Abstract
Objectives:
This retrospective analysis examined the efficacy and tolerability of nebivolol, a ß1-selective, vasodilatory β-blocker, in four different age groups of patients with hypertension.
Methods:
Data were pooled from three 12-week, randomized, placebo-controlled trials (placebo, n = 205; nebivolol [1.25–30/40 mg/day], n = 1811) and stratified into age quartiles (Group 1: 22–46 years; Group 2: 47–53 years; Group 3: 54–62 years; Group 4: 63–84 years). Only patients treated with placebo and the three commonly used nebivolol dosages (5, 10, and 20 mg/day) are presented. Baseline-to-endpoint changes in trough sitting diastolic blood pressure (DBP), systolic blood pressure (SBP), and heart rate (HR) were analyzed for each age quartile using an analysis of covariance (ANCOVA) model. Tolerability was assessed by means of adverse event (AE) rates.
Results:
The analysis comprised 205 placebo-treated patients and 1380 patients treated with nebivolol dosages of 5, 10, or 20 mg/day. Older age was associated with higher SBP values at baseline. In all age groups, each of the three most frequently used nebivolol dosages significantly reduced DBP, compared with placebo (–9.1 to −11.8 mmHg versus −3.4 to −5.9 mmHg; p ≤ 0.008 overall). For SBP, a statistically significant effect versus placebo was observed for all dosages and age groups except for 5 and 10 mg/day in Group 4. Within each group, treatment with nebivolol (all three dosages) and placebo resulted in similar AE rates (nebivolol: 26.1–36.6%; placebo: 36.2–42.6%) and AE-related discontinuation rates (1.8–3.8% versus 0–4.3%). In each age group, there were no significant nebivolol-placebo differences in the rates of patients who experienced clinically significant changes or abnormal endpoint levels of metabolic parameters.
Conclusions:
This retrospective analysis suggests that nebivolol monotherapy is efficacious and well tolerated across various age groups, with the efficacy in reducing SBP somewhat diminishing in patients over 62 years of age.
Keywords
Introduction
The prevalence of hypertension, a major risk factor for cardiovascular disease [World Health Organization, 2009], is strongly associated with age [Egan et al. 2010; Franklin et al. 1997, 2001]. In the US, hypertension affects approximately 35% (30 million) of those aged between 40 and 59 years and approximately 70% (40 million) of those who are 60 or older [Howden and Meyer, 2011; Egan et al. 2010; Vasan et al. 2002]. In addition to the overall prevalence, age is also associated with the severity and type of hypertension: patients with stage II hypertension comprise 5% of those aged 50–59 years and 32% of those 80 and older [Qureshi et al. 2005]; in individuals over 65, isolated systolic hypertension is the most common form of high blood pressure [Franklin et al. 2001; Kannel, 2000].
The changes in hypertension type, severity, and prevalence across age groups pose notable challenges in clinical practice. For example, older age is also associated with an increased prevalence of comorbidities and polypharmacy, whereas younger age is a key determinant of poor therapy adherence [Briesacher et al. 2008; Fischer et al. 2010]. In addition, antihypertensive medication itself is independently associated with a higher risk of adverse events (AEs) such as erectile dysfunction (ED) [Cordero et al. 2010; Düsing, 2005; Ko et al. 2002; Shiri et al. 2007] and orthostatic hypotension (OH) [Cleophas and van Marum, 2003], which may affect quality of life and lead to treatment discontinuation in both younger and older patients [Fogari and Zoppi, 2004]. Therefore, patient age should be carefully taken into consideration when selecting antihypertensive therapy [Carlberg and Nilsson, 2010].
Nebivolol, a ß1-selective blocker with vasodilatory properties that are thought to be mediated by nitric oxide (NO) [Münzel and Gori, 2009], is approved in the US for the treatment of hypertension. Nebivolol has been associated with a lower incidence of AEs compared with other β1-selective blockers [Ambrosioni and Borghi, 2005], and a number of studies suggest that sexual function in patients with hypertension is not adversely affected by nebivolol therapy [Boydak et al. 2005; Brixius et al. 2007; Cordero et al. 2010; Doumas et al. 2006]. Although available data suggest that treatment with older-generation β-blockers (mainly atenolol) is associated with a reduction of cardiovascular morbidity and mortality in younger (<60 years of age) but not older (≥60 years) patients with uncomplicated hypertension [Khan and McAlister, 2006; Messerli et al. 1998], there are no randomized, prospective, placebo-controlled trials that examined the effects of nebivolol treatment, short or long term, based on patients’ age. This post hoc analysis evaluated the efficacy and safety of nebivolol monotherapy in a pooled sample from three US registration trials [Greathouse, 2010; Saunders et al. 2007; Weiss et al. 2007], stratified by age quartiles.
Methods
Data collection
All data reported in this publication were pooled from three similarly designed phase III, randomized, double-blind, placebo-controlled, multicenter, parallel group trials: NEB-MD-202 [ClinicalTrials.gov identifier: NCT00145236; Saunders et al. 2007], NEB-MD-302 [Clinical Trials.gov identifier: NCT00200460; Weiss et al. 2007], and NEB-MD-305 [ClinicalTrials.gov identifier: NCT00200473; Greathouse, 2010]. Trials NEB-MD-202 and NEB-MD-302 were conducted in the US; trial NEB-MD-305 was conducted in the US, UK, Belgium, and the Netherlands.
Participants and trial design
The trial details have been published previously [Greathouse, 2010; Saunders et al. 2007; Weiss et al. 2007]. Participants were adult men and women of any race (except for study NEB-MD-202, which recruited black patients only) with a diagnosis of stage I–II hypertension (defined as an average untreated sitting diastolic blood pressure [SiDBP] ≥95 and ≤109 mmHg) at randomization (Day 0). Patients were not permitted to participate if they had a body mass index (BMI) ≥35 kg/m2 (NEB-MD-202 excluded patients with BMI ≥40 kg/m2), secondary or malignant hypertension, history or presence of respiratory problems (asthma, bronchospasm, or chronic obstructive pulmonary disease [COPD]), abnormalities in cardiac rate or rhythm, uncontrolled diabetes (hemoglobin A1C ≥10%), a cerebrovascular accident or myocardial infarction within 6 months of screening, heart failure requiring treatment, concomitant therapy with medications that may have affected blood pressure, or hypersensitivity to ß-blockers, or if they were pregnant or nursing.
In all three trials, a single-blind placebo run-in phase (2–6 weeks) was followed by a randomized, double-blind, fixed-dose treatment phase in which patients were randomized to receive placebo or nebivolol (NEB-MD-202: 2.5, 5, 10, 20, and 40 mg/day; NEB-MD-302: 1.25, 2.5, 5, 10, 20, and 30/40 mg/day; NEB-MD-305: 5, 10, and 20 mg/day) daily for 12 weeks. In NEB-MD-302, the patients randomized to receive the highest nebivolol dosage (30/40 mg/day) were given 30 mg/day for 2 weeks, at which point the dose was up-titrated to 40 mg/day if their sitting HR remained above 55 bpm. All measurements were performed in triplicate at trough (24 ± 2 hours postdose) and at peak (2–3 hours postdose). All studies were performed in accordance with the Declaration of Helsinki.
Assessments
Efficacy and tolerability data presented in this analysis are for the dosages of 5, 10, and 20 mg/day, which correspond to the doses most commonly used in clinical practice. The efficacy outcomes were changes from baseline in trough sitting diastolic blood pressure (DBP) and systolic blood pressure (SBP), which correspond to primary and secondary measures, respectively, in all three pivotal trials. The following safety and tolerability parameters are reported: AE rates, mean changes from baseline in heart rate (HR) and the levels of metabolic parameters (glucose, triglycerides, high-density lipoprotein [HDL], low-density lipoprotein [LDL], and total cholesterol), and the percentages of patients who had normal glucose levels (≤125 mg/dl) at baseline but high levels (>125 mg/dl) at the end of the trial. (Owing to low incidence, rates for ED and OH were examined for the entire dosage range: 1.25–30/40 mg/day.)
Data analysis
All data are presented for each age quartile (Group 1: 22–46 years; Group 2: 47–53 years; Group 3: 54–62 years; Group 4: 63–84 years). BP and HR analyses were based on the intent-to-treat (ITT) population and performed using an analysis of covariance (ANCOVA) model with treatment group as factor and baseline values, extensive- or poor-metabolizer status, diabetes status, race, and sex as covariates; missing data were imputed using the last observation carried forward approach (LOCF) approach. The changes from baseline in metabolic parameter levels (both absolute [mg/dl] and relative [%]) were also based on the ITT population (LOCF) and analyzed by means of an ANCOVA model with treatment group as factor and baseline parameter value as a covariate. The proportions of patients who attained high glucose levels were compared by means of the chi-squared test. No adjustments for multiple hypothesis testing were performed. Owing to the post hoc nature of this study, all references to statistical significance are nominal and for exploratory purposes only. AE rates were analyzed using descriptive statistics.
Results
Baseline characteristics and patient disposition
In all three trials, a total of 2016 patients were randomized to receive placebo (n = 205) or nebivolol (n = 1811). Dispositions of patients in each age quartile treated with placebo or nebivolol dosages of 5, 10, or 20 mg/day (n = 1585; 78.6% of total) are summarized in Figure 1. For the individuals treated with 5–20 mg/day nebivolol, the most common reasons for discontinuation by age quartile were loss to follow up (Group 1), consent withdrawal (Groups 2 and 4), and AEs (Group 3) (Figure 1). Discontinuation due to an AE (Figure 1) occurred in 1.8% of patients from Group 1, 1.6% of patients from Group 2, 3.2% of patients from Group 3, and 4.2% of patients from Group 4 (Figure 1).

Study flow, by age (placebo and selected nebivolol dosages only).
Baseline demographic and clinical characteristics for each age quartile are presented in Table 1. Baseline SBP/DBP values were similar within groups for placebo and 5, 10, and 20 mg/day dosages; of note, baseline SBP values appeared to increase with age (Table 1).
Baseline demographic and clinical characteristics (ITT population).

Abbreviations: BMI, body-mass index; DBP, diastolic blood pressure; ITT, intent-to-treat; SBP, systolic blood pressure; SD, standard deviation.
Blood pressure and heart rate
At week 12, all nebivolol dosages of 5–20 mg/day were significantly more effective than placebo in reducing DBP in all age groups (Figure 2A); all three dosages were also significantly more effective than placebo in reducing SBP in Groups 1, 2, and 3 (Figure 2B). In Group 4, the 20 mg/day dosage was significantly more effective than placebo in reducing SBP (Figure 2B).

Effect of nebivolol treatment (5–20 mg/day) on blood pressure, by age group (intent-to-treat population). Comparisons made using an analysis of covariance model with treatment as factor and baseline blood pressure, metabolizer status, diabetes status, sex, and race as covariates. *p < 0.05, **p < 0.01, ***p < 0.001, versus placebo Abbreviations: DBP, diastolic blood pressure; SBP, systolic blood pressure; SE, standard error of the mean.
In all age groups, nebivolol was associated with significant HR reductions (Figure 3).

Effect of nebivolol treatment (5–20 mg/day) on heart rate, by age group (intent-to-treat population). Comparisons made using an analysis of covariance model with treatment as factor and baseline blood pressure, metabolizer status, diabetes status, sex, and race as covariates. *p < 0.05, **p < 0.01, ***p < 0.001, versus placebo.
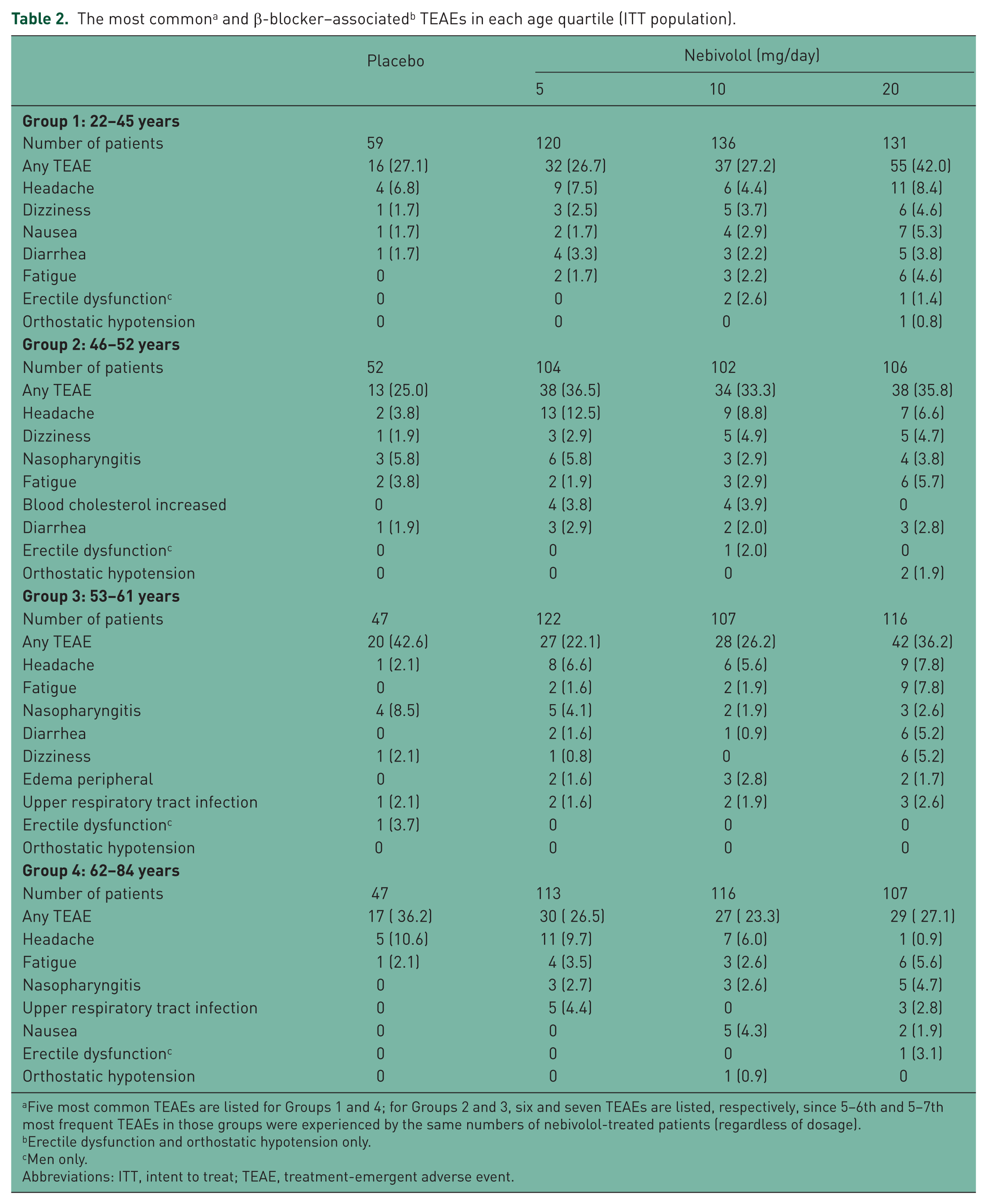
Most frequent treatment-emergent adverse events
The most common treatment-emergent AEs (TEAEs) per age quartile, by dose, are summarized in Table 2. In all groups, the most common TEAE was headache. Dizziness, the second most frequent AE in the younger half of the population, appeared to lose its prominence among older participants. Nausea, the third most common AE in the youngest group, appeared to follow a similar trend, whereas the trend seemed to be the opposite for fatigue, which was more prominent among older patients.
Five most common TEAEs are listed for Groups 1 and 4; for Groups 2 and 3, six and seven TEAEs are listed, respectively, since 5–6th and 5–7th most frequent TEAEs in those groups were experienced by the same numbers of nebivolol-treated patients (regardless of dosage).
Erectile dysfunction and orthostatic hypotension only.
Men only.
Abbreviations: ITT, intent to treat; TEAE, treatment-emergent adverse event.
Incidence of AEs associated with β-blockers
The observed TEAE rates (Table 2), and the fact that no patients discontinued treatment due to ED, indicate that ED was not associated with nebivolol therapy in any age group, No placebo-treated patient experienced OH, and the occurrence of OH among nebivolol-treated patients was low (a total of three patients in Groups 1 and 2, all treated with 20 mg/day, and one patient in Group 4 who was taking the 10 mg/day dosage; see Table 2). Two nebivolol-treated patients discontinued the trial due to OH: the patient in Group 4 and one of the patients in Group 2.
Metabolic parameters
Baseline levels and baseline-to-endpoint changes in metabolic parameters are summarized in Table 3. In Group 1, the only statistically significant metabolic effect of nebivolol (versus placebo) was a decrease in absolute (but not relative) levels of HDL cholesterol in patients randomized to the dosage of 10 mg/day (Table 3). In Group 2, treatment with 5 and 20 mg/day nebivolol appeared to be associated with a decrease in HDL cholesterol levels (both absolute and relative) and also a decrease (absolute and relative) in the levels of LDL and total cholesterol in patients treated with the 5/mg day dosage (Table 3). In Group 3, all three nebivolol dosages were associated with a decrease in HDL cholesterol levels and an increase in the level of triglycerides (Table 3). In Group 4, all metabolic parameters examined appeared to be influenced by at least one nebivolol dosage, ranging from a decrease in the levels of glucose and cholesterol (HDL, LDL, and total) to an increase in the levels of total cholesterol and triglycerides (Table 3). Finally, there were no statistically significant differences in the percentage of patients who had normal blood glucose at baseline but high levels (>125 mg/dl) at endpoint (Group 1: 3.6% [nebivolol, 5–20 mg/day] versus 7.0% [placebo], p = 0.233; Group 2: 4.4% versus 0%, p = 0.134; Group 3: 4.8% versus 2.2%, p = 0.427; Group 4: 3.7% versus 6.7%, p = 0.353).
Baseline-to-endpoint changes in glucose and lipid levels (ITT population).

Mean ± standard deviation.
Mean ± standard error of the mean.
Discussion
Study summary
Results of this post hoc analysis suggest that nebivolol may be effective in treating patients with stage I–II hypertension across various age groups, with the effect on SBP becoming less prominent with age. For all age groups, SBP and DBP were significantly reduced (versus placebo) by the three nebivolol doses most frequently used in clinical practice (5, 10, and 20 mg/day), with the exception of SBP in the oldest quartile, in which nebivolol was significantly more efficacious than placebo for the dosage of 20 mg/day only. Treatment with nebivolol was safe and well tolerated, with an AE profile that was similar across dosages and age groups and variations in glucose and lipid levels that were not clinically significant. In addition, nebivolol treatment was associated with low incidences of ED and OH, regardless of age.
Clinical relevance
Our data suggest that nebivolol is similarly effective across the entire adult age range in reducing DBP (Figure 2a), but also that its ability to reduce SBP diminishes with advancing age (Figure 2b). That observation, and the fact that the mean baseline SBP, but not DBP, was higher in the oldest quartile compared with other age groups (e.g. Group 4, 158 mmHg; Group 1, 146 mmHg; Table 1), are consistent with previous findings. For example, a post hoc analysis of the data from The Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT; N = 33,357) demonstrated that higher baseline SBP and older age are associated with a lower likelihood of achieving BP control [Cushman et al. 2002]. The BP results of our analysis are also partly consistent with the outcomes of a 12-week open-label postmarketing surveillance study of patients with hypertension and diabetes, conducted in Germany (N = 5071) [Ladage et al. 2010]. In that study, nebivolol (5 mg/day, administered as monotherapy [46%] or add-on therapy [54%]) was associated with significant reductions in both SBP and DBP across a range of age groups (<40 years, 40–49, 50–59, 60–69, 70–79, and >80 years), with the effect on both, as opposed to SBP only as in our analysis, diminishing with age [Ladage et al. 2010]. The mechanism for attenuation of the SBP effect of nebivolol in the elderly is most likely based on the fact that hypertension, and particularly the SBP component, in advanced age is driven by endothelial dysfunction and increased arterial stiffness [Safar, 2010], which are also the central physiological features affected by nebivolol’s proposed mechanism of action [Münzel and Gori, 2009]. In a situation where arterial stiffness is particularly high, higher doses of nebivolol would likely be required to produce a significant reduction in SBP. On the other hand, low levels of NO [Lyamina et al. 2003] and high levels of asymmetric dimethyl arginine, an inhibitor of NO synthase [Paiva et al. 2008], have been implicated in the pathogenesis of hypertension in younger individuals, which suggests that nebivolol, with its NO-mediated vasodilatory properties [McEniery et al. 2004], would be a well-suited antihypertensive therapy for patients <60 years of age. Prospectively designed randomized studies would be required to assess the physiological differences of response to nebivolol treatment between younger and older patients.
With regards to tolerability, the AE profile observed in our analysis suggests two possible trends. As mentioned in the Results section, there appears to be a shift in the AE profile with patients’ age (e.g. dizziness and fatigue appear to lose and gain prominence with age, respectively). This possible trend appears biologically plausible, as physiological response to medication is expected to show differences between younger and older people. The data also seem to suggest that age may affect dose dependence of certain AEs. For example, in the youngest quartile (Group 1), the rates of most AEs among patients randomized to 20 mg/day nebivolol were consistently higher than those observed among patients receiving dosages of 5 or 10 mg/day; a similar pattern emerged in Group 3, but it was largely absent from Group 2 and Group 4 (Table 2). However, this ‘intermittent’ effect of age on dose dependence does not seem biologically plausible and is likely an artifact of subdividing the pooled sample. A recent analysis of the entire pooled sample of these three trials suggested an overall dose dependence for fatigue, dizziness, and nausea, but not for other TEAEs [Weiss et al. 2011]. The aforementioned German study did not actively monitor AEs, other than stating the absence of ‘critical findings’, based on patient complaints [Ladage et al. 2010], and to best of the authors’ knowledge, there is no systematic study or review that evaluated β-blocker–associated AEs based on patients’ age. However, a recent cross-sectional observational study of hypertensive men treated with any β-blocker for at least 6 months (N = 1007; mean ± SD age: 57.9 ± 10.9 years) demonstrated that, compared with patients treated with atenolol, bisoprolol, metoprolol, or carvedilol, nebivolol-treated individuals had the lowest rates of ED and the highest scores on all subscales of the International Index of Erectile Dysfunction (encompassing domains of erectile function, orgasmic function, sexual desire, satisfaction with intercourses, and global satisfaction) [Cordero et al. 2010]. The results of our study are in agreement with these findings, as ED was experienced by only one placebo-treated man and five nebivolol-treated men.
In the three trials that were pooled in our analysis [Greathouse, 2010; Saunders et al. 2007; Weiss et al. 2007], the observed rates of ED (0.5% of men) and OH (0.2%) were relatively low. (OH is a phenomenon that has been estimated to occur in 5–33% of the elderly and may be caused by various antihypertensive medications, including β-blockers [Verhaeverbeke and Mets, 1997].) Such results are in agreement with previously published data indicating that nebivolol treatment is associated with lower AE rates compared with other β1-selective blockers [Ambrosioni and Borghi, 2005], other β-blockers in general, and other antihypertensive medications [Van Bortel et al. 2008], that it does not adversely affect sexual functioning [Boydak et al. 2005; Brixius et al. 2007; Doumas et al. 2006], and that it actually may result in positive pressor effects (i.e. reduced chances of OH) in elderly patients with mild hypertension [Cleophas et al. 2002].
In our data set, there were no statistically significant nebivolol-placebo differences in the rates of clinically significant metabolic changes, abnormal levels of metabolic parameters, or the rates of patients who attained blood glucose levels ≥125 mg/dl. However, we did observe a pattern of statistically significant changes in metabolic parameter levels in each age quartile, corresponding to variations from baseline of <5% for glucose and cholesterol, and <30% for triglycerides (Table 3). If one focuses on relative changes only (which facilitates comparisons and may be more relevant from the clinical standpoint), the vast majority of significant changes occurred in the three oldest quartiles (Table 3). The most consistent significant change was a decrease in HDL cholesterol levels by 3–4% in patients treated with nebivolol (Groups 2, 3, and 4), compared with 2–3% increase in patients treated with placebo (Table 3). The second most consistent change was an increase in triglyceride levels of 16–29% in nebivolol-treated patients (significance observed in Groups 3 and 4), compared with changes in the range from −5% to 1% among placebo-treated patients (Table 3). The remaining significant relative changes were observed in the oldest quartile (LDL and total cholesterol), and include variations from −0.3% to 1.9%, relative to baseline, among nebivolol-treated patients. The HDL cholesterol and triglyceride changes appear consistent and do suggest a real phenomenon. This effect is also mentioned in the prescribing information for nebivolol, which states that ‘in controlled monotherapy trials’, the drug was associated with a decrease in HDL cholesterol [Forest Laboratories, 2011]. The statistically significant increases in LDL and total cholesterol, observed in the oldest quartile, are even smaller in magnitude and less consistent than the HDL cholesterol changes; therefore, it seems reasonable to speculate that their impact on cardiovascular risk would be minimal. Of note, the German study reported statistically significant improvements on a number of metabolic parameters (HbA1c, triglycerides, HDL cholesterol, LDL cholesterol), and across all age groups examined [Ladage et al. 2010], which is consistent with the results of several previous trials that demonstrated neutral or beneficial metabolic effects of nebivolol [Fonseca, 2010]. A prospective, appropriately designed trial would be needed to investigate the metabolic effects of nebivolol treatment in regard to age.
Limitations
This was a post hoc analysis; therefore, all p values and references to statistical significance are nominal and for exploratory purposes only. In addition, in all three trials, patients were included based on their DBP values [Greathouse, 2010; Saunders et al. 2007; Weiss et al. 2007]. Such inclusion criteria would not be appropriate for a trial in which participants’ age corresponded to that in our oldest quartile, since at that age isolated systolic hypertension would be expected to be dominant [Franklin et al. 1997, 2001; Kannel, 2000]. Furthermore, in a sample closer to real-life situation, the percentages of patients with stage II hypertension in Groups 1–3 would be expected to be lower than those observed in our data set [Qureshi et al. 2005], and would be expected to steadily increase with age [Qureshi et al. 2005], which also appears not to be the case with our data (Table 1: Group 1, 45.5%; Group 2, 52.6%; Group 3, 55.1%; Group 4, 55.4%). Finally, the primary studies included in this analysis were not powered to detect efficacy and safety differences by age, particularly those related to ED and OH.
Conclusions
These data suggest that nebivolol is an efficacious and well-tolerated antihypertensive monotherapy across various age ranges; however, the efficacy in reducing SBP in the elderly appears to require higher nebivolol dosages (i.e. 20 mg/day), and the drug may be associated with a small decrease in HDL cholesterol levels in patients >45 years of age. The rates of ED and OH seen in our pooled analysis suggest that nebivolol may represent a well-tolerated treatment in patients who are at a particular risk for these AEs. Longer-term, active-comparator trials would be useful to further examine the potential benefit of this therapy in selected populations, relative to other commonly used antihypertensive medications.
Footnotes
Funding
This work was sponsored by Forest Research Institute, a subsidiary of Forest Laboratories, Inc., the US marketer of nebivolol.
Conflict of interest statement
F. Wilford Germino is a member of the speakers’ bureaus at Bristol-Myers Squibb Company, Pfizer Pharmaceuticals, Takeda, and Forest Laboratories, and has received grant support from Novartis Pharmaceutical Corporation, Forest Research Institute, and Daiichi-Sankyo. Yuhua Lin is an employee of Forest Research Institute; at the time of writing and submission of the manuscript, Lynn Bowen was also a Forest Research Institute employee. Vojislav Pejović is an employee of Prescott Medical Communications Group, a contractor of Forest Research Institute.