Abstract
Background and Objectives:
The recent SHARP trial clearly demonstrated that a reduction in low-density lipoprotein (LDL) cholesterol with a daily regimen of simvastatin plus ezetimibe safely reduced the incidence of major atherosclerotic events in patients with chronic kidney disease (CKD). We aimed to compare the efficacy of and adverse effects from statin uptitration versus statin in combination with ezetimibe since only a few studies have addressed this question.
Methods:
This was a randomized, open-label, multicenter trial that included 286 patients with CKD whose LDL cholesterol levels were not reduced below 120 mg/dl despite a minimum dose of statin therapy. Patients received double doses of statin or usual statin dose with the addition of ezetimibe 10 mg daily. The observation period was 1 year during which time patients were checked regularly in clinic for adverse effects as well as for usual laboratory examinations. The key prespecified outcome was the incidence of adverse effects, which included skeletal muscle complaints, myalgia, muscle weakness, and muscle cramps with and without elevated CK levels. Increases in alanine transaminase (ALT) or aspartate transaminase (AST) levels >2 times the upper limit of normal (ULN) were considered clinically significant adverse effects.
Results:
Adverse events occurred in 9/145 in the combination group and in 24/141 in the statin uptitration group (p < 0.01). Moreover, in patients with CKD of stages 3–5, rates were 6/58 in the combination group versus 20/52 in the statin uptitration group (p < 0.01). No serious adverse effects such as rhabdomyolysis were noted in either group. Serum creatinine levels remained essentially unchanged in both groups except in CKD stages 4 and 5. Reductions in LDL cholesterol were similar between the two groups at the start of and at the end of the study. During the study, no atherosclerotic events were reported in either group.
Conclusion:
When statin uptitration produces adverse effects such as myopathy, combination therapy with ezetimibe is recommended instead of statin alone.
Introduction
Muscle-related adverse effects from statins were reported to occur in 10–12% of patients although statins are well tolerated by most patients [Mikhailidis et al. 2011; Abd and Jacobson, 2011; Baumelou et al. 2005; Chertow et al. 2003]. Such adverse effects of statins would be more likely to occur in the presence of several factors, including compromised hepatic and renal function, hyperthyroidism, diabetes, and concomitant medication. Among those factors, severe renal insufficiency is an established risk factor for statin-induced myopathy [Sung et al. 1998]. On the other hand, it is well known that patients with chronic kidney disease (CKD) have a higher burden of cardiovascular disease (CVD) than the general population [Upadhyay and Weiner, 2012; Foley et al. 1998; Sharma et al. 2009]. Several large clinical trials and post hoc analyses examining lipid-lowering therapies and clinical outcomes in CKD have been published [Strippoli et al. 2008] and a previous recent meta-analysis [Foley et al. 1998] showed that lipid-lowering therapy decreased cardiac death and atherosclerosis-mediated cardiovascular events in persons with CKD. More recently the Study of Heart and Renal Protection (SHARP) clearly demonstrated that a reduction in low-density lipoprotein (LDL) cholesterol with simvastatin 20 mg plus ezetimibe 10 mg daily reduced the incidence of major atherosclerotic events in a wide range of patients with advanced CKD [Baigent et al. 2011]. However, in spite of the favorable data on lipid-lowering therapy, two important clinical issues remain unresolved. First, it is not clear which regimen results in the better reduction in LDL cholesterol levels in patients with CKD, a statin alone or a statin in combination with ezetimibe. It is also uncertain whether one regimen is safer and associated with a lower frequency of adverse effects than the other. In the present study, the aim was to compare the occurrence of adverse effects of statin uptitration versus stain in combination with ezetimibe in patients with CKD.
Methods
This trial was a prospective, multicenter, open-label, randomized trial in Japan. It was registered in the University Hospital Medical Intervention Network Clinical Trials Registry (UMIN-CTR) under the trial identification number UMIN000002935 and was approved by the Internal Review Board of Saitama Medical University Hospital. The trial was conducted in accordance with the Declaration of Helsinki Principles. Written informed consent was obtained prior to patient enrollment and after a thorough explanation of the trial’s objectives, duration and structure.
Participants
The inclusion criteria for this trial included fulfillment of all of the following: (1) age from >35 to <75 years; (2) undergoing treatment with low-dose statins; (3) LDL cholesterol >120 mg/dl; and (4) positive proteinuria or estimated glomerular filtration rate (eGFR) <60 ml/min/1.73 m2 for more than 3 months before enrollment. According to CKD Guidelines of the Japanese Society of Nephrology, eGFR was calculated by the following Modification of Diet in Renal Disease Study Group (MDRD) equation for Japanese people: eGFR =194 × Cr – 1.094 × age −0.287 (× 0.739, if female) [Matsuo et al. 2009; Horio et al. 2010].
Patients were excluded based on the presence of at least one of the following criteria: (1) undergoing dialysis therapy; (2) uncontrolled hypertension; (3) uncontrolled diabetes; (4) severe liver disease with alanine transaminase (ALT) levels >2 times the upper limit of normal (ULN); (5) triglycerides (TG) >400 mg/dl; (6) secondary hyperlipidemia or hyperlipidemia associated with the administration of a drug; (7) homozygous familial hypercholesterolemia; (8) unstable angina, myocardial infarction, surgical coronary intervention or stroke within 3 months of study entry; (9) pregnancy, possible pregnancy, desire to become pregnant during the study period, or lactation; (10) history of hypersensitivity to any ingredient in ezetimibe tablets; and (11) deemed inappropriate for study entry by the investigator.
Patients were considered to have diabetes if they had been diagnosed previously by a doctor judged using a diagnostic criteria (a fasting glucose of >126 mg or HbA1c >6.5%) or if they were receiving insulin or oral antidiabetic medications. The presence of hypertension was determined by either blood pressure levels greater than 140/90 mmHg or the use of antihypertensive drugs.
Study design
The choice of statins was at the discretion of the physician. Patients should receive a minimum dose of statin regardless of the statin. Patients were randomly assigned to a group with a doubling of the dose of statin (statin uptitration group) or receiving statin and ezetimibe in combination (combination group) based on stratified randomization: randomization was performed by using the dynamic allocation method after stratification [Signorini, 1993] by (1) male or female, (2) with or without hypertension and (3) with or without diabetes. The target serum LDL cholesterol level was <120 mg/dl. The follow-up period was 1 year.
Outcome measures
The primary objective of this trial was to assess the incidence of adverse effects, which included muscle complaints, myalgia, muscle weakness, and muscle cramps with and without elevated creatinine kinase (CK) levels. Increases in ALT and aspartate transaminase (AST) levels >2 times the ULN were considered to indicate liver toxicity.
The secondary outcomes were (1) changes in serum LDL and high-density lipoprotein (HDL) cholesterol levels, (2) changes in albumin/creatinine of urinary excretion (mg/gCr) and (3) the rate of decline in renal function. The change in eGFR (ml/min/1.73 m2) was calculated by the modified MDRD equation for Japanese people [Matsuo et al. 2009; Horio et al. 2010].
Definitions
In the present study, we employed the definitions set by the ACC/AHA/NHLBI [Pasternak et al. 2002], which are the most widely used in the literature. ‘Myopathy’ is a general term to describe all skeletal muscle-related adverse effects while ‘myalgia’ refers to muscle ache or weakness without CK elevation. ‘Myositis’ refers to muscle symptoms with an elevation in CK usually >2 times the ULN range [Pasternak et al. 2002; McKenney et al. 2006; Sewright et al. 2007].
Laboratory tests including those for total, LDL and HDL cholesterol, CK, ALT, AST, creatinine, urea nitrogen, electrolytes, total blood counts and HbA1c during the study period were performed at laboratories of each participating hospital and were scheduled to be performed just before the start of treatment, and 1, 3, 6 and 12 months after the start of the study. Urine creatinine and albumin were measured by enzymatic methods.
Statistics
Data are expressed as mean ± standard deviation (SD). Repeated-measurements analysis was used to analyze time-dependent data and multiple comparisons of continuous data were performed by analysis of variance. Statistical comparisons used to test differences between two independent groups were Student’s t-test or Mann–Whitney’s U-test, as appropriate.
In the present study, patients with CKD were eligible and the expected adverse effects were 25% in the statin uptitration group. If a 15% reduction in adverse effects in the combination group was expected, the necessary number of participants for assuring the noninferiority of the combination arm against the statin uptitration group was 150 for detecting a clinically significant risk ratio of 1.2 (two-sided a = 0.05; 1 – b = 0.80; drop-out rate = 10%).
In this study, muscle-related adverse effects produced by statin uptitration might be reduced by the addition of ezetimibe, 10 mg daily, to low doses of statins in patients with CKD of stages 1 to 5. In addition, it was expected that there would be no differences in the decrease in LDL cholesterol levels between the two groups.
Results
Demographics
In total, 356 subjects were screened and 296 patients were randomized for treatment (Figure 1), with 288 patients (96%) completing the study. Baseline characteristics for gender and age were comparable in the two treatment groups. Approximately 84% of patients were hypertensive and 34% of patients had diabetes. Systolic and diastolic blood pressure, LDL and HDL cholesterol and body mass index did not differ between the groups. There were no differences in the levels of eGFR and urinary excretion of albumin at the start of the study between the two groups (Table 1). There were no between-group differences in the prevalence of previous vascular diseases (Table 2).

Flow chart of patient enrollment and analysis.
Baseline characteristics of patients.

BP, blood pressure; eGFR, estimated glomerular filtration rate; HDL, high-density lipoprotein; LDL, low-density lipoprotein
Prevalence of previous vascular diseases.
Efficacy
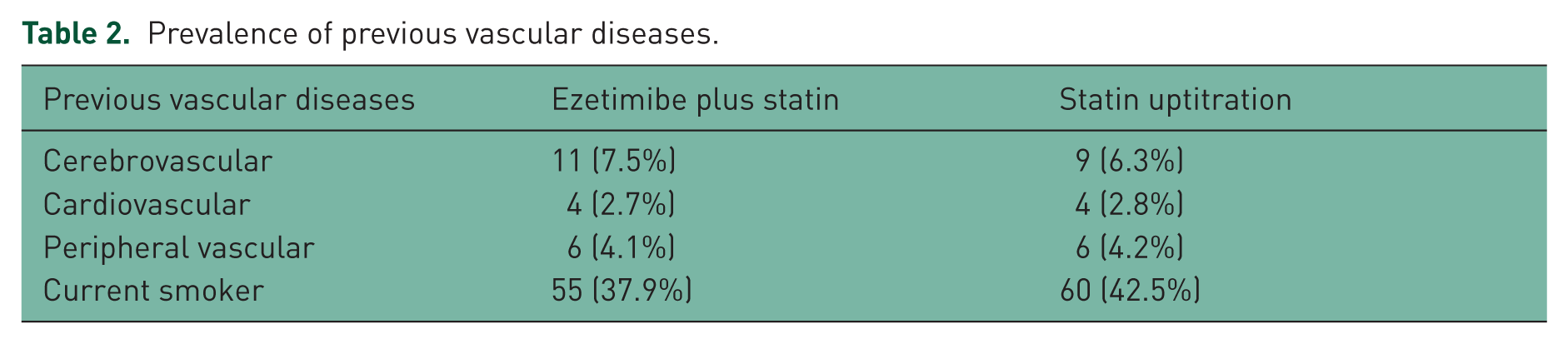
The addition of ezetimibe and uptitration of statins lowered LDL cholesterol in patients with CKD stages 1 and 2 and with CKD stages 3–5 by 15%, respectively (p < 0.05; see Figures 2 and 3). The proportional decreases in LDL cholesterol levels at 1 year in the two groups appeared similar among patients with CKD 1 and 2 and patients with CKD 3–5 (13 ± 5% versus 15 ± 6% decrease).
Levels of low-density lipoprotein (LDL) cholesterol in patients with chronic kidney disease (CKD) stages 1 and 2 in response to treatment with ezetimibe combined with statin or statin uptitration during the study period. Both treatments significantly reduced LDL cholesterol from baseline. *p < 0.05.

Levels of low-density lipoprotein (LDL) cholesterol in patients with chronic kidney disease (CKD) stages 3–5 and in response to treatment with ezetimibe combined with statin or statin uptitration during the study period. *p < 0.05.
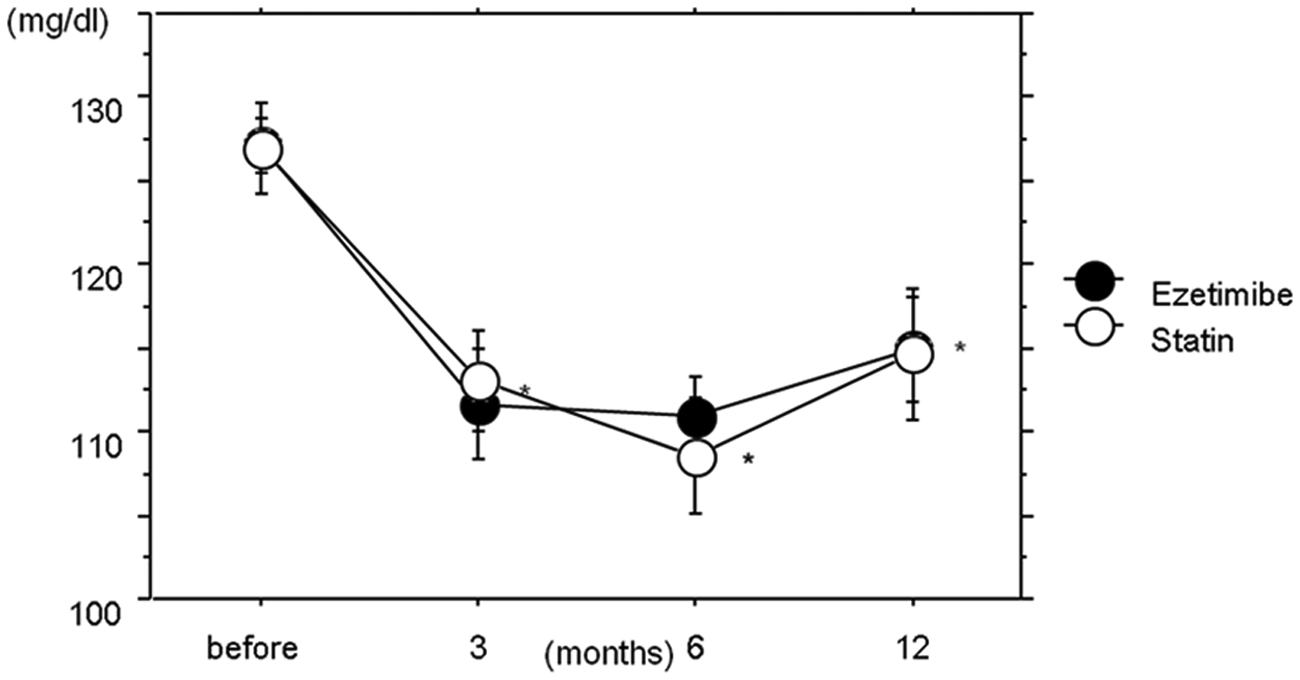
There was no significant effect on HDL cholesterol in the two groups throughout the study, although the relative increases at 6 months (10 ± 3%) were marginally nonsignificant (p = 0.07) in CKD stages 1 and 2 (Figure 4).

Levels of high-density lipoprotein (HDL) cholesterol in patients with chronic kidney disease (CKD) stage 1 and 2 in response to treatment with ezetimibe combined with statin or statin uptitration during the study period.*p < 0.05 between the two treatment groups. Treatment with ezetimibe combined with statin increased HDL cholesterol without significance (p = 0.07).
As shown in Figure 5, although the basal levels of HDL cholesterol in the combination group were significantly lower compared with those in the statin uptitration group (p < 0.05), at the end of the study, there was no difference between the two groups.
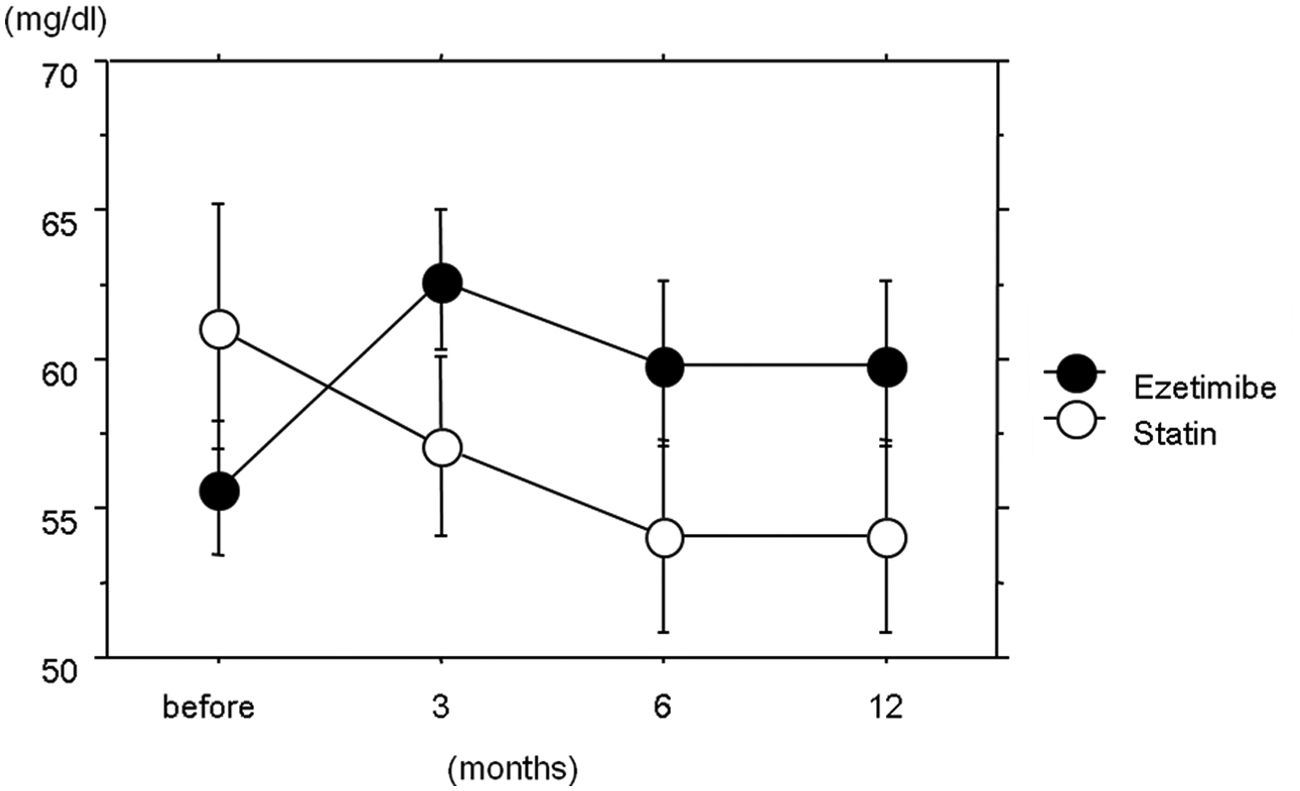
Levels of HDL cholesterol in patients with chronic kidney disease (CKD) stages 3–5 in with ezetimibe combined with statin or statin uptitration during the study period. Group differences by repeated-measures analysis of variance are shown in the figure. Data are means ± standard error of the mean.
Tolerability and safety
Approximately 95% of patients with CKD stages 1 and 2 receiving ezetimibe and statin in combination or statin uptitration completed the trial (Table 3). However, among patients with CKD stages 3–5, 20 of 52 (38%) patients in the statin uptitration group complained of muscle-related adverse effects compared with 6 of 58 (10%) patients in the combination group (Table 4). No serious cardiovascular events were observed during the study.
Adverse effects (chronic kidney disease stages 1 and 2) with duplication.

Adverse effects (chronic kidney disease stages 3, 4 and 5) with duplication.

p < 0.01
Biochemical safety
None of the patients in either group developed CK levels five times the ULN. There was no significant excess risk for abnormal hepatic transaminase levels (ALT) >2 times the ULN in CKD stages 1 and 2 (Table 3). However, in patients with CKD stages 3–5, 2 patients (3%) in the combination group versus 6 patients (12%) in the statin uptitration group had liver-related toxicity (Table 4). In patients with CKD stages 3–5, the two groups did not differ with regard to not having an excess incidence of deteriorating renal function (increase in serum creatinine >20% compared with the level at randomization; 4 of 58, 7%; 3 of 52, 5%). Also, there was no increased requirement for renal replacement therapy (data not shown). None of the patients with CKD stages 1 and 2 in either group had decreases in renal function.
The treatment modalities did not have any significant effects on urinary excretion of albumin (Tables 5 and 6) and blood pressure levels (data not shown) in patients with CKD stages 1 and 2 and patients with CKD stages 3–5.
Changes in eGFR and albuminuria in patients with chronic kidney disease stages 1 and 2.

eGFR; estimated glomerular filtration rate
Changes in eGFR and albuminuria in patients with chronic kidney disease stages 3, 4 and 5.

eGFR; estimated glomerular filtration rate
Discussion
Although patients with CKD frequently have multiple abnormalities in their lipid profile, LDL cholesterol reduction is the primary goal of therapy. The National Kidney Foundation (NKF) in the United States recommends LDL cholesterol <100 mg/dl for patients with CKD. Currently the NKF does not recommend a more aggressive LDL goal for patients with CKD and systemic atherosclerotic disease [Corsini and Holdaas, 2005]. In the present study, there were no differences between groups in the reduction of LDL cholesterol in patients with CKD, indicating that both treatments would be effective with respect to aspects of reduction of LDL cholesterol levels.
Previously, Mikhailidis and colleagues compared the efficacy of the addition of ezetimibe to statin versus doubling the statin dose using six randomized controlled trials and found that the reduction in LDL cholesterol levels attributed to the addition of ezetimibe during the first treatment period was greater than the reduction in LDL cholesterol levels with doubling the dose of statin in monotherapy during the second treatment period and that the difference between treatment arms was significant [Mikhailidis et al. 2011]. The difference between their study and ours might be due to several factors, including differences in ethnicity and populations.
While the therapeutic benefits from statins are related to the achievement of LDL reduction [Davidson et al. 1997], myopathy seems to be more related to the statin dose than to the degree of LDL reduction [Brewer, 2003]. For example, the incidence of myopathy with increasing simvastatin doses was 0.02%, 0.08% and 0.53% for 20, 40 and 80 mg, respectively [Joy and Hegele, 2009].
In the present study, among patients with CKD stages 1 and 2, adverse effects were reported in 3.4% of patients in the combination group and in 4.4% in the statin uptitration group. However, among patients with CKD stages 3–5, 10% in the combination group and 38.4% in the statin uptitration group reported adverse effects, indicating that adding ezetimibe might be safer for reduction of LDL cholesterol in patients with advanced stages of CKD.
The Prediction of Muscular Risk in Observational Conditions (PRIMO) study is the study that shed the most light on the actual incidence of myalgia in the general population [Bruckert et al. 2005]. High-dose statins, regardless of the particular statin, were included in our study. In this report, 10–12% of patients who received statin therapy developed muscle-related adverse effects. Myalgia is one of the most frequent causes of discontinuation of statin therapy [Sandhu et al. 2006]. Overall, muscle symptoms were reported by 10.5% of the cohort with a median time of onset of 1 month. However, 15% of those reporting muscle symptoms had symptoms that appeared 6 months after treatment initiation. In contrast, in the SHARP trial [Baigent et al. 2011], there were no differences in muscle pain between patients who took simvastatin and who took a placebo, although Armitage reported that severe renal insufficiency was an established risk factor for statin myopathy [Armitage, 2007]. There are many potential explanations for the lower rate of statin myopathy in clinical trials compared with observational studies. Clinical trials are more likely to exclude persons at high risk of developing statin myopathy, such as those with a previous myopathy or who were taking other drugs known to interact with statins. This difference also might be due to the lack of a consensus on the definition of statin myopathy at present.
People with this degree of renal impairment are at substantially increased risk of CVD and the limited trial data suggest the benefit of statins in these subgroups although such patients may be at increased risk of myopathy. In addition, the exact mechanisms of statin-induced myopathy remain unknown; if CKD is one of the major risk factors for development of statin-induced myopathy, further research would be needed to clarify its mechanism.
The proposed renal-protective mechanism is based on in vitro observations that statins impeded the normal reabsorption of albumin in the proximal tubule. In a recent meta-analysis, 22 placebo-controlled trials were identified that studied the renal benefit of statins. Statin reduced the rate of decline in GFR by 1.23 ml/min/1.73 m2/year compared with a placebo. This review, however, was limited by significant between-trial heterogeneity [Sandhu et al. 2006]. The proposed renal-protective mechanism is based on in vitro observations that statins impede the normal reabsorption of albumin in the proximal tubule. Mevalonate, a metabolite in the cholesterol synthetic pathway, is reduced in patients on statins and is necessary for the normal reabsorption of albumin in the proximal renal tubule [Agarwal, 2006].
However, in the present study, we did not compare the renoprotective effects between those who did and did not take statins. It is therefore uncertain from the present study whether statin therapy in patients with CKD might be effective for prevention of progression of renal dysfunction as well as reduction of urinary excretion of albumin.
Limitations
First, the absence of a validated questionnaire to assess statin-related muscle symptoms is another limitation for accurate definition of statin-related muscle effects and is an area in need of research. In the present study, low doses of statins, regardless of the statin, were employed as an initial therapy to reduce adverse events.
Although intensive-dose statin therapy was associated with a reduced risk of important cardiovascular events, it was also associated with an increased risk of statin-induced adverse events. Therefore, moderate-dose statin therapy may be the most appropriate choice for achieving cardiovascular risk reduction in the majority of individuals [Silva et al. 2007]. Second, the duration of the present study was too short to reveal the outcome of CVD; however, at present, adverse events were not reported systematically and many trial protocols did not specify standard methods to ascertain harm. In this regard, this study will shed light on the harm of the use of larger doses of statin in patients with advanced CKD. Third, it is well known that most patients with CKD have TG as well as HDL abnormalities along with elevated LDL (mixed dyslipidemia). Therefore, it remains uncertain if this therapy will lead to a reduction of CVD in patients with CKD.
Lastly, in this study, the levels of HDL were not taken into consideration at the time of randomization, which probably caused a significant difference in the levels of HDL between the two groups at the start of the study. As stated in the above limitations, these kind of abnormalities in lipid profile also might contribute to the findings of this study.
In conclusion, when statin uptitration produces adverse effects such as myopathy, combination therapy with ezetimibe is recommended instead of a statin alone.
Footnotes
Appendix: Participants
Akimoto Clinic: H. Akimoto; Soka Western Clinic: K. Iijima and S. Maeda, Mizuno Medical Clinic: K. Mizuno; Higashimatsuyama Medical Clinic: H. Nemoto; Noguchi Clinic: T. Noguchi; Kawamoto Medical Clinic: Y. Numajiri; Saito Clinic: H. Saito; Sawachika Clinic: M. Sawachika; Kouenbashi Clinic: J. Shoda; South Koshigaya Health Clinic: H. Shuto; K. Wakabayasi Clinic: K. Wakabashi; Ren Family Clinic; M. Yokoyama.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors have no conflicts of interest to declare.
