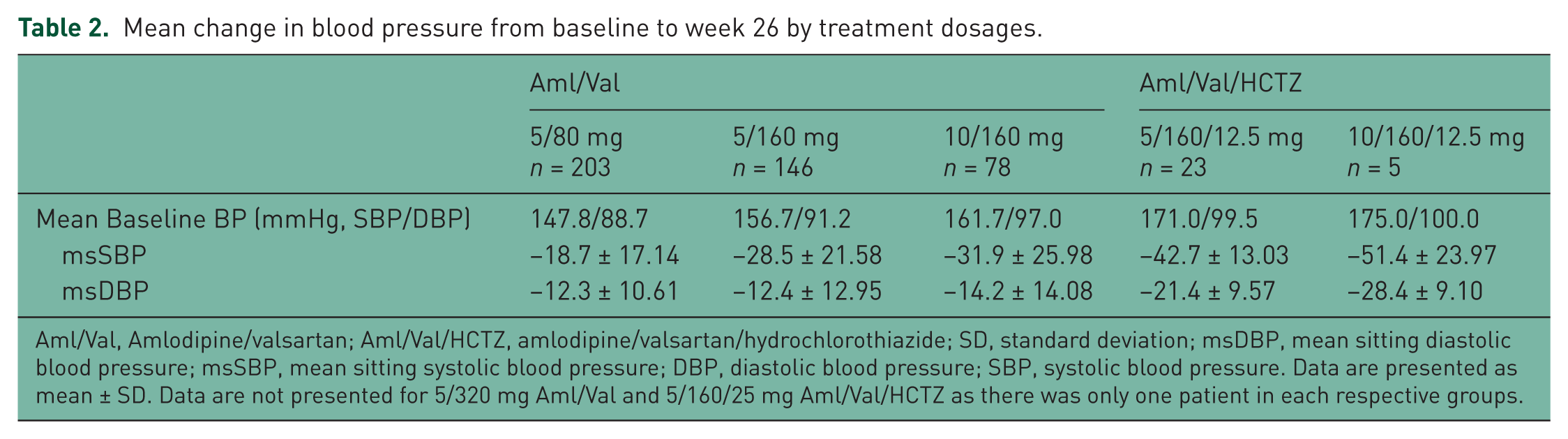
Aml/Val, Amlodipine/valsartan; Aml/Val/HCTZ, amlodipine/valsartan/hydrochlorothiazide; SD, standard deviation; msDBP, mean sitting diastolic blood pressure; msSBP, mean sitting systolic blood pressure; DBP, diastolic blood pressure; SBP, systolic blood pressure. Data are presented as mean ± SD. Data are not presented for 5/320 mg Aml/Val and 5/160/25 mg Aml/Val/HCTZ as there was only one patient in each respective groups.
Overall, some patients shifted therapy (either treatment or dosage) during observational period. At week 13 (visit 2), 78 patients (15.6%) and at week 26 (visit 3), 25 patients (5%) reported a change in the antihypertensive treatment prescribed at baseline (either medication or dosage). Unsatisfactory BP control was the most common reason for a change in treatment at visit 2. A total of 13 patients (3%) in the Aml/Val cohort switched therapy to Aml/Val/HCTZ combination by the end of study. At week 26, few patients shifted to use of higher dosages in the Aml/Val cohort compared with baseline, while patients on triple combination did not show any major shift in dosages during the study (Table 3).
Change in treatment dosages of Aml/Val and Aml/Val/HCTZ cohorts from baseline to end of study.

Aml/Val, amlodipine/valsartan; Aml/Val/HCTZ, amlodipine/valsartan/hydrochlorothiazide. Data are presented as number of patients in respective cohorts unless otherwise specified. Gray colored cells show shift to higher dosages; white colored cells represent no dosage shifts; blue colored cells show shift to lower dosages.
BP control and response rate
The BP goal according to the WHO criteria and diabetes status was reached by 237 patients (57.0%) in the Aml/Val cohort and by 16 patients (55.2%) in the Aml/Val/HCTZ cohort (Figure 4). A higher percentage of patients (n = 272, 65.5% in the Aml/Val cohort and n = 22, 75.9% in the Aml/Val/HCTZ cohort) achieved the BP goal, when defined independent of the diabetes status. A total of 55 (12%) patients in the Aml/Val cohort had already reached the therapeutic goal at baseline, and hence were excluded from the analysis. Similarly, 87 (17%) and 164 (33%) patients in the Aml/Val cohort reached the SBP response and DBP response criteria at baseline, respectively, and were also excluded from the analyses. The proportions of patients achieving SBP and DBP response rates for the respective cohorts (Aml/Val and Aml/Val/HCTZ) are presented in Figure 4.

Percentage of patients who achieved therapeutic blood pressure (BP) goal, SBP response and DBP response by week 26 endpoint.
With respect to dosage subgroups, the BP goal was achieved by 118 (58.1%), 80 (56.3%), and 38 (54.3%) patients in the 5/80, 5/160, and 10/160 mg dosage categories, respectively, in the Aml/Val cohort, and 12 (52.2%) and 4 (80%) patients in the 5/160/12.5 and 10/160/12.5 mg dosage categories, respectively, in the Aml/Val/HCTZ cohort. Similarly, the proportions of patients achieving SBP response were 138 (75.8%), 116 (87.2%), and 64 (94.1%) in the 5/80, 5/160, and 10/160 mg dosage categories, respectively, in the Aml/Val cohort, and 23 (100%) and 4 (80%) patients in the 5/160/12.5 and 10/160/12.5 mg dosage categories, respectively, in the Aml/Val/HCTZ cohort. Proportions of patients achieving DBP response were 111 (82.2%), 92 (86.8%), and 58 (89.2%) patients in the 5/80, 5/160, and 10/160 mg dosage categories, respectively, in the Aml/Val cohort, and 22 (95.7%) and 5 (100%) patients in the 5/160/12.5 and 10/160/12.5 mg dosage categories, respectively, in the Aml/Val/HCTZ cohort.
The effectiveness of hypertensive therapy was rated as ‘good’ or ‘very good’ by 71% of patients in the Aml/Val cohort and all patients in the Aml/Val/HCTZ cohort. Compliance was rated as ‘good’ or ‘very good’ by 66% and 100% of the patients in the respective cohorts.
Safety and tolerability
Overall, 40 (8%) patients reported at least one AE during the study period. The most common AEs were nausea (1.6%), vomiting (1.2%), headache (1.2%), and edema (1.2%) (Table 4). The incidence of edema was 12.1% (57 patients) at baseline and 9.1% (38 patients) at week 26 in the Aml/Val cohort and 24.1% (7 patients) at baseline and 3.4% (1 patient) at week 26 in the Aml/Val/HCTZ cohort.
Most commonly reported adverse events.

AEs, adverse events; Aml/Val, amlodipine/valsartan; Aml/Val/HCTZ, amlodipine/valsartan/hydrochlorothiazide.
Four patients (1%) reported nonfatal SAEs, the most common SAE was myocardial infarction (n = 3; 0.6%; patients had the medical history of myocardial infarction or heart attack or coronary heart disease and were treated with either 5/80 or 5/160 mg Aml/Val) followed by myocardial ischemia (n = 1; 0.2%) and chronic renal failure (n = 1; 0.2%). One patient who reported chronic renal failure which was suspected to be related to study drug. Three cases of death caused by myocardial infarction, myocardial ischemia, and road accident were reported during the study; none of them were considered by the investigator to be related to the study medication.
Tolerability was rated as ‘good’ or ‘very good’ by 66% of the patients treated with Aml/Val and 97% of the patients treated with Aml/Val/HCTZ.
Discussion
The EXCITE study demonstrates real-life effectiveness, safety, and tolerability of Aml/Val or Aml/Val/HCTZ SPC prescribed to patients with hypertension in Pakistan. The results are important for patients who are at a higher risk requiring two or more drugs for achieving effective control of BP [Allemann et al. 2008]. In these patients, a fixed-dose combination of amlodipine and valsartan (with or without a diuretic) may provide a safe and effective alternative treatment for hypertension.
Prior to study participation, 51% of patients in the Aml/Val group and 83% of patients in the Aml/Val/HCTZ group had unsatisfactory BP control with previous therapies. The unsatisfactory BP control with prior treatment was not surprising considering the fact that worldwide, low levels of BP control have been reported with antihypertensive treatment [Antikainen et al. 2006; Sever and Messerli, 2011; Wang et al. 2007; Wolf-Maier et al. 2003]. Even developed countries such as the USA and England had satisfactory BP control rates of only 54.5% and 40.3%, respectively, among patients using antihypertensive medications [Wolf-Maier et al. 2004]. International guidelines (JNC7 and European Society of Hypertension) consider that early combination therapy should be initiated in patients to achieve satisfactory levels of BP [Liu, 2011; Mancia et al. 2013]. In this study, patients were prescribed two or three drugs from different antihypertensive classes in a SPC to achieve effective BP control. This approach may lead to more effective and prompt lowering of BP, with improved compliance and usually fewer AEs compared with a single medication used at a higher dose as monotherapy [Hashmi et al. 2007]. Combination therapy is also helpful when phenotypes are not known. Wald and colleagues reported that the decrease in BP with combination of drugs belonging to different groups is five times greater than doubling the dose of a single drug [Wald et al. 2009].
Differences between the treatment cohorts were observed for the group size and baseline characteristics. The patients in the Aml/Val/HCTZ cohort presented a more chronic hypertensive profile (duration of hypertension was 9.3 years) compared with those in the Aml/Val cohort (duration of hypertension was 5.1 years). A higher percentage of patients in the Aml/Val/HCTZ cohort had cardiovascular risk factors such as diabetes, dyslipidemia, family history of hypertension, or BMI higher than 30 kg/m2 than those patients in the Aml/Val cohort. With the use of triple therapy, these patients showed a greater decrease in BP (43.9 mmHg reduction) as well as ‘good’ or ‘very good’ drug effectiveness as rated by physicians.
Approximately 9.2% of patients took concomitant antihypertensive medications during the study, which included mostly beta blockers. Although we did not capture reasons for prescription of beta blockers, it could be related to the underlying cardiovascular disease history of the patients participating in the study.
Overall, a large number of patients who needed combination therapy had diabetes. During the study duration, there were shifts in dosages within the cohorts mostly due to unsatisfactory BP control. Indeed, in our study, patients prescribed 5/80 mg of Aml/Val at baseline shifted to higher doses. However, the physicians’ prescribing habits leading to shifts in dosage cannot be ruled out.
The efficacy results of this real-life study correspond well with the previously reported data for Aml/Val (with or without HCTZ) from randomized trials. Data from randomized controlled studies have reported a mean reduction in msSBP of 12–24 mmHg with Aml/Val 5/80 and 5/160 mg doses, which was similar to those observed in this study [Huang et al. 2011; Wright et al. 2011]. In this study, a dose-dependent decrease in msSBP was observed with Aml/Val combination, which is consistent with the previously reported controlled studies [Philipp et al. 2007]. The mean reduction in msSBP was 19–32 mmHg (mean baseline BP 153.4/91.1 mmHg) with the 5/80, 5/160, and 10/160 mg doses of Aml/Val. However, other real-life studies report a mean SBP reduction of 29 to 44 mmHg (mean baseline BP 165.0/99.3 mmHg) with similar doses of Aml/Val [Chazova et al. 2011; Karpov et al. 2012]. The difference with the current study results with other real-world studies might be due to differences in the study population, which included pooled data from various countries, difference in baseline BP, and study duration. However, there are limited data on the efficacy of triple antihypertensive therapy. The mean reduction in BP in the Aml/Val/HCTZ group between this (43–51 mmHg in msSBP and 21–28 mmHg in msDBP; baseline BP: 171.6/99.3 mmHg) and another controlled study (40 mmHg in msSBP and 25 mmHg in msDBP at 10/320/25 mg dose; baseline BP: 169.6/106.4 mmHg) [Calhoun et al. 2009b] was similar irrespective of differences in doses and different study durations. Our results are comparable with the interim analysis of EXCITE pooled data, which showed a mean BP reduction of −28.7/−15.1 mmHg (baseline BP: 159.2/95.4) with Aml/Val and −36.6/−17.7 mmHg (baseline BP: 165.7/97.5) with Aml/Val/HCTZ [Shete et al. 2013]. BP goals were achieved by 54.3% and 55.4% of patients in the Aml/Val and Aml/Val/HCTZ groups, respectively, in the interim results of EXCITE pooled data [Shete et al. 2013] as compared with 57.0% and 55.2% of patients receiving Aml/Val and Aml/Val/HCTZ, respectively, in the present study. The mean reduction in BP was consistent in patients in the Aml/Val and Aml/Val/HCTZ groups regardless of diabetic/nondiabetic status, age, and BMI. These findings were similar to previously reported controlled efficacy studies [Allemann et al. 2008; Calhoun et al. 2013]. In the current study, triple therapy with Aml/Val/HCTZ produced reductions in msSBP and msDBP that were numerically better to that achieved with Aml/Val dual therapy regardless of age, diabetic/nondiabetic status, and BMI.
One of the challenges in improving BP control is patient compliance, which decreases with an increase in the number of medications. This challenge can be met to some extent by using a single pill containing two or more drugs [White, 2008]. In this study, patients in both the treatment cohorts were willing to continue the treatment, as they mostly rated the compliance and tolerability of the combinations as ‘good’ or ‘very good.’
In a real-life setting as seen in this study, the side effect profile of the combinations was mild to moderate and was consistent with the previously reported randomized and real-life clinical studies on Aml/Val with or without HCTZ [Allemann et al. 2008; Calhoun et al. 2009b, 2013; Chazova et al. 2011; Philipp et al. 2007; Smith et al. 2007].
Edema is a well-documented AE of CCBs and is the most frequent AE associated with amlodipine. It was one of the side effects reported in this study, but not to a noticeable extent. Incidence of edema was 12.1% in the Aml/Val cohort and 24.1% in the Aml/Val/HCTZ cohort at baseline, which can possibly be attributed to prior medications, especially calcium channel antagonists. The results on edema incidence by the end of study were in agreement with other real-life studies [Chazova et al. 2011; Karpov et al. 2012]. The other side effects reported in this study, such as nausea, vomiting, and headache, were mild in severity and thus the use of these drugs in combination can be considered safe.
Being a nonrandomized, uncontrolled, observational study, it has obvious limitations. There were some data anomalies which is not unusual in an observational study. The effectiveness and tolerability data were missing for 15–19% of the patients. Also, one needs to be cautious before deriving conclusions concerning comparative efficacy and tolerability of the study drug combinations due to the inherent nature of random variation associated with an observational study and the difference in patient numbers between cohorts [Carlson and Morrison, 2009]. This could result in a biased estimation of the effectiveness of the treatment of interest. Also, the number of patients in the Aml/Val/HCTZ cohort was less, although this reflects physicians’ prescribing habits to use dual or triple combination and also due to the observational design of the study. An additional limitation is the use of subjective evaluation scales for the investigator assessment of effectiveness, tolerability, and treatment compliance in this study.
Based on the above limitations, the findings of this observational study are limited to the cohorts that were studied and cannot be generalized to all patient populations.
Conclusion
In patients with hypertension, SPC treatment with Aml/Val and Aml/Val/HCTZ provided statistically significant reductions in both SBP and DBP from baseline. Reductions in BP were observed across all treatment dosages and patient subgroups regardless of diabetic/nondiabetic status, age, and BMI. In addition, both SPCs were generally well tolerated with few AEs. Almost half of all patients achieved a therapeutic BP goal of <140/90 mmHg (and <130/80 mmHg in patients with diabetes) in both treatment cohorts. The results provide evidence, from a real-life clinical practice, that Aml/Val and Aml/Val/HCTZ combinations are effective and well-tolerated SPC therapies for patients with hypertension from Pakistan.
Footnotes
Acknowledgements
The authors acknowledge all investigators at the participating centers and all patients for their commitment to the study that was supported by Novartis Pharma AG, Basel, Switzerland. The authors also acknowledge BIOP for data collection and data analysis. All authors participated in the development and writing of the manuscript and take full responsibility for the content of the article. The authors would like to thank Mr Vivek Khanna, M.Pharm, an employee of Novartis Healthcare Private Limited, India, for editorial assistance, preparation of figures and styling of the manuscript for journal submission. All authors approved the final draft that was submitted.
Funding
This work was supported by Novartis Pharma, Pakistan (cost center number 11E242244L).
Conflict of interest statement
Interim results of the EXCITE study were presented at the 23rd European Meeting on Hypertension and Cardiovascular Protection 2013 (ESH 2013), Milan, Italy, 14–17 June 2013. Dr Waheed Khan, Dr Nasir Moin, Dr Somia Iktidar, Dr Rizwan Abid, Dr Javed Afzal, and Dr Javed Sakrani have received honorariums and had consultancy agreement with Novartis Pharma, Pakistan, for the conduct of this trial. Dr Ahson Siddiqi, Dr Kishore Kumar, and Dr Neeta Maheshwary are employees of Novartis Pharma, Pakistan. In this study, the integrity of the study was not compromised by any financial interests. To assure that, investigators did not have financial arrangements that reward a particular study outcome, proprietary interest in the intervention being tested, significant equity in the manufacturer of the intervention, or significant payments of other sorts. The investigators were paid for the data filling and the examination of patient during the study period. Since this was a non-interventional study, patients were observed in real-life setting, and no support for any laboratory test or drug supply was provided to the investigators.
