Abstract
Background
Pruritus is a common symptom of intrahepatic cholestasis of pregnancy (ICP) and significantly reduces patients’ quality of life (QoL).
Methods
A multinational survey administered through ICP patient support groups was conducted in women with current or previous ICP.
Results
In total, 697 women from the UK, USA, Australia, Canada, New Zealand, and Ireland responded to the survey. 94% had ICP in a previous pregnancy. The median worst itch score (0–10) was 9. Pruritus severity was associated with a higher degree of sleep disturbance, fatigue, and mood changes. 59% reported that pruritus led to disruption of daily activities. 33% reported missing school or work. Most patients were taking ≥2 antipruritic medications and achieved only partial resolution of pruritus.
Conclusion
The results underscore the negative impact of pruritus on QoL in patients with ICP and substantiate the high unmet need for developing safe and effective antipruritic therapies in ICP.
Trial Registration
Trial registration is not applicable because this was not an interventional study.
Introduction
Intrahepatic cholestasis of pregnancy (ICP) 1 is the commonest gestational liver disorder, usually developing during the third trimester in women with pruritus and serum bile acid (sBA) concentrations >19 µmol/L. 2 The incidence of ICP ranges from 0.3% to 5.6% in various populations; in Europe and North America, ICP is encountered in 1–2% of pregnant women.2–4 It is most common in women with assisted reproduction and multifetal pregnancies.5,6 The etiology of ICP is not fully known, but increased levels of gestational hormones, genetics, and environmental influences are important contributing factors. 2 ICP is associated with adverse pregnancy outcomes, including spontaneous preterm birth, prolonged admission to the neonatal intensive care unit, and stillbirth. The extent of the elevation of maternal sBA is associated with the risk of these complications; specifically, sBA levels ≥40 µmol/L are associated with greater risk of preterm birth, and when sBA ≥ 100 µmol/L, there is a higher risk of stillbirth. 7
High sBA concentrations, which are typically observed in patients with ICP, are associated with the development of severe pruritus. 8 Although the precise pruritogen is not known, likely candidates in ICP include lysophosphatidic acid and progesterone sulfates.9,10 In a prospective Swedish cohort study to determine the relationship between sBA and fetal complications, the incidence of gestational pruritus was estimated at 2.1% (N > 45,485), and ICP diagnosis was confirmed in 1.5% of the study participants. 11
Pruritus in patients with ICP is a debilitating and disease-defining symptom that typically occurs in the second or third trimester of pregnancy. 2 Pruritus is more intense at night and may affect all parts of the body, but it is most common on the surfaces of the palms and the soles and is not associated with any specific skin manifestations or lesions.2,8 The impact of pruritus on the daily lives of patients with ICP can be significant and highly disruptive. Patients with ICP with pruritus experience insomnia, irritability, and mood changes, including depression and suicidal thoughts, which have a significant negative impact on patients’ quality of life (QoL).2,5
ICP is a reversible condition that spontaneously resolves within days or weeks following delivery; however, if pruritus and liver test abnormalities persist, other chronic liver disease conditions must be considered. 8 ICP has been linked to the development later in life of gallbladder diseases, particularly gallstones. Importantly, the ABCB4 gene, which is associated with ICP, has also been shown to increase the risk of gallstones.12,13 The chance of ICP recurring during a subsequent pregnancy is 60–90%.2,14 ICP can lead to iatrogenic delivery; alongside ICP-associated spontaneous preterm birth, this can be associated with significant perinatal morbidity.2,7 Patients with ICP continue to experience severe and debilitating pruritus until the resolution of the ICP or, potentially, other liver conditions. Other rarer symptoms, including abdominal pain, nausea, vomiting, fatty stool, and mild jaundice, have also been reported. 2
The main goal of pharmacotherapy in ICP is to reduce maternal symptoms (e.g., severe pruritus) and minimize the risk of fetal compromise, preterm birth, and stillbirth. 8 The primary standard of therapy for ICP is ursodeoxycholic acid (UDCA). However, there are limited data regarding the efficacy of UDCA for pruritus. 15 Other pharmacological treatments that are added or used as second-line therapy include S-adenosyl-L-methionine, bile acid sequestrants (e.g., cholestyramine), rifampicin, bezafibrate, naltrexone, phenobarbital, and selective serotonin reuptake inhibitors (e.g., sertraline).8,16 Topical creams and emollients are used with limited effectiveness. 13 Antihistamines are also used in patients with ICP to help with disturbed sleep, although they do not adequately address pruritus on their own.8,16 However, there are no approved medications for the effective management of pruritus related to ICP.
Considering the significance of pruritus in ICP as a disease-defining symptom and its adverse impact on patients’ QoL, as well as inadequate treatment options, we conducted a survey of patients with ICP to learn about the presentation and severity of pruritus in ICP and understand its broader impact on activities of daily living, emotional state, and overall QoL from a patient's perspective.
Methods
Survey questionnaire
A multinational survey used the patient advocacy website platform ICP Support (UK) along with reach out from institutions, including Imperial College London (UK), King's College London (UK), the University of Texas Medical Branch (USA), and Harvard University (USA). The web-based anonymous survey was posted on each organization's website from August 25 to September 10, 2020.
A complete list of questions is provided in Supplementary Section 1, and additional methods are provided in Supplementary Section 2.
Data analysis
The pruritus severity score was quantified by a numeric rating scale (NRS) ranging from 0 (no itch) to 10 (worst itch you can possibly imagine). The sleep disruption severity score was also measured using an NRS (0, no disruption; 10, no sleep).
All data were analyzed and presented using descriptive (e.g., numerical, percentage) statistical methods based on nonmissing data.
Ethical considerations
The patient advocacy group had an internal review team independent from other companies/agencies that reviewed and approved the questionnaire used for the survey. Ethical approval (other than the internal patient advocacy group review team) was not sought for the present study because it was a noninterventional, voluntary, and anonymous survey. The decision to participate in the survey was completely voluntary. Individuals who participated in the survey consented to the sponsor's use and publication of the collected data. The study was conducted in accordance with the Declaration of Helsinki. The study was sponsored by Mirum Pharmaceuticals, Inc.
Results
Participant information
A total of 697 adult females ≥18 years of age responded to this survey. Most participants resided in the UK (56%) and the USA (25%). More than half of the participants (52%) were between 25 and 34 years of age, and 94% had ICP in a previous pregnancy.
The participants’ demographics and medical background information are presented in Table 1.
Participants’ demographics and medical background.
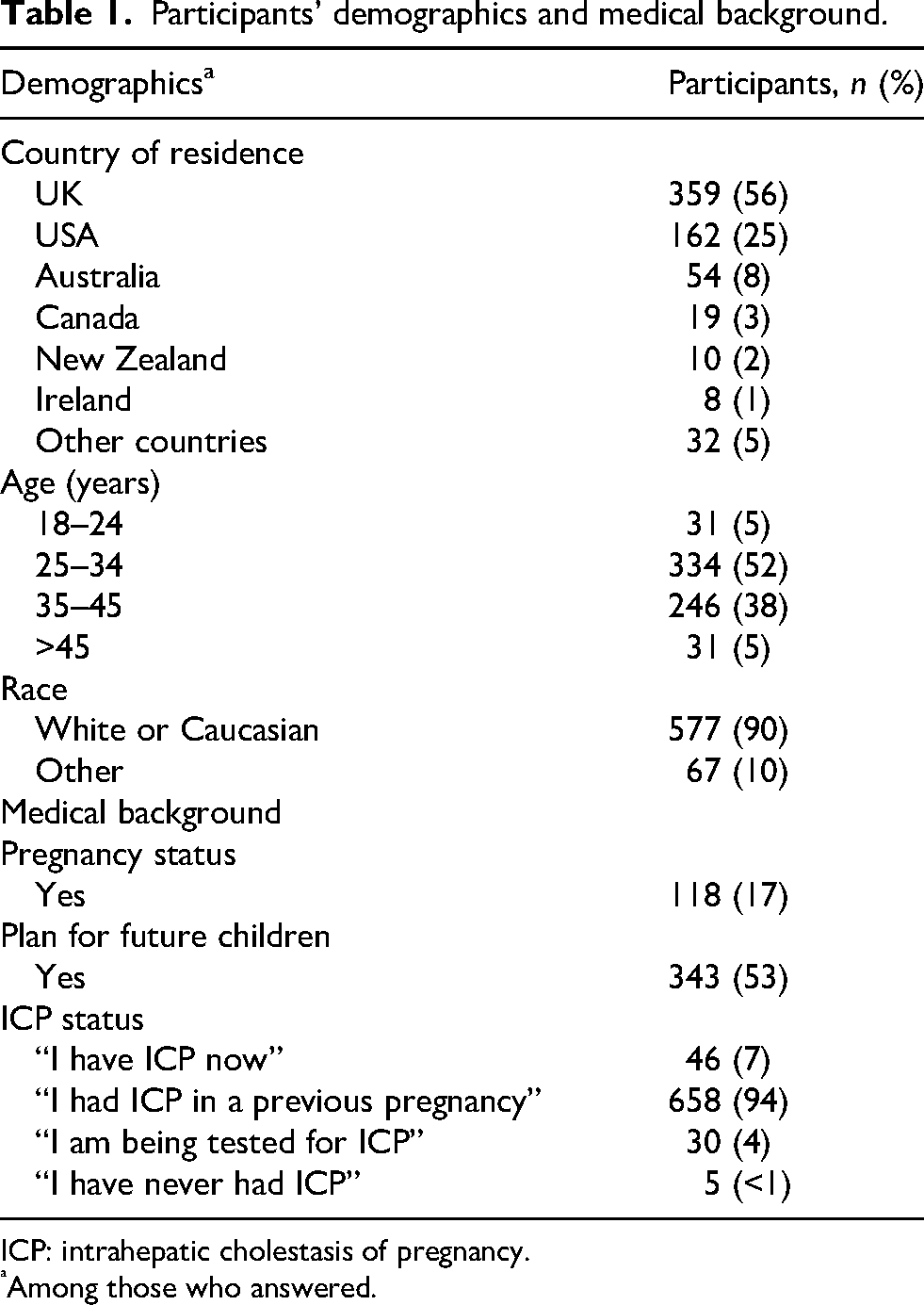
ICP: intrahepatic cholestasis of pregnancy.
Among those who answered.
Pruritus assessment
Ninety percent of participants (N = 688) reported a worst itch NRS score of ≥7, while 42% reported a score of 10 (Figure 1a). The median worst itch reported in the most recent ICP-associated pregnancy was 9. The first reported incidence of pruritus generally occurred from 21 to 34 weeks’ gestation (60%), and about 30% of participants experienced pruritus before 20 weeks’ gestation.
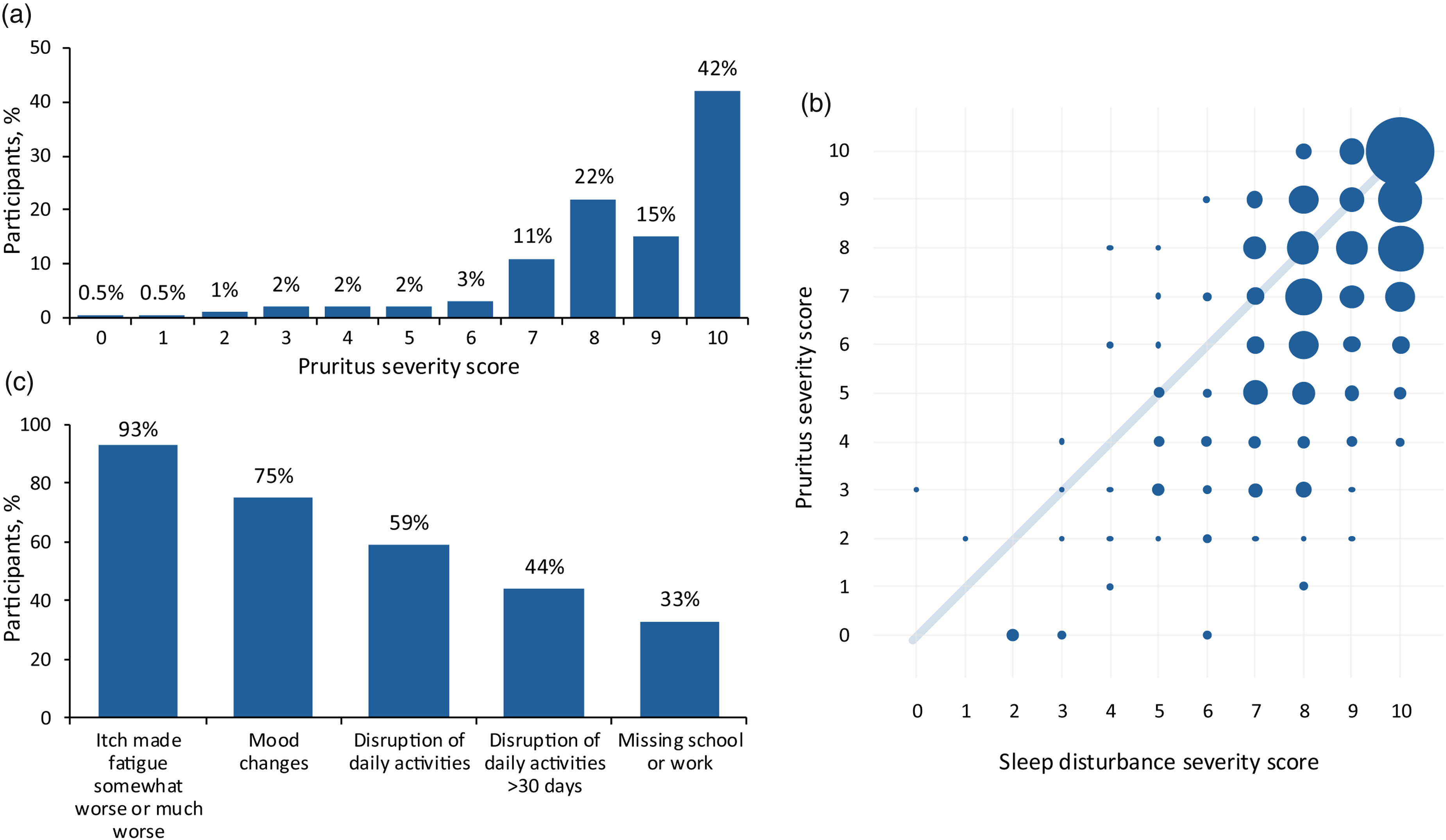
(a) Distribution of worst pruritus severity score. Pruritus scores were measured via a numeric rating scale ranging from 0 (no itch) to 10 (worst itch you can possibly imagine). (b) Impact of pruritus severity on sleep disturbance. Sleep disturbance scores were measured via a numeric rating scale ranging from 0 (no disruption) to 10 (no sleep). In the graph, larger circles represent a larger number of participants experiencing both severe pruritus and significant sleep disturbance. (c) Impact of pruritus on participants’ daily lives.
Participants across geographic regions had no apparent differences in mean pruritus severity (range, 8.1–9.0). The mean severity score for pruritus across participant age groups (range, 8–9.1) and gestational age groups (range, 7.7–9) also did not differ numerically. There was a trend toward higher pruritus severity scores in participants older than 45 years (9.1) compared with those in younger age groups (range, 8.0–8.7) from 12 to 28 weeks’ gestation.
Participants’ responses to the question “What is a meaningful decrease in pruritus severity score?” indicated the average meaningful decrease in score was 4.3 (range, 0.6–4.3), with a trend toward a greater decrease in score in participants reporting higher pruritus severity scores. When asked the above question in the context of a new pharmacotherapy, the average meaningful decrease in pruritus severity score was 3.4 (range, 1.5–3.7), with a trend toward a greater decrease in score in those with higher pruritus severity scores.
Only 29% of participants indicated that their antenatal or prenatal caregiver asked them about itching or mentioned it as a symptom to report. Of these, 89% of caregivers took one or more actions to manage the pruritic condition, including requesting lab/blood tests (94%), recommending antipruritic medication (35%), referring to another physician, such as a dermatologist (5%), or other unspecified actions.
Impact on sleep, fatigue, mood, and daily activities
Seventy-seven percent of participants reported more severe itching at night. Most participants (94%) experienced significant sleep disturbance due to their pruritus. The median severity score for sleep disturbance was 8. The severity of sleep disturbance correlated with the severity of pruritus (Figure 1b).
In participants who experienced fatigue unrelated to their sleep disturbance (n = 319), 30% reported “somewhat worse,” and 63% reported “much worse” fatigue. The majority of participants (75%) reported that pruritus had a negative impact on mood, including but not limited to increased anxiety, feelings of hopelessness, and/or irritability.
Disruption of daily activities and routines was commonly reported by participants (59%). Nearly half of the participants (44%) experienced disruptions lasting more than 30 days. About a third of participants reported missing school or work during the pruritus episodes. A summary of the significant burden of ICP-related pruritus on participants’ daily lives is presented in Figure 1c.
Antipruritic treatment and medications
The most common medications for managing pruritus among survey participants were UDCA, antihistamines, and creams/emollients. Fifty-eight percent of participants took at least two medications, and 29% took at least three. The list of common medications and treatment outcomes is provided in Table 2.
Antipruritic medications used by survey participants (N = 688).
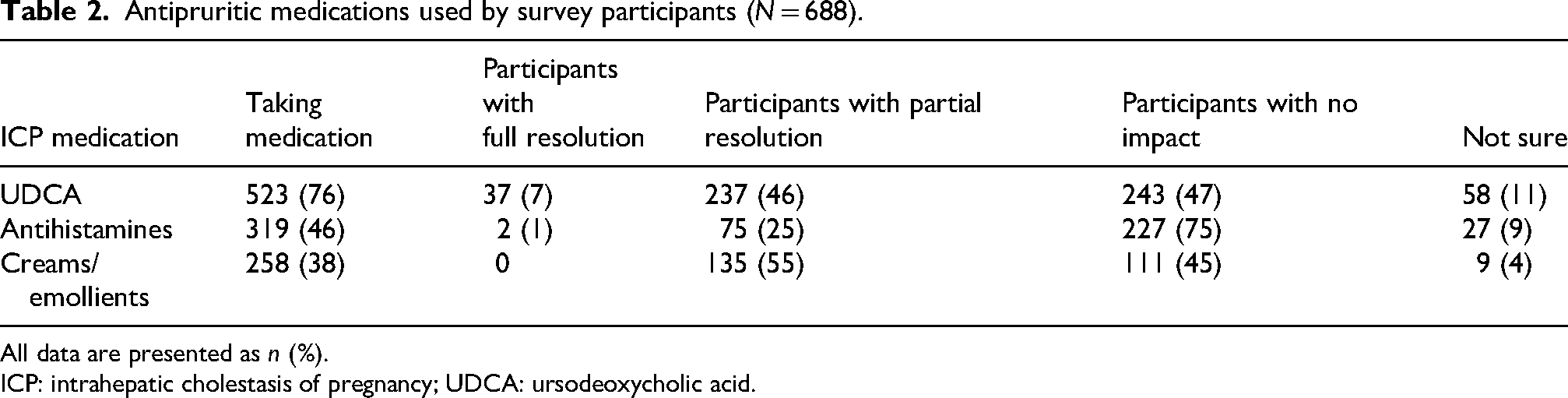
All data are presented as n (%).
ICP: intrahepatic cholestasis of pregnancy; UDCA: ursodeoxycholic acid.
The effectiveness of these medications was limited, and most participants experienced either no relief or partial relief. The use of various antipruritic medications by participants across the geographic regions was generally comparable. No apparent trends were detected when evaluating the effectiveness of medications by participant age or gestational age at the onset of pruritus.
Discussion
The results of this survey showed that most participants had experienced moderate to severe pruritus (NRS = 7–10) related to ICP during their pregnancy. Pruritus had significant adverse effects on participants’ QoL and daily living, including mood changes, sleep disturbances, disruption of daily responsibilities, and missing school or work.
The survey findings were generally consistent with those reported in the literature in patients with ICP.2,5,8 Pruritus was a common symptom of ICP reported by more than 90% of participants. Pruritus occurred more in the evenings and nights in the majority of respondents. Consistent with the literature regarding the gestational week of onset, pruritus was reported mostly during the second and third trimesters of pregnancy. Although ICP-related pruritus is generally self-limited and resolves in a few weeks after pregnancy, the majority of participants indicated that the duration of their itching episode lasted longer than 30 days and, at times, throughout the entire pregnancy, suggesting the variable nature of ICP-related pruritus. It also appeared that older participants had higher pruritus severity scores. The reason for this trend is unknown but may be related to participants’ pregnancy history, their evolving immune state associated with an underlying medical condition (e.g., an autoimmune disorder), and/or the natural aging process. The older participants were more likely to be parous; thus, it is possible that they could have had an earlier onset of ICP symptoms because they were more aware of pruritus, having experienced it in a previous pregnancy.
A limitation of the study is that many respondents were no longer pregnant and were grading their symptoms retrospectively, which may have reduced the accuracy of their responses. While the representation of women of a variety of nationalities is a strength of the study, approximately 90% were White, which is a limitation, as this survey did not provide data on the impact of pruritus on women from other ethnic groups. This is particularly important, as ICP is more common in women of Latin American and South Asian origin. 17
Patient–physician engagement and asking questions are critical to detecting signs and symptoms of ICP and helping to determine the best path forward to reduce the risk of adverse obstetric outcomes for the mother (severe and debilitating pruritus) and potential negative impact on the baby (preterm birth, fetal distress, and stillbirth). The survey results showed that most participants’ prenatal care providers did not proactively ask the participants about pruritus, which could limit the opportunity to provide relief when possible. However, of those physicians who asked about participants’ pruritus, the majority took one or more steps (e.g., ordering labs, referring to specialists) to better manage their patients. This finding is an opportunity to advocate for both patients and physicians to proactively communicate information about all signs and symptoms of ICP, including pruritus; optimize the care of patients with ICP; and raise awareness of the need for better treatments.
Another area of the patient–physician interaction is gaining insight into patients’ expectations related to the outcome of their treatment. In this survey, participants were asked about “meaningful” changes in pruritus severity, which directly impacts patients’ lives and daily activities. Per participants’ responses, a meaningful decrease in pruritus severity score, considering currently available medications (4.3) or future potential therapies (3.4), suggests that physicians should regularly ask their patients about the severity of pruritus. Physicians could then determine if continuing use of a current medication or prescribing a new medication would provide adequate effectiveness, balancing the decision against potential side effects of treatment.
ICP and its related pruritic symptoms have significant adverse effects on patients’ lives, such as experiencing insomnia, irritability, and mood changes, leading to a significant decline in QoL.2,5 Consistent with these reports, participants in this survey reported similar adverse consequences of ICP-related pruritus. The severity of pruritus negatively impacted both energy level and sleep quality. Daily activities, as well as going to work or attending school, were significantly impaired by pruritus. Mood changes (e.g., depression, anxiety) are also very important and could impact many aspects of participants’ lives, such as relationships, work and school attendance, and home care. These changes and disruptions lasted ≥30 days and sometimes up to 9 months, affecting the participants’ daily activities. These findings reinforce the need for developing ICP-specific QoL assessment tools and conducting additional studies to systematically evaluate patients’ physical and mental outcomes and the impact of severe symptoms like pruritus on their lives.
There are no approved medications for the management of pruritus in patients with ICP. In the absence of consistent clinical data, using available therapeutic options like UDCA is generally accepted in clinical practice, primarily based on evidence-based medicine or physicians’ clinical experiences. In this survey, participants used various medications commonly prescribed in medical practice to manage pruritus. Although more than 50% of participants were using two or more medications, the majority of participants were experiencing little or no relief. As previously noted, limited patient–physician engagement, as well as undefined expectations for both patients and physicians, could also limit the use of appropriate medication and achievement of better clinical outcomes. Research is ongoing to evaluate new medications with unique mechanisms of action, such as ileal bile acid transporter inhibitors 18 and rifampicin, 19 to find effective and safe treatments for pruritus in patients with ICP.
Conclusions
In summary, pruritus in patients with ICP remains a common and disease-defining clinical symptom with a significant adverse impact on patients’ lives. More research and better clinical tools are needed to understand the nature and course of pruritus in patients with ICP. Current ineffective treatment options demonstrate the significant unmet medical need to develop viable medications for the management of pruritus in patients with ICP.
Supplemental Material
sj-docx-1-obm-10.1177_1753495X251397321 - Supplemental material for Impact of pruritus in patients at high risk of significant intrahepatic cholestasis of pregnancy
Supplemental material, sj-docx-1-obm-10.1177_1753495X251397321 for Impact of pruritus in patients at high risk of significant intrahepatic cholestasis of pregnancy by Catherine Williamson, Jenny Chambers, Caroline Ovadia, George Saade, Andrew McKibben, Tiago Nunes, Jolan Terner-Rosenthal, Will Garner and Pamela Vig in Obstetric Medicine
Supplemental Material
sj-docx-2-obm-10.1177_1753495X251397321 - Supplemental material for Impact of pruritus in patients at high risk of significant intrahepatic cholestasis of pregnancy
Supplemental material, sj-docx-2-obm-10.1177_1753495X251397321 for Impact of pruritus in patients at high risk of significant intrahepatic cholestasis of pregnancy by Catherine Williamson, Jenny Chambers, Caroline Ovadia, George Saade, Andrew McKibben, Tiago Nunes, Jolan Terner-Rosenthal, Will Garner and Pamela Vig in Obstetric Medicine
Footnotes
Acknowledgments
The authors would like to thank Vyechi Low for managing the survey site at Harvard University in Cambridge, MA, USA. Editorial support was provided by StrongTower Consulting and Precision AQ in Bethesda, MD, USA, and was funded by Mirum Pharmaceuticals, Inc.
Ethical approval
Ethical approval (other than the internal patient advocacy group review team) was not sought for the present study because it was a noninterventional, voluntary, and anonymous survey. The study was conducted in accordance with the Declaration of Helsinki.
Consent to participate
Informed consent was not sought for the present study because participation in the survey was voluntary and anonymous.
Consent for publication
Individuals who participated in the survey consented to the sponsor's use and publication of the collected data.
Contributorship
All authors were involved in the conception, design, acquisition of data, data analysis, manuscript drafting and revision, and approval of manuscript submission to the journal.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Mirum Pharmaceuticals, Inc.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CW is a consultant and has a research grant from Mirum Pharmaceuticals, Inc. She was a funded seminar speaker for Ipsen and Advanz Pharma UK in 2023 and is a consultant for Ipsen. She was a consultant for GSK in 2019. JC and GS report no conflicts of interest. CO is a consultant for Mirum Pharmaceuticals, Inc. AM, TN, JT-R, WG, and PV are employees of and shareholders in Mirum Pharmaceuticals, Inc.
Data availability statement
Beginning 6 months and ending 5 years after publication, deidentified participant data might be made available to investigators whose proposed use of the data has been approved by a review committee, including the primary authors and the study funder. Proposals should be directed to grants@mirumpharma.com. Before being granted access, data requesters will be required to sign a data access agreement.
Guarantor
CW.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
