Abstract
We report the case of a 21-year-old woman at 29 weeks and four days of gestation who presented with one-week history of severe epigastric pain, pleuritic chest pain, dyspnoea, and chills. A peripherally inserted central catheter line had been placed at 14 weeks of gestation for outpatient management of hyperemesis gravidarum. Initial investigations included a transthoracic echocardiogram which revealed a large tricuspid valve vegetation with mild tricuspid regurgitation and CT pulmonary angiogram which identified multiple septic pulmonary emboli. Blood cultures grew methicillin-susceptible Staphylococcus aureus. Despite appropriate antimicrobial therapy, they remained bacteraemic and underwent percutaneous mechanical debulking of tricuspid valve infective endocarditis using an AngioVac aspiration thrombectomy device. Their clinical course post-AngioVac was complicated by the development of multiple pulmonary emboli and suspected left-sided empyema. They delivered by caesarean section at 32 weeks and one day of gestation due to maternal health reasons.
Keywords
Introduction
Infective endocarditis (IE) is a rare but serious disease in pregnancy associated with high maternal and fetal morbidity and mortality. The diagnostic and management principles for IE in pregnancy are similar to non-pregnant women. Intravenous (IV) antibiotics are the mainstay of treatment of IE. Surgery is reserved for cases complicated by acute heart failure, to reduce risk of embolic phenomenon, or for refractory cases. AngioVac (AngioDynamics, USA) is an aspiration thrombectomy device which represents a percutaneous alternative to cardiac surgery to debulk right-sided vegetations due to IE in a less invasive manner in pregnancy. The AngioVac system is composed of venous drainage and return cannula and an extracorporal circuit with pump and filter.1,2 Blood and vegetations are aspirated and filtered, and venous blood is then returned to the patient's circulation.1,2
Case report
A 21-year-old immunocompetent woman in her first pregnancy was transferred to our tertiary care hospital at 29 weeks four days of gestation with a one-week history of severe epigastric pain radiating to the back with accompanying chills and subjective fevers. They also reported a seven-day history of pleuritic chest pain, dyspnoea, presyncope, and palpitations. Their pregnancy was complicated by severe hyperemesis gravidarum (HG), necessitating six emergency department visits and three prior hospitalisations for IV hydration, electrolyte replacement, and IV antiemetics. Their symptoms were refractory to scheduled doxylamine and pyridoxine, oral metoclopramide, oral ondansetron, and oral/rectal dimenhydrinate. They had a peripherally inserted central catheter (PICC line) inserted at 14 weeks of gestation at a peripheral hospital to facilitate outpatient administration of intravenous fluids and IV antiemetics.
On initial presentation, their temperature was 37.0°C, blood pressure 123/55 mmHg, heart rate 142 beats/minute, respiratory rate 24 breaths/minute, and oxygen saturation 95% on room air. The patient appeared uncomfortable. No murmurs were present. There were no petechiae. Bilateral crackles to the mid-lung fields were noted. They had a gravid uterus with tenderness to the epigastrium and left upper quadrant. There was no costovertebral angle tenderness. Bilateral calf tenderness was present with mild right foot oedema. No erythema or purulence was observed around the PICC line.
Initial investigations revealed a haemoglobin of 115 g/L, white blood cell count of 23.2 × 109/L, and platelet count of 95 × 109/L. Liver transaminases were mildly elevated: AST 40 U/L, ALT 78 U/L. Bilirubin, haptoglobin, lipase, INR, creatinine, and urine albumin-to-creatinine ratio were normal. Fibrinogen was elevated. High-sensitivity troponin was 1121 ng/L (normal 0–16 ng/L) and ECG showed sinus tachycardia.
Over the next 12 hour the patient became febrile and hypoxaemic. Chest x-ray showed bilateral patchy opacities and piperacillin-tazobactam and azithromycin were started due to clinical suspicion of severe pneumonia. CT pulmonary angiogram showed bilateral multifocal consolidative opacities as well as a filling defect within the distal segmental right lower lobe suggestive of septic emboli. Bilateral lower leg Dopplers were negative for deep vein thrombosis. Transthoracic echocardiogram (TTE) revealed a large independent mobile mass on the septal leaflet of the tricuspid valve measuring 24 × 10 mm and mild tricuspid regurgitation (Figure 1). Blood cultures grew methicillin-susceptible Staphylococcus aureus and a diagnosis of PICC-associated tricuspid valve endocarditis (IE) with pulmonary septic emboli was made.
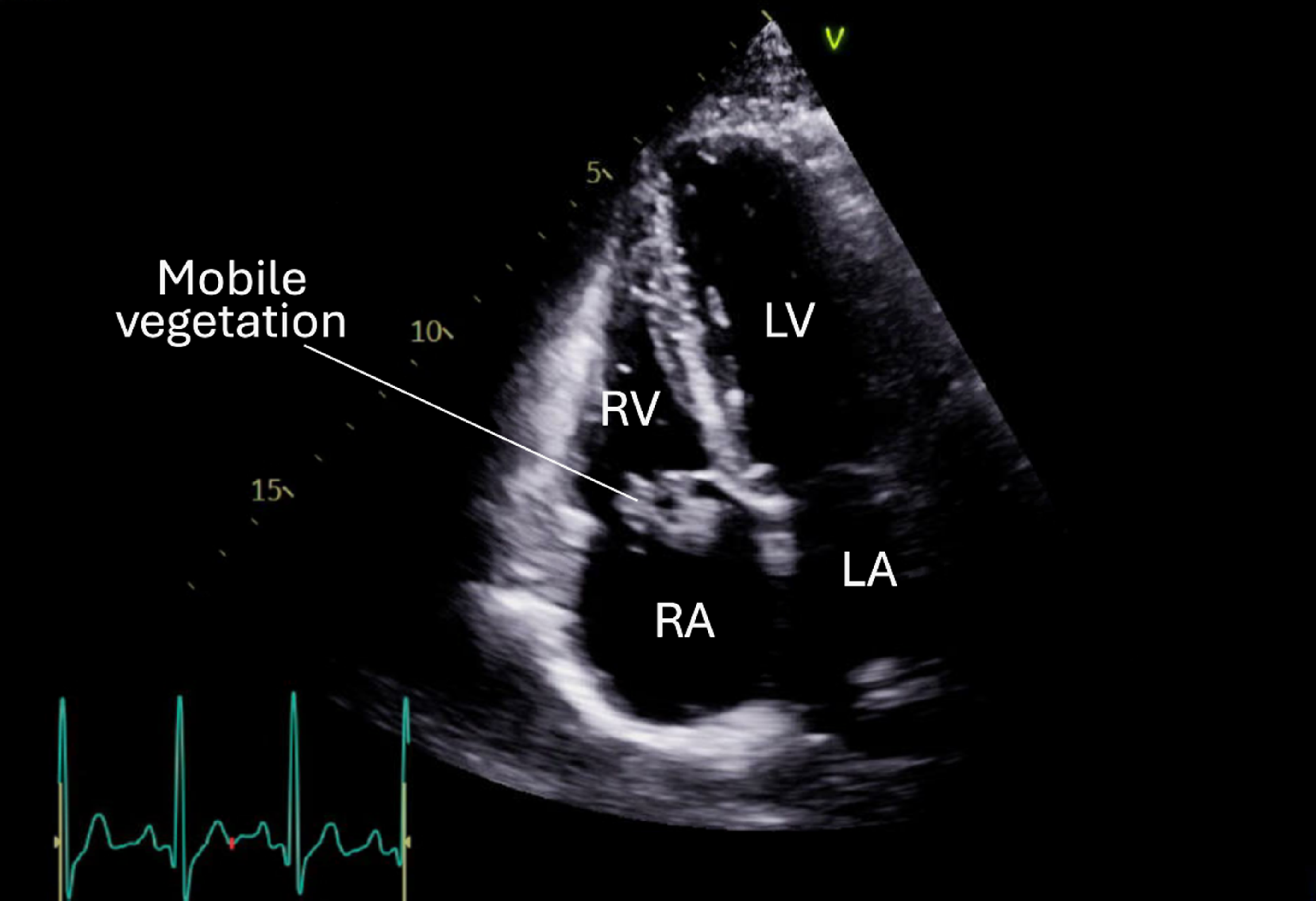
Transthoracic echocardiogram showing a large vegetation on the septal leaflet of the tricuspid valve.
The patient was started on IV cefazolin and the PICC line was removed. Blood cultures remained positive after 7 days of antimicrobial treatment and antimicrobials were changed to IV cloxacillin at the recommendation of the Infectious Disease team. MRI spine was normal. Repeat TTE on day 8 of admission showed progressive enlargement of the tricuspid valve vegetation (23 × 19 mm) with mild-to-moderate tricuspid regurgitation. Clinically the patient had ongoing dyspnoea and tachycardia related to persistent bacteraemia and septic pulmonary emboli.
The risks of cardiac surgery during pregnancy were considered prohibitive by the Cardiac Surgery team. AngioVac was considered as an alternative temporising measure to achieve source control. AngioVac debulking of the tricuspid valve vegetation was performed at 31 weeks four days of gestation, on day 14 of admission.
Post-AngioVac the patient developed recurrent pleuritic chest pain and dyspnoea. Repeat CT pulmonary angiogram showed multiple pulmonary emboli, interval cavitation and necrosis of the bilateral consolidations, and a small-to-moderate loculated left pleural effusion. Therapeutic anticoagulation was initiated. IV heparin was preferred by Maternal Fetal Medicine due to the patient's tenuous circumstances and unknown timing of delivery. The loculated left pleural effusion was not amenable to percutaneous drainage. Amoxicillin-clavulinic acid was started for the treatment of suspected empyema.
Obstetric ultrasounds identified a small for gestational age fetus (fifth percentile) with normal biophysical profile. Maternal Fetal Medicine initially recommended delivery at 35 weeks of gestation, however a multidisciplinary decision was made to proceed with caesarean section under general anaesthesia at 32 weeks and one day of gestation for maternal health reasons, including persistent maternal tachycardia (heart rate 120 to 140 beats/min), pain related to positioning of the fetus, difficulty achieving therapeutic PTT on IV heparin, and maternal emotional distress. There were no signs of fetal distress. Delivery under general anaesthesia was preferred by the anaesthesia team due to use of IV heparin, although there were no absolute contraindications to spinal or epidural anaesthesia. A 1615-g female infant was delivered.
The patient's tachycardia and pain improved postpartum. A six-week total course of IV antibiotics was completed for IE as well as six-weeks of amoxicillin-clavulanic acid for suspected empyema. Therapeutic dalteparin was recommended for a total of three months postpartum for provoked pulmonary emboli. Repeat CT chest showed improvement of the necrotising pneumonia and resolution of the loculated pleural effusion. TTE eight months postpartum showed normal biventricular function, normal tricuspid leaflets, and moderate tricuspid regurgitation. No further cardiac interventions are planned.
The patient's daughter was admitted to NICU for almost eight weeks postpartum, though had no significant medical complications.
Discussion
IE in pregnancy is very rare with an estimated incidence of 0.006% 3 and is associated with high maternal and fetal mortality (11–33% maternal, 14.6–29% fetal).3,4 Risk factors for IE include IV drug use, congenital heart disease, rheumatic heart disease, prosthetic valve, and prior IE.4,5 IE is diagnosed based on the Modified Duke criteria. 5
This case highlights iatrogenic harms patients may experience due to medical care. Although our patient had severe HG, there was no history of difficult IV access. Additionally, our patient had received limited PICC care in the community and had been self-administering IV fluids at home, further increasing the risk of bacteraemia. While PICC lines may be used for the administration of IV fluids and parenteral nutrition for patients with severe HG, studies have shown high rates of maternal complications associated with antenatal PICC line use, including bacteraemia and thrombosis, 6 so judicious use is recommended. If PICC lines are used patients and healthcare providers should monitor for complications associated with their use and PICC lines should be discontinued promptly when no longer medically necessary.
IE in pregnancy should be treated the same way as in non-pregnant women, including early IV antimicrobial therapy, source control, and managing complications. 7 A multidisciplinary approach to the care of pregnant women with IE should be undertaken with involvement of specialists in the field of Maternal Fetal Medicine, Infectious Disease, Cardiology, Cardiac Surgery, and Obstetric Medicine. Clinicians should monitor for signs of complications which may warrant surgical intervention including acute heart failure, resistant organisms, recurrent embolic phenomenon, perivalvular abscess, or persistent bacteraemia refractory to medical management. 7 Cardiac surgery in pregnancy carries high risk of maternal complications and very high risk of fetal mortality. A recent meta-analysis showed maternal mortality of 7.3% after cardiac surgery and fetal mortality of 26.5%, with highest fetal mortality documented if surgery is performed in the first trimester (44.8%) and lowest with surgery in the third trimester (10.3%). 8 For patients with right-sided IE with contraindications to cardiac surgery percutaneous debulking of vegetations by AngioVac may be beneficial.1,9 In a 2019 meta-analysis of use of AngioVac for extraction of venous thromboses and endocardial vegetations, this procedure has a 74.5% success rate and an operative mortality rate of 14.6% in non-pregnant women. 1 However, more recent studies have shown higher procedural success of over 92.6% and lower all-cause mortality rates of 7.3% or less.9,10 Case reports of AngioVac use in the third trimester of pregnancy have been described in patients with right-sided IE secondary to substance abuse in whom surgical risk was prohibitive.11–13 Potential complications of AngioVac procedure include distal embolisation of thrombus or vegetation, injury to blood vessels or valves, and local or systemic infection.
To our knowledge this is the first case report of PICC-associated tricuspid valve IE in pregnancy managed with AngioVac. Data on the safety and efficacy of AngioVac in pregnancy remains limited, and further research is needed.
Footnotes
Acknowledgments
We would like to acknowledge our patient who provided consent to share their story.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This case report was granted an exemption for ethics review by the Queen's University Health Sciences and Affiliated Teaching Hospitals Research Ethics Board (HSREB) per the Tri-Council Policy Statement: Ethical Conduct for Research Involving Humans (TCPS 2).
Informed consent
The patient provided written informed consent for the publication of this case report.
Guarantor
Lindsay Anderson guarantees the authenticity of this clinical case.
Contributorship
Lindsay Anderson drafted the manuscript; Lisa Nguyen critically reviewed and edited the commentary for content and accuracy. All authors gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
