Abstract
This article reviews anatomical and physiological changes and alterations in reference intervals for laboratory tests in healthy pregnancy, pertinent to investigation of the gastrointestinal system. The safety of procedures and radiological investigations relevant to the investigation of gastrointestinal disorders in pregnancy are also reviewed.
Introduction
From the time of conception, significant physiological alterations occur affecting all organ systems in pregnancy. Awareness of anatomical and physiological changes in the gastrointestinal system in pregnancy is important for the correct interpretation of investigations. Knowledge of the alterations in reference intervals for laboratory investigations is crucial for correct interpretation of results in diagnosis and management. The need for imaging and procedures frequently cause maternal concern with regard for the safety of the fetus. Familiarity with the evidence regarding these examinations is important to assist the pregnant woman in making informed decisions regarding consent, as well as providing reassurance.
Physiological and anatomical changes
Liver volume measured by magnetic resonance imaging (MRI) increases by a mean of 15% between 12–16 weeks’ gestation and 32–36 weeks’ gestation. 1 A longitudinal cohort study of 24 healthy women with normal singleton pregnancies found that mean liver stiffness increased from 3.8 kPa at 18–20 weeks’ gestation to 5.9 kPa at 36–38 weeks’ gestation, with 2 women (8%) having an elastography measurement of greater than 7.9 kPa in third trimester. 2 By 8 weeks postpartum, liver stiffness had decreased to early second trimester values. Similarly, the mean controlled attenuation parameter, a non-invasive measure of quantifying hepatic steatosis, increased from 186 dB/m in early second trimester to 215 dB/m in late third trimester, and reversed postpartum. 2 Total liver blood flow increases by 163% during third trimester due to increased portal venous flow. 3 Combined with the 60% increase in blood volume in pregnancy, this may result in worsening portal hypertension in women with chronic liver disease. Hepatic arterial flow is unchanged, with reduced hepatic artery resistance. 3
The diameter of the common hepatic duct is unchanged in pregnancy compared with non-pregnant females, with an upper limit of normal being 5 mm. 4 Gallbladder contractility and emptying in pregnancy is reduced, mediated by increased levels of progesterone, resulting in an increase in residual gallbladder volumes. 5 Spleen size increases significantly in pregnancy with increasing gestational age. 6 Awareness of the physiological increase in splenic size is important regarding the diagnosis of portal hypertension in women with chronic liver disease. A Nigerian study of healthy pregnant women found that splenic volume increased by 60% from first to third trimester. 7 Normograms for splenic volume and length at specific gestation are available.
Early in pregnancy, lower oesophageal sphincter pressure falls under the influence of oestrogen and progesterone, returning to preconception levels by 1–4 weeks postpartum. 8 Gastro-oesophageal reflux symptoms are reported by 40–85% of pregnant women, usually from the end of first trimester, and persisting for the remainder of pregnancy. Gastric emptying is unchanged throughout pregnancy compared with non-pregnant individuals, though may be delayed in the immediate postpartum period depending on the analgesia and anaesthesia used for delivery. 9 Gastric emptying is accelerated in hyperemesis gravidarum. 10 Point-of-care ultrasound is a reliable method in assessing for gastroparesis during pregnancy. 11 Elastogastrogram studies have found an increased prevalence of gastric dysrhythmias in nauseated pregnant women. Small bowel transit times are prolonged in the second and third trimesters of pregnancy when compared to the first trimester of pregnancy or postpartum period. 12 Colonic transit times have not been studied in pregnancy.
Laboratory investigations
Liver disease
Pregnancy is associated with mild falls in alanine aminotransferase (ALT), aspartate aminotransferase (AST), gamma-glutamyl transferase (GGT), bilirubin, and albumin13,14 (Table 1). Some authors have reported that lactate dehydrogenase (LDH) may progressively rise from first trimester, normal values in third trimester being up to double values prepregnancy. 13 Other authors report no change in LDH. 14 An LDH:AST ratio of greater than 22.12 provided accurate discrimination between diagnoses of pregnancy-associated thrombotic thrombocytopenic purpura and haemolysis, elevated liver enzymes, and low platelet (HELLP) syndrome in third trimester in women presenting with haemolysis, abnormal liver enzymes, and thrombocytopenia. 15
Sample reference intervals in pregnancy.
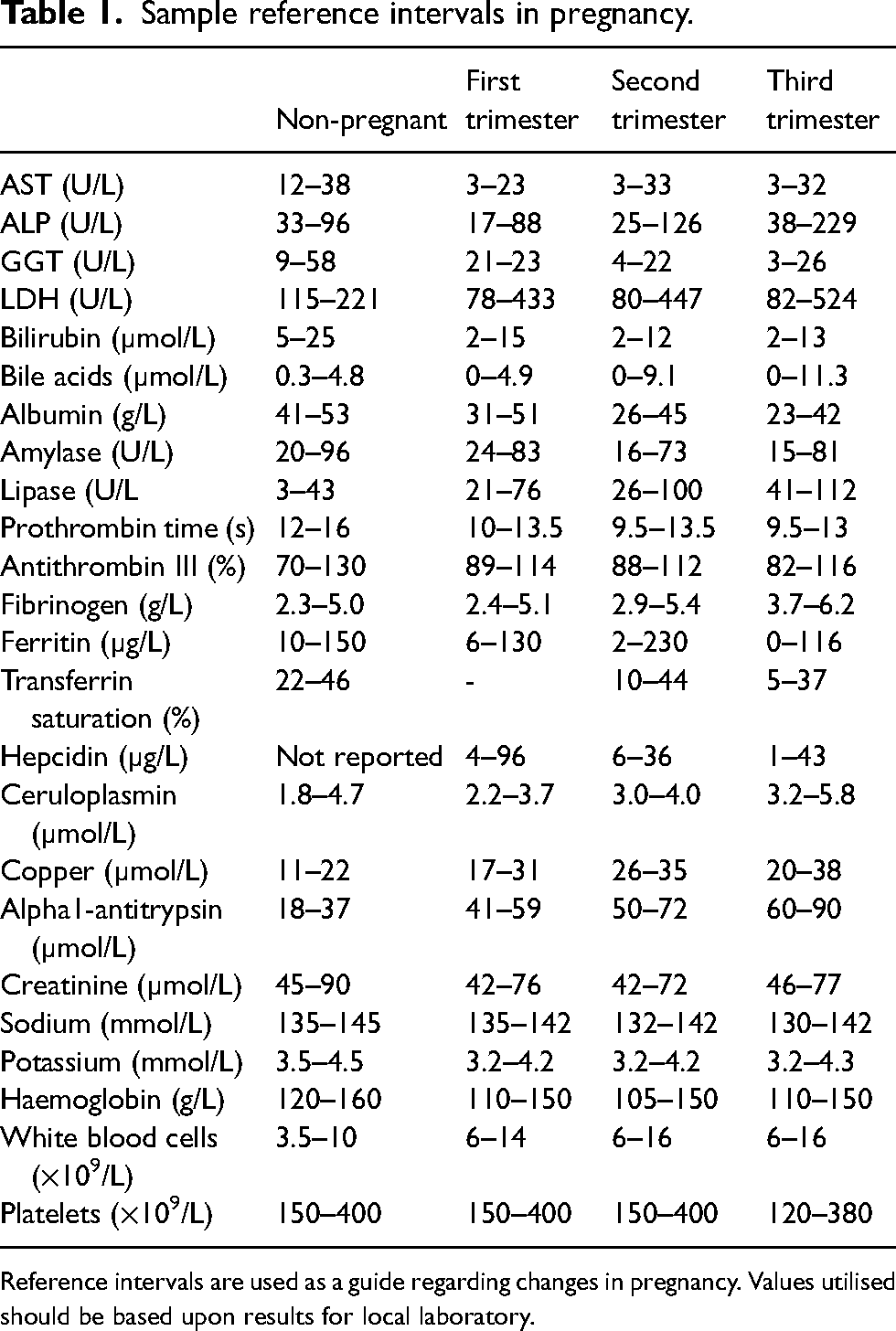
Reference intervals are used as a guide regarding changes in pregnancy. Values utilised should be based upon results for local laboratory.
Fasting and postprandial total serum bile acids (TSBA) rise progressively from preconception values, peaking in late pregnancy.16,17 TSBA and liver enzymes may lack sensitivity in diagnosis of intrahepatic cholestasis of pregnancy (ICP), the onset of pruritus predating biochemical abnormalities in 21% of women, by a mean of 4.5 weeks.18,19 Elevated TSBA is also not specific for ICP, being elevated in a variety of hepatic disorders including non-alcoholic fatty liver disease, hyperemesis gravidarum, and pruritus of unknown origin. 20
Levels of alkaline phosphatase (ALP) rise due to production of the placental isoenzyme, as well as a significant increase in bone isoenzyme in third trimester.13,14 An acute rise in ALP may signify placental damage or infarction. Rarely, marked elevations in ALP up to 30 times the upper end of the pregnancy reference interval may occur without adverse maternal or fetal outcomes and may be managed expectantly in the absence of other feto-maternal concerns. 21 Elevated placental ALP may also be seen with antiphospholipid syndrome. 22
Prospective studies in Wales and India each found abnormal liver enzymes in 3.2% of pregnant women.23,24 The majority of cases of abnormal liver enzymes in both studies were related to pregnancy-specific liver disease (Table 2). More recently, 13.9–29.7% of pregnant women admitted to a hospital with COVID-19 infection had elevated liver enzymes. 21 An audit of 329 cases of marked transaminitis (ALT and/or AST > 300U/L – normal < 30) during pregnancy at a tertiary maternal hospital in Brisbane over a 13-year period found the major aetiologies identified to be ICP (37%), preeclampsia with HELLP syndrome (22%), preeclampsia without haemolysis or low platelets (11%), acute fatty liver of pregnancy (AFLP, 5.2%), hyperemesis gravidarum (3.6%), cholecystitis/pancreatitis (3%), prepregnancy non-alcoholic fatty liver disease (1.8%), and bacterial sepsis (1.5%) (unpublished data).
Causes of abnormal liver function tests (LFTs) in pregnancy.
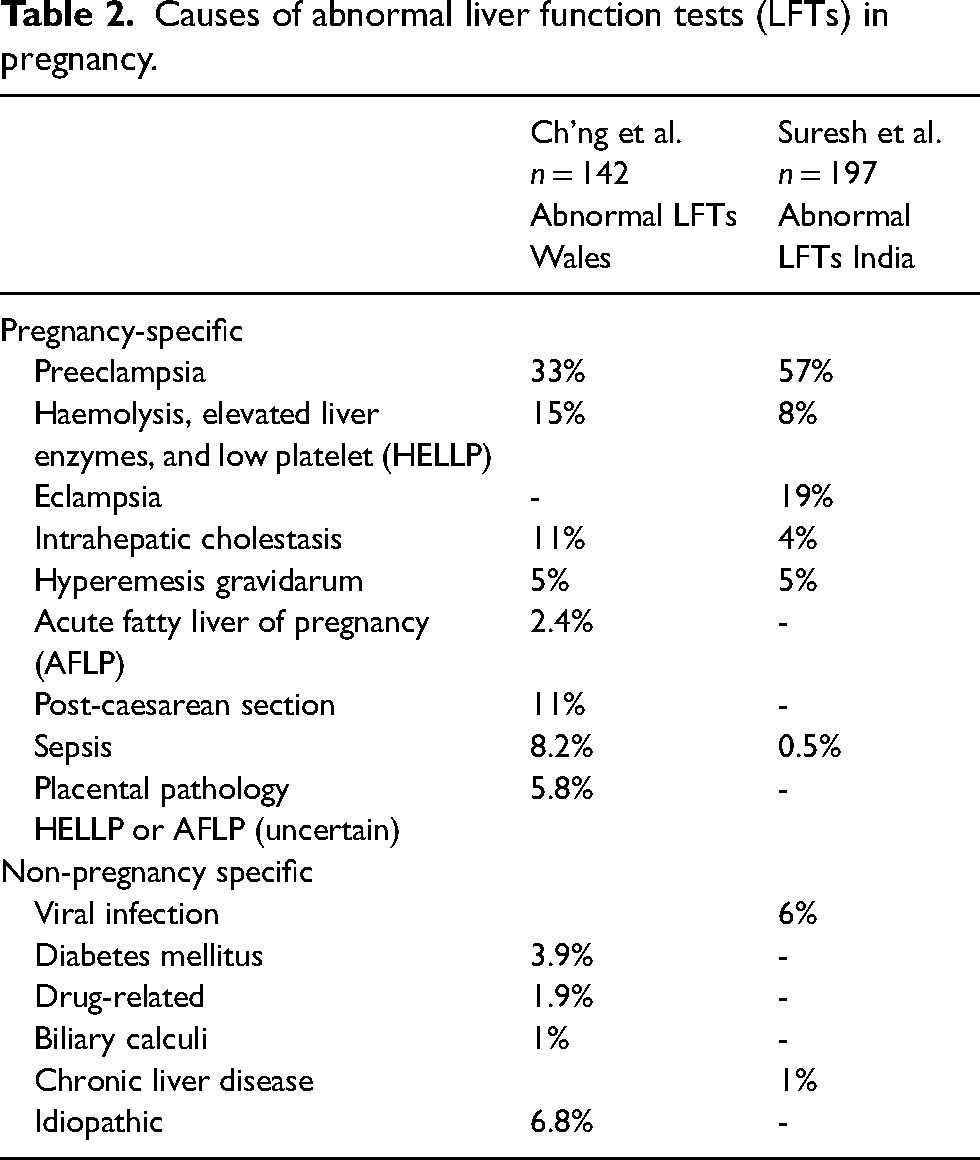
The aetiology of acute liver failure (ALF) in pregnancy varies according to the population examined. A study of 70 pregnant women with ALF in the United States found the predominant causes to be paracetamol-induced liver injury (30%), HELLP syndrome (23%), and AFLP (20%). 25 Other significant causes included herpes simplex hepatitis (6%), autoimmune hepatitis (4%), cancer (4%), and drug-induced liver injury (3%). 25 A study of 249 pregnant women with ALF in India found that 59.4% of cases were due to hepatitis E virus, without any cases of AFLP. 26
Prothrombin time falls progressively from the second trimester such that values are approximately 10–20% lower than preconception values in the third trimester. 27 This may be significant when considering abnormal hepatic synthetic function regarding disorders such as AFLP. Similarly, activated partial thromboplastin time (APTT) and thrombin time fall by 10–20% from the first to third trimester. 27 Fibrinogen levels rise significantly during healthy pregnancy, and the use of non-pregnant reference intervals may underestimate the prevalence of disseminated intravascular coagulation (DIC). A fibrinogen level less than 3 g/L together with a platelet count less than 50 × 109/L and prolonged PT/APTT is consistent with DIC in pregnancy. A pregnancy-modified score considering the physiological changes in PT, fibrinogen, and platelets reported a sensitivity of 88% and specificity of 96% for the diagnosis of DIC. 28 Studies described either no change or a mild fall in antithrombin III (ATIII) during healthy pregnancy. Low levels of ATIII may be useful for distinguishing between AFLP and HELLP syndrome.29,30 A review of 61 women with AFLP found a mean ATIII of 16.1% compared with mean levels of 62–80% in HELLP.30,31 Low ATIII levels, however, do not distinguish AFLP from other causes of acute hepatic failure.
The incidence of maternal Wilson disease in pregnancy ranges between 1 in 30,000 and 1 in 100,000 live births. 32 The diagnosis of Wilson disease in non-pregnant individuals is based upon a combination of clinical symptoms and signs, measures of copper, ceruloplasmin, and liver enzymes, hepatic copper and histology, and genetic testing. Serum ceruloplasmin levels and total serum copper progressively rise during healthy pregnancy from the sixth week of gestation such that third trimester serum copper levels are approximately double those of prepregnancy levels. 33 These physiological changes may complicate the diagnosis of Wilson disease in pregnancy. Twenty-four-hour urine copper levels in healthy pregnant women are not significantly different from those in healthy non-pregnant women. Twenty-four-hour urinary copper excretion greater than 1.6 µmol/24 h is diagnostic of Wilson disease in symptomatic patients; however, levels may also be increased in preeclampsia and other liver diseases. 34 Wilson disease may imitate HELLP syndrome by presenting with abnormal liver enzymes, thrombocytopenia, haemolysis, and coagulopathy. 35
While an ALP-to-total bilirubin ratio less than 4 may help to make a diagnosis of Wilson disease in non-pregnant patients presenting with ALF, the physiological rise in ALP in pregnancy may render this unhelpful. 36 Serum non–ceruloplasmin-bound copper level greater than 1.6 µmol/L is indicative of Wilson disease in non-pregnant individuals; however, there are no reference intervals for serum copper levels diagnostic of Wilson disease pregnancy. 37 Liver biopsy to estimate hepatic parenchymal copper and histology is not performed routinely in pregnant women due to risks involved and the lack of specificity in diagnosis. Routine use of molecular genetic analysis is not recommended for diagnosis, as most affected patients are compound heterozygotes, resulting in a complex process for identification.
The author is unaware of any guidelines regarding target levels of 24-h urine copper excretion in women treated with chelating agents during pregnancy.
Hereditary haemochromatosis (HH) may be associated with left ventricular systolic and diastolic dysfunction and pulmonary hypertension. 38 The correlation between iron study parameters and echocardiographic indices is relatively weak, and iron studies cannot be used to select which individuals with HH should have echocardiography performed. 39 Left ventricular ejection fraction has been shown to be reduced in recently diagnosed HH compared with healthy controls. 39 In the absence of consensus guidelines, previous authors recommend that all patients with HH undergo echocardiography at diagnosis and every 1–2 years thereafter, and that cardiac MRI be performed if echocardiographic abnormalities are detected.40,41 Echocardiography should be considered preconception or in first trimester in all women with HH who have not had echocardiography in the previous 12 months. HH may also be associated with elevated maternal lead levels, increasing the risk of gestational hypertension, preeclampsia, reduced birth weight, neurodevelopmental disorders, and subclinical brain injury. 42 It would seem prudent to measure maternal serum lead levels in each trimester in women with HH. A normal term pregnancy results in the loss of 1 g of iron from the mother. Sokoloff et al. 43 described a progressive fall in ferritin while transferrin saturation remained unchanged in a pregnant woman with HH.
Alpha-1 antitrypsin levels rise steadily throughout pregnancy such that third trimester values are approximately 2.5–6 times those preconceptions. 44 Prenatal and preimplantation diagnosis of alpha-1 antitrypsin deficiency is available, although it is not recommended as results do not predict prognosis or disease severity.
Pancreas
Serum amylase activity is unchanged throughout pregnancy. Serum lipase activity is significantly lower in the first trimester, though unchanged in second and third trimesters compared with non-pregnant women. 45 Hyperemesis gravidarum has been associated with significantly elevated levels of lipase and amylase in the absence of clinical or radiological evidence of acute pancreatitis. 46 The most common causes of acute pancreatitis in pregnancy are gallstones (66%), alcohol excess (12%), idiopathic (17%), and hypertriglyceridaemia (4%). Less common causes in pregnancy include hyperparathyroidism, trauma, betamethasone therapy, and AFLP. 47 During pregnancy, there is a two- to fourfold increase in plasma triglyceride levels related to increasing insulin resistance and reduced activity of lipoprotein lipase. The risk of acute pancreatitis is increased when plasma triglyceride levels are >11.3 mmol/L.
Individuals with hypertriglyceridaemic pancreatitis may have falsely normal lipase and amylase levels due to hypertriglyceridaemia interfering in vitro with the laboratory assay. 48
Inflammatory bowel disease
Faecal calprotectin levels remain unchanged in healthy pregnancy, though calprotectin levels rise in parallel with disease activity in pregnant women with inflammatory bowel disease (IBD). 49 Most authors state C-reactive protein remains unchanged during pregnancy. Erythrocyte sedimentation rate (ESR) rises progressively during healthy pregnancy, values being as high as 112 mm/h in third trimester. 50 ESR is therefore not useful as a marker of infection or inflammation in pregnancy.
White blood cell count (WBC) increases significantly in uncomplicated pregnancy due to increased neutrophils, with typical reference interval of 6–16 × 109/L in second and third trimesters. 51 Pregnancy-related changes in WBC persist for 6–8 weeks after delivery. 51
Serum total vitamin B12 levels fall significantly in healthy pregnancy, and active B12 levels should be measured. 51
There is no published data on the use of the presence of faecal occult blood or faecal immunohistochemical testing in pregnancy; however, positive salivary occult blood testing occurs after the gentle use of a soft-bristle toothbrush in 65% ± 17% of pregnant women. This suggests faecal testing for occult blood is unlikely to be helpful. 52 Additionally, 10–43% of women report rectal bleeding in pregnancy, due to benign perianal disease in most cases. 53
Tumour markers
Carcinoembryonic antigen and carbohydrate antigen 19-9 rise in third trimester in healthy women though usually remain in the normal range. 54 The significant physiologic rise in alpha fetoprotein during pregnancy is relevant where these are being used for screening for hepatocellular carcinoma. 54 Gastrin levels remain unchanged during pregnancy. 55
Procedures
The general principles of procedures in pregnancy are summarised in Table 3. 56
General principles of procedures in pregnancy.

No currently used anaesthetic agents have been shown to have any teratogenic effects in humans when used in standard doses at any gestational age. 57 There is no evidence that in utero exposure to anaesthetic or sedative drugs has any effect on the developing fetal brain. Propofol is generally the anaesthetic agent of choice during pregnancy due to its long history of safety during pregnancy and its unchanged pharmacokinetics in early pregnancy. 56 Midazolam is the preferred benzodiazepine in pregnancy.
Esophagogastroduodenoscopy
The risk of oesophagogastroduodenoscopy (EGD) in pregnancy is similar to the general population and should be performed if strongly indicated and likely to change management. 58 Fetal heart rate remains normal during EGD, Apgar scores in infants of mothers who underwent EGD were similar to those of infants whose mothers did not undergo EGD, and adverse fetal adverse outcomes reported following maternal EGD appeared to be related to high-risk pregnancies and not temporally related to maternal EGD. EGD has not been shown to be associated with congenital malformations or to precipitate preterm labour. 59
Strong indications for EGD in pregnancy with high diagnostic yield include major or continued upper gastrointestinal bleeding and recent onset dysphagia with weight loss and suspected malignancy. EGD is rarely helpful or indicated for intractable vomiting/hyperemesis gravidarum as the diagnostic yield is low and EGD did not lead to a significant change in management.58,60
Therapeutic EGD
The risk of variceal bleeding is highest in second trimester, when portal venous pressure peaks, and during labour, due to increased vascular resistance with Valsalva manoeuvres. A systematic review and meta-analysis found the rate of variceal bleeding in pregnancy with non-cirrhotic portal hypertension was 9.6%, without any maternal mortality. 61 A meta-analysis of pregnancy outcomes in women with liver cirrhosis reported acute variceal haemorrhage in 4%. Variceal haemorrhage in pregnant women with cirrhosis is associated with maternal mortality in 12% of cases. 62 Endoscopic variceal ligation is preferred to endoscopic sclerotherapy in pregnancy, due to higher efficacy and lower risk of complications. 63
The most common causes of non-variceal upper gastrointestinal bleeding in pregnant women are Mallory–Weiss tear (25%), gastritis (12%), peptic ulcer disease (6.8%), oesophagitis (2.5%), anastomotic ulcer (0.2%), and arteriovenous malformation (0.1%). A non-endoscopic approach in pregnant women with non-variceal upper gastrointestinal bleeding is reasonable, as conservative management is not associated with worse maternal or fetal outcomes. 64
Achalasia symptoms may be exacerbated during pregnancy, postulated to be due to the elevation of the diaphragm by the fetus, reduction in smooth muscle motility and tension secondary to increased progesterone concentrations, and oesophagitis due to reflux. High-resolution oesophageal manometry is safe in pregnancy, although there are no pregnancy-specific reference values. 65 Five cases of bougie dilatation, seven cases of pneumatic dilatation, five cases of botulinum toxin injection, and one case of laparoscopic Heller's myotomy have been described in pregnancy. 65 While there are no controlled trials regarding the safety of botulinum toxin administration in pregnancy, intramuscular administration does not lead to significant systemic concentrations, and the large molecular size should prevent botulinum toxin crossing the placental barrier. The use of botulinum type A toxin has been described in more than 50 pregnancies in the management of migraine, cervical dystonia, and cosmetic surgery without adverse maternal, fetal, or childhood outcomes. 66 Peroral endoscopic myotomy has been performed for achalasia in one pregnant woman with mitochondrial disease.
Endoscopic ultrasound
Endoscopic ultrasound (EUS) is safe in pregnancy, though may require longer anaesthetic time. 67 EUS should be considered if there is an intermediate probability of common bile duct stone and magnetic resonance cholangiopancreatography (MRCP) is not desirable. EUS may be done in the same session as endoscopic retrograde cholangiopancreatography (ERCP), or ERCP may be performed through linear echo-endoscope ultrasound guidance.
Enteroscopy
Enteroscopy in pregnancy is complicated by compression of small bowel by the gravid uterus and the risks of placental abruption due to pressure from the enteroscope, prolonged procedure time, and the requirement for higher anaesthetic doses. A single case of double-balloon enteroscopy in second trimester in a pregnant woman with Peutz–Jeghers syndrome has been described. 68
Capsule endoscopy
No trials have been performed regarding the safety of capsule endoscopy in pregnancy. Concerns relate to possible damage to the fetus from electromagnetic waves emitted from the capsule transmitter and the risk of capsule retention due to prolonged small bowel transit time in pregnancy. Three cases of capsule endoscopy have been described in pregnancy, one in each trimester. 69 Due to the lack of long-term safety data, pregnancy is regarded as a relative contraindication to capsule endoscopy.
Endoscopic cystogastrostomy
Three cases of endoscopic cystogastrostomy have been performed for pancreatic pseudocyst in second trimester, two under abdominal ultrasound guidance and the third appeared to be with EUS. 70
Percutaneous endoscopic gastrostomy and percutaneous endoscopic gastrojejunostomy
Percutaneous endoscopic gastrostomy/jejunostomy (PEG/J) has been described in 14 pregnancies in the management of refractory hyperemesis gravidarum, restrictive eating disorders, swallowing impairment, and chronic intestinal pseudo-obstruction. 71 PEG/J placement has been described as early as 8 weeks and as late as 29 weeks’ gestation. Ultrasound-guided marking of the upper limit of the uterus should be performed prior to PEG/J to prevent uterine puncture. Stomal leak and pain may occur secondary to gastric compression by the uterus. To date, no adverse effect on pregnancy outcome has been described after PEG placement.
Endoscopic retrograde cholangiopancreatography
A systematic review of 24 studies reviewed 327 endoscopic retrograde cholangiopancreatography (ERCP) procedures in pregnant women. 72 Ninety (27%) of the procedures were performed in first trimester, 108 (33%) in second trimester, and 108 (33%) in third trimester. ERCP was performed without fluoroscopy in 88 procedures (26.8%). Maternal complications were reported in 44 women (14.3%). Twenty-one women (6.8%) developed post-ERCP pancreatitis. Premature delivery was reported in 14 women (4.6%). Fetal complications were reported in 13 pregnancies (5.5%) – fetal loss abortion in 5 pregnancies, elective abortion of 2 pregnancies, low birth weight in 4 infants, and fetal growth restriction in 1 case. The authors concluded that ERCP may be employed safely in pregnancy with a low risk of complications. Similarly, a subsequent systematic review of 1307 ERCP procedures concluded that ERCP is a relatively safe procedure during pregnancy. 73
The technical success rates and risk of bleeding, infection, and perforation with ERCP in pregnant women are similar to that in non-pregnant populations. 74 Pregnant women appear to be at increased risk of post-ERCP pancreatitis, reported in 6.8–16% of pregnant patients, compared with 5% in non-pregnant individuals. 75
Three studies found no long-term complications in a total of 56 offspring of women who underwent ERCP in pregnancy, although exposures were all in second and third trimesters, and the number of cases performed without fluoroscopy was not recorded.76–78 Multiple studies have estimated the average radiation exposure to the fetus as between 0.01 and 5.77 mGy, a level below that which could potentially cause fetal harm.79,80
A single case of percutaneous transhepatic biliary drainage at 12 weeks’ gestation for common bile duct obstruction due to a 9 mm calculus has been described. 81 The drainage tube was left in place until ERCP was performed 2 weeks postpartum.
The use of percutaneous cholecystotomy and percutaneous gallbladder aspiration have been described as a temporary measure in 15 women with acute cholecystitis in first or third trimester. 82 The authors concluded percutaneous cholecystotomy was a safe alternative treatment modality in pregnant patients with co-morbid conditions making surgery high-risk or in third trimester to enable delay of laparoscopic cholecystectomy until the postpartum period.
Liver biopsy
Liver biopsy should be deferred until postpartum if possible. Liver biopsy should be considered where the diagnosis is unclear, presentation is severe and atypical, and where the results may affect management or timing of delivery. A nationwide population-based cohort study linking data from the Swedish Medical Birth Registry between 1992 and 2011 with those from the Swedish Patient Registry identified liver biopsy in 23 pregnancies. 83 Apart from a moderately increased risk of preterm birth and small for gestational age, there was no association between liver biopsy during pregnancy and adverse pregnancy outcome. Westbrook et al. 84 described liver biopsy under ultrasound guidance without complication in 16 pregnant women who had previously undergone liver transplantation.
Light microscopy of liver biopsies in healthy pregnancy shows no specific changes compared with non-pregnant individuals.
Flexible sigmoidoscopy and colonoscopy
A systematic review (n = 164), one prospective (n = 42), three case–control (n = 118), and two retrospective cohort studies (n = 64) found no adverse obstetric or fetal events with flexible sigmoidoscopy or colonoscopy in pregnancy.85–90 Approximately 75% of procedures in these studies were flexible sigmoidoscopies and 25% colonoscopies. Strong indications for sigmoidoscopy or colonoscopy during pregnancy include haematochezia, severe chronic diarrhoea, sigmoid/rectal mass, or stricture. Lower abdominal pain and altered bowel habit are regarded as weak indications. A study of 45 pregnant women undergoing sigmoidoscopy in pregnancy found the most common diagnoses were reactivated or newly diagnosed IBD, other colitis, or bleeding internal haemorrhoids. 88 Change in management as a result of sigmoidoscopy/colonoscopy ranged from 35% to 78%. Polyethylene glycol has been shown to be safe in pregnancy and is preferred to sodium phosphate for bowel preparation. Due to the greater amount of safety data, sigmoidoscopy with tap water enemas is preferred over colonoscopy. 58
Imaging
Ultrasound is the imaging modality of choice during pregnancy in view of the lack of exposure of the fetus to ionising radiation. A study of the use of gastrointestinal ultrasonography (GIUS) in monitoring IBD found adequate colonic views were obtained in 91% of scans throughout pregnancy. 91 Adequate ileal views were obtained in 93% of scans prior to 20 weeks’ gestation and 59% of scans between 20 and 26 weeks’ gestation. 91 A positive correlation between calprotectin and bowel thickness was demonstrated, and GIUS provided a specificity of 83%, sensitivity of 74%, and negative predictive value of 90% compared with calprotectin.
MRI with magnet strength of 3 T or less in any trimester without gadolinium is not associated with increased risk of harm to the fetus or in early childhood. 92 Gadolinium should be avoided as maternal exposure at any stage of pregnancy is associated with increased risk of stillbirth or neonatal death, as well as an increased risk of childhood inflammatory, rheumatologic, or infiltrative skin conditions. 92
The background rate of childhood malignancy is approximately 1 in 3000. Fetal exposure to doses of 10–20 mGy of ionising radiation may increase the risk of childhood leukaemia by a factor of 1.5–2.0 above this background risk. 93 With the exception of the possible increased risk of later carcinogenesis, exposure to less than 50 mGy of ionising radiation at any gestation is associated with negligible additional risk to the fetus. The estimated threshold radiation dose for congenital anomalies or growth restriction during organogenesis (2–8 weeks after conception) is 200 mGy. The estimated threshold dose for later intellectual disability is 60–310 mGy between 8–15 weeks and 250–280 mGy from 16 to 25 weeks. All the radiological procedures commonly performed in pregnancy fall below the threshold of 50 mGy (Table 4). External shielding of the maternal pelvis reduces the radiation dose by only 3% at all stages of gestation. There is no evidence for teratogenic or mutagenic risk with iodinated contrast in pregnancy, and no cases of fetal or neonatal hypothyroidism have been reported. Iodinated and gadolinium contrast are safe to use in lactating patients as the systemic doses absorbed by the infant from breast milk are less than 0.01% and 0.0004% of the maternal dose, respectively. 94
Estimated fetal dose with radiological examinations.

Positron emission tomography (PET) scanning results in less than 10 mGy fetal dose exposure and may be performed with MRI or comprised tomography (CT). 95 More than 100 cases of 18-fluorodeoxyglucose-PET with CT or MRI have been reported in pregnancy, without adverse effect on the neonate or during childhood.
Conclusion
Physiological changes in pregnancy result in alterations in reference intervals of pathology tests and anatomical changes which need to be taken into consideration when investigating gastroenterological disorders. Except for the uncertainty regarding capsule endoscopy and the need to avoid gadolinium, gastroenterological procedures and radiological investigations appear to be safe for the mother and fetus.
Footnotes
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethics approval was waived by the Mater Human Research Ethics Committee.
Informed consent
Not applicable.
Guarantor
AM.
Contributorship
AM researched and wrote the manuscript.
