Abstract
Objective:
Oxidative stress is currently considered to be the key factor in doxorubicin-induced cardiotoxicity. Comparatively small quantity of the endogenous antioxidant content of the heart is assumed to be the predisposing factor for doxorubicin-induced cardiotoxicity. The present research was designed to evaluate the antioxidant potential and tumor necrosis factor alpha-(TNF-α) inhibiting activity of sesame oil against acute doxorubicin-induced cardiotoxicity.
Methods:
Male Wistar albino rats (180–200 g) were administered sesame oil in two dissimilar doses (5 and 10 ml/kg body weight, orally) for 30 days, followed by a single dose of doxorubicin (30 mg/kg s.c.).
Results:
In the doxorubicin-treated group, increased oxidative stress was proven by a significant rise of thiobarbituric acid reactive substances level and a decrease of myocardial superoxide dismutase, catalase and reduced glutathione content. Histopathological studies showed myocardial necrosis with accumulation of inflammatory cells, vacuolization and overall enlargement of the myocardium. Western blot analysis showed marked expression of TNF-α in the myocardium. Alteration in biochemical parameters by doxorubicin administration was prevented significantly (p < 0.0001) in the 5 and 10 ml/kg sesame oil treated rat hearts. Treatment with 5 and 10 ml/kg of sesame oil reduced the doxorubicin-induced TNF-α expression in the myocardium, which was associated with reduced myocyte injury. The overall effect of sesame oil was comparable with probucol, which shows similar protection.
Conclusion:
The chronic oral administration of sesame oil prevents acute doxorubicin-induced cardiotoxicity by enhancing cardiac endogenous antioxidants and decreasing myocardial TNF-α expression.
Introduction
Doxorubicin (DOX) is a very effective anticancer antibiotic used in the treatment of many malignancies and solid tumors. DOX has a high toxicity profile including cardiotoxicity and bone marrow suppression, which limits the clinical usefulness of DOX chronically. In particular, cardiotoxicity produced by DOX is dose dependent, which leads to irreversible heart failure and cardiomyopathy [Van Acker et al. 2000; Alkreathy et al. 2010]. Of several mechanisms involving in the development of DOX-induced cardiotoxicity, such as the generation of reactive oxygen species (ROS), calcium accumulation and the dysfunction of mitochondria, the free radical mechanism is the most important and also well documented [Van Acker et al. 2000; Alkreathy et al. 2010]. Generation of free radicals in the myocardium may cause more damage when compared with other tissues due to a low level of defensive antioxidant enzymes such as superoxide dismutase (SOD) and catalase (CAT) [Van Acker et al. 2000; Mukherjee et al. 2003]. Thus, administration of exogenous antioxidants as drugs may protect the myocardium from DOX-induced free radical damage [Alkreathy et al. 2010; Patil and Balaraman, 2011]. In the past, several studies have concluded that antioxidants such as α-tocopherol (αTC), a-phenyl-tert-butyl-nitrone, gave protection from DOX-induced myocardial injury without disturbing its anticancer action [Patil and Balaraman, 2011].
Tumor necrosis factor alpha (TNF-α) is an inflammatory biomarker for cardiovascular disease. Cardiotoxicity through oxidative stress mediates the expression of different cytokines, particularly TNF-α [Mukherjee et al. 2003; Zanwar et al. 2013]. The expression of TNF-α in myocardium is greater during DOX administration. The inhibition of TNF-α expression during oxidative stress through the administration of drugs has possible therapeutic value [Mukherjee et al. 2003].
Sesame oil is one of the major cooking oils used in human diets with known antioxidant constituents [Gauthaman and Saleem, 2009; Saleem et al. 2012a]. It is obtained from the seeds of Sesamum indicum L. (Pedaliaceae) and contains different fatty acids and nonfat antioxidants, as well as tocoferol, sesamin, sesamolin and sesamol [Fukuda, 1990]. Ahmad and colleagues studied the antioxidant and neuroprotective effects of sesame oil [Ahmad et al. 2012]. Karatzi and colleagues reported a beneficial effect of daily intake of sesame oil in endothelial dysfunction in hypertensive men [Karatzi et al. 2013]. Recently, several researchers reported the antioxidant role of sesame oil in experimental models and also showed that it protected the heart by eliminating risk factors [Arumugam and Ramesh, 2011; Alipoor et al. 2012].
In view of the above literature, the present study was designed to investigate the cardioprotective role of sesame oil against DOX-induced cardiotoxicity in terms of inhibition of oxidative stress, TNF-α expression and myocardial injury.
Materials and methods
Drugs and chemicals
Sesame oil was obtained from VV & Sons edible oil Ltd, Viruthunagar, India as a gift sample. All chemicals were of analytical grade purchased from Sigma chemicals, USA.
Experimental animals
Male Wistar albino rats of body weight 180–200 g (3 months old) were obtained from the Institute Animal House. The animals were fed with a standard pellet diet (Sai Durga Feeds and Foods, Bangalore) and had access to water ad libitum. The rats were assimilated in the department animal house and followed the guidelines given by the Committee for the Purpose of Supervision and Control of Experiments on Animals (CPCSEA), India. The research protocol was approved by the Institute animal ethical committee (1220/a/08/CPCSEA/ANCP).
DOX-induced myocardial necrosis
A total of 30 healthy rats were divided into 5 different groups (n = 6) and fed with sesame oil by the oral route once a day for 30 days except the saline control and probucol-treated group. At the end of the treatment period the rats from all groups except the control group were administered DOX 30 mg/kg s.c., to induce myocardial injury. After 24 hours of DOX administration the rats were anesthetized with pentobarbitone sodium (60 mg/kg−1), hearts were isolated and subjected to biochemical and histopathological studies. All of the animals carefully monitored for mortality during treatment periods.
Treatment protocol
The groups studied were divided according to the following treatment protocols.
Group C: Saline-treated (10 ml/kg) rats.
Group DOX: Saline-treated rats that received DOX (30 mg/kg).
Group SO-1+DOX: Sesame-oil-treated (5 ml/kg) rats that received DOX (30 mg/kg).
Group SO-2+DOX: Sesame-oil-treated (10 ml/kg) rats that received DOX (30 mg/kg).
Group PRO+DOX: Probucol-treated (10 mg/kg) rats that received DOX (30 mg/kg).
Biochemical parameters
The following biochemical parameters were estimated in the heart homogenate.
Myocardial thiobarbituric acid reactive substances
Thiobarbituric acid reactive substances (TBARS) levels in the myocardium were measured by a method of Ohkawa and colleagues [Ohkawa et al. 1979]. Hearts were homogenized in 10% trichloroacetic acid at 4°C. Then, 0.2 ml homogenate was pipetted into a test tube, followed by the addition of 0.2 ml of 8.1% sodium dodecyl sulphate (SDS), 1.5 ml of 20% acetic acid (pH-3.5) and 1.5 ml of 0.8% thiobarbituric acid (TBA). Tubes were heated for 60 min at 95°C and then chilled on ice. Double-distilled water (1.0 ml) and n-butanol:pyridine (15:1 v/v) combination (5.0 ml) were added to the test tubes and they were then centrifuged at 4000g for 10 min. The absorbance of color formed in the organic layer was measured at 532 nm. Data are expressed as nMole of TBARS/g wet wt.
Myocardial reduced glutathione (GSH)
Glutathione (GSH) was estimated by the method of Ellman [Ellman, 1959]. The reaction mixture contained 0.1 ml of supernatant, 2.0 ml of 0.3 M phosphate buffer (pH 8.4), 0.4 ml of double-distilled water and 0.5 ml of 5,5-dithiobis-2-nitrobenzoic acid. The reaction mixture was incubated for 10 min and the absorbance was measured at 412 nm. Data are expressed as Mole/g wet wt.
SOD
SOD levels in the hearts were determined by the method of McCord & Firdovich modified by Kakkar and colleagues [Kakkar et al. 1984]. A sample (0.6 ml) was added to sodium pyrophosphate buffer (pH 8.3) followed by the addition of 0.1 ml of 186 M phenazine methosulphate, 0.3 ml of 300 mM nitroblue tetrazolium and 0.2 ml of 780 M nicotinamide adenine dinucleotide. The reaction mixture was incubated for 90 s at 30°C and stopped by adding 1 ml of acetic acid. n-Butanol (4 ml) was then added and the mixture was centrifuged at 3000g for 10 min. The absorbance of the organic layer was measured at 560 nm. Data are expressed as units per milligram of protein.
CAT
CAT was estimated by the method described by Aebi [Aebi, 1974]. A sample was added to a 3 ml cuvette that contained 1.95 ml of 50 mM phosphate buffer (pH 7.0). Then 1 ml of 30 mM hydrogen peroxide was added and changes in absorbance were followed for 30 s at 240 nm at intervals of 15 s. Data are expressed as units per milligram of protein.
Protein
Protein estimation for the tissue sample of SOD and CAT were done by the method of Bradford [Bradford, 1976]. A sample was added up to 20 µl with double-distilled water, 50 µl in sodium hydroxide and 1 ml of Bradford reagent and kept aside for 10 min after vortexing. The absorbance was measured at 595 nm.
Cardiac marker enzymes
Lactate dehydrogenase (LDH), creatine kinase (CK) and aspartate transaminase (AST) levels in the myocardium were estimated by using respective kits as per the manufacturer’s instruction booklet (Transasia Bio-Medicals Limited, Solan).
Western blot analysis
The heart tissues were homogenized in 0.1 mM phosphate buffer containing 5% SDS, 1% mercaptoethanol and 0.1 mM phenyl methyl sulfonyl fluoride (PMSF) for 4 s using a tissue homogenizer at 4°C. The homogenate was then strained through a 27 Gauge needle, followed by centrifugation at 14,000g for 10 min. The supernatant, representing protein lysate, was divided into small aliquots and stored at −70°C until use. At the time of analysis, samples were thawed and volumes were pipetted to allow loading of approximately 100 µg of protein per lane on a slab gel. Proteins were separated by SDS-polyacrylamide-gel electrophoresis on 1 mm thick 12.5% acrylamide gels. After electrophoresis, proteins on the gel were transferred to nitrocellulose membranes by electro elution. Protein transfer was confirmed by employing prestained molecular weight markers. Following the transfer, the membrane was blocked by nonfat dry milk, and the nitrocellulose membranes were incubated with rabbit polyclonal anti-TNFα (Santa Cruz Biotechnology, USA) at a dilution of 1:250 followed by incubation with secondary antibody, which was conjugated with antigoat IgG (Santa Cruz Biotechnology, USA; 1:200 dilution). Subsequently, the membranes were incubated with avidin–biotin HRP complex (Santa Cruz Biotechnology, USA). The signal was detected using diamino benzidine. The membrane was developed and the levels of TNF-α were calculated.
Histological examination
The rat hearts were removed, washed immediately with saline and then fixed at 10% buffered formalin. The hearts were embedded in paraffin sections cut at 5 µ stained with hematoxylin and eosin. These sections were then examined under a light microscope for histological changes.
Statistical analysis
All values are expressed as mean ± SEM for six animals in each group. Data for various biochemical parameters were analyzed using analysis of variance (ANOVA) using GraphPad (Version 3.06, La Jolla, CA, USA). Significance is set at p < 0.05.
Results
There was no mortality observed in any of the group during treatment periods.
Heart weight:body weight ratio
There was a significant decrease in heart weight:body weight ratio (Table 1) of animals treated with DOX (group DOX) when compared with control rats (group C). There was no significant decrease in the heart weight:body weight ratio of animals treated with sesame oil (group SO-1-DOX and group SO-2-DOX) and probucol (group PRO-DOX) when compared with group DOX.
Results of heart weight:body weight ratio of sesame oil + DOX-treated rats.

All values are expressed as mean ± SEM [n = 6].
p < 0.0001 versus group C.
DOX, doxorubicin; PRO, probucol; SO, sesame oil.
Myocardial LDH, CK and AST
There was a significant decrease in the level of LDH, CK (p < 0.0001) and AST (p < 0.001) in heart homogenate of rats treated with DOX (group DOX) when compared with group C rats (Table 2). There was significant (p < 0.0001) increase in the level of LDH, CK and AST in group SO-1-DOX, SO-2-DOX and PRO-DOX in comparison with the DOX group.
Effect of chronic administration of sesame oil on LDH, CK and AST in rat heart homogenate.
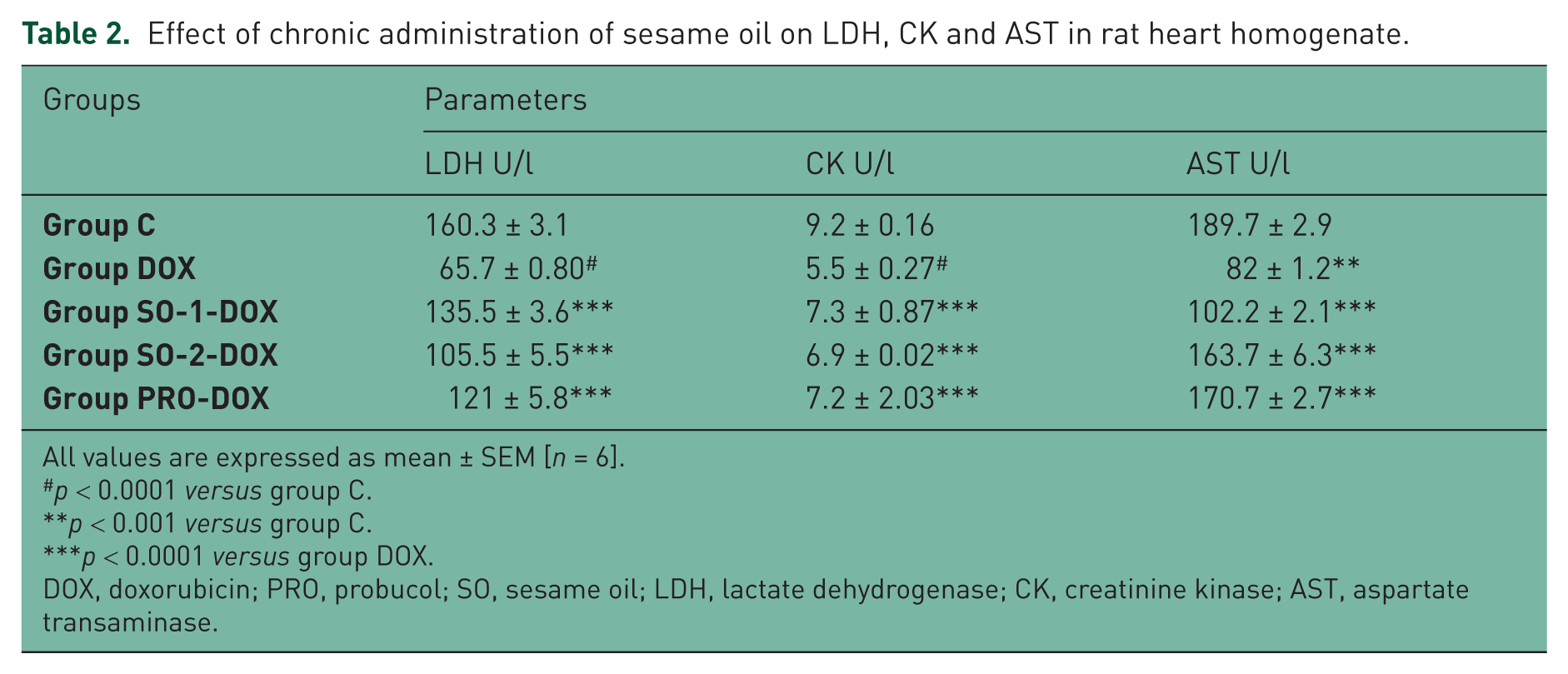
All values are expressed as mean ± SEM [n = 6].
p < 0.0001 versus group C.
p < 0.001 versus group C.
p < 0.0001 versus group DOX.
DOX, doxorubicin; PRO, probucol; SO, sesame oil; LDH, lactate dehydrogenase; CK, creatinine kinase; AST, aspartate transaminase.
Myocardial TBARS
There was a significant increase in the level of TBARS (p < 0.0001) in heart homogenate of rats treated with DOX (group DOX) when compared with group C rats (Table 3). There was a significant (p < 0.0001) decrease in the level of TBARS in group SO-1-DOX, SO-2-DOX and PRO-DOX in comparison with the DOX group.
Effect of chronic administration of sesame oil on TBARS, GSH, SOD and catalase in rat heart homogenate.
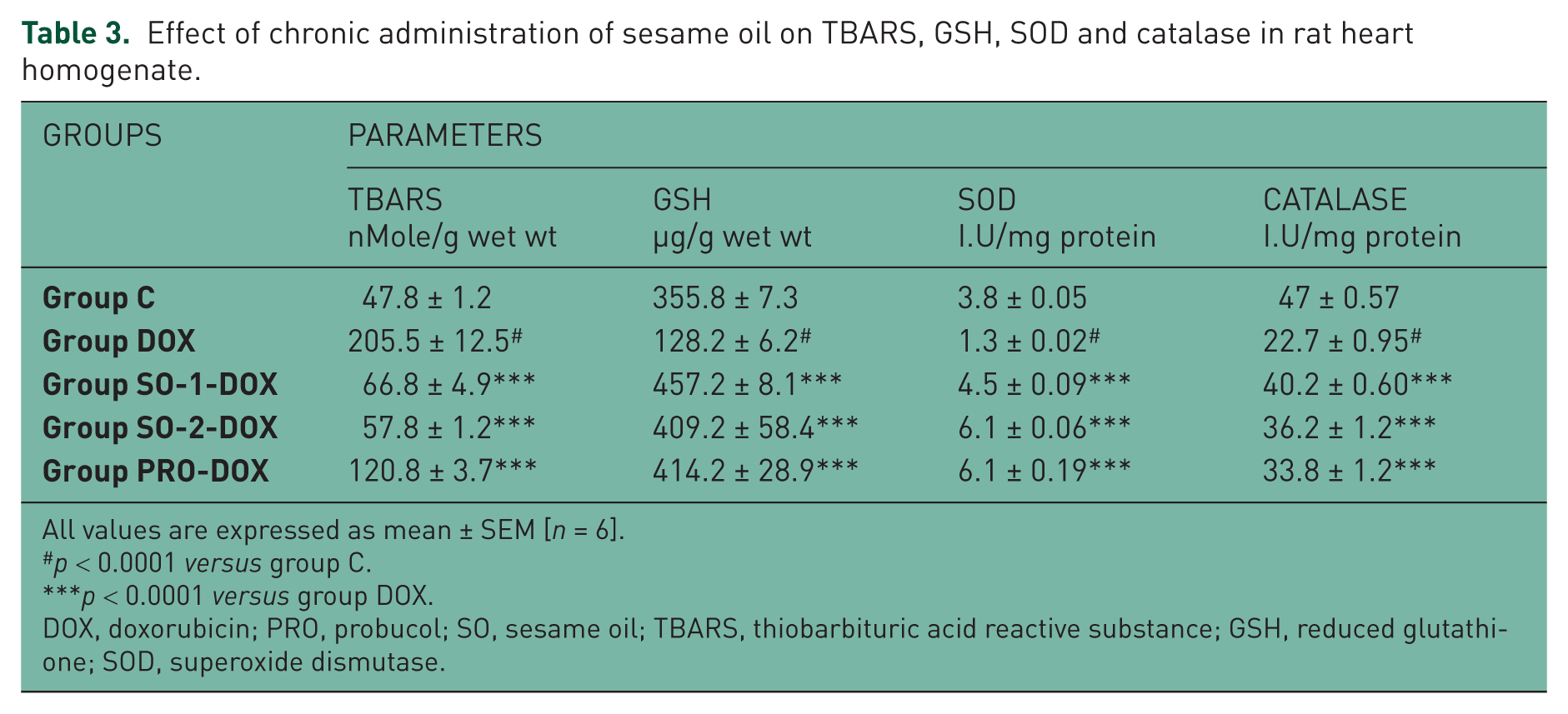
All values are expressed as mean ± SEM [n = 6].
p < 0.0001 versus group C.
p < 0.0001 versus group DOX.
DOX, doxorubicin; PRO, probucol; SO, sesame oil; TBARS, thiobarbituric acid reactive substance; GSH, reduced glutathione; SOD, superoxide dismutase.
Myocardial GSH, SOD and CAT
There was a significant (p < 0.0001) decrease in the level of GSH, SOD and CAT in heart homogenate of rats treated with DOX (group DOX) when compared with group C rats (Table 3). There was a significant (p < 0.0001) increase in the level of GSH, SOD and CAT in group SO-1-DOX, SO-2-DOX and PRO-DOX in comparison with the DOX group.
Myocardial TNF-α expression
There was a marked expression of TNF-α in DOX group when compared with group C (Figure 1). The expression of TNF-α in rats treated with sesame oil was significantly decreased when compared to DOX group. Expression of TNF-α in probucol treated group was slightly higher when compared to sesame oil treated groups and lower than DOX group.

Results of Western blot analysis of tumor necrosis factor α of sesame-oil-treated rats. DOX, doxorubicin; PRO, probucol; SO, sesame oil.
Histopathological changes
Rats treated with vehicle showed the normal myofibrillar structure (group C); see Figure 2. Rats treated with DOX showed myocardial necrosis with accumulation of inflammatory cells, vacuolization and overall enlargement of the myocardium (group DOX). Rats treated with sesame oil (group SO-1-DOX and SO-2-DOX) and probucol (group PRO-DOX) showed a well-preserved myocardium when compared with the DOX group.
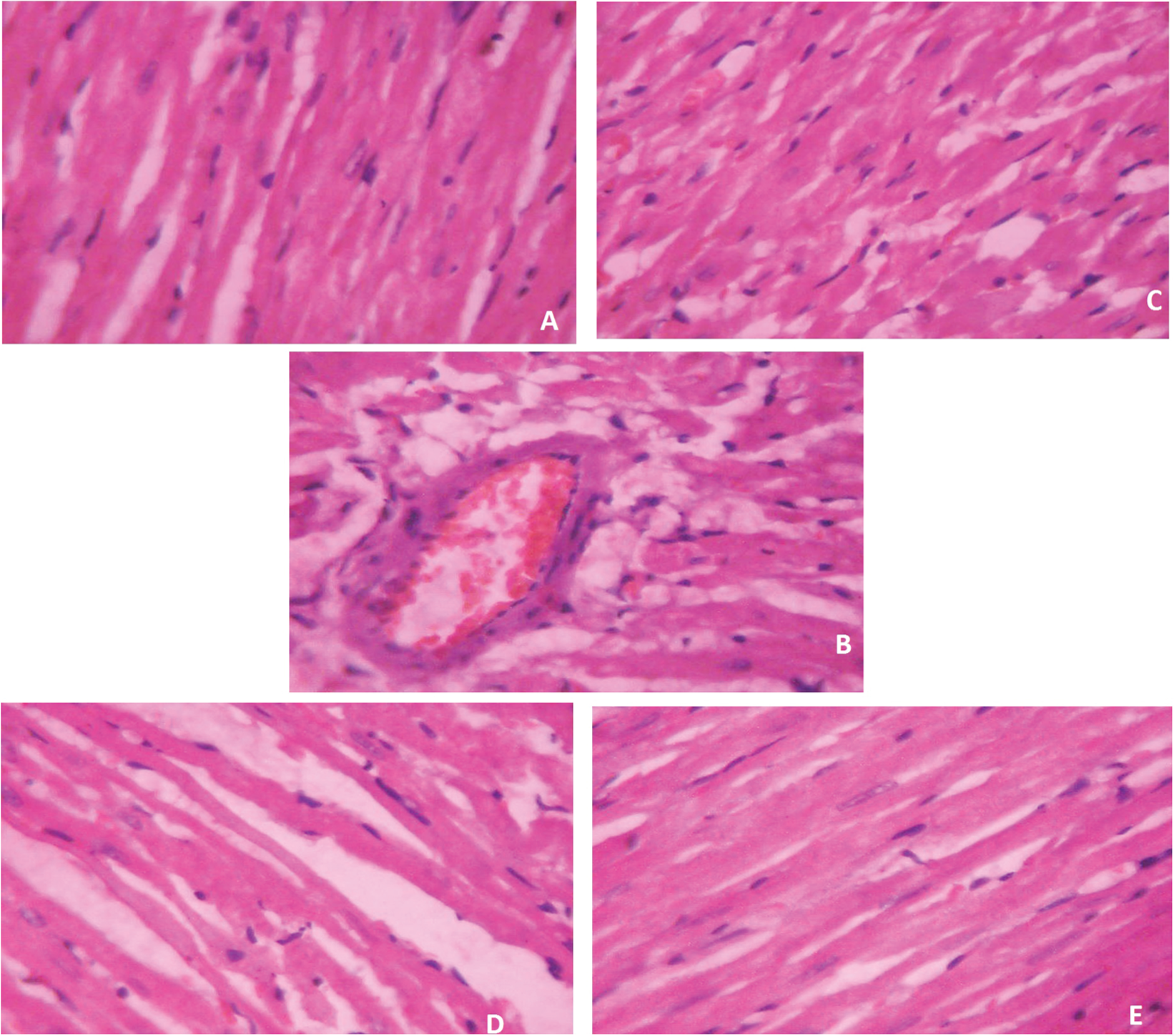
Histopathological report. (A) Rats treated with vehicle show normal myofibrillar structure (group C). (B) Rats treated with DOX show myocardial necrosis with accumulation of inflammatory cells, vacuolization and overall enlargement of myocardium (group DOX). (C–E) Rats treated with sesame oil (group SO-1-DOX and SO-2-DOX) and probucol (group PRO-DOX) show well-preserved myocardium when compared with the DOX group.
Discussion
The present research is undertaken to find out the cardioprotective effect of sesame oil against DOX-induced cardiotoxicity.
Oxidative stress shows a key role in the progress of DOX-induced cardiotoxicity [Karim et al. 2001; Viswanatha Swamy et al. 2011]. Generation of ROS by DOX is mediated by nonenzymatic and enzymatic pathways [Vander Heide and L’Ecuyer, 2007]. During the nonenzymatic pathway, DOX reacts directly with iron to form iron (Fe2+) radical complex. Fe2+ reacts with hydrogen peroxide to form the highly reactive hydroxyl (OH•.) radical [Van Acker et al. 2000; Alkreathy et al. 2010]. During the enzymatic pathway, DOX reacts with cardiolipin of the mitochondrial membrane, causing respiratory-chain defects that lead to the generation of greater amounts of free radicals [Vander Heide and L’Ecuyer, 2007]. The role of free radicals in the mechanism of DOX-induced cardiotoxicity has been well documented [Allen, 1992; Goebel and Kaplan, 1992; Van Acker et al. 2000]. Another study reported that the generation of superoxide anions and their derivatives induces the peroxidation of lipids in the cell membrane [Alkreathy et al. 2010; Viswanatha Swamy et al. 2011]. In the present study administration of DOX-induced oxidative stress, which is evidenced by increased levels of lipid peroxidation in terms of TBARS. Sesame oil (5 and 10 mg/kg) and probucol significantly decreased the level of TBARS, indicating the reduction of oxidative stress by the administration of these drugs.
Endogenous antioxidants such as SOD and CAT protect the myocardium from oxidative damage. In some studies, it was reported that transgenic expression of SOD and CAT protected the myocardium from free radical damage [Vander Heide and L’Ecuyer, 2007]. Depletion of antioxidant enzymes GSH, SOD and CAT were observed in DOX-treated rats when compared with the control group, indicating the loss of these defensive mechanisms in the heart. Similar changes have been observed by other investigators on DOX-induced cardiotoxicity [Mukherjee et al. 2003; Barman et al. 2013]. Administration of sesame oil and probucol enhanced the myocardial antioxidant enzymes, indicating the protective role of these drugs against oxidative stress through its putative antioxidant property. The antioxidant property of probucol in small animal models is well documented [Li et al. 2000; Mukherjee et al. 2003]. Previously it was reported that the chronic administration of sesame oil augments the endogenous antioxidants in ischemic myocardium [Saleem et al. 2012b]. A similar effect was observed in the present study, confirming the putative antioxidant property of sesame oil in the heart.
Free radicals such as superoxide anions and hydroxyl radicals may damage several intracellular components. Oxygen or nutritional deficiency may alter the myocardial cells and the cell membrane may become more penetrable and breaches, resulting in an outflow of myocardial marker enzymes such as LDH, CK and AST [Viswanatha Swamy et al. 2011]. Abnormal levels of these myocardial markers in blood and heart after myocardial injury indicates the magnitude of myocardial damage [Alkreathy et al. 2010]. Depletion of myocardial LDH, CK and AST in DOX-administered rats confirmed the myocardial necrosis caused by DOX. Administration of sesame oil and probucol significantly elevated these myocardial marker enzymes in heart homogenate, indicating that sesame oil protected the myocardium from DOX damage.
Expression of TNF-α during oxidative stress of the heart develops several pathological conditions such as cardiomyopathy, myocytes apoptosis and the worsening of myocardial function [Kubota et al. 1997; Bozkurt et al. 1998]. The relationship between oxidative stress and expression of TNF-α has been reviewed and well documented. ROS activate nuclear factor kappa B (NFκB) and p38 mitogen activated protein kinase (MAP kinase), which further stimulate the release of TNF-α in the myocardium [Bogoyevitch et al. 1996; Barnes and Karin, 1997]. Administration of DOX enhanced the expression of TNF-α in myocardium. Similar findings have been observed by several investigators [Mukherjee et al. 2003; Zanwar et al. 2013]. Sesame oil (5 and 10 mg/kg) and probucol reduced the myocardial expression of TNF-α following DOX administration. This result confirmed that sesame oil reduced the oxidative myocardial injury via inhibition of TNF-α expression.
This biochemical report was further supported by histopathological analysis. DOX-administered heart shows myocardial necrosis with accumulation of inflammatory cells, vacuolization and overall enlargement of the myocardium. Rats treated with sesame oil and probucol showed a well-preserved myocardium, indicating the protective role of these drugs against DOX-induced cardiotoxicity.
Conclusion
Sesame oil shows protective action against DOX-induced cardiotoxicity through enhancement of endogenous antioxidants, reduction of lipid peroxidation and TNF-α in rat myocardium. The present research confirmed the nutritional value of sesame oil in the prevention of cardiovascular diseases.
Footnotes
Acknowledgements
The authors are grateful to the management team and principal of the Annamacharya College of Pharmacy, Rajampet, for providing facilities for the present work.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors have no potential conflicts of interest to declare with respect to the research, authorship, and/or publication of this article.
